Abstract
Background:
The Rutgeerts score (RS) is widely used to predict postoperative recurrence after ileocolonic resection for Crohn’s disease (CD) based on the severity of lesions at the neoterminal ileum and anastomosis (RS i0–i4). However, the value of anastomotic ulcers remains controversial.
Objectives:
Our aim was to establish a nomogram model incorporating ileal and anastomotic lesions separately to predict the long-term outcomes of CD after ileal or ileocolonic resection.
Design:
A total of 136 patients with CD were included in this retrospective cohort study.
Methods:
Consecutive CD patients who underwent ileal or ileocolonic resections with postoperative ileocolonoscopy evaluation within 1 year after the surgery were included. The primary endpoint was postoperative clinical relapse (CR). An endoscopic classification separating ileal and anastomotic lesions was applied (Ix for neoterminal ileum lesions; Ax for anastomotic lesions). A nomogram was constructed to predict CR. The performance of the model was evaluated by the receiver-operating characteristic (ROC) curve and decision curve analysis (DCA).
Results:
CR was observed in 47.1% (n = 64) of patients within a median follow-up of 26.9 (interquartile range, 11.4–55.2) months. The risk of CR was significantly higher in patients with an RS ⩾ i2 assessed by the first postoperative endoscopy compared with patients with an RS ⩽ i1 (p < 0.001). Moreover, the cumulative rate of CR was significantly higher in patients with ileal lesions (I1–4) compared with patients without (I0) (p < 0.001). Besides, patients with anastomotic lesions (A1–3) had significantly higher rates of CR than patients without (A0) (p = 0.002). A nomogram, incorporating scores of postoperative ileal or anastomotic lesions, sex, L2-subtype and perianal disease, was established. The DCA analysis indicated that the nomogram had a higher benefit for CR, especially at the timeframe of 24–60 months after index endoscopy, compared to the traditional RS score.
Conclusion:
A nomogram incorporating postoperative ileal and anastomotic lesions separately was developed to predict CR in CD patients, which may serve as a practical tool to identify high-risk patients who need timely postoperative intervention.
Introduction
Crohn’s disease (CD) is a chronic inflammatory disease that can affect any locations of the digestive tract and its pathophysiology is complicated. Within 10 years after the diagnosis, nearly 50% of patients need surgery treatment, which brings a heavy burden to the patients. 1 However, postoperative recurrence (POR) is still common due to the nature of CD. 2 Thus, the recognition of those patients with potential higher POR is crucial to improve patients’ quality of life.
The postoperative endoscopic recurrence (ER) score, commonly referred to as the Rutgeerts score (RS, i0–4), was designed to predict risk of clinical relapse (CR) in CD patients undergoing ileocolonic resection based on early endoscopic findings at the anastomosis and in the neoterminal ileum.2,3 ER can be defined as RS ⩾ i2. 3 The patients with i0 or i1 category tend to have a favorable disease, while those with i3 or i4 category have a higher risk of POR. 3 However, the patients with the i2 category, including aphthous lesions in the neoterminal ileum as well as ileocolonic anastomosis lesions, had a heterogeneous recurrence risk.4–7
Since anastomotic ulcers were suspected to be postsurgical sequelae, a modified Rutgeerts score (mRS) was proposed: i2a, lesions confined to the anastomosis ±<5 isolated aphthous ulcers in the ileum; i2b, more than five aphthous ulcers in the ileum with normal mucosa in between, ± anastomotic lesions.4,5,8 Till now, whether this scoring index can predict subsequent disease courses is still conflicting. Bayart et al. 5 found that the rate of postoperative CR is not different in i2a and i2b patients. It was supported by another larger cohort (n = 365) conducted in Europe. 7 However, Ollech et al. 6 showed that patients with i2b had an increased risk of endoscopic progression and recurrent surgery, whereas patients with isolated anastomotic ulcers (i2a) did not have a statistically significant increased risk of either outcome when compared with i0–1. Since the current modified RS category gained discrepant results, the REMIND (groupe de REcherche sur les Maladies INflammatoires Digestives) group recently conducted a prospective multicenter study and built an endoscopic score describing the anastomotic and ileal lesions separately. 9 However, the score system has not been validated by other group and the prediction efficacy is not well understood.
