Abstract
Objectives
To explore effectiveness and safety of polyglycosides of Tripterygium wilfordii (GTW) and mesalazine (5-aminosalicylic acid [5-ASA]) in preventing postoperative clinical and endoscopic recurrence of Crohn’s disease.
Methods
In this prospective, single-centre, single-blind study, postoperative Crohn’s disease patients in remission were randomized to receive 1 mg/kg GTW daily, orally, or 4 g 5-ASA daily, orally, for 52 weeks. Patients underwent physical examinations, ileocolonoscopies and biochemical analyses at baseline and weeks 13, 26 and 52, or when clinical recurrence was suspected. Outcome measures were proportion of patients showing clinical or endoscopic recurrence at week 52, and changes in Rutgeerts’ and Crohn’s Disease Activity Index (CDAI) scores.
Results
Twenty-one patients were assigned to receive GTW and 18 to 5-ASA; two patients on GTW and one on 5-ASA were withdrawn. Clinical and endoscopic recurrences were less common in the GTW group (n = 4) versus the 5-ASA group (n = 9). There were improvements in Rutgeerts’ scores for those taking GTW. Mean between-group CDAI scores were similar. No serious adverse events were reported.
Conclusion
These findings indicate that GTW appears to be an effective, well-tolerated prophylactic regimen, superior to oral 5-ASA, for preventing clinical and endoscopic recurrence in postsurgical Crohn’s disease.
Keywords
Introduction
Crohn’s disease is a chronic, granulomatous, inflammatory bowel disease that can affect any segment of the gastrointestinal tract from the anus to the mouth. No definitive medical or surgical cure is available for Crohn’s disease patients. 1 Thus, the principal treatment goal is to control active symptoms and maintain or prolong remission, preventing the disease from significantly compromising the patient’s health-related quality of life. 2
Surgery is required at least once in ∼75% of patients with Crohn’s disease during the natural course of their disease. 3 Surgical intervention is likely to resolve some complications (such as intestinal obstruction, fistula and abscess) that are refractory to medical treatment. Relapse of the disease postoperatively is common, however, as current prophylactic measures against postoperative recurrence are not particularly effective in the long term, with 70–90% of patients showing endoscopic recurrence within the first 12 months of surgery. 4
Tripterygium wilfordii (TW) is a vine used in traditional Chinese medicine that has been reported to be therapeutically efficacious in the treatment of autoimmune and inflammation-related diseases. 5 A chloroform/methanol extract of TW, T2, principally contains polyglycosides of TW (GTW). 6 A prospective study demonstrated that T2 appeared to be effective in inducing remission of active Crohn’s disease after 12 weeks’ treatment, although only in a small number of patients. 7 Remarkable clinical benefits have been achieved in the absence of severe toxicity, suggesting favourable safety and tolerability profiles for GTW. 5 Mesalazine (also known as 5-aminosalicylic acid [5-ASA]) has been widely used as first-line prophylaxis against postsurgical recurrence of Crohn’s disease. 8 Uniquely for 5-ASA compounds, mesalazine delivers the active substance to the neoterminal ileum, which is the site most commonly involved in Crohn’s disease recurrence. Current evidence shows that 5-ASA (≥2 g/day) lowers postoperative recurrence in small-bowel disease, although its efficacy is marginal. 9
To date, no randomized study has investigated the effectiveness and safety of GTW in preventing postoperative recurrence of Crohn’s disease. The present prospective study, conducted in postsurgical patients with Crohn’s disease in remission, evaluated the effectiveness and tolerability of GTW compared with 4 g/day 5-ASA as maintenance therapy in preventing the clinical and endoscopic recurrence of Crohn’s disease.
Patients and methods
Study Population
This was a prospective, single-centre, randomized, single-blind study. Patients who had undergone terminal ileectomy, partial colectomy or ileocolectomy with ileocolonic anastomosis, due to histologically diagnosed active Crohn’s disease, were enrolled at Jinling Hospital, Nanjing University Medical School, Nanjing, China, between May 2009 and December 2011. Patients aged between 18 and 60 years were eligible for the study if they had remained in clinical remission since resection (which had to have been undertaken within the previous 2 weeks) and had a Crohn’s Disease Activity Index (CDAI) score 10 ≤150 in the preceding 1–2 weeks. CDAI values ≤150 are associated with quiescent disease; values >150 indicate active disease and values >450 are seen with extremely severe disease.
