Abstract
Percutaneous biliary drainage and radiofrequency ablation (RFA) have long been used for malignant biliary obstruction (MBO). Endoscopic radiofrequency ablation (eRFA) has been performed for this condition, and it has also been performed in combination with endoscopic biliary stenting, and/or chemotherapy. Although eRFA is apparently being used in a wide variety of applications, there are insufficient reports on its use, and mostly from retrospective studies. This article summarizes and seeks to clarify the status of RFA for MBO. eRFA for MBO with endoscopic biliary stenting was shown in a recent meta-analysis to improve overall survival (OS) at 6 months of follow-up, but there was no improvement of stent patency. A combination of eRFA and chemotherapy reportedly improved OS and progression-free survival, especially for patients with locally-advanced biliary tract cancer. When eRFA was performed for occluded self-expandable metal stents (SEMSs), the time to recurrent obstruction in the eRFA group was significantly longer than that in the patients treated by uncovered SEMS placement alone. eRFA has also been performed for inoperable ampullary tumors, and the median OS was significantly longer in an eRFA group than in a stenting alone group, and there was improvement of obstructive jaundice. eRFA reportedly has a high clinical success rate for patients after endoscopic papillectomy. Future studies should examine the synergistic effects of using immune-checkpoint inhibitors and eRFA together. eRFA has been shown to have therapeutic effects in various applications, but further large prospective research is needed to improve the level of evidence.
Plain language summary
Percutaneous biliary drainage and radiofrequency ablation have long been used for malignant biliary obstruction. Endoscopic radiofrequency ablation (eRFA) has been performed for this condition, and it has been also performed in combination with endoscopic biliary stenting, and/or chemotherapy. Although eRFA is apparently being used in a wide variety of applications, there are insufficient reports on its use, and mostly from retrospective studies. This article summarizes and seeks to clarify the status of RFA for malignant biliary obstruction. eRFA for malignant biliary obstruction with endoscopic biliary stenting was shown in a recent meta-analysis to improve overall survival at 6 months of follow-up, but there was no improvement of stent patency. A combination of eRFA and chemotherapy reportedly improved overall survival and progression-free survival, especially for patients with locally-advanced biliary tract cancer. When eRFA was performed for occluded self-expandable metal stents, the time to recurrent obstruction in the eRFA group was significantly longer than that in the patients treated by uncovered self-expandable metal stent placement alone. eRFA has also been performed for inoperable ampullary tumors, and the median overall survival was significantly longer in an eRFA group than in a stenting alone group and there was improvement of obstructive jaundice. eRFA reportedly has a high clinical success rate for patients after endoscopic papillectomy. Future studies should examine the synergistic effects of using immune-checkpoint inhibitors and eRFA together. eRFA has been shown to have therapeutic effects in various applications, but further large prospective research is needed to improve the level of evidence.
Keywords
Introduction
In cases of late-stage malignant biliary obstruction (MBO), chemotherapy is sometimes thought to be the only option when surgical resection is impossible, but the efficacy is limited. Biliary stenting is sometimes used as a palliative procedure to relieve biliary obstructions; a self-expandable metal stent (SEMS) or a plastic stent can be inserted as treatment for MBO. 1 SEMS are perhaps preferable to plastic stents because of the longer stent patency, but as survival time is extended by chemotherapy, stent occlusion can sometimes occur before patients’ deaths.
Meanwhile, percutaneous biliary drainage has been used for MBO for some time, and radiofrequency ablation (RFA) has also been used. As a palliative and alternative treatment for MBO, some centers use endoscopic radiofrequency ablation (eRFA). Since the development of a type of catheter for eRFA, its use has expanded worldwide in the treatment of MBO. 2 However, the efficacy of eRFA for MBO is unclear, and reported results differ. Furthermore, eRFA apparently has a wide variety of applications such as in cases of stent occlusion, in combination with chemotherapy, and in cases with an ampullary tumor, but reports are insufficient. In this article, we therefore summarize the role of eRFA for biliary diseases and summarize how it has been applied.
