Abstract
Background:
Current literature still lacks studies evaluating the effectiveness and safety of switching from Infliximab originator to SB2 biosimilar in Inflammatory Bowel Diseases (IBDs). We aimed to verify the ability of SB2 to maintain the clinical and biochemical response induced by originator after switching. As secondary outcome, we aimed to verify safety, tolerability and immunogenicity of SB2 biosimilar compared with its IFX originator.
Methods:
We prospectively enrolled all patients who switched from originator to SB2 at three Italian IBD Units from August 2018 to April 2020. We collected clinical and biochemical data at the time of switch (T0), and at the first (T1) and the second (T2) visits after switching (mean time from switching: 135 and 329 days, respectively). In addition, data regarding therapeutic drug monitoring at T0 and T1 were recorded.
Results:
Eighty-five IBD patients (28 with Ulcerative Colitis and 57 with Crohn’s Disease) were included in the study. At T1, we observed statistically significant modifications in clinical activity of disease (70 patients were in clinical remission at baseline and 60 at T1 p = 0.02), but not at T2 (p = 0.3). Fecal calprotectin values were not different both at T1 and T2 (both p = 0.9) as well as the rate of concomitant treatment with steroids (p = 0.2 and p = 0.1) or immunosuppressants (p = 0.1 and p = 1.0). Moreover, the need for therapeutic optimization from T0 to T1 and from T1 to T2 was found significant (both p = 0.01). No anti-drug antibodies were identified at T1, and no serious adverse events were recorded.
Conclusions:
Overall, our data show that most of the patients switching from Infliximab originator to SB2 maintain the clinical and biochemical remission for at least 1 year. Further data are necessary to understand the clinical implications of these findings in the long term.
Introduction
Inflammatory Bowel Diseases (IBDs), including Ulcerative Colitis (UC) and Crohn’s Disease (CD), are chronic disorders of the intestinal tract characterized by relapsing and remitting intestinal inflammation and are associated with a reduced quality of life.1–6 The introduction of biological drugs has radically changed the therapeutic approach and management of IBD patients. 1
In particular, anti-tumor necrosis factor alpha (TNF-α) therapies, such as Infliximab (IFX), have greatly improved treatment expectations in patients with IBD refractory or intolerant to standard treatments,7,8 allowing to achieve and maintain clinical and biochemical remission as well as mucosal healing.9–12 However, one-third of patients are primary non-responders and approximately 10–50% of patients have a loss of response (secondary non-responders) to the drug during treatment.13,14 There are several possible causes of lack or loss of response to anti-TNF alpha therapy,13,15 including the presence of low levels of serum drug concentrations and high titers of anti-drug antibodies. 15 Thus, immunogenicity could play a relevant role in determining favorable outcome in IBD patients. 16
Infliximab was first authorized in the European Union (EU) in August 1999 under the name of Remicade®. 17 After the expiration of its patent, several biosimilars have been developed and authorized with substantial cost reductions for IBD-related healthcare.18–20 In June 2013, the European Medicines Agency’s Committee for Medicinal Products approved the use Inflectra® and Remsima® as biosimilar medicinal products containing IFX CT-P13. 21 The second biosimilar to IFX gaining marketing authorization in Europe (as Flixabi®) in April 2016 was SB2. 22
While on CT-P13 there is substantial and robust literature regarding its effectiveness and safety in patients with IBD, 23 on SB2 biosimilar there is a paucity of data in the IBD setting, as their approval for indications other than rheumatologic disease is based on the principle of extrapolation.24–28 That is one of the reasons why, as with other anti-TNF biosimilars already on the market, real-life data and pharmacovigilance studies are needed to develop long-term evidence on effectiveness and safety of these drugs in the IBD population.
Therefore, in the present multicenter study, our primary aim was to prospectively verify the ability of SB2 to maintain the clinical and biochemical response induced by IFX originator after switching. As secondary outcome, we aimed to verify safety, tolerability and immunogenicity of SB2 biosimilar compared with its IFX originator.
