Abstract
The purpose of this article is to provide an overview of white light colon capsule endoscopy’s current clinical application, concentrating on its most recent developments. Second-generation colon capsule endoscopy (CCE2) is approved by the FDA for use as an adjunctive test in patients with incomplete colonoscopy and within Europe in patients at average risk, those with incomplete colonoscopies or those unwilling to undergo conventional colonoscopies. Since the publication of European Society of GI Endoscopy guidelines on the use of CCE, there has been a significant increase in comparative studies on the diagnostic yield of CCE. This paper discusses CCE2 in further detail. It explains newly developed colon capsule system and the current status on the use of CCE, it also provides a comprehensive summary of systematic reviews on the implementation of CCE in colorectal cancer screening from a methodological perspective. Patients with ulcerative colitis can benefit from CCE2 in terms of assessing mucosal inflammation. As part of this review, performance of CCE2 for assessing disease severity in ulcerative colitis is compared with colonoscopy. Finally, an assessment if CCE can become a cost-effective clinical service overall.
Keywords
Introduction
Over 1.9 million new colorectal cancer (CRC) cases and 935,000 deaths were expected in 2020, based on global cancer statistics. 1 In terms of incidence, CRC ranks third but is second in mortality. CRC commonly develops from benign adenomatous polyps (90%) that grow into cancer. Statistically, the risk of developing CRC is reduced by 70–90% due to screening and subsequent polypectomy.2 –4 To enhance the adoption of cancer screening, the European Commission introduced a new approach in February 2021, focusing on the early detection of cancers. As a result of this new EU directive, based on the best scientific evidence and current developments, Member States must offer breast, cervical and CRC screening by 2025 to 90% of EU citizens who qualify. 5 However, compared with breast, cervical and prostate screening programs, participation in the CRC screening program is still dismally low.6,7 Although colonoscopy is often the preferred CRC screening method in certain healthcare settings, it fails to meet the criteria for widespread population screening due to its invasive nature. The procedure carries a small but definite risk of serious complications such as bowel perforation, haemorrhage and cardiovascular incidents.8,9 Fear of unfamiliar settings, sedation and invasion of personal privacy may also contribute to reduced participation in screening colonoscopies. To address the limitations associated with colonoscopy and optimize the allocation of screening resources, alternative options have been explored. 8 In recent years, there have been a variety of screening options available for CRC. Non-invasive alternatives include faecal immunochemical test (FIT), multitarget stool DNA testing (available under the brand name Cologuard), computed tomography (CT) colonography (previously called virtual colonoscopy), guaiac-based fecal occult blood testing and colon capsule endoscopy (CCE). These tests have varied the degree of evidence supporting their use. FIT, which detects occult blood, is a simple and widely implemented screening modality. However, false positives and the need for subsequent colonoscopy present challenges. 10 Research suggests that using cut-off values of 45, 80, 125, 175 and 350 ng Hb/mL would require 24, 19, 16, 14 and 10 colonoscopies, respectively, to detect one advanced neoplasm.10,11 It often results in a high number of negative colonoscopies, draining valuable resources and exposing patients with negative results to the potential risks associated with the procedure. In an effort to optimize resources and reduce unnecessary colonoscopies, CCE has been proposed. CCE offers a safe, repeatable and affordable screening option that minimizes discomfort and serious adverse events. The National Health Service (NHS) England, for example, suggests offering CCE to individuals with FIT concentrations in the 10–99 ng Hb/mL range. 12 This approach aims to improve efficiency and resource allocation while maintaining the effectiveness of early CRC detection. In summary, CRC screening programs aim to provide safe, accessible and effective methods for early detection and prevention. While colonoscopy remains the reference standard, non-invasive alternatives like FIT and CCE are being explored to enhance screening efficiency, reduce unnecessary invasive procedures and improve patient experience. In this paper, we review the current status of CCE for colon cancer screening, compare its diagnostic yield (DY) to that of CT colonography (CTC), and consider the potential for use in inflammatory bowel disease. Additionally, CCE innovation and clinical translation will be discussed.13,14
Colon capsule endoscopy
The first CCE was released by Given® Imaging Ltd (Yoqneam, Israel) in 2006, and through a series of acquisitions, Medtronic, USA. 13 The CCE1 (PillCam® COLON, Medtronic, USA) is 11 × 31.5 mm in size, and it has two cameras with a 156° angle of view on both sides. 14 Three key components are needed to conduct CCE: an ingestible capsule endoscope, a data recorder worn by the patient during the procedure and imaging software.