Basic technical details of RFA
RFA is based on the principle of delivering alternating electrical current through an electrode placed in the target tissue (Figure 1). 3 The current induces ionic agitation, which generates frictional heat and leads to controlled coagulative necrosis of the surrounding cells. The extent of ablation depends on factors such as electrode design, tissue impedance, and the duration of energy delivery. Modern RFA systems often incorporate temperature monitoring, with impedance feedback to enhance safety and efficacy by ensuring consistent thermal injury while minimizing collateral damage.
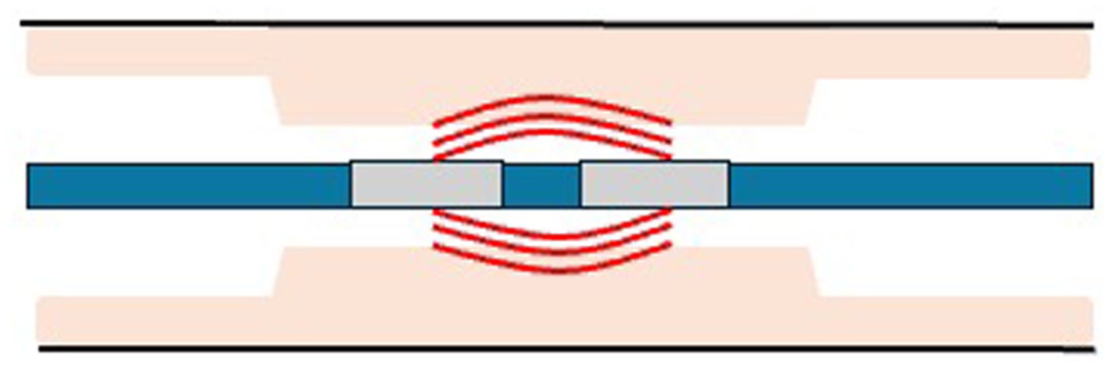
Image of radiofrequency ablation.
Potential complications of RFA
RFA is generally safe and minimally invasive, but it is not without risks. Complications can sometimes arise from unintended thermal injury to adjacent structures, leading to strictures, perforation, or bleeding. Endobiliary RFA may induce cholangitis (6.3%–20%), pancreatitis (0%–20%), or cholecystitis (0.5%–5.2%). 4 The risk profile is influenced by tumor location, underlying anatomy, and patient comorbidities. Complication rates can be significantly reduced by careful patient selection, optimized procedural technique, and the use of adjunctive measures such as antibiotics and stent placement.
Activation of tumor immunity by RFA
RFA can induce hyperthermic cell injury and cause coagulative necrosis and cell death. Necrosis results from a loss of plasma membrane integrity and a release of necrotic-cell intracellular antigens as well as damage-associated molecular patterns, including heat shock proteins, parts of intracellular organelles, and nucleotide particles. These debris components can contain tumor antigens that can be recognized and targeted by the host immune system, leading to its activation. 5 Meanwhile, the ablated area becomes densely populated by neutrophils, macrophages, dendritic cells, natural killer cells, and B and T lymphocytes, which increase cytokine production.6,7 According to several human and animal studies, the immune response is not necessarily limited to the local site of ablation, and there may also be a systemic reaction. 8 A systemic antitumor T-cell immune response, which is characterized by increased levels of CD8+ and CD4+ T cells, has been repeatedly observed in relation to hepatocellular carcinoma.9,10 However, despite an apparently great deal of research activity, there are currently few reports on the use of eRFA for MBO. 2
Currently available equipment and instrument for RFA
Two kinds of RFA catheters have been clinically used for MBO: Habib EndoHPB bipolar radiofrequency catheter (EMcision UK, London, UK; Boston Scientific, Marlborough, MA, USA), and ELRA (EndoLuminal Radiofrequency Ablation; Taewoong Medical, Seoul, Korea). Habib Endo HPB catheter has an 8 Fr outer diameter, an effective length of 180 cm and two electrodes (Figure 2(a)). It has the potential to produce an ablation depth of 4 mm and an ablation zone of around 25 mm for up to 120 s. ELRA has a 7 Fr outer diameter, and an effective length of 175 cm. Unlike Habib Endo HPB catheter, it has four types of electrode length (11, 18, 22, 33 mm) (Figure 2(b)), so endoscopists can choose from them to suit the individual case (Table 1).