Postoperative recurrence risk assessment was performed for enrolled patients using the following criteria: 11 (1) very low, defined as longstanding, first surgery, short stricture; (2) low–moderate, i.e. <10-year history of Crohn’s disease, presence of a long stricture or any case of inflammatory Crohn’s disease; (3) high risk, defined as presence of penetrating disease or patients with repeated intestinal resection (more than twice). A patient’s risk status was not part of the study inclusion criteria.
Patients were excluded from the study if they had any of the following: a malignant tumour; received immunosuppressant agents (methotrexate, cyclosporine, 6-mercaptopurine [6-MP], azathioprine [AZA] or 6-thioguanine) since resection; received inhibitors of tumour necrosis factor (TNF)-α, or other biologic therapies indicated for Crohn’s disease since resection; received corticosteroids or oral antibiotics (such as metronidazole or ciprofloxacin) for >2 weeks since resection; received nonsteroidal anti-inflammatory drugs (NSAIDs) other than low-dose acetylsalicylic acid (75 mg) within the preceding 2 weeks; received paracetamol; exhibited an elevation in levels of alanine aminotransferase and/or aminotransferase; a serum creatinine level >130 µmol/l.
The study protocol was approved by the Institutional Review Board at Jinling Hospital (Ethics Committee reference number: 2009SJJ-036). Patients volunteered for the study and provided written informed consent before enrolment.
Study Design
Patients were randomized in a 1 : 1 ratio to receive either 1 mg/kg per day GTW tablets (Huangshi Feiyun Pharmaceutical Co., Taizhou, China), orally, three times per day after food, or 4 g/day 5-ASA tablets (Salofalk®, Losan Pharma GmbH, Neuenburg am Rhein, Germany), orally, four times per day after food. The randomization schedule was generated by Jinling Hospital Pharmacy, using a self-compiled computer randomization program written in Visual Basic, version 6 (Microsoft®, Redmond, VA, USA). Patients were medicated in an investigator-blinded manner within 2 weeks of surgery if they could tolerate oral intake.
Concomitant use of the following medications was not permitted during the study: immunosuppressants; allopurinol; oxipurinol; thiopurine; TW-containing or 5-ASA-containing drugs other than the study drug; oral antibiotics for >4 weeks or more than three cycles of 2 weeks; NSAIDs for >2 weeks; corticosteroids; cimetidine. Patients who developed symptoms suggestive of clinical recurrence were expected to continue the assigned treatment until the final follow-up visit at week 52. In cases of tolerable adverse events, the medication was continued at the discretion of the principal investigator. Patients were able to withdraw from the study at any time for any reason, including an adverse event.
The intent-to-treat (ITT) population consisted of patients who met the inclusion/exclusion criteria and were randomized to receive (and took at least one dose of) study drug. Patients who met the inclusion/exclusion criteria, who were randomized to receive study drug and who completed the course of study treatment to week 52 constituted the per-protocol population and were included in the final analysis.
Study Assessments
During the study period, all patients were instructed to record their intake of concomitant medications and their abnormal symptoms or events, including elevated body temperature measured by thermometer, increased stool frequency and type, and abdominal pain or discomfort. This information was then reported to the investigators at each study visit. Patients attended hospital for a physical examination and biochemical analyses at baseline (day 0) and weeks 13, 26 and 52. The CDAI was calculated at each visit.
Patients were required to fast overnight for 8–12 h prior to venous blood samples being taken on the morning of the visit. Blood samples were added to 1.5 mg/ml ethylenediaminetetra-acetic acid as an anticoagulant, and were used to determine the complete blood count and C-reactive protein (CRP) level. Blood samples were also added to 3.8% sodium citrate as an anticoagulant, to determine the erythrocyte sedimentation rate (ESR). Analyses were carried out using a Sysmex XE-2100™ analyser (Sysmex, Kobe, Japan).
