Abstract
Background:
Faecal biomarkers are increasingly utilized for disease assessment in inflammatory bowel disease (IBD).
Objectives:
To characterize the relative and combined accuracy of faecal calprotectin (FC) and faecal immunochemical testing (FIT) for detecting endoscopic and histologically active disease in Crohn’s disease (CD) and ulcerative colitis (UC), subdivided by disease location.
Design:
A prospective cohort study.
Methods:
Patients with confirmed IBD undergoing routine ileocolonoscopy for activity assessment were prospectively recruited and performed both FC and FIT ±30 days of ileocolonoscopy. Endoscopic activity was assessed via the simplified endoscopic score for CD, Mayo endoscopic score for UC and histological activity graded as nil/mild/moderate. Receiver-operator curve analyses were utilized to assess the performance of FC and FIT per disease subtype and location.
Results:
In all, 137 (79 CD, 57 UC) patients were recruited. FC was more sensitive than FIT in detecting active endoscopic (CD: 91% versus 69%, UC: 94% versus 82%) and histological (CD: 86% versus 55%, UC 88% versus 56%) disease. However, FIT was more specific than FC in detecting active endoscopic (CD: 94% versus 56%, UC: 85% versus 69%) and histological (CD: 93% versus 55%, UC: 96% versus 70%) diseases. FIT was more sensitive and specific than FC in detecting active colonic CD (endoscopic activity: 94% versus 93%, histological activity: 92% versus 77%, respectively); however, it was poorly sensitive for active ileal CD (43% versus 89%).
Conclusion:
FC demonstrated higher sensitivity and FIT higher specificity for active IBD. Hence, dual testing was synergistic, displaying excellent performance characteristics across most IBD locations and subtypes, holding promise for future clinical application.
Trial registration:
Not applicable.
Introduction
Inflammatory bowel diseases (IBDs), comprising Crohn’s disease (CD) and ulcerative colitis (UC), are a group of chronic autoimmune conditions with increasing prevalence worldwide.1,2 Despite differing phenotypes, UC and CD share many similarities including methods of disease activity monitoring, disease-related treatment targets and the medical therapies applied in both conditions.3–6 Recently, Selecting Therapeutic Targets in Inflammatory Bowel Disease-2 (STRIDE-2) guidelines in IBD were developed and advocated for frequent periodic activity assessment with ileocolonoscopy remaining the gold standard of disease evaluation.7,8 Furthermore, treatment targets now include both defined levels of endoscopic activity and histological healing, the latter particularly in UC. 8 Yet with demand outpacing supply as well as patient-related risk factors, ileocolonoscopy remains an impractical assessment modality for frequent, serial disease monitoring as promulgated by recent international consensus. Thus, highly sensitive non-invasive disease activity biomarkers remain an unmet need.
Faecal calprotectin (FC) is a validated non-invasive measurement of IBD activity.9,10 Calprotectin is a protein found predominantly in neutrophils but has also been detected in monocytes and macrophages.11,12 In IBD, the inflamed intestinal mucosa results in increased permeability to migrating neutrophils, facilitating the quantitative measurement of calprotectin as a surrogate marker of intestinal inflammation in stool.11,12 Despite its widespread use, there are conflicting data regarding the accuracy of FC in ileal versus colonic IBD.13–15 By contrast, faecal immunochemical testing (FIT) provides quantitative measurement of haemoglobin in faeces. 16 FIT is performed worldwide and is the backbone of multiple national bowel cancer screening programs with a positive result usually necessitating endoscopic examination.17,18 Moreover, FIT has also been explored as a faecal biomarker in IBD assessment albeit published data are sparse.19–22 FIT levels are putatively representative of intestinal inflammation given the inflamed intestinal mucosa results in increased permeability facilitating haemoglobin loss, which is then detected in the stool in active forms of IBD. 16
Hence, the primary aims of this study were (1) to determine the accuracy of FIT versus FC for identifying endoscopically and histologically active disease in CD and UC and (2) to assess the performance of FIT and FC in combination for detecting active endoscopic and histological disease in CD and UC. A secondary aim was to assess the comparative accuracy of FC and FIT with respect to disease location, that is, colonic IBD versus small bowel CD.