(a) Endoscopic radiofrequency ablation catheter (Habib EndoHPB, Boston Scientific, Marlborough, MA, USA). © 2025 Boston Scientific Corporation. All rights reserved (b) Endoscopic radiofrequency ablation catheter (ELRA, Taewoong Medical, Seoul, Korea). © 2025 Taewoong Medical co, Ltd. All rights reserved.
Endoscopic radiofrequency ablation catheters.

ELRA, EndoLuminal Radiofrequency Ablation; RFA, radiofrequency ablation.
Percutaneous endobiliary RFA
Percutaneous transhepatic cholangiodrainage with stent placement has been performed for MBO, and there are recent reports on the combination of percutaneous RFA and stent placement (Table 2).11–17 No significant difference in overall survival (OS) was seen in the study with the largest number of cases between RFA plus stent insertion and stent insertion alone (12.3 months vs 11.8 months,
Summary of the latest studies about percutaneous endobiliary radiofrequency ablation.

BTC, biliary tract cancer; CC, cholangiocarcinoma; GC, gallbladder cancer; ICC, intrahepatic cholangiocarcinoma; OS, overall survival; PC, pancreatic cancer; RFA, radiofrequency ablation; SEMS, self-expandable metal stent.
Performance of eRFA
In eRFA, a catheter is inserted via guidewire into the bile duct during endoscopic retrograde cholangiopancreatography. After confirming the location of the malignant biliary stricture by inducing contrast medium into the bile duct, the RFA catheter is inserted, and the electrode is aligned with the stricture caused by tumor (Figure 3). The RFA catheter is set at the location of the stricture, and then eRFA is performed, and a plastic stent or SEMS is introduced across the stricture. Before and after eRFA, peroral cholangioscopy images are shown in Figure 4(a) and (b). The burned tumor can be observed in the bile duct. Endoscopic retrograde cholangiography before and after eRFA could show improvement of bile duct stricture (Figure 4(c) and (d)).

Image of endoscopic radiofrequency ablation for unresectable cholangiocarcinoma.
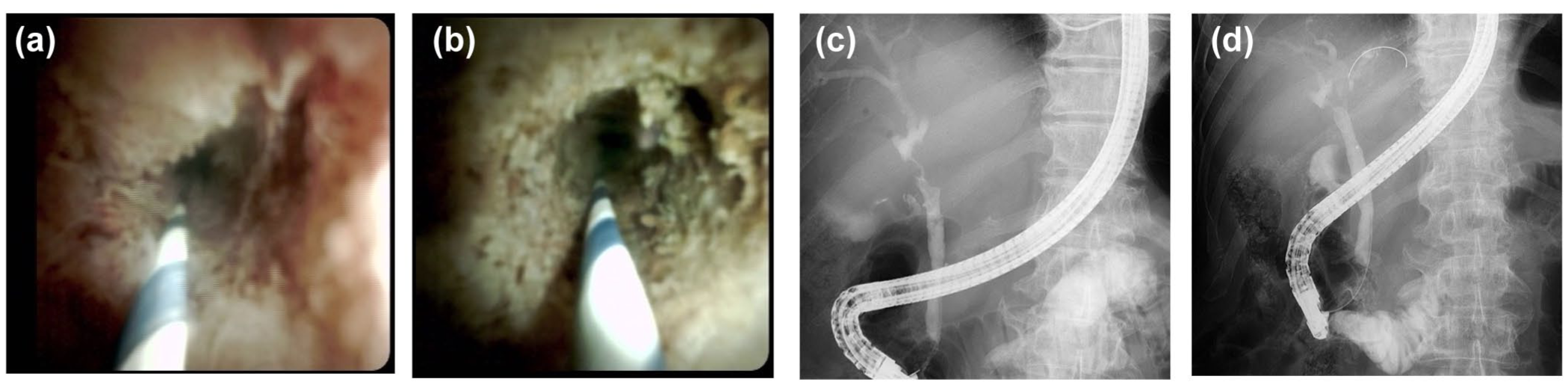
(a) Still image of biliary tract cancer before radiofrequency ablation. (b) Still image of biliary tract cancer after radiofrequency ablation. (c) Still image of endoscopic retrograde cholangiography before radiofrequency ablation. (d) Still image of endoscopic retrograde cholangiography after radiofrequency ablation.