There has been an advancement in technology, and the second generation of CCE is now on the market. PillCam™ Colon 2 (CCE2) was introduced in 2009 by Given® Imaging (eventually purchased by Medtronic, an American medical device company based in Ireland) to replace PillCam™ Colon 1. Among the improvements in CCE2 are higher camera frame rates, a greater field of view and simplified data recording procedures. 15
Having an approximate battery life of 10 h, the CCE2 capsule measures 11.6 mm × 31.5 mm. In CCE2, two cameras with 172° field of view are mounted, along with light-emitting diodes to illuminate the area around the camera and bidirectional wireless communication technology. Because the cameras have an adaptive frame rate, CCE2 can take more images in the colon when moving and fewer when stationary. Approximately the size of a hand, the data recording device consists of a small screen and a sensor lead connection socket. In addition to a real-time viewer, the data recorder can also provide bidirectional communication with the capsule. Images from the capsule are transmitted wirelessly and stored in the data recorder. As soon as the study is completed, it can be downloaded to a workstation with RAPID software and viewed by the reader. For enhanced visualization of mucosal patterns and vasculature, the software also has a polyp size estimation function and a flexible spectral imaging colour enhancement.16,17 It is crucial to highlight that although CCE is a non-invasive procedure, achieving excellent visualization of polyps relies on high-quality bowel preparation. This preparation surpassed the regimens typically used for standard colonoscopy and CTC. 18 It involves a bowel cleansing protocol lasting 1 or 2 days, which utilizes polyethylene glycol (PEG) along with additional booster preparations like sodium phosphate (NaP) or ascorbic acid. Moreover, patients are required to adhere to a fasting period and abstain from consuming solid food for 1 or 2 days before undergoing CCE, which contributes to the overall patient burden (Figure 1).17,18

Newly developed colon capsule system
As of 2022, several manufacturers produce capsule endoscopes. PillCam capsule endoscopes by Medtronic are among the most popular capsule endoscopes on the market. 19 Although Medtronic (following the sequential acquisition of Given® Imaging and Covidien) remains at present the only CCE manufacturer, this will change drastically by the end of the year (2022) as the two leading China-based capsule manufacturers are poised to present their respective systems and claim part of the international market (currently monopolized by Medtronic). Another less conventional system (CapsoCamPlus, CapsoVision, Saratoga, CA, USA) is said to have completed recruitment for a multi-centre, prospective, open-label, non-significant risk, pivotal pilot study of their CE system compared to colonoscopy (https://clinicaltrials.gov/ct2/show/NCT04246632). Below is the list of current available CCE (Table 1 and Figure 2)
Available types of colon capsule endoscopes and operating characteristics.

CCE, colon capsule endoscopy.

(a, b) Taken with OMOM CC100, Jinshan Science & TechnologyLtd, Yubei, China.
