Abstract
Background:
Immunogenicity to antitumor necrosis factor alpha agents, such as infliximab (IFX), may lead to therapeutic failure.
Objectives:
This study evaluated the relationship between free and total antibodies-to-infliximab (ATIs), trough levels (TLs) of IFX, and the response to dose intensification.
Design:
We performed a prospective, observational study including pediatric patients with Crohn’s disease (CD) receiving IFX maintenance therapy without dose intensification.
Methods:
We compared clinical and laboratory outcomes according to the presence of free and total ATIs. Factors associated with response to IFX dose intensification were investigated by analyzing IFX TLs and free and total ATIs.
Results:
Of the 98 patients, 9 patients had detectable free ATIs and 38 patients had total ATIs. Patients with free ATIs had significantly lower TLs (0.7 versus 5.1 µg/mL, p < 0.001) than patients without free ATIs. However, there was no difference in the IFX TLs according to the presence of total ATIs (p = 0.2523). Analysis of the 38 samples with total ATIs showed that response to dose intensification was significantly lower in patients with free ATIs than those without free ATIs (22.2% versus 65.5%, p < 0.001). In addition, free ATIs were the only factor with poor response to dose intensification [odds ratio (OR): 14.15, 95% confidence interval (CI): 1.31–151.97, p = 0.0140]. According to the receiver operating characteristic analysis, the optimal cutoff level indicating non-response to IFX dose intensification was 30.0 AU/mL for free ATIs concentration (area under curve, 0.792; 95% CI: 0.590–0.942; sensitivity, 60.0%; specificity, 96.7%; p = 0.0241).
Conclusion:
Free ATIs, but not total ATIs, have a negative impact on the course of CD. Free ATIs are potential reliable biomarker for predicting the effect of dose intensification in patients with loss of response to IFX. Future studies based on serial and proactive therapeutic drug monitoring are required in the future.
Keywords
Introduction
Infliximab (IFX) is a purified, recombinant DNA-derived chimeric human–mouse immunoglobulin G monoclonal antibody blocking the effect of tumor necrosis factor alpha (TNFα). 1 IFX was the first monoclonal antibody therapy shown to modify disease course of inflammatory disease including Crohn’s disease (CD).2,3 However, up to 40% CD patients do not respond to anti-TNFα agents during induction phase (primary non-response) and 23–46% patients undergo secondary loss of response (LOR) during maintenance therapy. 4 The mechanisms of secondary LOR include immunogenic or nonimmunogenic. One of the most important causes for secondary LOR is immunogenic failure, the development of anti-drug antibodies (ADAs) followed by decreased in trough level (TL) of anti-TNFα agents.5,6
Immunogenicity to anti-TNFα develops when the immune system of patients recognizes drugs as antigens and triggers the formation of ADAs. ADAs accelerate drug clearance by the reticuloendothelial system and neutralize drugs by binding to anti-TNFα. 5 Several studies reported that up to 60% of patients with CD treated with anti-TNFα develop ADAs. 7 In patients with inflammatory bowel disease (IBD) treated with anti-TNFα agents, the association between presence of ADAs, low TLs, and LOR has already been revealed.8,9 In addition, ADAs have been known to be associated with adverse effects such as infusion reaction as well as therapeutic failure.7,10
Evaluating the immunogenicity of anti-TNFα is complicated due to potential analytical interference of the drug with assay, variability in antibody properties such as affinity and isotype, and variable time course of the ADAs response. 11 There are two types of assay for ADAs detection: drug-tolerant assay which measure ADAs that are free or bound to the anti-TNFα, that is, total ADAs, and drug-sensitive assay which measure ADAs that are not bound to the drug, that is, free ADAs. Information on which assay is used is essential to accurately interpret ADAs results and clinical relevance. Drug-sensitive ADAs assays typically only detect ADAs when drug levels are below the clinically effective threshold. Therefore, the strongest associations between ADAs and therapeutic efficacy were mainly established using drug-sensitive assays.12,13 On the other hand, drug-tolerant ADAs assays can detect low titers of ADAs, helping proactive therapeutic drug monitoring (TDM) strategy. However, the usefulness of free or total ADAs measurements is still controversial and more complicated due to lack of standardized assay.