Methods
Study design
A multicentre prospective study was conducted across two tertiary IBD centres in Australia between March 2021 and August 2022 inclusive. Patients ⩾16 years or older with known CD or UC referred for routine endoscopic evaluation of disease by their treating physician were invited to participate and each provided a single stool sample within 30 days of ileocolonoscopy on which quantitative FIT and FC were performed. The study complied with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 23
Colonoscopy protocol
Colonoscopy was performed using a standardized bowel preparation protocol as proscribed by the study hospital under anaesthetist-administered deep sedation, with Olympus 190 series high-definition colonoscopes by experienced gastroenterologists accredited by the Conjoint Committee for the Recognition of Training in Gastrointestinal Endoscopy (CCRTGE) of Australia. Colonoscopy reporting was performed using Olympus Endobase© software. CD and UC activity was reported segmentally in real-time upon visual inspection using the simplified endoscopic score for Crohn’s disease (SES-CD) and Mayo endoscopic score (MES) for UC. The active endoscopic disease was defined as a total SES-CD ⩾3 for CD and if the total sum of all colonic segments scored by the MES was ⩾3 for UC. The active colonic CD was defined as an SES-CD ⩾3 from colonic segments only. The active ileal-only CD was defined as an SES-CD ⩾3 within the ileum with no points scored from colonic segments. The active colonic IBD was defined as either active colonic CD with an SES-CD ⩾3 from colonic segments or if the total sum of all colonic segments scored by the MES was ⩾3 for UC. The distal colonic IBD was defined similarly but the active segments were confined distally to the splenic flexure. Gastroenterologists performing each colonoscopy were blinded to respective faecal biomarker data.
Histology protocol
Histology was obtained during the endoscopic examination with biopsy forceps with a minimum of two samples per bowel segment. The location of segmental biopsies was divided into the terminal ileum and colon (encompassing the caecum, ascending colon, transverse colon, descending colon, sigmoid colon and rectum) which applied to all analyses and descriptions throughout the study.
All histology specimens were reported by gastrointestinal pathologists who had obtained a fellowship from the Royal Australian College of Pathologists. Pathologists were blinded to clinical and endoscopic disease activity data as well as FC and FIT data. Due to a lack of a widely accepted histological scoring system in UC and CD, histology specimens were graded as follows (in accordance with previous literature) 22 : inactive if no granulocytic reaction was observed in the epithelial or stomal layers of the bowel wall, mild if a granulocytic reaction (predominantly neutrophils) was seen in the lamina propria and crypt epithelium (cryptitis) and moderate if abundant inflammatory cells were observed and granulocytic reaction (predominantly neutrophils) was seen in the crypt lumen (crypt abscesses) in addition to the changes seen above in mild disease. The segment of inflammatory activity on colonic or ileal biopsies with the highest grading of inflammation was used in all analyses.
FIT and FC protocol
Samples for FC and FIT were taken up to 30 days prior to or after the colonoscopy. No faecal samples were taken during the period of bowel preparation commencing 24 h prior to the colonoscopy. In addition, no faecal samples were taken for 5 days following the colonoscopy to avoid potential erroneous results, for instance, caused by the bowel preparation, bleeding from endoscopic interventions and/or from performing biopsies during the colonoscopy.
FIT and FC testing was performed on the same faecal sample by two accredited laboratories locally. Two FIT samples were collected from the index stool sample and the highest of the quantitative results of the two was used in all related analyses. All FC levels were tested according to the manufacturer’s instructions and measured using the automated LIAISON Calprotectin assay (DiaSorin Inc., Stillwater, MN, USA). FIT was performed using the OC-Sensor PLEDIA assay (Eiken Chemical Company, Tokyo, Japan).