Bowel prep
CCE requires a larger preparation than colonoscopy since laxatives help clean the bowel and encourage capsule excretion. A small number of debris could affect the colon capsule`s ability to detect colonic polyps and, ultimately, the outcome of the procedure. 24 It is essential that completion rates (CRs) and adequate cleanliness rates (ACRs) are improved to meet the standards for optical colonoscopy (OC) established by the European Society of GI Endoscopy (ESGE). As recommended by ESGE, CR and ACR should reach 90%. 25 A minimum unadjusted caecal intubation rate of ⩾90% and a target rate of ⩾95% are expected as a measure of the completeness of the colonoscopy examination. A minimum of ⩾90% procedures and a target of ⩾95% procedures should have adequate bowel preparation. 26 By focusing on achieving high-quality bowel preparation and adherence to established performance measures, the effectiveness and reliability of CCE can be enhanced, leading to improved diagnostic accuracy and patient outcomes. 27 Meeting the standards set for OC ensures that CCE can provide comparable levels of completeness and cleanliness, further establishing it as a valuable tool in gastrointestinal endoscopy. Bjoersum-Meyer et al. 28 conducted the largest meta-analysis on CCE bowel preparation regimens and found both CRs and ACRs to be suboptimal for CCE. The most commonly used laxatives were PEGs and NaP boosters, but they did not increase CRs or ACRs. Spada et al. 18 found 100% sensitivity and 93% specificity for detecting polyps with good to excellent preparation but only 54% and 78% with suboptimal to inadequate preparation. Multiple booster and cleansing agents have been proposed in the literature in an attempt to improve CCE excretion and bowel preparation rates.29,30 NaP is a most commonly utilized booster; this preparation may be associated with major adverse events related to electrolyte abnormalities and renal failure in elderly patients and in those with underlying cardiac and renal comorbidities. Therefore, it is prescribed in low dose. 31
Among new boosters, the use of prucalopride as an additional booster agent in CCE practice has been studied. Prucalopride is a high affinity 5-HT4 (5-Hydroxytryptamine receptor 4) receptor agonist 32 that targets the impaired motility associated with chronic constipation. 33 Deding et al. 34 conducted a study in which a subgroup of 406 individuals underwent CCE in 2021. The first half (control) received the standard bowel preparation, and the second half (prucalopride) was supplemented with 2 mg of prucalopride. The CRs of CCE was 74.9% in the prucalopride group compared to 56.7% in the control group. Both proportions of complete transits within the battery lifetime of the capsule, and the proportion of acceptable bowel preparations were higher in the prucalopride group.
Gastrografin as an alternative to sodium booster has been considered recently. One significant advantage of Gastrografin is its high osmolality, which makes it a hyperosmotic laxative. In comparison to blood plasma, Gastrografin has approximately nine times higher osmolality; this property enables Gastrografin to effectively promote bowel movement unlike NaP, which can potentially increase the serum level of inorganic phosphate due to its high phosphate content. Gastrografin is minimally absorbed from the intestinal tract into the bloodstream. Only a small fraction, approximately 2%, is excreted unchanged in the urine without undergoing significant metabolism. As a result, Gastrografin administration is less likely to cause electrolyte imbalances. 35 A study conducted by Togashi et al. 36 evaluated the use of Gastrografin, substituted for NaP, in CCE bowel preparation. The study also compared the colon transit time between Gastrografin and a sulfate solution, with Gastrografin exhibiting a longer median transit time (165 min). This extended transit time allows for sufficient observation of the colon during capsule endoscopy. The polyp detection rate using Gastrografin as a booster was high (52%), indicating its effectiveness in detecting colon abnormalities. To increase patient acceptance of CCE, it is essential to address patient tolerability of preparation regimens. Schelde-Olesen et al. 37 conducted a single-centre retrospective comparative cohort study comparing a very low-volume PEG laxative to a conventional high-volume laxative. CR and ACR of 77% and 67% were reported in the high-volume group and 72% and 75% in the very low-volume group, respectively. A very low-volume bowel preparation regimen was non-inferior to a high-volume regimen before CCE in terms of CR and ACR. In a study led by Spada et al., 38 it was found that a split-dose bowel preparation regimen combining a 2 L PEG) solution in the evening before and 2 L in the morning, along with a low dose of NaP boosters, proved to be highly effective for capsule endoscopy (CCE). The cleansing regimen used in this study showed favourable levels of cleansing and successful excretion rates, leading to a significant portion of patients (78%) achieving sufficient cleansing. Additionally, the capsule was successfully excreted by 83% of the patients.