According to the TDM guidelines, in patients with subtherapeutic TL with negative ADAs (pharmacokinetic failure), dose intensification could be attempted to recapture response, especially for pediatric patients with limited option for biological agents. 14 However, in the case of secondary LOR caused by immunogenic failure, dose intensification would not be of benefit and switching to an alternative anti-TNFα and/or the addition of an immunomodulatory may be indicated. Therefore, for patients with clinically suspected LOR, it is recommended to measure both drug TL and ADAs. However, studies on the associations between immunogenicity of anti-TNFα agents and response to dose intensification are limited to date. Therefore, we aimed to evaluate the relationships between free and total antibodies-to-IFX (ATIs), IFX TLs, and response to dose intensification.
Materials and methods
Patients and data collection
This prospective cohort study was conducted between February 2018 and June 2021 in the Department of Pediatrics of Samsung Medical Center.
This study included pediatric patients <18 years of age with a diagnosis of moderate-to-severe luminal CD who received IFX maintenance therapy without dose intensification. Patients with missing IFX TLs and ATIs data and primary non-responders to IFX were excluded. CD was diagnosed according to the revised Porto criteria of the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition. 15
Clinicodemographic and treatment-related data, including sex, diagnosis age, disease phenotype, family history of IBD, concomitant medication, and initial simple endoscopic score for CD 16 were obtained from the electronic medical records. The following data were collected together with IFX TL and ATIs results from the electronic medical records: Pediatric Crohn’s Disease Activity Index (PCDAI) score, white blood cell count, hematocrit, platelet count, serum albumin, erythrocyte sedimentation rate, C-reactive protein (CRP) and fecal calprotectin (FC) levels. Serum samples for IFX TL and ATIs had been obtained during IFX maintenance therapy before the scheduled infusion. Samples had been obtained not serially but just once for all patients immediately after study enrolment, while for those with positive free or total ATIs at initial tests additional samples were obtained after 1 year.
Dose intensification of IFX was allowed in cases of suspected secondary LOR, which was defined as worsening of clinical symptoms (PCDAI ⩾ 10) and a significant increase in serum CRP (⩾0.3 mg/dL) or FC levels (⩾250 mg/kg) at two consecutive visits plus a status that required dose intensification or switching the therapy. In patients positive for free or total ATIs, the clinical course and outcomes were further observed over the following 12 months.
Laboratory analyses
IFX TLs and free and total ATIs were analyzed using the IDKmonitor® Infliximab drug-level enzyme-linked immuosorbent assay (ELISA), the drug-sensitive IDK monitor® infliximab-free ADAs, and the drug-tolerant IDK monitor® infliximab-total ADAs (all from Immunodiagnostik AG, Bensheim, Germany), respectively. The quantifications using ELISA kits were performed following the manufacturers’ instructions,17,18
Free ATIs assay measures only ATIs that are freely circulating in serum and does not detect ATIs bound to IFX. However, total ATIs assay measures both free and IFX-bound ATIs. To measure IFX-bound ATIs, a pre-treatment acid dissociation step is used to separate ATIs from IFX. The assay then follows a standard ELISA method using recombinant therapeutic antibody as a capture and detection ATIs. For both assays, the manufacturer established a positivity threshold by linear dilution of sera with high concentrations of IFX until no further linear dilution was possible. The manufacturer’s recommended positivity threshold is 10 AU/ml.
Statistical analysis
For statistical comparisons between groups, Student’s t-test or Wilcoxon’s rank-sum test was used for continuous variables and chi-squared or Fisher’s exact test was used for categorical variables. Comparative data for continuous variables are reported as medians and interquartile ranges (IQRs) or means and standard deviations. Univariate and multivariate logistic regression analyses were performed to investigate factors associated with poor response to dose intensification. Univariate logistic regression analysis was performed to investigate the crude odds ratio (OR) for each factor; factors with p value <0.1 in the univariate analysis were included in the multivariate analysis. The results are expressed as adjusted OR and 95% confidence interval (CI). Receiver operating characteristic curve analysis was performed to determine the optima cutoff of factors that could best stratify patients with and without response to IFX dose intensification. Statistical significance was defined as p value ⩽0.05. All statistical analyses were performed by Rex (Version 3.6.0, RexSoft Inc., Seoul, Korea).
The reporting of this study conforms to the STROBE statement (Supplemental Appendix 1). 19
Results
Baseline characteristics
A total of 118 patients were receiving maintenance treatment with IFX during the study period. Among them 98 patients met the inclusion criteria and were eligible for the analysis. The median age at diagnosis was 13.9 ± 2.8 years, and 80.6% (79/98) of the patients were male. Non-stricturing nonpenetrating (B1) disease was observed in 85.7% (84/98) of patients, and 68.4% (67/98) of patients had perianal disease modifiers. Among them, 90 patients (91.8%) received concomitant azathioprine. Detailed baseline characteristics are summarized in Table 1.