The present study aimed to establish a nomogram based on postoperative ileal and anastomotic lesions separately to predict the long-term outcomes of CD after ileal or ileocolonic resection.
Methods
Study population
This retrospective study was conducted in the First Affiliated Hospital, Sun Yat-Sen University (China) consecutively, including CD patients underwent ileal or ileocolonic resections between 2005 and 2021. Inclusion criteria included (a) consecutive adult CD patients; (b) the patients had undergone an ileal or ileocolonic resection with primary anastomosis; (c) the patients had postoperative ileocolonoscopy evaluation within 1 year after the surgery; (d) the patients had >6 months of documented clinical follow-up since the index endoscopic evaluation. Patients were excluded if they had (a) a stoma; (b) incomplete follow-up colonoscopies that did not reach the anastomosis. All patient details have been de-identified.
Definitions and outcome
The primary outcome was CR, which was confirmed by imaging, endoscopy, or therapeutic intensification (treatment optimization or drug switch), CD-related complication, CD-related hospitalization, and/or subsequent surgery. 9
Data collection
Patient demographic, clinical, laboratory, and endoscopic data were collected by a comprehensive review of their electronic medical records. The following baseline characteristics were collected: age at diagnosis, gender, disease duration, CD location, and behavior according to the Montreal classification, smoking status, previous surgery, and bowel anastomosis technique (end-to-end, side-to-side, or end-to-side). All follow-up colonoscopies that reached the anastomosis were recorded. A further endoscopic evaluation with a classification separating anastomotic and ileal lesions was also performed. 9 Specifically, the anastomotic lesions were scored according to the size of ulcerations and whether stenosis existed: normal or mild inflammation (A0), at least semi-circumferential anastomotic ulcerations (A2) or less (A1), stenosis with or without ulcerations (A3). 9 The ileal lesions were scored based on the RS: no lesions (i0); ⩽5 apthous lesions (i1); >5 apthous lesions with normal mucosa between the lesions (i2); diffuse apthous lesions with diffusely inflamed mucosa (i3); diffuse inflammation with larger ulcers, nodules and/or narrowing (i4) in the neoterminal ileum. 9 Exposure to treatments (5-aminosalicylic acid, steroids, immunosuppressive agents, and biologics) after each subsequent ileocolonoscopy was also recorded. The reporting of this study conforms to the STROBE statement (Supplemental Table 1). 10
Statistical analysis
The baseline demographic and clinical characteristics were expressed in medians and interquartile range (IQR) for continuous variables and frequency distributions for categorical variables. The cumulative probabilities of CR were estimated using the Kaplan–Meier survival method. Differences between curves were compared using the log-rank test. Univariate and multivariate bi-direction stepwise Cox regression were used to screen the variables associated with CR and to build prediction models, respectively. A nomogram was constructed to predict CR based on the above prediction model. The receiver-operating characteristic (ROC) analysis and decision curve analysis (DCA) were then performed to evaluate the performance of the nomogram model.
All statistical analyses were performed using SPSS 26.0 (IBM Inc., NY, USA) and R v4.1.0 and a p value < 0.05 was considered statistically significant.
Results
Baseline characteristics
One hundred thirty-six patients were included. The characteristics of the study population are detailed in Table 1. The median disease duration was 4.5 years (IQR, 1.7–8.3) at inclusion. Ninety-five (69.9%) patients were men and 18 (13.2%) patients were smokers. According to the Montreal classification, the majority of patients ranged between 16 and 40 (79.4%) years old, having a penetrating phenotype (n = 86, 63.2%) and an ileocolonic location (n = 109, 80.1%). Perianal disease was found in 56 (41.2%) patients at diagnosis. An end-to-side anastomosis (n = 68, 62.4%) was the most common technique. The majority of patients (n = 122, 89.7%) received immunosuppressive agents as postoperative prophylaxis treatment. The postoperative index colonoscopies were performed within a median of 6.2 (IQR, 3.9–8.2) months after surgery. The RS scores at index colonoscopies were as follows: 34 patients (25.0%) scored i0, 15 patients (11.0%) scored i1, 65 patients (47.8%) scored i2, 4 patients (3.0%) scored i3, and 18 patients (13.2%) scored i4.