Methods
Study design
This is a multicenter prospective study, coordinated by the IBD Unit of Padua University (Veneto, Italy), with the involvement of two other Italian IBD centers (Pisa, Tuscany and Santorso, Veneto, Italy). Consecutive IBD patients in therapy with IFX originator at the time of biosimilar drug SB2 approval were prospectively enrolled and switched to SB2, independently from disease status and clinical conditions, from August 2018 to April 2020. Of note, after regional approval of SB2 use, all IBD centers in Tuscany and Veneto were invited to switch all patients taking IFX originator to SB2, and the long period of inclusion was determined by the selection of patients who were at their “first switch” from originator to SB2 biosimilar. The study was approved by our Ethics Committee as part of a larger study that aimed to evaluate disease course and characteristics of IBD patients from the introduction of biologics in clinical practice (N. 3312/AO/14). Written informed consent was obtained from all eligible participants or their legal representatives before participation. The study protocol was performed accordingly to the ethical guidelines of the 1964 Declaration of Helsinki (6th revision, 2008) as reflected in
Data collection
For the purpose of our study, demographic, clinical and biochemical data were collected at the time of the switch, and therefore before SB2 biosimilar administration (T0), at the first follow-up visit after switching (T1), and finally at the second follow-up visit (T2). To note, five patients were included despite not reaching this at the time of publication. The following baseline data were collected for each patient: age, sex, date of start of the IFX originator, dose of the last IFX originator administration, fecal calprotectin (FC) value, use of steroids, use of immunosuppressants (azathioprine, 6-mercaptopurine, methotrexate) and adverse events. Clinical activity was measured by using partial Mayo (p-Mayo) Score and Harvey–Bradshaw Index (HBI) for UC and CD, respectively.17,29–31 According to the medical literature, clinical activity was classified into remission, mild, moderate and severe according to the following values of p-Mayo for UC: 0–1 remission, 2–4 mild disease, 5–6 moderate disease, 7–9 severe. The following values of HBI were used for CD patients: <5 remission, 5–7 mild disease, 5–15 moderate, >16 severe. C-Reactive Protein (CRP) levels (increased if >6 mg/l) and FC values >250 µg/g were also evaluated at the same timepoints. 32 Optimization rate for both the IFX originator and SB2 was recorded at T0, T1 and T2. The allowed methods of therapeutic optimization were: 10 mg/kg every 8 weeks, 5 mg/kg every 4 weeks, 5 mg/kg every 6 weeks, and 5 mg/kg every 10 weeks. The need for therapeutic optimization was evaluated at T1 based on clinical scores (p-Mayo and HBI) and biochemical activity (FC values).
Finally, treatment failure was defined as discontinuation of biological therapy due to adverse events (AEs), lack of clinical response and need for hospitalization/surgery. All AEs, not only those that led to discontinuation of therapy, were recorded.
Pharmacokinetic sub-study
At both T0 and T1 we collected serum samples of a subgroup of patients (n = 55) from Padova (Veneto, Italy) and Pisa (Tuscany, Italy) Hospital in order to evaluate serum trough levels (TLs) and anti-drug antibodies (ADAs). After obtaining informed consent, venous blood samples were collected in covered test tubes (Vacutainer SST II Advance, Roborough, Plymouth, UK). The tubes containing whole blood were left undisturbed at room temperature for 15–30 min and then centrifugated at 3000×
Statistical analysis
The data were analyzed using the STATA11 software (Stata Corp., College Station, TX, USA). Continuous variables were reported as medians with range of values, categorical variables were reported as frequencies and percentages. To determine if there was a statistically significant difference in proportion between paired data we used McNemar’s Test, while the comparison between ordinal or continuous values over the study period (T0 vs. T1 and T0 vs. T2) was performed using the Wilcoxon signed-rank test. A Kaplan–Maier curve was calculated to evaluate the drug survival, considering the switching date as the study start date, whereas we considered the latest date as the minimum dates between the drug interruption at T1 or T2 and the date of exit, arbitrarily defined 8 months after the end of the study. The statistical significance was set for values of p ⩽ 0.05.
Results
Study population and disease characteristics at baseline
A total of 85 patients were enrolled, 28 (32.9%) with UC and 57 (67.1%) with CD on IFX originator therapy at the time of enrollment. The main characteristics of the population are reported in Table 1.
Study population characteristics at baseline.

CD, Crohn’s disease; UC, ulcerative colitis;
Based on clinical scores, HBI and p-Mayo for CD and UC respectively, at baseline: 70 (82.3%) patients were in remission, 12 (14.1%) had mild disease activity, 3 (3.5%) moderate and none of the patients had severe disease (Figure 1). The study population had a median FC value of 68.5 µg/g (range 22–199 µg/g). Regarding the anti-TNF dosage at T0: 43 patients (50.6%) were on 5 mg/kg every 8 weeks regimen; 13 (15.3%) on 10 mg/kg every 8 weeks or 5 mg/kg every 4 weeks regimen; 16 (18.8%) on 5 mg/kg every 6 weeks regimen; 13 (15.3%) on 5 mg/kg every 10 weeks regimen (Figure 2). Two patients (2.3%) were on steroid therapy and 14 (16.5%) were taking immunosuppressants at baseline.