Baseline characteristics.
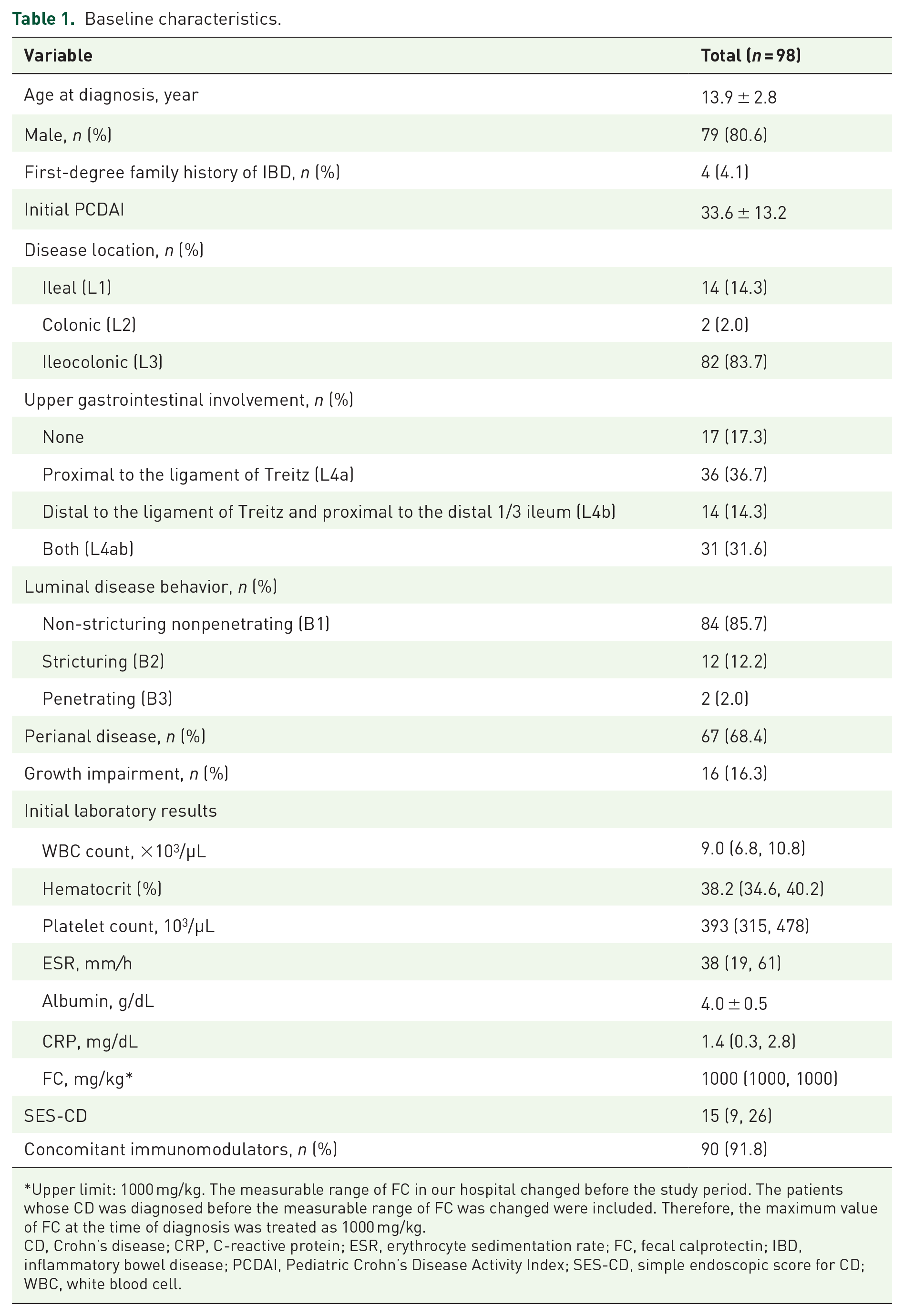
Upper limit: 1000 mg/kg. The measurable range of FC in our hospital changed before the study period. The patients whose CD was diagnosed before the measurable range of FC was changed were included. Therefore, the maximum value of FC at the time of diagnosis was treated as 1000 mg/kg.
