Abstract
Background:
Exposure–response studies have shown that higher infliximab concentrations are associated with better outcomes in inflammatory bowel disease. There is little agreement about the optimal time to measure infliximab levels in children.
Objectives:
We aimed to evaluate whether trough levels at week 6 or week 14 predict sustained remission. The secondary aim was to define target trough levels at weeks 6 and 14.
Design:
We used routinely collected electronic healthcare data of 70 anti-tumour necrosis factor naïve children with inflammatory bowel disease treated with a standard infliximab induction- and variable maintenance scheme.
Methods:
Trough levels and blood and faecal markers for disease activity were measured before every infliximab administration. Sustained remission was defined as the absence of symptoms and low inflammatory markers between weeks 26 and 52 after the start of infliximab therapy. Optimal infliximab levels at weeks 6 and 14 were determined using the receiver operating characteristic curve.
Results:
The median infliximab level at week 6 was not significantly higher in children who achieved sustained remission compared to those who did not (16.9 mg/L versus 12.0 mg/L; p = 0.058) but the median infliximab level at week 14 was significantly higher in those with sustained remission (7.7 mg/L versus 3.8 mg/L; p = 0.006). The area under the receiver operating characteristics curves at weeks 6 and 14 to predict sustained remission was 0.67 (95% CI 0.51–0.83) and 0.75 (95% CI 0.60–0.90), respectively. Target trough levels at weeks 6 and 14 were ⩾13.2 and ⩾6.9 mg/L, respectively.
Conclusion:
An infliximab measurement at week 14 with a target through level ⩾6.9 mg/L best predicted sustained remission.
Introduction
Infliximab, a monoclonal antibody against tumour necrosis factor-alpha (TNF-α), has been shown to induce and maintain mucosal healing and its use improves long-term outcomes in paediatric patients with inflammatory bowel disease (IBD). 1 Infliximab is now considered the first choice for children with active perianal fistulizing disease or children with luminal disease and increased risk of poor outcome, whereas this therapy was initially restricted to patients who failed conventional treatment.2–4
Long-term sustained remission is the ultimate treatment goal to reduce complications, surgery and malignancy but a substantial number of patients lose response to therapy.5–8
Several studies have shown that the effectiveness of infliximab can be prolonged by measuring drug levels followed by dose optimization.9–12 However, there is little agreement about the optimal time to measure infliximab levels in children to predict sustained durable remission (SDR). Singh et al. measured trough levels at weeks 14 and 54, and demonstrated that trough levels at week 14 were associated with therapeutic outcomes. We assessed whether we could confirm this association in a cohort of Dutch paediatric patients.11,13,14 We aimed to evaluate whether trough levels at week 6 and/or week 14 (respectively TL6 and TL14) predict sustained clinical and biochemical remission between 26 and 52 weeks after the initiation of infliximab therapy in paediatric IBD patients. Our secondary aim was to define target trough levels at weeks 6 and 14.
Materials and methods
Patients and study design
We performed a retrospective study that included consecutive biologic-naïve paediatric IBD patients who started infliximab treatment at Zuyderland Medical Center or Maastricht University Medical Center in the Netherlands between March 2019 and May 2022. All children had diagnostic esophagogastroduodenoscopy and ileocolonoscopy at diagnosis. The majority of patients were first treated conventionally. Patients with active luminal Crohn’s disease (CD) were treated with steroids (to a maximum of 40 mg/day) and gradual dose tapering, or with exclusive enteral nutrition for 6 weeks. Patients with active ulcerative colitis (UC) were treated with steroids and aminosalicylate dose escalation. Maintenance therapy was started at the same time as induction therapy and included azathioprine (2–2.5 mg/kg/day), while in UC this was combined with aminosalicylate (60–80 mg/kg/day). After the failure of conventional therapy, no additional endoscopy was performed before stepping up to infliximab. A minority of patients (exclusively CD) started with upfront infliximab (n = 13). All participating patients received a standard dose of 5 mg/kg bodyweight on weeks 0, 2 and 6, irrespective of the disease phenotype. No dose adjustments were made during the induction phase. The first post-induction interval was 8 weeks for all patients. The decision to shorten the interval after 14 weeks was based on proactive therapeutic drug monitoring (TDM), to keep infliximab levels above 5 mg/L. The study complied with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 15
Data collection
At the time of each infliximab administration, disease activity was determined using the Paediatric Ulcerative Colitis Activity Index (PUCAI) 16 or the Paediatric Crohn’s Disease Activity Index (PCDAI), 17 according to the disease phenotype. In addition, C-reactive protein (CRP), faecal calprotectin (FC), erythrocyte sedimentation rate (ESR), albumin, infliximab levels and antibodies-to-infliximab (ATI), if any, were measured.