Combination of eRFA and endoscopic biliary stenting
A recent meta-analysis of nine studies compared eRFA with endoscopic biliary drainage (EBS) versus EBS alone in patients with MBO (Table 3).18–27 Five of the nine studies conducted an analysis to consider stent patency at 3 months, none of which reported a significant difference in stent patency at 3 months between their two groups. Specifically, stent patency at 3 months in the eRFA with EBS group was not significantly longer than that in EBS-only group (RR: 1.01, 95% CI 0.92–1.11,
Combination eRFA and endoscopic biliary stenting (latest meta-analysis).

AC, ampullary cancer; CC, cholangiocarcinoma; CT, chemotherapy; EBS, endoscopic biliary drainage; eRFA, endoscopic radiofrequency ablation; GC, gallbladder cancer; NR, not reported; PC, pancreatic cancer.
A recent meta-analysis analyzed survival times across five papers.
18
The median survival time for patients undergoing eRFA + EBS was 8.1 months (95% CI 5.2–13.2), while for EBS without eRFA, it was 6.8 months (95% CI 4.8–9.2 months). Another meta-analysis reported that eRFA significantly improved survival at 6 months of follow-up (RR: 0.84, 95% CI 0.73–0.96;
These results assumed that eRFA could not prevent recurrent biliary obstruction by prolonging stent patency. However, it could directly burn the tumor and reduce the tumor volume, which could be a factor in prolonging the median survival time.
No difference in the rate of overall adverse events (under 30 days) was observed (RR: 0.82, 95% CI 0.61–1.09;
Combination of eRFA and chemotherapy
For many years, biliary tract cancer (BTC) has been treated with gemcitabine, cisplatin, or tegafur gimeracil oteracil potassium (S-1). Chemotherapy for BTC is rapidly evolving. More recently, immune-checkpoint inhibitors have emerged, and their effectiveness has been reported for BTC.28,29
Meanwhile, RFA is a minimally invasive treatment for cancer management. It can reduce cancer cells and provide palliation for unresectable cancers and thereby reducing tumor burden. 30 Additionally, RFA can activate anti-cancer immunity, making it suitable for combination with immunotherapy to enhance anticancer effects. 31 The combination of eRFA and chemotherapy has been recently reported in a randomized control study, 32 and in two retrospective studies (Table 4).33,34
Summary of recent studies about combination of CT and eRFA.

CT, chemotherapy; eRFA, endoscopic radiofrequency ablation; ExCG, extrahepatic cholangiocarcinoma; Gem+Cis, gemcitabine and cisplatin; OS, overall survival; PFS, progression-free survival; RCT, randomized controlled trial; S-1, tegafur gimeracil oteracil potassium.
In the randomized control study, eRFA with chemotherapy was compared with eRFA alone, and the median OS was longer in the eRFA + S-1 group than in the RFA alone group (16.0 months (95% CI 13.1–19.0) vs 11.0 months (95% CI 9.7–12.3);
eRFA plus chemotherapy (gemcitabine + cisplatin) groups have been compared with chemotherapy only groups.33,34 In one study, the median time to recurrent biliary obstruction (TRBO) was significantly longer in the chemotherapy with eRFA group (10.7 vs 5.2 months,
Another study reported that the median OS was 17.3 months in their chemotherapy with eRFA group (95% CI 10.9–23.8) versus 8.6 months in their chemotherapy only group (95% CI 4.9–12.4). 34 PFS was 12.9 months in the chemotherapy with eRFA group (95% CI 7.8–18.0) vs 5.7 months (95% CI 4.0–7.4) in the chemotherapy only group. 34 Median OS in patients with non-metastatic disease was 20.9 months for the chemotherapy with eRFA group (95% CI 17.9–24.0) versus 12.4 months (95% CI 3.7–21.0) in the chemotherapy only group, and in patients with metastatic disease it was 15.0 months (95% CI 4.7–25.3) versus 8.6 months (95% CI 4.3–13.0), respectively. 34 OS and PFS were shown to be significantly longer in the chemotherapy with eRFA group.