CD, Crohn’s disease; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; FC, fecal calprotectin; IBD, inflammatory bowel disease; PCDAI, Pediatric Crohn’s Disease Activity Index; SES-CD, simple endoscopic score for CD; WBC, white blood cell.
Comparison of factors according to free ATIs positivity
Nine of 98 patients (9.2%) had detectable free ATIs and 38 of 98 had total ATIs (38.8%). Comparison of factors between patients according to free ATIs positivity revealed that clinical and laboratory outcomes were significantly worse in patients with free ATIs than those without free ATIs (Table 2): PCDAI (27.5 versus 0, p < 0.001), CRP (0.33 mg/dL versus 0.04 mg/dL, p = 0.0124), and FC (991.0 mg/kg versus 84.6 mg/kg, p = 0.0102).
Comparison factors between patients stratified according to the presence of free ATIs.
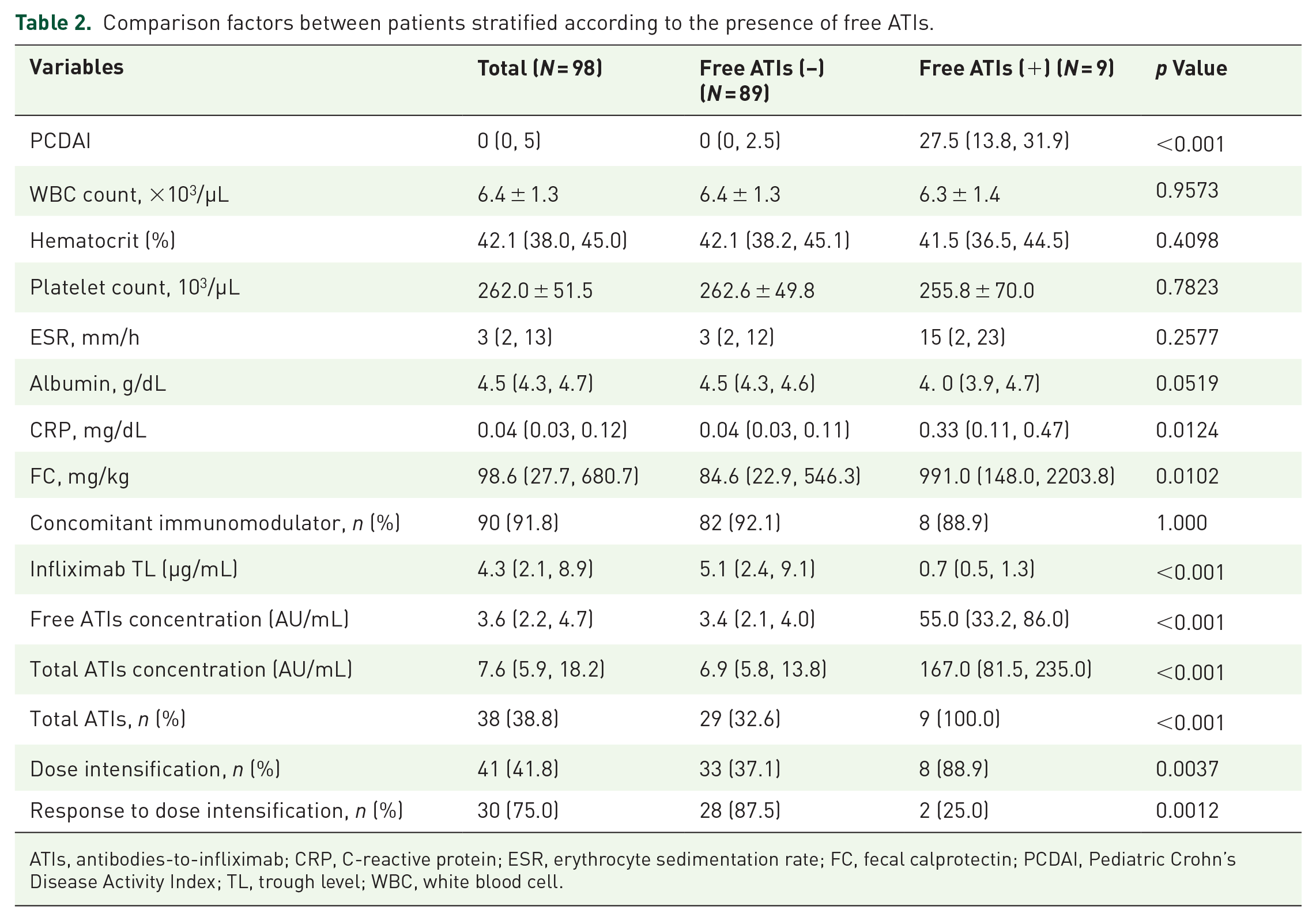
ATIs, antibodies-to-infliximab; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; FC, fecal calprotectin; PCDAI, Pediatric Crohn’s Disease Activity Index; TL, trough level; WBC, white blood cell.