Serum was prepared for the detection of alkaline phosphatase, total bilirubin, alanine aminotransferase, aspartate aminotransferase, creatinine and urea. Blood samples were allowed to stand for ≥20 min at room temperature to allow clotting, after which the serum was collected by centrifugation at 2796
To determine each patient’s Rutgeerts’ score, 12 an endoscopist from the Endoscopy Centre (Jinling Hospital), who was blinded to treatment assignment, performed ileocolonoscopy at weeks 26 and 52 (or earlier if a symptom of suspected clinical recurrence was observed). Rutgeerts’ score was estimated as follows: grade i0, no lesions on endoscopy; grade i1, five or fewer aphthous lesions; grade i2, more than five aphthous lesions with normal mucosa between the lesions, or skip areas of larger lesions, or lesions confined to the ileocolonic anastomosis (<1 cm in length); grade i3/i4, diffuse aphthous ileitis with diffusely inflamed mucosa, or diffuse inflammation with larger ulcers, nodules and/or narrowing.
Patients were withdrawn prematurely from the study for any of the following reasons: if they did not have complete follow-up data available for the outcome evaluation; if they developed serious clinical recurrence or adverse events requiring termination of treatment; if they failed to take the medication as prescribed, switched to any other agent without permission from the investigators, or started/resumed cigarette smoking or alcohol consumption.
Outcome Measures
Endoscopic recurrence was defined as the occurrence of a new lesion (small aphthous ulcers, deep linear ulcers, mucosal inflammation, fistulae and strictures) on endoscopy, or a Rutgeerts’ score of i2 or above. Clinical recurrence was defined as postoperative occurrence of symptoms attributable to Crohn’s disease, or a CDAI score >150, requiring medical or surgical treatment in the context of endoscopic recurrence.
The primary outcome measure was the proportion of patients showing clinical recurrence at week 52. Secondary outcome measures were the proportion of patients with endoscopic recurrence at week 52 and the change in Rutgeerts’ score, change in the CDAI and elevations in CRP levels and the ESR between baseline and week 52.
Adverse Events
Safety data were collected and evaluated at each follow-up visit for each patient. Adverse events were documented by study staff at each visit, based on verbal information provided by the patients, and were reported immediately to the principal investigator. Adverse events were categorized in terms of severity (mild, moderate or severe) and causative relationship with the study medication (certain, probable/likely, possible, unlikely, conditional/unclassified or unassessable/unclassifiable). Patients and investigators were asked to assess global tolerability as very good, good, satisfactory or poor.
Statistical Analyses
Statistical analyses were performed using SPSS® software, version 13.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Continuous data were expressed as mean ± SD; categorical data were expressed as frequency. Between-group differences in continuous data were compared using Student’s t-test or repeated-measures analysis of variance, and differences in categorical data were compared using Fisher’s exact test. A P-value < 0.05 was considered statistically significant.
Results
In total, 45 patients were enrolled, of whom 39 were eligible for randomization and were included in the ITT population; 21 were assigned to receive GTW and 18 to receive 5-ASA treatment. The ITT population consisted of 21 males and 18 females (mean ± SD age, 35 ± 9 years). The two groups were similar in terms of baseline characteristics including age, sex, history of Crohn’s disease, disease location, disease severity, previous surgical history, concomitant medications, surgical complications and procedures received. The overall proportion of patients with penetrating disease was 76.9% (30/39), and the proportions of patients who underwent multiple (i.e. at least two) bowel resections in the treatment and control groups were 66.7% (14/21) and 61.1% (11/18), respectively, suggesting a high risk of recurrence.
13
At week 13, two patients were prematurely withdrawn from the GTW treatment group due to their failure to take the study medication as prescribed. At week 26, one patient on 5-ASA treatment was lost to follow-up. All patients remained in remission and free of any symptom suggestive of clinical recurrence between baseline and week 26, except for one patient in the 5-ASA group. Crohn’s disease symptoms recurred between weeks 26 and 52 in one patient on GTW and three patients on 5-ASA. None of these four patients was withdrawn prematurely from the study. Thirty-six patients (19 [90.5%] in the GTW group and 17 [94.4%] in the 5-ASA group; Figure 1) completed the course of study treatment to week 52; these patients constituted the per-protocol population eligible for analysis. The data reported here were all from the per-protocol population.