Disease activity at baseline (T0), at the first follow-up visit (T1) and at the second follow-up visit after switching (T2) in patients with UC (a) and CD (b).
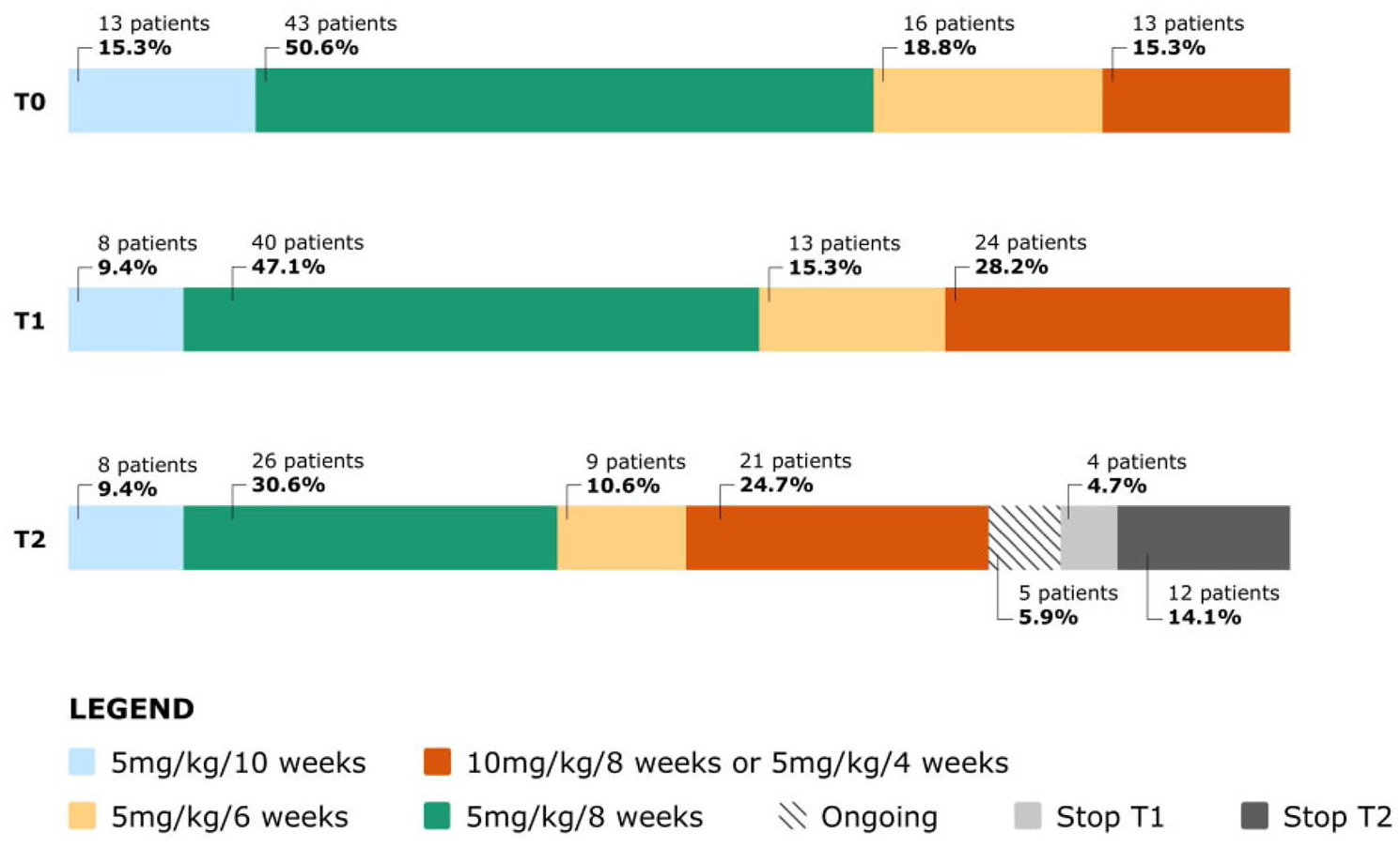
Anti-TNFα dosage at baseline, before switching (T0), at the first follow-up visit (T1) and at the second follow-up visit after switching (T2).