Baseline demographic and clinical characteristics (n = 136).

CR
After a median duration of follow-up of 26.9 (IQR, 11.4–55.2) months, CR was observed in 47.1% (n = 64) of patients. The median CR-free survival was 56.1 (IQR, 32.0–80.3) months. The probability of CR was significantly higher in the RS ⩾ i2 group (52/87, 60.0%) compared with the RS ⩽ i1 group (12/49, 24.5%) (HR 3.37, 95% CI: 1.79–6.32, p < 0.001) [Figure 1(a), log-rank p < 0.001].

Kaplan–Meier curve of the risk of clinical relapse stratified by the modified endoscopic score after ileal or ileocolonic resection. Kaplan–Meier curve showed that the risk of CR was significantly higher in the RS ⩾ i2 group compared with the RS ⩽ i1 group [part label (a), log-rank p < 0.001]. Moreover, patients with ileal lesions (I1–4) had a significantly higher risk of CR compared with patients without (I0) [part label (b), log-rank p < 0.001]. Besides, patients with anastomotic lesions (A1–3) had significantly higher rates of CR than patients without (A0) [part label (c), log-rank p = 0.002].
Moreover, CR-free survival was significantly shorter in patients with ileal lesions (I1–4) irrespective of their severity compared with patients without (I0) (median survivals: 104.5 versus 20.0 months for I0 versus I1–4, respectively; HR 2.73, 95% CI: 1.60–4.66, p < 0.001) [Figure 1(b), log-rank p < 0.001]. Besides, patients with anastomotic lesions (A1–3) had significantly higher rates of CR than patients without (A0) (HR 2.44, 95% CI: 1.37–4.37, p = 0.003) [Figure 1(c), log-rank p = 0.002].
To determine the potential factors associated with CR, univariate Cox regression was used to screen the associations between variables (including RS score, scores of ileal and anastomotic lesions, and other covariates) and CR (Table 2). We found that RS score (HR 1.71, 95% CI: 1.40–2.10, p < 0.001), modified ileal score (HR 1.87, 95% CI: 1.49–2.35, p < 0.001) and anastomotic score (HR 1.54, 95% CI: 1.21–1.95, p < 0.001) were associated with higher risk of postoperative CR (Table 2). We further built several prediction models based on the traditional RS score or modified ileal and anastomotic scores, respectively (Table 3). As a basic model for reference, Model 1 and 2 contained only RS score and modified ileal and anastomotic scores, respectively. Model 3 and 4 were constructed by bi-direction stepwise Cox regression. Model 3 included RS score, L2 type and perianal disease, while Model 4 contained modified ileal and anastomotic scores, sex, L2 type and perianal disease (Table 3).
Factors associated with clinical relapse using univariate analysis.

CI, confidence interval; B1, nonstricturing, nonpenetrating type; B2, stricturing type; B3, penetrating type; L2, isolated colonic Crohn’s disease; RS, Rutgeerts score.
The bold font represents statistical significance at p<0.05.
Predicting models based on Rutgeerts score or modified ileal and anastomotic scores for clinical recurrence of postoperative patients with Crohn’s disease.

A, postoperative anastomotic lesions; I, postoperative ileal lesions; L2, isolated colonic Crohn’s disease; RS, Rutgeerts score.