Flow chart of postoperative patients with Crohn’s disease, detailing patient numbers at study enrolment, study medication (receiving 60 mg/day polyglycosides of Tripterygium wilfordii [GTW] orally or 4 g/day mesalazine [5-aminosalicylic acid; 5-ASA] for 52 weeks) and follow-up.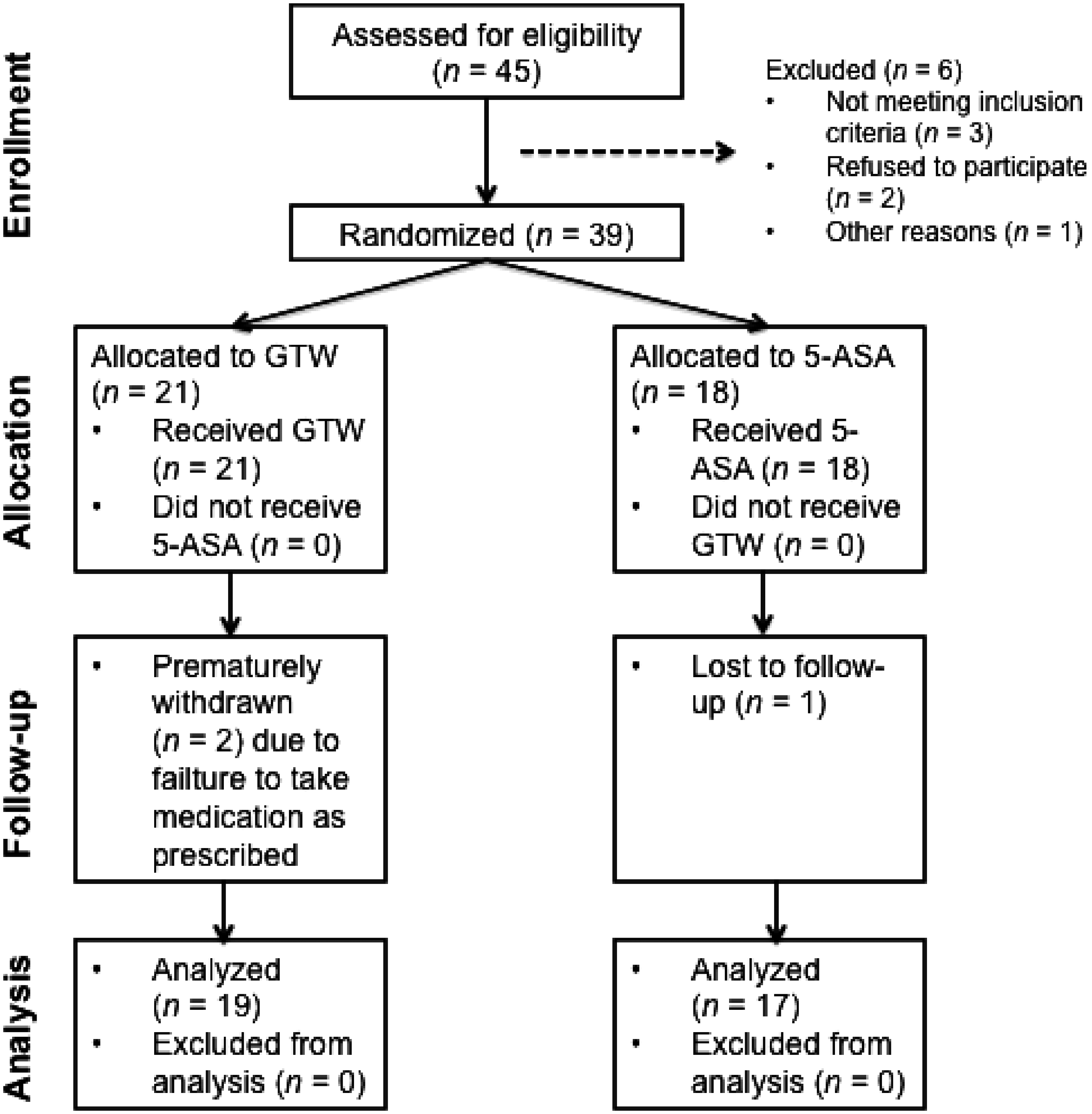
Profile of clinical and endoscopic recurrence of postoperative Crohn’s disease in patients (n = 36) receiving 