Statistical analyses
Continuous and categorical variables were depicted via medians (range) and frequencies (%), respectively. Non-parametric statistics were assumed and applied to all relevant analyses. Grouped FC and FIT data per grades of endoscopic and histologic activity in UC and CD were assessed with the Kruskal–Wallis test. The optimal cut-off values of FC and FIT for correctly attributing the ‘gold standards’ endoscopic and histologically active disease in UC and CD were gleaned via receiver operating characteristic (ROC) curve analyses with area under the curve statistics also derived. In each case, the actual cut-off was selected based on the highest likelihood ratio calculated from the tangent of the ROC curve at a single point of the curve on each respective ROC curve for FC and FIT. This likelihood ratio represents the proportion of positive tests confirmed with active disease by the gold standard, divided by the proportion of positive test results in those with inactive disease per the gold standard. Thus, it favours a cut-off that minimizes the inclusion of false negatives and hence favours sensitivity over specificity which was deemed most clinically relevant to the utility of faecal biomarkers assessing active disease. 24 For each category for FC and FIT, sensitivity, specificity, negative predictive value (NPV) and positive predictive value (PPV) were presented. A significance level of p < 0.05 was applied throughout the analyses in this study. All statistics were calculated using GraphPad PRISM software version 10.0 (La Jolla, CA, USA).
Results
Description of population
In all, 137 patients were consecutively recruited into this prospective observational study, 79 patients with CD and 57 patients with UC. The median duration between ileocolonoscopy and collection of faecal samples in the CD and UC cohorts was 13 and 14 days, respectively. Further characteristics of the cohort, subdivided by IBD subtype, are shown in Table 1.
Characteristics of Crohn’s disease and ulcerative colitis cohorts.
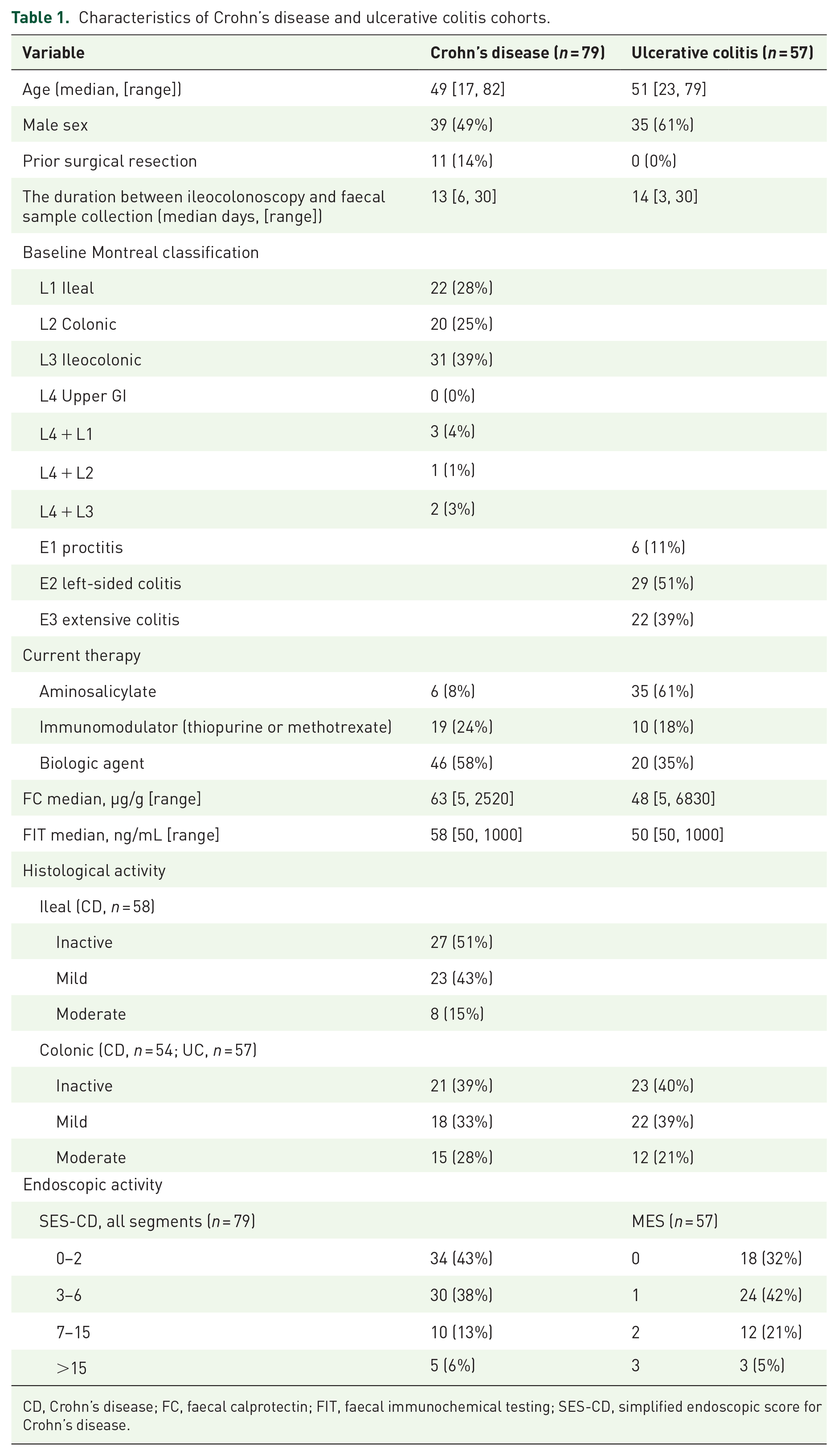
CD, Crohn’s disease; FC, faecal calprotectin; FIT, faecal immunochemical testing; SES-CD, simplified endoscopic score for Crohn’s disease.