Nomogram construction
A prognostic nomogram to identify early CR after ileal or ileocolonic resections of CD patients was established based on Model 4 in Table 3 and points were assigned to the predictive factors according to their regression coefficients (Figure 2). As shown in the nomogram plot, female patients with non-L2 subtype, perianal disease, and postoperative ileal/anastomotic lesions were more likely to have CR. By summing the scores of each variable and projecting the total score to the bottom axis, the probability of CR can be calculated.

A nomogram plot for prediction of clinical relapse based on the Model 4 in Table 3. As shown in the nomogram plot, female patients with non-L2 subtype, ileal lesions, anastomotic lesions, and perianal disease were more likely to have CR after ileal or ileocolonic resections. The total points obtained from the sum of the scores of each variable in the model could quantitatively reflect the risk of CR.
The performance of the nomogram model
To evaluate the performance of the nomogram, different models were compared using the ROC curve (Figure 3) and DCA plot (Figure 4). We found that Model 4 had a relatively higher area under the curve (AUC = 0.764) compared with other models (AUC = 0.703, 0.715, and 0.757 in Model 1, 2, and 3, respectively) (Figure 3), although no statistical difference was found in the ROC test when comparing to other models. Further DCA analysis was performed to explore the benefit gained by each model in clinical decision. The Model 4 showed a generally higher area under the decision curve (AUDC) compared with other models, especially at the timeframe of 24–60 months after index endoscopy, indicating a higher benefit of the nomogram based on Model 4 than other models in the time period (Figure 4).
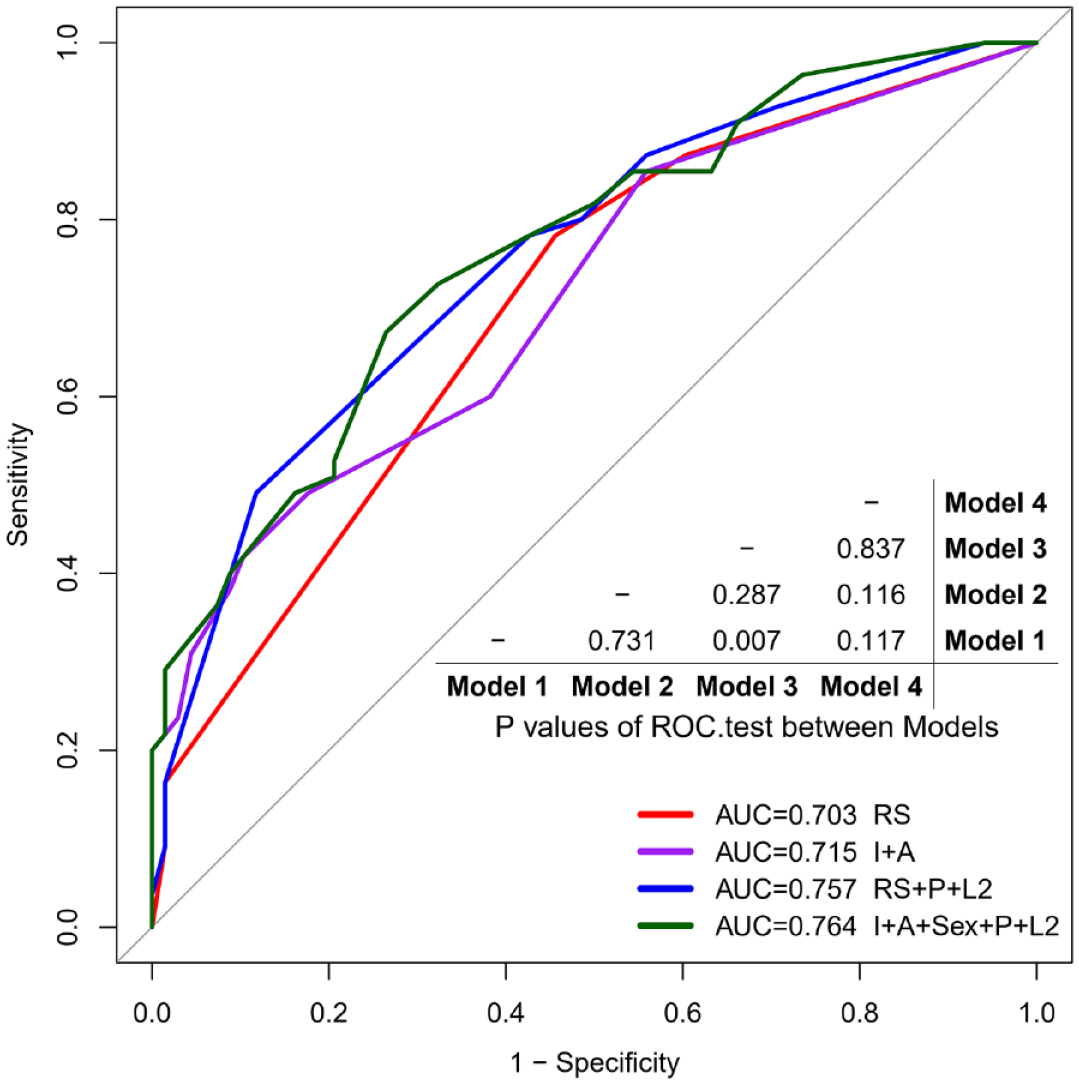
The ROC curve of different models in Table 3. A higher AUC represents a better prediction accuracy. The prediction performance of each model was compared with other models, and the results were summarized in the table.