1 mg/kg per day polyglycosides of Tripterygium wilfordii (GTW) orally or 4 g/day mesalazine (5-aminosalicylic acid; 5-ASA) orally for 52 weeks. a
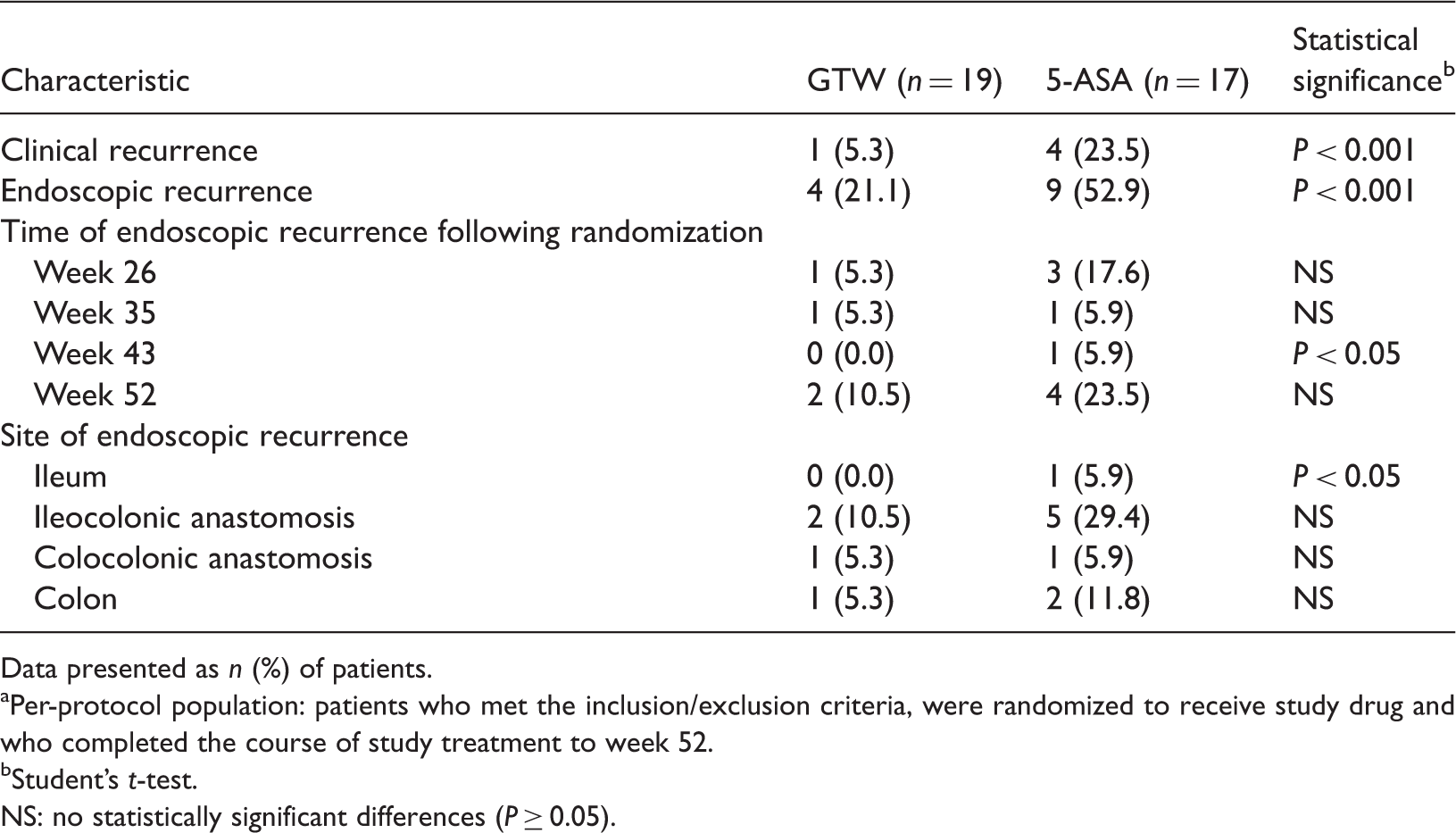
Data presented as n (%) of patients.
Per-protocol population: patients who met the inclusion/exclusion criteria, were randomized to receive study drug and who completed the course of study treatment to week 52.
Student’s t-test.