Of the 79 patients with CD, 16 (20%) had isolated, histologically active ileitis and 31 (39%) had histologically active colitis only. Of the 57 patients with UC, 34 (60%) had histologically active colitis. Endoscopically, 45 (57%) patients with CD had active disease with 28 (of the total 79, 35%) having isolated active ileal disease and 25 (32%) having active colonic disease. In all, 39 (68%) patients with UC had endoscopically active disease (see Table 1).
Accuracy of FC and FIT representing endoscopic and histological activity in CD and UC
There was a significant difference in median FC and FIT between incremental grades of endoscopic and histological activity when incorporating both UC and CD (see Figure 1). After establishing the optimal cut-off for each biomarker via ROC analyses for each scenario, the performance of FC and FIT in various disease locations pertaining to this cohort is shown in Table 2.
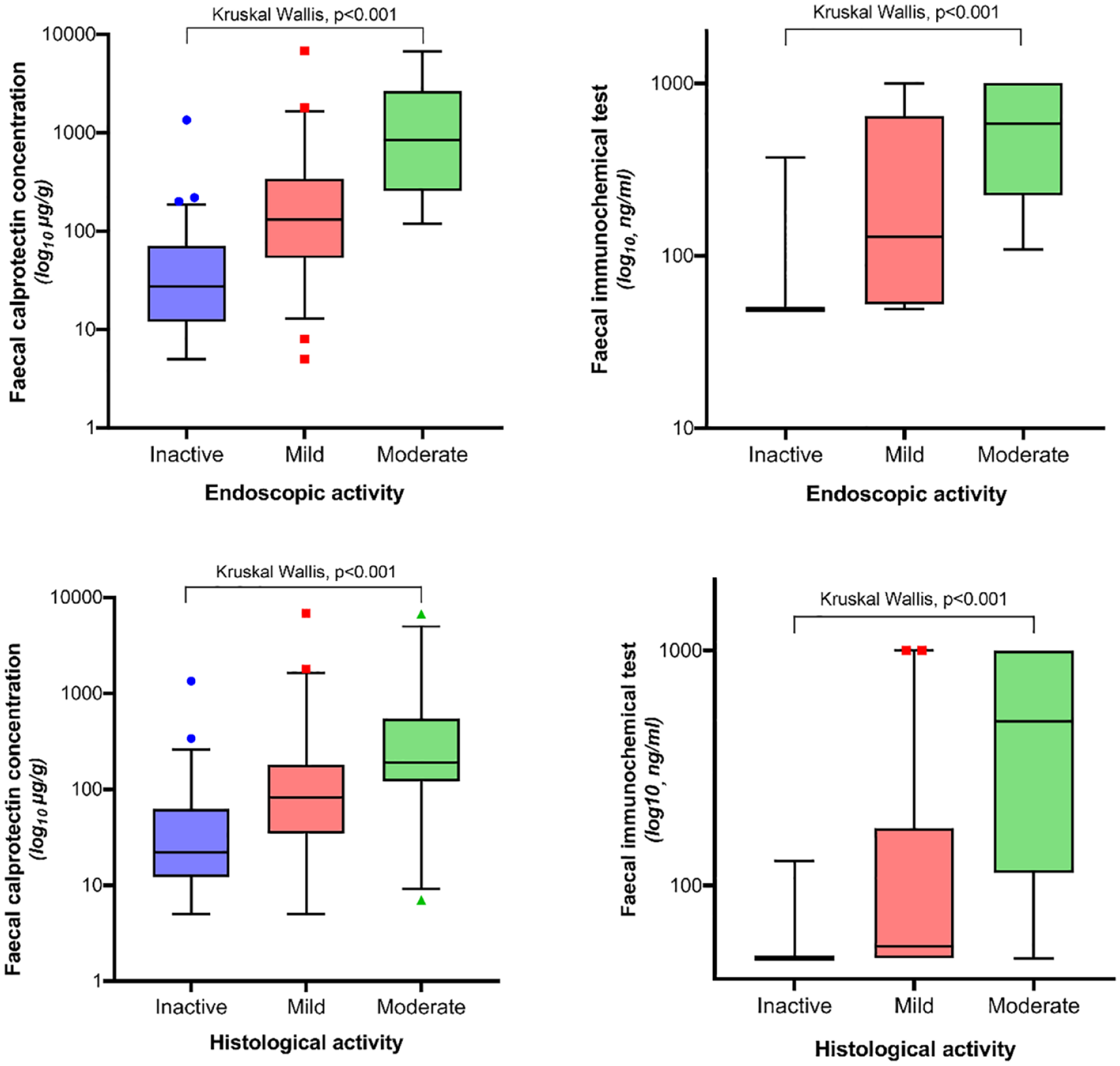
FC and FIT by endoscopic and histological grading, demonstrating a correlation between each faecal biomarker concentration and increasing endoscopic/histological activity (inclusive of both CD and UC).
Sensitivity, specificity, PPV and NPV for each disease subgroup/location of FC and FIT at optimal cut-off values, based on receiver-operator curves, in each scenario.
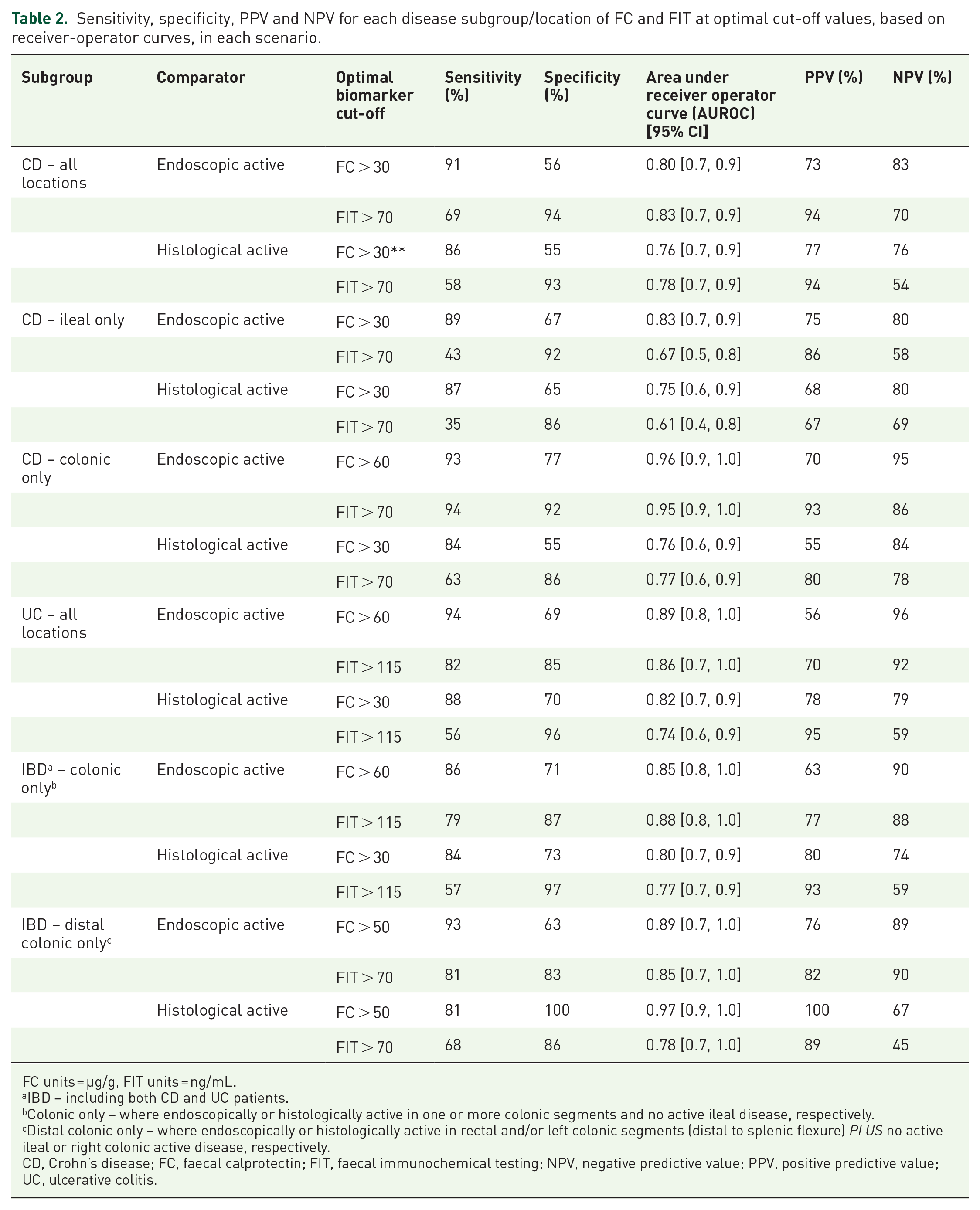
FC units = µg/g, FIT units = ng/mL.
IBD – including both CD and UC patients.