Clinical and biochemical data at the first follow-up visit after switching to SB2 biosimilar
At T1 (with a mean time from switching of 135 days, ranged from 56 to 2011 days), we reassessed the disease clinical activity: 60 patients (70.6%) were in remission, 17 (20%) had mild disease, eight (9.4%) presented moderate disease, and none had severe disease (p = 0.02 clinical activity T0 vs. T1) (Figure 1). The study population had a median FC value of 79 µg/g (range 19.5–194.5 µg/g) at T1. The comparison between T0 and T1 FC values was not statistically significant (p = 0.9). At T1, 12 out of 85 patients had an increased CRP compared with 11 out of 85 at T0, and this comparison was not statistically significant (p = 0.8; median value with range at T0: 11.9 mg/l, 2.4–45 mg/l; median value with range at T1: 10.1 mg/l, 1.4–54 mg/l). From T0 to T1, a not statistically significant (p = 0.2) increase of patients who needed to add steroid therapy (n = 5 at T1) to biological therapy was recorded. A not statistically significant (p = 0.1) increase in the number of patients who needed to add immunosuppressive therapy with azathioprine (n = 16 at T1) was detected as well.
Regarding the need for drug optimization, the number of patients undergoing treatment optimization (10 mg/kg every 8 weeks, 5 mg/kg every 4 weeks and 5 mg/kg every 6 weeks) increased from 29 patients at T0 to 37 patients a T1 in a statistically significant manner (p = 0.01) (Figure 2). Of the 29 patients at T0 who required therapeutic drug optimization, one patient returned to the standard drug dosage, while another nine patients required new dosage optimization. Of these newly optimized patients four were in clinical remission, two had mild disease and three presented moderate disease. At switch time these three patients had out of average calprotectin values as well.
At T1, 4 (4.7%) patients discontinued therapy: two for lack of efficacy, two for disease remission. One of the patients who interrupted therapy at T1 for lack of efficacy resumed therapy with IFX originator, obtaining complete biochemical and clinical remission.
Clinical and biochemical data at the second follow-up visit after switching to SB2 biosimilar
Seventy-six patients, from the 85 initially enrolled, had a second follow-up visit after switching, as four patients had discontinued SB2 therapy at T1 and five have not yet booked a second visit (ongoing).
At T2 (with a mean time from switching of 329 days, ranged from 188 to 411 days), we newly reassessed the disease clinical activity of these 76 patients: 59 patients (69.4%) were in remission, 15 patients (17.6%) had mild disease, and two (2.4%) presented severe disease (T0 vs. T2: p = 0.3) (Figure 1). In particular, looking at T0 vs. T2 disease clinical activity, out of the 70 patients in remission at baseline at T0, 52 maintained remission, 11 developed mild disease, none had moderate or severe disease activity. Of the 12 patients with mild disease at baseline, five achieved remission, three did not improve maintaining a mild disease status, and two developed severe disease. Of the three patients with moderate disease at baseline, two achieved remission and one passed from moderate to mild disease.
Regarding the need for drug optimization (10 mg/kg every 8 weeks, 5 mg/kg every 4 weeks and 5 mg/kg every 6 weeks) from T0 to T2, of the 21 patients at T1 who required drug therapeutic optimization, two patients returned to the standard dosage, while another 11 patients required new drug optimization. From T0 to T2 the need for optimization increased another time in a statistically significant manner (p = 0.01 T0 vs. T2).
Median FC was 57 µg/g (range 15–234 µg/g), without statistically significant difference from baseline to T2 (p = 0.9). Again, the comparison between CRP values at T0 and T2 (10/76 patients at T2 had an increased CRP) was not statistically significant (p = 0.7; median value with range at T0: 11.9 mg/l, 2.4–45 mg/l; median value with range at T2: 9.2 mg/l, 3.2–56 mg/l). Five patients at T2 were on steroid and 14 on immunosuppressive therapy, and even in this case without statistically significant difference (p = 0.1 for steroids and p = 1.0 for immunosuppressants, T0 vs. T2) (Table 2).
Concomitant drugs and reason for interruption at baseline (T0), at the first follow-up visit (T1) and at the second follow-up visit (T2) after switching.

T0, at baseline, which is the time of the switching; T1, at the first follow-up visit after switching (mean time after switching: 135 days); T2, at the second follow-up visit after switching (mean time after switching: 329 days).