Decision curve analysis of different models in Table 3. (a) Decision curve analysis on the benefit of patients when predicting risk of CR at five different time points (from 12 to 60 months) by different models. A higher DCA curve at a time point (a higher Net Benefit) represents a more accurate prediction of risk of CR on a patient at that time. ‘ALL’ means that all patients were divided into high-risk population. ‘None’ means that no model was used for stratification. (b) A summary of DCA analyses of different models. A higher AUDC of a model at a time point means a better average performance at the time point. (c) Comparison of AUDC between RS and each of other models. A higher curve represents a more obvious improvement of the prediction model compared to RS.
Discussion
In this retrospective study, the risk of CR was significantly higher in patients who scored an RS ⩾ i2 on the postoperative endoscopic evaluation. More importantly, we found a higher risk of CR in patients with ileal lesions at early postoperative endoscopy irrespective of the severity (I1–4) compared with patients without (I0) (p < 0.001). Besides, patients with anastomotic lesions (A1–3) had significantly higher rates of CR than patients without (A0) (p = 0.002). A nomogram was established using bi-direction stepwise regression results and can be used to predict CR after ileal or ileocolonic resections in CD patients.
The REMIND group recently built an endoscopic score describing the anastomotic and ileal lesions separately and showed that CR-free survival was significantly shorter in patients with ileal lesions (I1–4) compared to patients without (I0). 9 Our study came to a similar result. As for anastomotic lesions, the REMIND group showed severe anastomotic lesions (A2–3) had a significantly higher risk of occlusive complications and a trend toward a higher risk of CR. 9 In our study, patients with A1–3 were significantly associated with shorter CR-free survival than patients with A0 (p = 0.002). These studies suggested that anastomotic lesions are indicative of disease recurrence. Indeed, a retrospective cohort study (n = 182) found that anastomotic ulcers were common after ileocolonic resection and were associated with an increased risk of disease recurrence. 11 Similarly, another retrospective study (n = 116) showed that the risk of ER was significantly higher in patients with anastomotic ulcers. 12 However, the etiology of these anastomotic lesions is unknown. It has been suggested that postoperative relative ischemia, microbiome, immune dysfunction, and altered lymphatics might be associated with ulcerations at the anastomotic site.8,11,12 Further studies are warranted in this setting.