Colonic only – where endoscopically or histologically active in one or more colonic segments and no active ileal disease, respectively.
Distal colonic only – where endoscopically or histologically active in rectal and/or left colonic segments (distal to splenic flexure) PLUS no active ileal or right colonic active disease, respectively.
CD, Crohn’s disease; FC, faecal calprotectin; FIT, faecal immunochemical testing; NPV, negative predictive value; PPV, positive predictive value; UC, ulcerative colitis.
Within the entire CD cohort, the optimal FC and FIT values for classifying both endoscopically and histologically active disease were an FC >30 µg/g and FIT >70 ng/mL, respectively. FC maintained a higher sensitivity than FIT for both endoscopic (91% versus 69%) and histological (86% versus 58%) activity but conversely, there were higher specificities achieved by FIT compared to FC for both endoscopic (94% versus 56%) and histologically (93% versus 55%) active CD. Moreover, the PPVs in CD were higher with FIT compared to FC; yet, the NPVs were higher with FC (see Table 2).
Within the entire UC cohort, the optimized FC for classifying endoscopically and histologically active disease was >60 and >30 µg/g, respectively. The optimal FIT cut-off for classifying both active UC endoscopically and histologically was >115 ng/mL. FC maintained a higher sensitivity than FIT for both endoscopic (94% versus 82%) and