In summary, eRFA has been shown to have a higher rate of cholecystitis, but other complication rates were not different from those without eRFA. In addition, the combination of chemotherapy and eRFA may be expected to prolong OS and PFS. Taken together, these results suggest that eRFA has the potential to prolong OS and PFS in patients with locally advanced BTC, but not in those with metastatic BTC.33,34 Patients with locally advanced cancer are perhaps good candidates for eRFA.
ERFA for occluded biliary stent
eRFA has been reported to prolong stent patency when performed for stent occlusion (Table 5).35,36 One study evaluated the efficacy of in-stent RFA followed by uncovered SEMS placement for the management of occluded SEMSs.
35
The median time to TRBO for the eRFA group was significantly higher than that treated by uncovered SEMS placement alone (117 days vs 82.5 days,
eRFA for occluded stent (stent patency).

BTC, biliary tract cancer; CT, chemotherapy; eRFA, endoscopic radiofrequency ablation; FCSEMS, full-covered self-expandable metal stent; GC, gallbladder cancer; NR, not reported; PC, pancreatic cancer; UCSEMS, uncovered self-expandable metal stent.
Based on these three reports, eRFA for occluded stent is a seemingly safe procedure that plays an important role in prolonging TRBO, but they each had a comparatively small number of patients. The effectiveness of eRFA for stent occlusion requires examination in randomized controlled studies.
ERFA for ampullary tumor
Ampullary carcinoma is a rare kind of cancer comprising just 0.2% of all gastrointestinal cancers. Recent studies reported eRFA plays a important part in extending OS for unresectable ampullary carcinoma.
One such paper reported the OS and clinical success rate of eRFA for advanced ampullary cancer.
37
Clinical success was defined as a decrease in the total bilirubin level to less than normal total bilirubin level within 4 weeks after the procedure. The median OS was significantly longer in the eRFA group than in the stenting alone group (16.9 vs 9.8 months,
Endoscopic papillectomy can be considered in ampullary adenoma, but not for ampullary cancer.
39
Endoscopic papillectomy is preferable because it is more effective than pancreatoduodenectomy in terms of mortality and morbidity. However, recurrence occurs in 30% of cases.37,38,40–43 A recent meta-analysis showed the advantages of eRFA for residual lesions and as a complementary therapy for an intraductal extension.
44
The clinical success rate of eRFA after endoscopic papillectomy was 75.7% (95% CI 65–88;
Summary and future perspectives
eRFA may play an important role for the improvement of patient prognosis in patients with MBO. If endoscopic retrograde cholangiopancreatography can be performed, it is perhaps feasible for patients with MBO. The reports in the literature suggest that eRFA can extend median survival time but not stent patency. Its addition to chemotherapy can prolong OS and PFS, especially in patients with locally advanced BTC. Nonetheless, further large-scale prospective studies on the additive effect of eRFA on chemotherapy are required to prove this. eRFA has also been utilized in cases in which metal stent occlusion has occurred. TRBO (stent patency) is prolonged by the use of eRFA in cases of stent occlusion after inserting SEMS. In patients with pancreatic cancer and BTC, we suggest that reducing the incidence of stent obstruction will alleviate the burden on patients. Furthermore, eRFA is also being applied to the treatment of ampullary tumors. eRFA is effective in inoperable cases with jaundice and those with intraductal extension, and it prolongs OS for patients with inoperable ampullary tumors, and it has become an alternative treatment for residual lesions after endoscopic papillectomy.
There are nonetheless some outstanding issues in relation to eRFA. First, direct observation with peroral cholangioscopy is necessary to ensure that eRFA has successfully ablated the tumor in the bile duct. However, significant costs and long procedure time would be required to determine whether the bile ducts could be cauterized after eRFA with peroral cholangioscopy. eRFA has the potential to improve the prognosis of patients with locally advanced BTC, so a simple means of determining whether a BTC has been appropriately ablated with eRFA is warranted. Secondly, eRFA may elevate the risk of cholecystitis. The cause of cholecystitis is thought to be related to the location of tumor ablation with eRFA, but there are currently no detailed studies on the relation of the ablated site to adverse effects. Finally, the combination of chemotherapy and eRFA is still not widely reported in prospective observational studies. Future studies should ideally examine the synergistic effects of using immune-checkpoint inhibitors and eRFA together. eRFA has been shown to have therapeutic effects for a variety of applications, so further large-scale prospective research is needed to improve the level of evidence.