To further identify possible predictors for POR, stepwise regression was conducted using variables including gender, age, disease behavior, location, perianal disease, smoking, type of anastomosis, and endoscopic scores. However, only non-L2 subtype, female, perianal disease, RS score, ileal lesions, and anastomotic lesions were associated with CR based on stepwise regression in our study. As indicated by previous studies, smoking has been a well-established risk factor for clinical recurrence. 13 Our study did not find an increased risk in smokers which may be explained by the relatively small number of smokers since our center has established an effective clinical follow-up system and provided reasonable health education for the patients, such as no smoking. Although an Australian group found that ileocolonic end-to-end anastomoses increased the risk of early postoperative ER in CD patients with an emergency resection and suggested that end-to-end anastomoses should be avoided in this subgroup of patients, 14 our study failed to reveal a significant difference among types of anastomosis which partly due to the small sample size of the end-to-end group (this kind of anastomosis procedure has been avoided in our center these years). Isolated colonic CD (L2) has been considered a distinct disease when compared to ileal or ileocolonic CD.15,16 Ileal involvement was associated with an increased risk of change in disease behavior and developing an intestinal complication relative to colonic CD. 17 We found L2 subtype was a protective factor, which was consistent with the previous studies.
To the best of our knowledge, our study is the first to establish a nomogram incorporating ileal and anastomotic lesions to predict the long-term outcome of CD patients after ileal or ileocolonic resection. As shown in the nomogram plot (Figure 2), female patients with non-L2 subtype, perianal disease, ileal lesions, and/or anastomotic lesions were more likely to have CR after ileal or ileocolonic resections. By summing the total score, CR can be predicted. Further, the ROC curve showed a relatively higher AUC of the nomogram when compared to other prediction models, which was confirmed by the DCA analysis. Nevertheless, the weight of the L2 subtype in the nomogram seemed heavy due to the limited number of L2 patients enrolled in our study, and none of them experienced CR during the follow-up. According to the previous studies, isolated colonic CD (L2) had relatively low surgery rate when compared with other phenotypes 18 and showed a protective role of disease behavior progression. 17 Further larger unbiased cohort is needed to validate our preliminary findings in this setting.
Objective outcomes are important when evaluating patients with CD because the correlation between symptoms and endoscopic disease activity is poor. 19 This might be especially true in the postoperative CD patient, as a recent study showed that the type of anastomosis may drive clinical symptoms without evidence of endoscopic disease recurrence. 20 Thus, in the present study, we used a combined outcome with objective index including treatment intensification and endoscopic or surgical intervention. 9
Our study has certain limitations. First, it was conducted in a single-center retrospective setting. To mitigate recall risks, objective outcomes defining CR such as treatment escalation and recurrent surgery were retained to restrict the observational bias. Second, the reproducibility of the RS is moderate, which may lead to incorrect therapeutic decisions in >10% of patients. 21 Two senior gastroenterologists blind reviewed all colonoscopy reports and images to reduce this risk, and discrepancies were solved by a third investigator when indicated. Third, the rate of CR in patients with i0 or i1 RS scores was higher than in the previous literature. 3 It might be caused by the admission bias. As a large tertiary referral center, the patients in this cohort tend to have a more severe disease course. Besides, although postoperative prophylaxis, the majority of the patients used immunosuppressive agents rather than biologics, which may partly explain the relative high rate of CR in this cohort. Last but not least, the lack of an external cohort to validate the established nomogram is one of the major limitations of our study. Further prospective studies with larger cohorts are warranted.
In conclusion, a nomogram based on postoperative ileal and anastomotic lesions was developed to predict CR in CD patients, which may serve as a practical tool to identify high-risk subgroups who need timely postoperative intervention.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231198933 – Supplemental material for A nomogram incorporating ileal and anastomotic lesions separately to predict the long-term outcome of Crohn’s disease after ileocolonic resection
Supplemental material, sj-docx-1-tag-10.1177_17562848231198933 for A nomogram incorporating ileal and anastomotic lesions separately to predict the long-term outcome of Crohn’s disease after ileocolonic resection by Shanshan Xiong, Jinshen He, Baili Chen, Yao He, Zhirong Zeng, Minhu Chen, Zhihui Chen, Yun Qiu and Ren Mao in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