histological (88% versus 56%) activity but conversely, there were higher specificities achieved by FIT compared to FC for classifying both endoscopic (85% versus 69%) and histologic (96% versus 70%) CD activity. Moreover, the PPVs in UC were generally higher with FIT compared to FC; yet, the NPVs were higher with FC (see Table 2).
Hence, the combined application of both FC and FIT biomarkers in CD accurately classified endoscopically and histologically active diseases across all subgroups with a range (highest of lowest and highest of highest pair in each case) of sensitivity of 84–94%, specificity of 86–94%, PPV of 67–94% and NPV of 76–96%. The combined applications of FC and FIT in the UC cohort across both targets gleaned a range of sensitivity of 88–94%, specificity of 85–96%, PPV of 70–95% and NPV of 79–96%, indicating similar performance characteristics of FC and FIT in CD versus UC.
Accuracy of FC and FIT representing endoscopic activity- and histological-activity-specific disease activity locations within IBD
There was a pattern of reduced median concentrations of both FC and FIT in relation to endoscopically active disease isolated in the ileum, versus proximal or distal colonic IBD which was significant for FC (p < 0.001) but not FIT (p = 0.08) (Figure 2).

FC and FIT levels by ileal, proximal colon and rectal only disease locations (in relation to endoscopically active disease, both CD and UC included).