Median IFX TLs were significantly lower in patients with free ATIs compared with those without free ATIs (0.7 μg/mL versus 5.1 μg/mL, p < 0.001), but there was no statistically significant difference in IFX TLs with or without total ATIs (4.8 μg/mL versus 4.3 μg/mL, p = 0.2523) (Figure 1). In addition, patients with free ATIs had higher concentration of total ATIs than those without free ATIs (167.0 AU/mL versus 6.9 AU/mL, p < 0.001). All patients with free ATIs showed positive for total ATIs; however, 32.6% (29/89) of patients without free ATIs showed positive for total ATIs. During follow-up period, proportion of patients who received dose intensification was significantly higher in patients with free ATIs than those without free ATIs (88.9% versus 37.1%, p = 0.0037). In contrast, proportion of patients who showed response to dose intensification was significantly lower in patients with free ATIs than those without free ATIs (25.0% versus 87.5%, p = 0.0012).
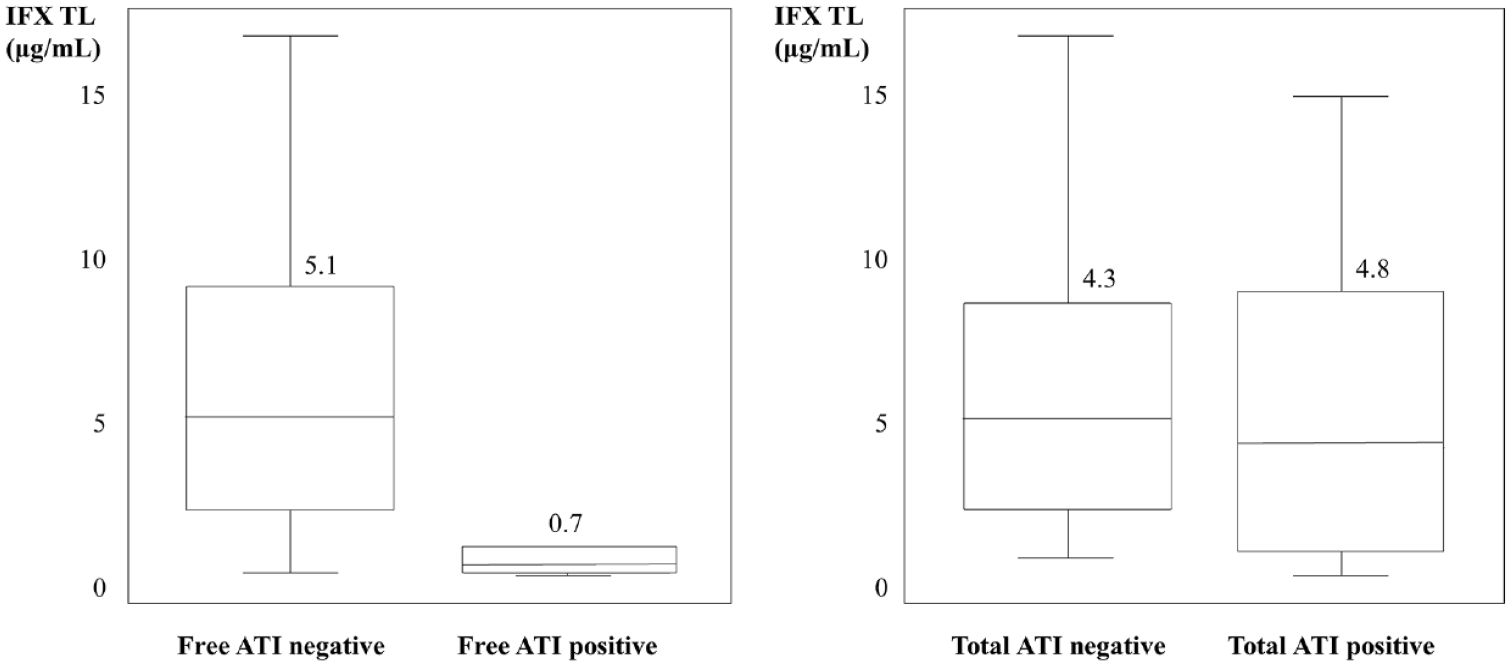
Infliximab TLs according to development of free or total ATIs.