The study suggests that delivering the bowel preparation as a split dose similar to colonoscopy, along with the addition of NaP boosters, can optimize the outcomes of CCE.
Influential factors affecting CR in CCE
The standardized use of CCE in daily practice is still limited despite potential clinical indications. CCE’s accuracy and generalization will be enhanced by obtaining good capsule transit times and high CRs. To achieve completion within the battery time, the capsule transit time must be fast but not too fast to miss lesions. A recent study conducted in Nederland by Moen et al. 39 examined the factors affecting transit times in CCE. The study, which included 451 participants, is the largest to date to investigate possible predictors of CCE transit times. The following diagram summarizes the factors significantly associated with longer CCE transit times and lower CRs (Figure 3).

influential factors associated with longer CCE transit times and lower completion rates.
Current status on the use of CCE
The current status in the United States does not recommend using CCE in clinical scenarios other than incomplete colonoscopies. Based on the ESGE guidelines, CCE2 is recommended for use in patients with average risk, patients who have had an incomplete colonoscopy previously due to difficult scope progression, unrelated to inadequate bowel preparation, patients unwilling to undergo conventional colonoscopies, and patients unable to undergo or incompatible with conventional colonoscopies. 14 One of the main reasons ESGE could not strongly recommend enough CCE against its ‘rival’ CTC was that by the time these guidelines were compiled, the number of studies and information available to back CCE dwarfed the comparison of the evidence CTC had behind it. Since the publication of ESGE guidelines on the use of CCE, 14 there has been a significant increase in comparative studies on the DY of CCE versus CTC.
Use of CCE in clinical practice
CCE in CRC screening
There has been widespread adoption of colon screening programs for detecting adenomas and early stages of cancer, with the optimal age decreasing over the years, resulting in a greater demand for screening services.40 –42 CCE is an attractive option for patients undergoing colon screening due to its minimally invasive nature.
The use of CCE can assist in prioritising individuals with polyps, aiding in the management of demand. One approach is to prioritize colonoscopic intervention for individuals with large polyps measuring 10 mm or greater. On the other hand, individuals with diminutive or small polyps can be safely managed through routine pathways, resulting in a ‘downgrade’ in priority. 43 When CCE detects malignant pathology or large polyps (⩾10 mm), luminal assessment within an appropriate timeframe becomes necessary, provided the patient is fit enough for further investigation or treatment. To address intermediate and diminutive polyps, a pragmatic approach is required, especially when the current focus of endoscopy resources is on detecting CRC. 44 This approach allows endoscopy units to have more flexibility in scheduling appointments and promotes better workflow. CCE is being increasingly considered in diagnostic settings, such as low-risk abdominal symptoms. 45 By combining indications like urgent referral from general practitioners for patients with alarming symptoms and negative FIT (this cohort has a very low prevalence of advanced adenomas or carcinoma), 46 surveillance for hereditary CRC, and follow-up after polypectomy with a FIT triage, CCE can identify a significant proportion of referred patients.45,47 In the United Kingdom, a national pilot is underway to evaluate CCE as a first-line diagnostic test for patients with a positive FIT test. 12