Moreover, across all disease location subgroups (isolated ileal CD, colonic IBD and distal colonic IBD), there were numerically higher sensitivities of FC for both endoscopic and histological activity compared to FIT at the derived optimal cut-offs. FC achieved higher sensitivity than FIT in terms of detecting active ileal disease (sensitivity of FC >30 µg/g: 89%, 87% versus FIT >70 ng/mL: 43%, 35% for endoscopic and histological activity, respectively). Across both CD and UC, FC also achieved higher sensitivity than FIT for histologically active colonic disease (sensitivity FC >30 µg/g: 84% versus FIT >115 ng/mL: 57%) and in distal colonic disease (sensitivity FC >50 µg/g: 81% versus FIT >70 ng/mL: 68%), difference in sensitivity for detecting endoscopically active colonic and distal colonic IBD was of lesser magnitude. Conversely, in all but one disease location subgroup (histologically active distal IBD), there were numerically higher specificities achieved by FIT in each scenario, compared to FC. Moreover, the PPVs were generally higher with FIT in all but two of the subgroups (histologically active isolated CD and histologically active distal IBD); yet, the NPVs were generally higher with FC apart from one subgroup (endoscopically active distal IBD) (see Table 2).
Discussion
This study highlights the value of dual faecal biomarker testing for detecting both endoscopic and histologically active IBD and to our knowledge is the first study to assess histological CD activity using FIT. FC is a universally established biomarker that has been extensively evaluated in both CD and UC, although the optimal cut-off values to detect endoscopic and histological activity, particularly with reference to disease location, that is, small versus large bowel segments, are not yet established.9,10 FIT is a widely used marker in colorectal cancer screening worldwide with early promise in the assessment of IBD activity; however, it has yet to be incorporated into routine clinical practice in this setting.
In this study, FC and FIT levels were both able to discriminate between incremental grades of endoscopic and histological activity in UC and CD (as shown in Figure 1). But generally, FC consistently outperformed FIT as a biomarker in terms of higher sensitivity and higher NPV, which, in reality, are the most critical parameters for accurate assessment of disease activity where false negatives should be minimized as much as possible. FC was more sensitive than FIT within all but one CD location subgroup where they were essentially equivalent (colonic CD: 93% versus 94%). Similar to previous studies showing FIT did not accurately reflect isolated active ileal CD, the sensitivity of FC was far superior to FIT (endoscopic activity: 89% versus 43%, histological activity: 87% versus 35% respectively) in this subgroup.19,25 This is surmised to reflect the innate differences of inflamed colonic mucosa which typically invokes a more haemorrhagic response than the inflamed ileum. Moreover, ileal inflammation is often transmural and thus less mucosally dominant plus often shorter in segment length which is detectable through FC (usually at lower titres than colonic inflammation) yet does not produce the ‘minimal threshold’ of mucosal haemorrhage detectable by FIT.
As expected, compared with endoscopically isolated colonic CD, the optimal cut-off for identifying endoscopically active, isolated ileal CD was lower (60 versus 30 µg/g). Thus, the application of the manufacturer’s often recommended cut-off level of 50 µg/g into real-world practice could result in under-reporting of active disease isolated to the small bowel, especially in generally milder active disease as was frequently encountered in this cohort. Moreover, optimal FC cut-off levels were deemed higher for endoscopic versus histologically active colonic IBD (60 versus 30 µg/g) which may reflect the more subtle nature of milder histological activity in IBD, which often may not be appreciable with endoscopic assessment. 26 Also, compared to proximal and distal colonic segments, significantly lower FC levels were gleaned from endoscopically isolated ileal CD, whereas FIT was unable to demonstrate separation between these disease locations (see Figure 2). Nevertheless, the optimal FC and FIT values derived in this cohort were remarkably stable between endoscopically and histologically active targets. For instance, an FC >30 µg/g cut-off was optimal for classifying both endoscopic and histological activity in CD (all in), ileal CD and the same cut-off applied to histologically active colonic CD, colonic IBD and UC (all in). A FIT >70 ng/mL optimal cut-off applied to endoscopically and histologically active CD (all in), ileal CD, colonic CD and distal colonic IBD. Finally, an FC >60 µg/g was optimal for active UC (all in), colonic CD and colonic IBD, with a similar FC >50 µg/g cut-off for distal colonic IBD for both targets. Overall, the FC values within our cohort are lower than recent guidelines which refer to levels between 100 and 250 µg/g as cut-offs to discriminate between active and inactive disease. 8 Our findings are likely explained by the fact that >70% of the entire study population had inactive or mild disease; hence, FC values were predominantly low (median calprotectin values of 63 and 48 µg/g in the CD and UC cohorts, respectively) and, in turn, lower FC cut-off values were derived.