Sub-analysis of patients with total ATIs according to free ATIs positivity
Among 98 patients, 38 patients developed total ATIs (38.8%). Of the patients with total ATIs, 29 were discordant with free ATIs (total ATIs positive but free ATIs negative). When analyzing these 38 patients according to the presence of free ATIs, patients with positive free ATIs had lower IFX TLs than those with negative free ATIs (Table 3). During follow-up period, there was no significant difference in the number of patients required dose intensification (88.9% versus 69.0%, p = 0.3985); however, response to dose intensification was statistically significant lower in patients with free ATIs than those without free ATIs (22.2% versus 65.5%, p < 0.001).
Sub-analysis of patients according to discrepancy of free and total ATIs.

Discrepancy (+): total ATIs positive but, free ATIs negative.
Discrepancy (−): total and free ATIs positive.
ATI, antibodies-to-infliximab; IFX, infliximab; TL, trough level.
Factors associated with poor response to IFX dose intensification
According to multivariate logistic regression analysis, free ATIs positivity was the only significant factor associated with poor response to dose intensification (Table 4, OR: 14.15, 95% CI: 1.31–151.97, p = 0.0140). Meanwhile, total ATIs positivity was not associated with response to dose intensification within 1 year after positive total ATIs had been confirmed.
Logistic regression analyses of factors associated with poor response to dose intensification in patients with Crohn’s disease.

ATI, antibodies-to-infliximab; IFX, infliximab; TL, trough level.
A further analysis was carried out to reveal the optimal cutoff of factors that could best stratify patients according to non-response to IFX dose intensification. According to receiver operating characteristic curve analysis, the optimal cutoff level indicating non-response to IFX dose intensification was 30.0 AU/mL for free ATIs concentration (area under curve, 0.792; 95% CI: 0.590–0.942; sensitivity, 60.0%; specificity, 96.7%; positive predictive value, 85.7%; negative predictive value 87.9%; p = 0.0241) (Figure 2).

ROC curve of free ATIs concentration stratifying patients according to response to dose intensification of IFX.
Clinical course and outcomes in patients with free and total ATIs
All patients who had developed free ATIs showed secondary LOR (n = 9). Immunomodulator was added in one patient and IFX dose intensification was performed in eight of these nine patients. In these eight patients, secondary LOR occurred in six patients within a year, and then IFX was switched to adalimumab, while clinical remission was achieved in the other after dose intensification. Initial free ATI concentration of these two patients who responded to IFX dose intensification were 16.8 and 22.3, respectively.
Among 38 patients who had developed total ATIs, 29 patients were negative for free ATIs. Clinical remission was achieved in nine patients, while 20 patients experienced secondary LOR, and then IFX dose intensification was performed. In these 20 patients, secondary LOR occurred in one patient within a year, and so the patient was switched to adalimumab, while clinical remission was achieved in the other 19 patients after dose intensification.
Discussion
In the era of personalized treatment, interest in factors related to treatment response and prognosis is increasing along with the design of personalized treatment strategies through risk stratification. In this prospective study, we demonstrated the utility of free ATIs as a reliable biomarker that can predict the effect of dose intensification in pediatric CD patients with secondary LOR to IFX. In addition, we provided the evidence that free ATIs, not total ATIs have a negative effect on the course of pediatric CD. To the best of our knowledge, this is the first pediatric study to demonstrate the clinical impacts of free ATIs on response to IFX dose intensification in CD patients.
TNF-α plays an important role in the development of immune-mediated inflammatory diseases, including IBD as well as rheumatic diseases such as ankylosing spondylitis, rheumatoid arthritis, juvenile idiopathic arthritis, even systemic sclerosis.20,21 Inhibition of TNF-α downregulates progressive and abnormal inflammatory process, resulting in sustained clinical remission, improved quality of life, and prevention of target organ damage.22,23 There are currently five anti-TNFα agents approved by the Food and Drug Administration and the European Medicines Agency: IFX, adalimumab, etanercept, golimumab, and certolizumab pegol. IFX is a chimeric IgG1 antibody, adalimumab and golimumab are fully human IgG1 antibodies, certolizumab pegol is a PEGylated Fab fragment of a humanized anti-TNF antibody, and etanercept is a fusion protein between a human IgG1 Fc-tail and the TNF-receptor type 2. Etanercept is known to be the least immunogenic anti-TNFα agent, 24 with a report of ADAs to etanercept detected in only about 6% of patients. 25 In other words, many patients treated with anti-TNFα agents other than etanercept develop ADAs.