NS: no statistically significant differences (P ≥ 0.05).
Rutgeerts’ scores in postoperative Crohn’s disease patients (n = 36) receiving 1 mg/kg per day polyglycosides of Tripterygium wilfordii (GTW) orally or 4 g/day mesalazine (5-aminosalicylic acid; 5-ASA) orally for 52 weeks. a

Data presented as n (%) patients.
Per-protocol population: patients who met the inclusion/exclusion criteria, were randomized to receive study drug and who completed the course of study treatment to week 52.
Student’s t-test.
Rutgeerts’ score estimated as follows: grade i0, no lesions on endoscopy; grade i1, five or fewer aphthous lesions; grade i2, more than five aphthous lesions with normal mucosa between the lesions, or skip areas of larger lesions, or lesions confined to the ileocolonic anastomosis (<1 cm in length); grades i3/i4, diffuse aphthous ileitis with diffusely inflamed mucosa, or diffuse inflammation with larger ulcers, nodules, and/or narrowing.
NS: no statistically significant differences (P ≥ 0.05).
Improvement in the mean CDAI score from baseline to week 52 was similar between the GTW group and the 5-ASA group. Both groups showed a continuously decreasing trend, and there was a significant difference between the two groups at week 52 (P < 0.05 for both) (Figure 2). Levels of CRP and the ESR remained relatively unchanged from baseline until week 13, but both indices showed a slightly increasing trend through the remaining 39 weeks (Figure 3). The increase was slightly greater in the 5-ASA group than in the GTW group, although this did not reach statistical significance. Elevations of ESR >20 mm/h, and CRP >10 mg/l, were observed in nine patients (GTW versus 5-ASA, three of 19 [15.8%] versus six of 17 [35.3%]; P < 0.01) and eight patients (GTW versus 5-ASA, three of 19 [15.8%] versus five of 17, [29.4%]; P < 0.01) at week 52, respectively.
Crohn’s Disease Activity Index (CDAI) scores for postoperative Crohn’s disease patients (n = 36) receiving 1 mg/kg/day polyglycosides of Tripterygium wilfordii (GTW) (n = 19) orally or 4 g/day mesalazine (5-aminosalicylic acid; 5-ASA) (n = 17) orally for 52 weeks. Data presented as mean ± SD. *P < 0.05 for the GTW group versus the 5-ASA group, Student’s t-test. (A) Erythrocyte sedimentation rate (ESR) and (B) C-reactive protein (CRP) levels for postoperative Crohn’s disease patients (n = 36) receiving 1 mg/kg per day polyglycosides of Tripterygium wilfordii (GTW) (n = 19) orally or 4 g/day mesalazine (5-aminosalicylic acid; 5-ASA) (n = 17) orally for 52 weeks. Data presented as mean ± SD. There were no statistically significant differences between the two groups; Student’s t-test.

No severe adverse events occurred in either group. No patients were withdrawn prematurely from the study due to adverse events. The overall incidence of adverse events was not significantly different between the two groups (GTW versus 5-ASA patients, seven of 19 [36.8%] versus seven of 17 [41.2%]). The most commonly reported adverse events in patients were: leucocytopenia (GTW versus 5-ASA patients, <4.0 × 109/l; five of 19 [26.3%] versus four of 17 [23.5%]); mild elevation (1–4 × upper limit of normal) of ALT (GTW versus 5-ASA, one of 19 [5.3%] versus one of 17 [5.9%]); diarrhoea (GTW versus 5-ASA patients, one of 19 [5.3%] versus two of 17 [11.8%]). The overall tolerability of both treatments was reported to be ‘good’ by the majority of patients: (GTW versus 5-ASA patients, 14/19 [73.7%] versus 12/17 [70.6%]).