Treatment was discontinued in 12 (14.1%) patients: six for lack of efficacy, five for adverse reactions, and one for pregnancy.
Tolerability profile of drugs
The whole number of AEs has been recorded at the different study time points and reported in Supplementary Table 1. Overall, four and five AEs were recorded at T1 and T2, respectively. All the AEs recorded at T2 led to drug discontinuation (Table 2 and Supplementary Table 1).
Trough levels and anti-drug antibodies sub-analysis
Of the total study population (n = 85), 55 patients accepted to be included in the pharmacokinetic sub-study. At baseline, 20 patients showed subtherapeutic TLs (36.3%) with a median value of 4.6 μg/ml (0.06–19.5 μg/ml range). Only two patients had detectable levels of ADA (3.7%). We collected the same identical data at T1: 20 patients showed subtherapeutic levels with a median value of 4.1 (0.1–15.7), and the same two patients of T0 showed detectable levels of ADAs (3.7%). Of the 20 patients with subtherapeutic TLs at T0, five patients returned to therapeutic TLs at T1 and five instead lost their therapeutic TLs (Table 3). The drug survival curve for treatment persistency is provided in Figure 3.
Trough levels (TLs) and anti-drug antibodies (ADAs) at baseline (T0) and at the first follow-up visit (T1) after switching.

T0, at baseline, which is the time of the switching; T1, at the first follow-up visit after switching (mean time after switching: 135 days).

Biosimilar drug survival curve during follow-up period after switching from IFX originator.
Discussion
The recent development and authorization of various IFX biosimilars for the treatment of IBD largely changed our daily clinical practice, opening new possibilities, even though with some concerns about their use. Indeed, currently data to properly support their use in the IBD setting is still lacking. Thus, publication of studies reporting effectiveness and safety of these drugs has been advocated. The aim of this study was to verify the ability of SB2 biosimilar to maintain the clinical and biochemical response induced by IFX originator in consecutive IBD patients who underwent switching to biosimilar. In addition, we aimed to evaluate the safety and tolerability profile of SB2 biosimilar compared with its IFX originator. We found that patients in clinical remission dropped from 70 out of 85 at the time of last originator infusion to 59 out of 76 patients at 1 year after switching to SB2. Moreover, two patients transitioned from mild to severe disease, eight patients (9.4%) required optimization, and 16 discontinued therapy (18.8%). Furthermore, addition of steroid therapy was necessary in 10 patients. Finally, we did not observe significant differences in terms of TLs or ADAs development after switching.
To the best of our knowledge, this study evaluates, in a real-life setting, the largest cohort of patients to from IFX originator to SB2 biosimilar to date, showing that SB2 has a similar efficacy, immunogenicity and safety profile to that of IFX originator. Indeed, to date, only few studies with even a modest population evaluated the switch from IFX originator to SB2. Macaluso
We found a statistically significant increase in need for therapeutic drug optimization both at T1 and at T2. Data on the need for therapeutic drug optimization are currently scarce in the scientific literature. However, a previous study from our research group comparing the effectiveness and safety of Infliximab originator, CT-P13 and Adalimumab between patients with CD and UC showed that, at T2, 31.1% of ADA, 16.7% of IFX originator, and 36.2% of CT-P13 biosimilar patients needed treatment optimization. 36 In addition, at T2, 17 out of 76 (22.3%) patients had a recurrence of the disease. Surely, we do not know whether this failure was related to the switch to SB2 biosimilar or might be secondary to the physiologic loss of response recorded with this type of drug and already reported in the medical literature (i.e. one-third of patients are primary non-responders and approximately 10–50% of patients experience loss of response, secondary non-responders, during treatment).13,14
It is known that cross-immunogenicity can develop between IFX originator and SB2, and it has been already demonstrated that anti-IFX originator antibodies in patients with IBD inhibit CT-P13 to a similar degree, confirming similar immunogenicity and shared immunodominant epitopes on these two IFX agents. 37 In keeping with this, it has been hypothesized that Remicade®-treated ADA-positive patients should not switch to the biosimilar, since antibodies can interact with biosimilar and loss of response may occur. 37 The BIOSIM-01 study is a retrospective cohort study conducted in Italy on a cohort of patients with IBD which analyzed cross-reactivity of anti-IFX originator antibodies with CT-P13 and SB2 molecules. 38 Similarly, the cross-reactivity of anti-CT-P13 antibodies with IFX originator and SB2 was evaluated. The study demonstrated a complete cross-reactivity: antibodies directed against IFX originator react identically against CT-P13 and SB2. 38 The study also investigated the possibility to use the IFX originator serum levels assays for the biosimilars SB2 and CT-P13, concluding that the accuracy for the three drugs was the same. These data are in favor of “biosimilarity” and endorse the use of these assays in monitoring of SB2 therapy for optimization of the administration rate. 38 As to our data, we evaluated at T0 and T1 both TLs and antibodies against IFX originator as well as SB2, observing no significant implication of these pharmacological features in therapeutic failure. Interestingly, one of the patients who interrupted therapy at T1 for lack of efficacy resumed therapy with IFX originator, obtaining complete biochemical and clinical remission. This has been also recently observed in a retrospective study by Mahmmod and coworkers.39 These authors showed that reverse switching from IFX biosimilar to originator was applied in 9.9% (75 out of 758) of patients, predominantly for biosimilar-attributed adverse effects. Switching back to originator IFX seemed effective in patients who experienced adverse effects, worsening gastrointestinal symptoms or loss of response after switching from originator IFX to CT-P13.