In Zuyderland Medical Center, infliximab levels were measured with a commercially available enzyme-linked immunosorbent assay (ELISA) kit (apDia, Turnhout, Belgium), which was implemented on an automated ELISA processor at the laboratory for clinical chemistry and haematology. When a patient’s infliximab concentration was <1.0 mg/L, ATIs were determined with the in-house ELISA kit of Sanquin Diagnostics (Amsterdam, the Netherlands) as described elsewhere. 18
Blood samples collected at Maastricht University Medical Center were sent to Sanquin Diagnostic Services (Amsterdam, the Netherlands) for evaluation of infliximab and ATI concentration. The infliximab assays of both hospitals agreed sufficiently to be used interchangeably. 19
Definitions of sustained clinical and biochemical remission
We used composite scores to define ‘clinical and biochemical remission’. In CD, this required a Mucosal Inflammation Non-Invasive index below 8. 20 In UC, clinical and biochemical remission was a PUCAI below 10 in combination with an FC concentration below 250 µg/g. Sustained remission was reached when the above-defined targets were maintained between 26 and 52 weeks after initiation of infliximab therapy.
Statistical analysis
Numerical variables were summarized as the median and interquartile range (IQR), that is, the 25th and 75th percentiles. They were compared across independent groups using the Mann–Whitney U tests. Categorical variables were summarized as frequency and percentage and were compared across independent groups using the chi-square test. Associations between numerical variables were assessed with Spearman’s rank correlation coefficient. Receiver operating characteristics (ROC) analysis with Youden’s J-statistics was used to determine infliximab thresholds that best predicted sustained clinical and biochemical remission. The overall prediction quality was estimated using the area under the ROC curve (AUROC) and reported together with a 95% confidence interval (CI). The association between potential covariates (CRP, ESR, FC, albumin and co-medication) and TL6 or TL14 was assessed with Pearson’s correlation coefficients (r).
Results
Patient characteristics
A total of 70 biologic-naïve children with IBD were identified between March 2019 and May 2022, with boys and girls equally divided (Table 1). One patient developed ATI after the second induction dose and discontinued infliximab. In all, 69 patients reached the maintenance phase at week 14. In all, 22 of 69 patients missed TL6 or TL14 and were excluded from the analyses. Four patients discontinued infliximab before week 52 and were analysed in the ‘no sustained remission’ group, bringing the total in this group to 16. The total number of patients in the ‘sustained remission’ group is 31 (see Supplemental Digital Content, Figure S1).
Baseline characteristics of 70 paediatric IBD patients.