Immunogenicity to anti-TNFα agents occurs when the immune system of the host recognizes them as antigens and develops specific ADAs against them. 26 In adult IBD patients, the formation of free ATIs has been reported in 63−65%,27,28 while in pediatric patients, it was reported to be 8−43%. 29 The results of our study are consistent with these results, as free ATIs were detected in 9.2% and total ATI in 38.8%. In addition, since the proportion of patients treated with concomitant immunomodulators in this study is higher than in previous studies, free ATIs formation in this study might be relatively low.
The drug interference complicates accurate quantification of ADAs, comprehension of the assay format used for ADAs detection is essential for correct interpretation of ADAs results. Drug-sensitive assays measure free ADAs with very low drug tolerance, thereby underestimating the levels of immunogenicity. Most drug-tolerant assays use dissociation buffer, that is, an acid dissociation step, to separate therapeutic drug-ADAs complex.30–32 In contrast to drug-sensitive assays, drug-tolerant assays can also measure ADAs bound to drug through this step. Considering the effect of ADAs on drug levels, ADAs can be functionally classified into two types: neutralizing ADAs and non-neutralizing ADAs. Neutralizing ADAs directly bind to the pharmacologically active site and physically prevents the drug from binding to its target, thereby lowering the drug TLs. On the other hand, non-neutralizing ADAs form immune complex with the drug to sequester the therapeutic agent and indirectly decrease drug levels by increasing drug clearance rate due to immune complex recognition by FcR. 33 Therefore, since ADAs–drug complexes can be cleared from the circulation more rapidly, even a complete drug-tolerant assays will underestimate the actual amount of ADAs formation.
Several studies have reported that ATIs cause a decrease in IFX TLs, which results in LOR during treatment.34–36 In our study, same result was also observed that IFX TLs were significantly lower (0.7 µg/ml versus 5.1 µg/mL, p < 0.001) and the proportion of patients with dose intensification was higher (88.9% versus 37.1%, p = 0.0037) in patients with free ATIs compared to those without free ATIs. Furthermore, PCDAI score, CRP and FC were significantly higher in patients with free ATIs, indicating the negative impact of the presence of free ATIs on clinical and biochemical remission as well as endoscopic remission. Unexpected immunogenicity is a challenge in the management of patients treated with biological agents. To be able to provide precision medicine, the results of ADAs on treatment efficacy and adverse events should be identified.
There is growing interest in proactive TDM, which optimizes treatment based on drug level and ADA, regardless of symptoms during maintenance therapy. Recent randomized controlled trial-the Trough Concentration Adapted Infliximab Treatment trial revealed that there were no differences in clinical or biochemical remission at 1 year between proactive (69%) and reactive TDM-based groups (66%). 37 Nevertheless, proactive TDM-based group showed fewer relapses during follow-up compared to reactive group (7% versus 17%, p = 0.018). Using the drug-tolerant assays, it was found that the majority of patients treated with IFX develops immune responses toward this biological agent.38,39 In our study, 38.8% of patients were found to develop total ATIs, which is larger proportion than those with free ATIs. Recently, it has been reported that drug-tolerant assays can evaluate the presence of ATIs more accurately and earlier than drug-sensitive assays, which has led to the prevailing opinion that total ATIs is more suitable for proactive TDM than free ATIs.40,41 However, drug-tolerant assay, that is, total ADAs are not necessarily more useful in predicting poor clinical outcome. Drug-tolerant assays also detect ATIs that would not have caused a clinical relevant decrease in IFX TLs, while drug-sensitive assays will typically only detect ATIs when IFX TLs are below the clinically effective threshold. Our results are line with published data that IFX TLs are not significantly different according to the presence of total ATIs (IFX TLs in patients with total ATIs 4.84 versus IFX TLs in those without total ATIs 4.25, p = 0.2523). Therefore, the strongest association between ATIs and clinical implication has been established mainly using drug-sensitive assays.8,12
Current recommendations for medical management of pediatric CD advise early screening of TLs of anti-TNFα and then only checking for ADAs in samples with lower TLs than tolerated by the drug-sensitive (free) ADAs assays.42,43 Therapeutic strategies should be changed based on the results of TDM and causes of secondary LOR including pharmacokinetic, pharmacodynamic, and immunogenic LOR. According to the guideline, dose intensification is recommended for pharmacokinetic (low TLs without ADAs) or immunogenic LOR with low ADAs titer; however, switch in-class of anti-TNFα is recommended for patients experiencing pharmacodynamic (adequate TLs) or immunogenic LOR with high ADAs titer. 13 In adult patients with CD, the choice of biological agents is relatively wider than pediatric patients, so it seems reasonable to switch biological agents in the case of pharmacodynamic or immunogenic LOR with high ADAs titer. However, compared to adult patients, pediatric CD patients have narrower biologic options and longer duration of disease, requiring careful switching of biological agents in real-world setting. Due to this limitation, the addition of immunomodulators, dose intensification, or both are often considered and tried fist in pediatric patients suffering from secondary LOR, regardless of type of LOR, in clinical practice. Therefore, prognostic factors associated with response to dose intensification are elucidated in pediatric patients with IBD.