Again, within the UC cohort, FC demonstrated a superior sensitivity for both histologically active (88% versus 56%) and endoscopically (94% versus 82%) active UC compared to FIT and were consistent with previously published literature.25,27 For instance, this study used the same histological grading system as Karling et al. 22 and achieved an identical sensitivity and specificity of 56% and 96%, respectively, in the UC cohort.
In what is perhaps the most salient finding of this study, though the sensitivity of FC typically outperformed FIT, the complementary utility of FIT was demonstrated via consistently higher specificities and PPVs in all but two subgroups (histologically active ileal CD and distal colonic IBD). From a clinical perspective, this illustrates the potential application of dual testing of both FC and FIT on a single sample, harnessing the accuracy of FC for correctly identifying active disease and FIT correctly identifying those with inactive disease. For instance, a combined testing approach across all patients with CD yielded a sensitivity of 91% and specificity of 94% for endoscopically active disease, thus minimizing the likelihood of false negatives or positives. Therefore, whilst the addition of FIT adds a small cost to FC alone, the complementary nature of its increased specificity compared to FC may ultimately mitigate the costs of further unnecessary diagnostic tests (i.e. ileocolonoscopy).
This study was subject to several potential limitations. The sample size restricted the ability to confidently assess the accuracy of the biomarkers in certain subgroups, such as distal colonic IBD. However, with the prospective data collection and rigorous phenotyping and classification of targets, we are confident that the data are robust and biases are minimized. Secondly, the endoscopic data collection was performed in real time by multiple gastroenterologists who performed the endoscopy (albeit all experienced and certified in IBD endoscopy), rather than via a central reading process, which may have impacted the accuracy of activity grading documented. Moreover, although the pragmatic histological scoring system has been used in a similar study by Karling et al., 22 it has not been formally validated. Finally, the faecal samples were collected up to a strict maximum of 30 days prior to or after the colonoscopy. Whilst no therapy change(s) occurred in any of the patients during the time between the faecal sample and endoscopy, inherently their disease activity would be subject to fluctuation which may have resulted in discordant results and have detrimentally affected the data. Similarly, it is well known that a one-off faecal biomarker sample is subject to variability and may not be reflective of the true underlying disease state, 28 but serial biomarker assessment was beyond the scope of this study.
Conclusion
This study has shown both FC and FIT as accurate surrogate biomarkers of endoscopic and histological activity in IBD. It has advanced the understanding of their respective potential in both CD and UC, according to specific disease locations in predominantly mild–moderate phenotypes, within a prospectively acquired, well-defined cohort. The most notable finding was the synergism apparently achieved with dual FC and FIT testing which typically enhanced both sensitivity and specificity (plus NPV and PPV) in this cohort, for both endoscopic and histologically assessed active disease. Moreover, the small additional cost of adding FIT to FC on a single sample suggests the potential cost–benefit of this approach whilst providing more incisive disease assessment and thus clinical decision-making in IBD. Further prospective evaluation of dual FC and FIT is eagerly awaited in different IBD clinical scenarios along with the application of rigorous endoscopic and histological indices, prior to incorporation into widespread practice.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241237895 – Supplemental material for The synergy of dual faecal immunochemical and faecal calprotectin testing for accurate assessment of endoscopic and histological activity in inflammatory bowel disease
Supplemental material, sj-docx-1-tag-10.1177_17562848241237895 for The synergy of dual faecal immunochemical and faecal calprotectin testing for accurate assessment of endoscopic and histological activity in inflammatory bowel disease by Anuj Bohra, Nicholas Batt, Krishneel Dutt, Diana Lewis, Jonathan P. Segal, Olga Newiadomski, Abhinav Vasudevan and Daniel R. Van Langenberg in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