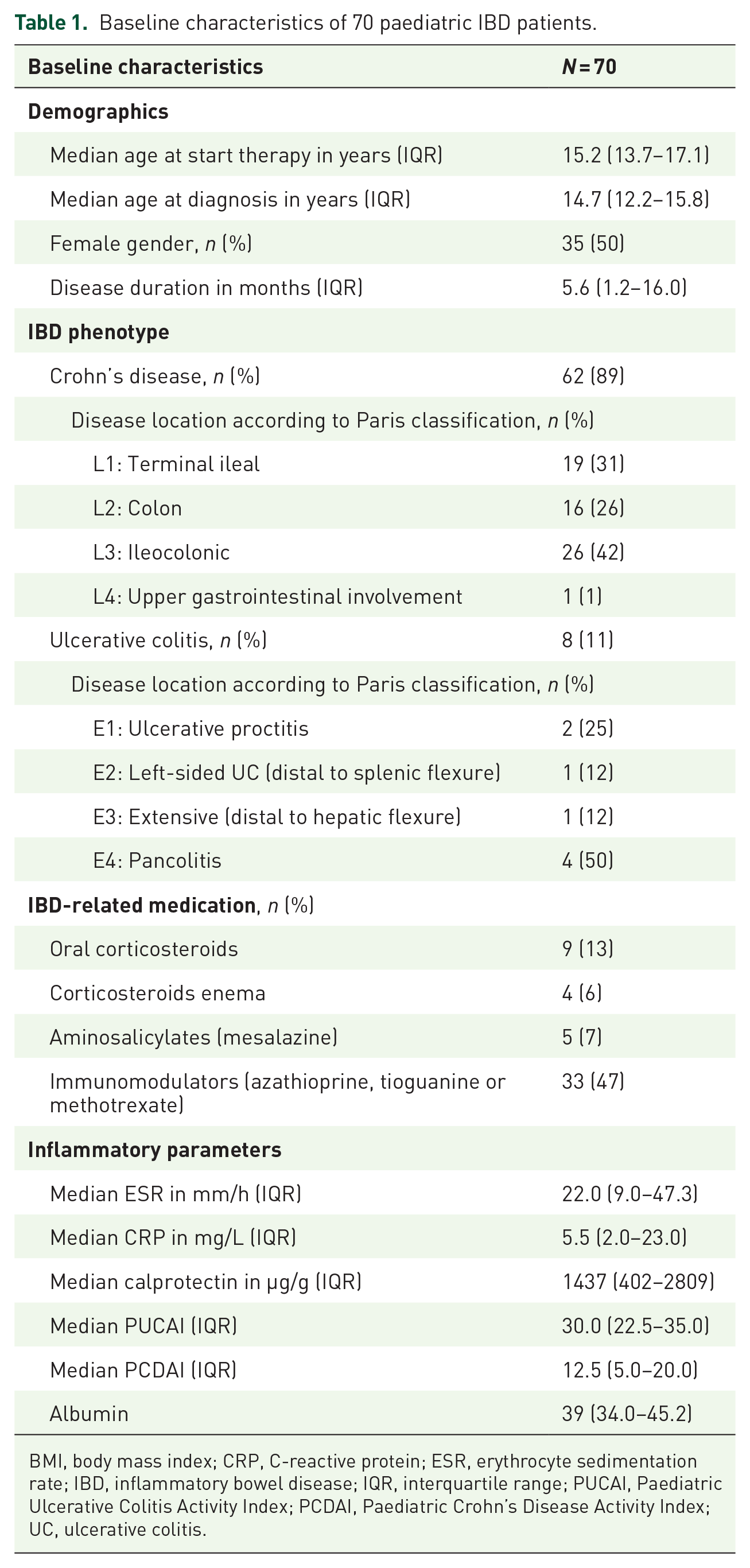
BMI, body mass index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IBD, inflammatory bowel disease; IQR, interquartile range; PUCAI, Paediatric Ulcerative Colitis Activity Index; PCDAI, Paediatric Crohn’s Disease Activity Index; UC, ulcerative colitis.
The median (IQR) age at diagnosis for the cohort was 15.2 (13.7–17.1) years, the median disease duration was 5.6 (1.2–16.0) months and 87% of patients had the diagnosis CD. Indications for initiation of infliximab included the presence of predictors of poor outcome (n = 21, 30%), failure of conventional medical treatment (n = 45, 64%), steroid-refractory disease (n = 2, 3%) and perianal fistulizing disease (n = 2, 3%).2 Median infliximab dose at baseline was 5.3 mg/kg (IQR 5.0–6.0). Forty-five percent of the final included patients used immunomodulators at baseline.