Discussion
To our knowledge, this is the first prospective randomized study to evaluate the prophylactic effects of GTW on postoperative recurrence in Crohn’s disease patients. Over a 1-year period, GTW was more effective than 5-ASA in minimizing the postoperative clinical and endoscopic recurrence of Crohn’s disease. The 5-ASA group showed a 1-year clinical recurrence rate of 23.5% and an endoscopic recurrence rate of 52.9%, suggesting a relatively high risk of postoperative relapse of Crohn’s disease in the patient population, even compared with a high-risk population receiving prophylaxis with AZA versus 5-ASA; this was suggested to be attributable to a higher overall proportion of patients with penetrating disease (76.9%). 14
Although the CDAI is commonly used to assess severity of disease and outcome of treatment in Crohn’s disease patients, its role in evaluating the progress of postsurgical patients is limited, as there is a poor correlation between CDAI score and endoscopic recurrence at 1 year postoperatively. 11 The present study showed a continuously decreasing trend in mean CDAI for both treatments, although the incidence of endoscopic recurrence increased. Both ESR and CRP are reported to be useful for predicting response to medical treatment in Crohn’s disease patients. 3 These indices remained relatively constant in patients with Crohn’s disease in remission, but increased significantly during recurrence.
Historically, a variety of agents have been evaluated for the prevention of postoperative recurrence of Crohn’s disease including 5-ASA, corticosteroids, antibiotics, probiotics, immunomodulators and immunosuppressors, and monoclonal antibodies against TNF-α (such as infliximab and adalimumab).15,16 In a meta-analysis, use of 5-ASA in patients with Crohn’s disease was confirmed to have good compliance and a low risk of adverse events, 17 but seems to provide only a small reduction in clinical and endoscopic recurrence. 18 Until recently, clinical data regarding the immunosuppressants AZA and 6-MP were limited. AZA and 6-MP have been shown to be superior to 5-ASA for the prevention of postsurgical recurrence of Crohn’s disease; most therapeutic failures result from adverse effects requiring discontinuation of treatment.14,19 These two regimens are, therefore, recommended for high-risk patients, such as those with ileocolonic anastomoses and moderate or severe endoscopic recurrence. 14 Infliximab is reported to be effective in the prevention of endoscopic and histologic recurrence of Crohn’s disease, but the high cost of treatment limits its accessibility. 18
Although GTW has anti-inflammatory and immunosuppressive activities, its underlying mechanism of action is less well understood. 7 GTW is reported to induce apoptosis in T-lymphocytes and dendritic cells through the inhibition of the nuclear factor-κB signalling pathway, and reducing production of the proinflammatory cytokines TNF-α and interleukin-1.20,21 Thus, GTW is expected to ameliorate the intestinal inflammatory response, decrease intestinal permeability and (consequently) protect intestinal barrier function, in postoperative Crohn’s disease patients. 22 Research has suggested that GTW significantly improved symptoms and delayed postsurgical recurrence of Crohn’s disease. 23
The present study demonstrated that GTW had similar efficacy to 5-ASA in preventing clinical and endoscopic recurrence of Crohn’s disease in high-risk patients, with a favourable safety profile. No major adverse events were identified in GTW-treated patients during the 52-week observation period: the majority of adverse events were mild, and of transient duration. Clinical laboratory evaluations detected moderate effects of GTW on the haematological system and in the liver. The safety and tolerability profile of GTW was not inferior to that of the widely used 5-ASA.
There were some limitations to the present study. As discussed above, patients enrolled in the study were primarily considered to be at high risk of recurrence, although this was not an inclusion criterion or a direct aim of the study. Additionally, 5-ASA might be considered an alternative treatment option for patients with a low, rather than high, risk of clinical recurrence. 24 Most of the patients had a penetrating phenotype or had undergone multiple intestinal resections. Thus, using 5-ASA as the comparator in this study may not have been fair. Despite this, the authors believe that the results contribute to the investigation of the efficacy and safety of GTW, and that they may have a future impact on clinical practice. Some limitations of the study design should be addressed in any future studies: the pharmaceutical properties of GTW do not allow double-blind medication; in addition, a relatively small number of patients from a single centre was studied. A multicentre, randomized controlled trial is planned, following ethical approval.
In conclusion, the present study showed that GTW is more efficient than 5-ASA in preventing postoperative Crohn’s disease recurrence in high-risk patients over the course of 1 year, with minimal toxicity. These data suggest that GTW can reduce clinical and endoscopic recurrence rates, and may also delay the onset and severity of postoperative recurrence.
Footnotes
Declaration of Conflicting Interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by a grant from the Climb Program of the Natural Science Foundation for Distinguished Scholars, Jiangsu, China (BK2010017) and the Advanced Training Program for Research Talents of Jiangsu Province, China (BRA2011232).