Our study has some limitations. First of all, the sample size, even though this study represents the largest population of IBD patients switched to SB2 biosimilar in a real-life setting (28 UC and 57 CD), is limited. Furthermore, the follow-up is not long enough to gather reliable data on the long-term response. The baseline population was remarkably heterogeneous, including biologic monotherapy and concomitant immunosuppressant and steroid therapy. The TLs and ADAs sub-analyses considered only 55 patients of the total population, and were performed from the two academic hospitals (Padua and Pisa), implicating a possible selection bias. However, we do not feel that this might impact our results as therapeutic drug monitoring (TDM) was performed in consecutive patients and without any clear indication for doing such analysis (i.e. unselected cases). Moreover, we did not collect TDM data at 1 year, because most of the data currently published on the role/value of TDM suggests its utility in the early phases of treatment (post-induction) rather than during long-term follow-up. Nevertheless, this study also has some strengths. This is the multicenter prospective real-life study including the largest population on switching from IFX originator to SB2 biosimilar in the medical literature. In addition, until this study, data to verify cross-immunogenicity between IFX originator and SB2 had not been published. Finally, data were collected from multiple operating units, making the study population representative of multiple geographical areas and limiting single-center clinical bias.
In conclusion, our data showed that switching from originator to biosimilar is safe in terms of both effectiveness and tolerability. Indeed, our patients maintained clinical response to therapy after switching and did not develop serious AEs. In addition, we did not find differences in immunogenicity between IFX originator and SB2. Surely, it would be desirable that more multicenter real-life studies with a larger sample, and a longer follow-up, should be conducted in order to obtain more consistent data and further support the use of biosimilar drugs in clinical practice.
Supplemental Material
sj-docx-1-tag-10.1177_17562848211023384 – Supplemental material for Switching from Infliximab Originator to SB2 Biosimilar in Inflammatory Bowel Diseases: A Multicentric Prospective Real-Life Study
Supplemental material, sj-docx-1-tag-10.1177_17562848211023384 for Switching from Infliximab Originator to SB2 Biosimilar in Inflammatory Bowel Diseases: A Multicentric Prospective Real-Life Study by Davide Massimi, Brigida Barberio, Lorenzo Bertani, Francesco Costa, Antonio Ferronato, Sonia Facchin, Romilda Cardin, Linda Cingolani, Cesare Casadei, Renata D’Incà, Fabiana Zingone and Edoardo Vincenzo Savarino in Therapeutic Advances in Gastroenterology
Footnotes
Author Contributions
DM, BB, FZ, EVS: study concept and design, analysis and interpretation of data, drafting of the manuscript, critical revision of the manuscript for important intellectual content. DM, BB, LB, FC, AF, LC, CC, AG, MG, GL: acquisition of data, and critical revision of the manuscript. RD: critical revision of the manuscript for important intellectual content. FZ: statistical analysis.
Conflict of interest statement
- Edoardo Vincenzo Savarino served as a speaker, consultant, and advisory board member for AbbVie, MSD, Takeda, Ferring.
- Renata D’Incà served as a speaker, consultant, and advisory board member for AbbVie, MSD, Takeda, Ferring.
- The other authors have no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor of the Article
EVS and FZ are guarantors.
Ethics Committee Approval
The study was approved by University of Padova’s Ethics Committee as part of a larger study aimed to evaluate disease course and characteristics of IBD patients from the introduction of biologics in clinical practice (N. 3312/AO/14).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