Infliximab trough concentrations
The median infliximab trough level 6 weeks after the study baseline (TL6) was 15 (9.9–20.0) mg/L, and 14 weeks after the study baseline (TL14) 5.5 (IQR 3.0–9.6) mg/L (n = 55). The median trough levels at weeks 26 and 52 after the study baseline were 5 (IQR 2.5–9.5) mg/L (n = 56) and 6.2 (IQR 3.1–9.8) mg/L (n = 38), respectively.
Sustained clinical and biochemical remission between 26 and 52 weeks
Steroid-free sustained remission was achieved in 31 of 47 (66%) of participants. At the study baseline, the patients in this group were not significantly different from those in the ‘no sustained remission’ group (Table 2).
Characteristics of participants who achieved sustained remission and who did not.

ATI’s, antibodies towards infliximab; BMI, body mass index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IQR, interquartile range; TL, trough level.*p value < 0.005.
Table 2 and Figure 1 show that the median infliximab level at week 6 was not significantly higher in children who achieved sustained remission compared to those who did not (16.9 mg/L versus 12.0 mg/L; p = 0.058) but the median infliximab level at week 14 was significantly higher in those with sustained remission (7.7 mg/L versus 3.8 mg/L; p = 0.006).

Median infliximab trough levels and interquartile ranges at 6 and 14 weeks for patients who achieved sustained remission (blue) and those who did not (red).
ROC-curve analyses (Figure 2) show that TL14 better predicts sustained remission than TL6 [AUROC 0.75 (95% CI 0.60–0.90) compared to 0.67 (95% CI 0.51–0.83)]. The ROC-based optimal cut-off level to predict sustained remission at week 6 was 13.2 mg/L with a sensitivity of 68%, a specificity of 69%, a positive predictive value of 81% and a negative predictive value of 52%. The optimal cut-off level to predict sustained remission at week 14 was 6.9 mg/L with a sensitivity of 58%, a specificity of 81%, a positive predictive value of 86% and a negative predictive value of 50%.

ROC curve for infliximab trough level at 6 weeks (green line) and 14 weeks (red line) predicting sustained remission from 26 until 52 weeks after the start of infliximab therapy.
Covariate analysis
TL6 correlated significantly with immunosuppressive co-medication at that timepoint (r = −0.314, p = 0.032) and with biomarkers of inflammation [CRP (r = −0.399, p = 0.007), ESR (r = −0.333, p = 0.022) and calprotectin (r = −0.345, p = 0.039)]. Univariable logistic regression showed that concomitant immunomodulator use at baseline, 6 weeks and 14 weeks was not predictive for sustained remission from 26 weeks until 52 weeks (Table 2).
Discussion
In this Dutch paediatric IBD cohort, we showed that an infliximab concentration higher than 6.9 mg/L measured 14 weeks post-induction best predicted sustained remission between 26 and 52 weeks after the initiation of infliximab therapy. This finding is in accordance with the observations done in a paediatric retrospective cohort described by Singh et al. 11 They concluded that a trough level 14 weeks after initiation of infliximab should ideally be above 5.1 mg/L to predict SDR from week 22 through week 54.
Comparison with existing literature
Table 3 shows that multiple studies have demonstrated an exposure–response relationship of infliximab in children with IBD. At week 6 of the induction phase (i.e. before the third infusion), an infliximab concentration between 8.3 and 13.5 mg/L predicts clinical remission in the early postinduction phase.13,14,21 An infliximab concentration higher than 5–7 mg/L measured at week 14 (i.e. before the fourth infusion) not only predicts short-term biochemical or endoscopic remission but also a sustained effect that lasts until week 52.11,22,23,24,25
Overview of pre- and postinduction infliximab target concentrations and therapeutic outcomes in paediatric patients with IBD.

CD, Crohn’s disease; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; FC, faecal calprotectin; IBD, inflammatory bowel disease; IFX, infliximab; PCDAI, Paediatric Crohn’s Disease Activity Index; PUCAI, Paediatric Ulcerative Colitis Activity Index; SES, Simple Endoscopic Score; TDM, therapeutic drug monitoring; TL, trough level; UC, ulcerative colitis; wPCDAI, Weighted Paediatric Crohn’s Disease Activity Index.