In recent years many authors48 –54 systematically reviewed the validity of CCE in clinical practice, specifically its performance in CRC screening. Below is a comprehensive summary of systematic reviews on the implementation of CCE in CRC screening from a methodological perspective. In 2021, Vuik et al. 50 reported a systematic review of CCE, comprising 13 studies and 2485 patients. The polyp detection rate of CCE was 24–74%. For polyps >6 mm, the sensitivity of CCE was 79–96% and specificity was 66–97%. For polyps ⩾10 mm, the sensitivity of CCE was 84–97%; the CRC detection rate for completed CCEs was 93%. CRs were 57–92%, depending on the booster used. These studies, primarily designed to explore the use of CCE as a filter for colonoscopy following a positive FIT test, indicate that CCE appears to be an effective modality for detecting CRC and polyps. In another interesting systematic review and meta-analysis of clinical trials by Möllers et al., 49 the pooled sensitivities and specificities for polyps ⩾6 mm were 87% (95% CI: 83–90%) and 87% (95% CI: 76–93%) in eight studies, respectively. For polyps ⩾10 mm, the pooled estimates for sensitivities and specificities were 87% (95% CI: 83–90%) and 95% (95% CI: 92–97%) in nine studies, respectively. CCE2 is demonstrated to be an effective and safe method in the setting of CRC screening. The diagnostic performance of CCE2 was similar for polyps measuring at least 6 and 10 mm. Sulbaran et al. 48 conducted a systematic review of CCE2 accuracy for CRC screening. CCE2 is demonstrated to be an effective and safe method in the setting of CRC screening. The diagnostic performance of CCE2 was similar for polyps measuring at least 6 and 10 mm. Likelihood ratios suggest that CCE2 may substantially impact screening, adequately filtering patients who would benefit the most from colonoscopy.
CCE is also currently being evaluated for polyp detection utilizing Artificial intelligence to enhance pathways for utility (Table 2)55,56:
Available meta-analysis and systematic review on the performance of CCE in CRC screening from a methodological perspective.

CCE, colon capsule endoscopy; CR, completion rate; DA, diagnostic accuracy; DY, diagnostic yield; MA, meta-analysis; PDR, polyp detection rate; SR, systematic review.
Use of CCE for UC
Ulcerative colitis (UC) is a chronic, inflammatory disease of the colonic mucosa characterized by a relapsing–remitting course. As CCE2 is relatively non-invasive and does not involve direct trauma to the mucosa or inhalation of air, it offers several benefits for patients with UC for assessing mucosal inflammation. 57
In a study conducted by San Juan-Acosta et al. 58 the feasibility of CCE 1 and 2 in 42 patients with UC were examined, bowel preparation was considered adequate in 80% of the patients, and no serious adverse events related to the CCE procedure, or bowel preparation were reported. The correlation between OC disease severity and that estimated by the CCE, as well as the estimation of disease extent by the two modalities, were substantial (κ = 0.79; 95% CI: 0.62–0.96) and (κ = 0.71; 95% CI: 0.52–0.90), respectively. Forty patients with histologically confirmed diagnoses of UC were enrolled in the study led by Hosoe et al. 59 CCE2 procedure was completed within 8 h in 69% of the patients. Matts endoscopic scores determined by CCE2 showed a strong correlation with scores obtained by conventional colonoscopy (average ρ = 0.797). A total of 26 with confirmed UC were enrolled in a prospective single-centre study by Ye et al. 60 There was a significant correlation in the severity (κ = 0.751, p < 0.001) and extent (κ = 0.522, p < 0.001) of UC between the CCE and conventional colonoscopy. In addition, the excellent to good rate of colonic cleanliness in CCE was 80%. There were no remarkable adverse events during the study. CCE provides notable performance in the detection of the severity and extent of active UC.
Table 3 compares the performance of CCE2 and colonoscopy in assessing UC severity.
Performance of CCE2 for assessing disease severity in UC as compared to colonoscopy.

CCE, colon capsule endoscopy; UC, ulcerative colitis; CSUC, capsule scoring of UC; UCEIS, endoscopic index of severity.