Several studies have published the relationship between free ATIs and LOR, but as far as we know, there is no research on the relevance of free ATIs and response to IFX dose intensification in pediatric patients with secondary LOR.9,44,45 Our study demonstrated that free ATIs have a negative impact on response to dose intensification as well as poor clinical outcome in pediatric CD patients treated with IFX. In addition, of those patients undergoing empiric IFX dose intensification at time of secondary LOR, 75.0% (30/41) of patients recaptured response. However, patients with free ATIs showed significantly poor response to IFX dose intensification compared to those without free ATIs (25.0% versus 87.5%, p = 0.0012). We suggested cutoff value of free ATIs associated with non-response to IFX dose intensification as 30.0 AU/mL (area under curve, 0.792; 95% CI: 0.590–0.942; sensitivity, 60.0%; specificity, 96.7; p = 0.0241). Therefore, in patients with free ATIs whose concentration is 30 AU/mL or higher, there is a high possibility that remission may not be achieved even if the IFX dose is increased, therefore measurement of IFX TLs and ATIs titers through proactive TDM should be considered.
Our study has some limitations. First, we measured ATIs through ELISA methods which are prone to give false-positive results because of nonspecific binding of cross-reacting antibodies. However, published comparisons for ATIs measurement using various platforms such as ELISA, radioimmunoassay, homogeneous mobility shift assay, and functional cell-based reporter gene assay have reported comparable correlations despite method differences (Pearson’s r = 0.77−0.96, p < 0.0001). 46 Second, we lacked serial laboratory results for ATIs and IFX TLs starting from IFX. It is known that 90% of patients with a sustained ATIs response against IFX do so within the first 12 months of treatment, while transient ATIs are detected throughout the period of IFX treatment. 47 In our study, since the blood samples for measurement of ATIs were taken at a time period of 2.5 ± 1.2 years from IFX initiation, the proportion of patients who had actually developed ATIs may not be accurate. Third, we did not analyze risk factors for immunogenicity with pharmacogenomics such as human leucocyte antigen (HLA) polymorphism. However, formal pharmacokinetic or prospective interventional studies based on HLA polymorphism have not been completed, and it is unclear how this approach fits into pediatric patients or TDM guidelines.
In conclusion, free ATIs, not total ATIs, have a negative effect on the course of CD and can be used as a reliable biomarker that can predict the effect of dose intensification in patients with LOR to IFX. In addition, proactive TDM should be considered in patients with free ATIs whose concentration is 30 AU/mL or higher.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231170948 – Supplemental material for Free antibodies-to-infliximab are biomarker for predicting the effect of dose intensification in pediatric Crohn’s disease patients with secondary loss of response
Supplemental material, sj-docx-1-tag-10.1177_17562848231170948 for Free antibodies-to-infliximab are biomarker for predicting the effect of dose intensification in pediatric Crohn’s disease patients with secondary loss of response by Eun Sil Kim, Yiyoung Kwon, Yon Ho Choe and Mi Jin Kim in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848231170948 – Supplemental material for Free antibodies-to-infliximab are biomarker for predicting the effect of dose intensification in pediatric Crohn’s disease patients with secondary loss of response
Supplemental material, sj-docx-2-tag-10.1177_17562848231170948 for Free antibodies-to-infliximab are biomarker for predicting the effect of dose intensification in pediatric Crohn’s disease patients with secondary loss of response by Eun Sil Kim, Yiyoung Kwon, Yon Ho Choe and Mi Jin Kim in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