The optimal timing of trough-level measurement is debated throughout the literature. Induction trough levels are usually determined before the third infliximab infusion, whereas post-induction trough levels are usually determined before the fourth infusion. Suboptimal trough levels at week 6 possibly give rise to insufficient therapeutic response and increase the risk for antibody formation prior to the first maintenance dose. 26 By contrast, the prospective personalised anti-TNF therapy in Crohn’s disease study ([PANTS], involving anti-TNF-naïve patients aged 6 years and older) 27 found that the sole factor independently associated with primary nonresponse was low drug concentration at week 14 [odds ratio 0·35 (95% CI 0·20–0·62), p = 0.00038]. Their findings align with the conclusions drawn from our study.
Limitations
A limitation of our study was that we did not confirm the presence of endoscopic remission at 52 weeks. By contrast, our definition of sustained remission was based on repeated symptom scores and FC concentrations within the normal range between 26 and 52 weeks. When patients fulfil these criteria, we usually do not expose them to another colonoscopy. In a post hoc analysis of the Tailored Treatment With Infliximab for Active Crohn’s Dease (TAILORIX) clinical trial, in which the correlation between FC levels and endoscopic disease activity in IBD patients was examined, calprotectin correlated well with endoscopic outcomes. 28 Furthermore, the Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE)-initiative acknowledges in the STRIDE-II that an FC level in the range of 100–250 mg/kg can be used as an intermediate treatment target in both UC and CD. 29 Second, the retrospective design of this study bears the risk of overestimating the predictive power of TL14 for sustained remission. Several other studies did find comparable optimal therapeutic threshold levels which supports our results. Third, our patient cohort included only two cases with perianal involvement and our infliximab thresholds can probably not be applied to children with exclusive perianal disease. A recent study showed that children with perianal CD require higher trough levels for fistula closure. 30
Implications for paediatric practice and further research
As we found TL14 ⩾ 6.9 mg/L to be strongly associated with favourable outcomes, we suggest measuring infliximab concentrations before the fourth infusion at 14 weeks and in case of concentrations lower than 6.9 mg/L to escalate the dosage or to shorten the interval.
The validity of our study findings could be enhanced through replication in a prospective, multi-centre trial featuring two arms: one employing conventional treatment with reactive TDM for insufficient clinical responses, and the other utilizing postinduction therapeutic drug levels with treatment adjustments. While population pharmacokinetic modelling may hold significance, it falls outside the scope of this manuscript.
Conclusion
An exposure–response relationship of infliximab in paediatric IBD was confirmed in the current study. The infliximab levels measured 14 weeks after the start of therapy outperformed the levels measured 6 weeks after the start of therapy in predicting sustained clinical and biochemical remission from 26 until 52 weeks after starting infliximab. The optimal therapeutic threshold level at 14 weeks after the start of therapy was 6.9 mg/L.
As drug concentrations are modifiable factors in treatment effectiveness, we suggest monitoring and adjusting infliximab concentrations to improve treatment outcomes.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231222337 – Supplemental material for Early infliximab trough levels in paediatric IBD patients predict sustained remission
Supplemental material, sj-docx-1-tag-10.1177_17562848231222337 for Early infliximab trough levels in paediatric IBD patients predict sustained remission by Nanja Bevers, Arta Aliu, Dennis R. Wong, Bjorn Winkens, Anita Vreugdenhil, Marieke J. Pierik, Luc J. J. Derijks and Patrick F. van Rheenen in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848231222337 – Supplemental material for Early infliximab trough levels in paediatric IBD patients predict sustained remission
Supplemental material, sj-docx-2-tag-10.1177_17562848231222337 for Early infliximab trough levels in paediatric IBD patients predict sustained remission by Nanja Bevers, Arta Aliu, Dennis R. Wong, Bjorn Winkens, Anita Vreugdenhil, Marieke J. Pierik, Luc J. J. Derijks and Patrick F. van Rheenen in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We would like to thank Adrienne Hansen, Jeannie Ruiters and Esmee Picavet for their help in collecting information from the electronic healthcare files from included patients.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