Comparison with CTC
Based on the recent consensus of the ESGE and the European Society of GI and Abdominal Radiology (ESGAR) guideline, CTC is the most appropriate radiological examination for patients with colorectal neoplasia who cannot undergo endoscopy or have an incomplete colonoscopy. 66 Several studies have compared colon capsules and CTC so far (Table 4). The VICOCA study 67 was a prospective, single-centre, randomized trial conducted from March 2014 to May 2016; out of 349 individuals, 290 individuals agreed to participate: 147 in the CCE group and 143 in the CTC group. Regarding detecting significant neoplastic lesions, the sensitivity of CCE and CTC was 96.1% and 79.3%, respectively. The detection rate for advanced colorectal neoplasm was higher in the CCE group than in the CTC group (100% and 93.1%, respectively; relative risk (RR) = 1.07; p = 0.08). Although both techniques seem similar in detecting patients with advanced colorectal neoplasms, CCE is more sensitive for detecting any neoplastic lesion. In another multicentre comparison study led by Cash et al. 68 on the DY of CCE versus CTC in a screening population, out of 320 enrolled, data from 286 (89.4%) subjects were evaluated. CCE was superior to CTC in detecting polyps ⩾6 mm and non-inferior for polyps ⩾10 mm.
Comparison studies on the diagnostic yield of CCE versus CTC.

CCE, colon capsule endoscopy; CTC, CT colonography; gFOBT, guaiac-based fecal occult blood testing; OC, optical colonoscopy; gFOBT, guaiac faecal occult blood test.
CE innovation and clinical translation
Certain developments in other countries known to have led (and keep showing) CE innovation and clinical translation, such as Scotland and England, should be highlighted:
Scottish Capsule Programme
In Scotland, following a prolonged period of review and discussions with NHS Scotland, CCE is being implemented as part of the Scottish Capsule Programme (SCOTCAP), 73 which has been designed to address demand and capacity pressures within the NHS in Scotland. Piloting of full clinical service based on CCE started in June 2019 (ScotCap first phase, Highlands and NHS Islands of Scotland between NHS Highlands and Corporate Health International UK). Since then, the service has been fully available to all boards across Scotland. As part of the SCOTCAP trial, symptomatic and surveillance patients were recruited from multiple sites across Scotland. Detailed results of the study are provided below. Overall, it indicated that CCE could reduce the number of patients requiring a colonoscopy by using a safe, well-tolerated diagnostic test (Tables 5–7).
Scottish Capsule Programme (SCOTCAP). 73

CCE, colon capsule endoscopy.
Outcome of SCOTCAP.

CCE, colon capsule endoscopy; CTC, CT colonography; SCOTCAP, Scottish Capsule Programme.
Outcome of SCOTCAP in those required further test.

SCOTCAP, Scottish Capsule Programme.
NHS England
To mitigate pressures on endoscopy services, the British Society of Gastroenterology Endoscopy Committee set up a COVID-19 Research Group to consider potential roles for novel alternative diagnostic technologies and NHS England has provided £6 million to deliver CCE services and committed to a pilot scheme of 11,000 capsules via hospital-based delivery. 74
Green endoscopy: Our practice and its carbon footprint
Healthcare systems worldwide are making significant commitments to reduce carbon emissions and achieve sustainability goals. For instance, the United Kingdom’s NHS has pledged to achieve a net-zero carbon footprint for its direct emissions by 2040 and extend this to include its supply chain by 2045. 75 Similar commitments have been made by other healthcare systems during the COP26 summit. 76
Within healthcare, gastrointestinal endoscopy is recognized as a specialty that has a substantial environmental impact. This is attributed to factors such as high caseloads, patient travel, generation of non-renewable waste, use of single-use consumables and resource-intensive decontamination processes. While a comprehensive assessment of the carbon footprint specifically in endoscopy is yet to be conducted, estimations indicate a significant environmental burden. In the United States alone, it is approximated that endoscopy contributes around 86,000 tons of CO2 equivalent emissions annually. Furthermore, the specialty generates considerable waste, with each endoscopy bed day estimated to produce approximately 3 kg of waste, resulting in 13,500 tons of plastic waste annually in the United States. 77
To address these environmental concerns, exploring home-based and non-invasive alternatives and diagnostic tools like faecal calprotectin and FITs is being considered. These approaches aim to enhance the efficacy of invasive investigations while reducing the need for invasive procedures like colonoscopy. 78 One potential alternative highlighted is CCE, which has the potential to decrease the number of symptomatic patients requiring colonoscopy significantly.
Cost-effectiveness of CCE
Having put all this info on one side, the question of the price of the service and cost-efficiency arises. The European standard recommends a participation rate of 45% for the nationwide CRC screening program to be cost-effective, yet the latest published rate in France was 34%. 79 In the French study conducted by Barré et al., 80 eight screening strategies were compared, based either on a screening test (guaiac or FIT testing, blood-based, stool DNA, CTC, colon capsules and sigmoidoscopy) followed by complete colonoscopy if positive or direct colonoscopy. FIT every 2 years appears to be the most cost-effective CRC screening strategy considering a real-world participation rate of 34%. CCE use for CRC screening may be cost-effective if compliance with screening programs is increased through this technology, and if long-term downstream costs are considered. Another report from as early as 2008 (Hassan et al. 81 ) estimated that the cumulative cost of the CCE, including the reading process, was $950 (2008 prices). The exercise aimed to calculate the cost-effectiveness of CCE in CRC screening. When equal compliance was simulated, the colonoscopy program was more effective and less costly than a strategy based on capsule endoscopy. It is already known from previous reports in the field that the cost-effectiveness of CCE for use in patients referred for CTC is $26,750 per life-year, assuming an increased sensitivity of CCE. Replacement of CTC with CCE is associated with moderate costs to the health care system. In conclusion, the cost-effectiveness of capsule endoscopy depends mainly on its ability to improve compliance with CRC screening. However, looking at reports from France, one must remain cautious about the potential of achieving this (Table 8).
Cost Effectiveness results. 80

CCE, colon capsule endoscopy; CRC, colorectal cancer; CTC, CT colonography; FIT, faecal immunochemical test; gFOBT, guaiac-based fecal occult blood testing.
Transitioning to CCE in colonoscopy-centric environments: Adoption challenges
The incorporation of CCE into screening/surveillance algorithms by a medical community that earns income from colonoscopy procedures faces various obstacles. One primary challenge is the resistance to change within the medical field, as traditional colonoscopy has long been the established method for identifying colorectal abnormalities. 82 Concerns about the accuracy and reliability of CCE compared to conventional methods also contribute to the slow adoption. Financial considerations play a significant role as well. The revenue generated from colonoscopy procedures creates hesitancy to shift to CCE, potentially reducing the demand for traditional colonoscopy. The financial implications, coupled with the costs of implementing CCE technology and training healthcare professionals, create resistance within the community reliant on colonoscopy for income. The absence of standardized guidelines and regulations for CCE implementation further hampers its adoption. Without clear guidelines and established reimbursement structures, healthcare providers are hesitant to incorporate CCE into their practices, fearing financial losses and legal complications.
Overall, the slow adoption of CCE in screening/surveillance algorithms can be attributed to resistance towards change, concerns about accuracy, financial factors and the absence of standardized guidelines. However, as research continues to support the effectiveness and cost-efficiency of CCE, there is potential for gradual acceptance of this advanced technology in the times ahead.
The layer cake theory
For successful clinical implementation, we need to draw all possible conclusions from:
(a) Colonoscopy referral and outcome registries (local)
(b) The information from the SCOTCAP and English experience
(c) Existing literature review from national (or international guidelines) and relevant meta-analyses with limited real-world data.
With the above measures and more data available, CCE will be suitable for widespread clinical implementation. Increasing CRs and reducing re-investigations through a better patient selection process can be instrumental to CCE’s clinical implementation. CCE could offer alternatives to in-hospital care to reduce long waiting lists for colonoscopy and move away from hospital (Figure 4).
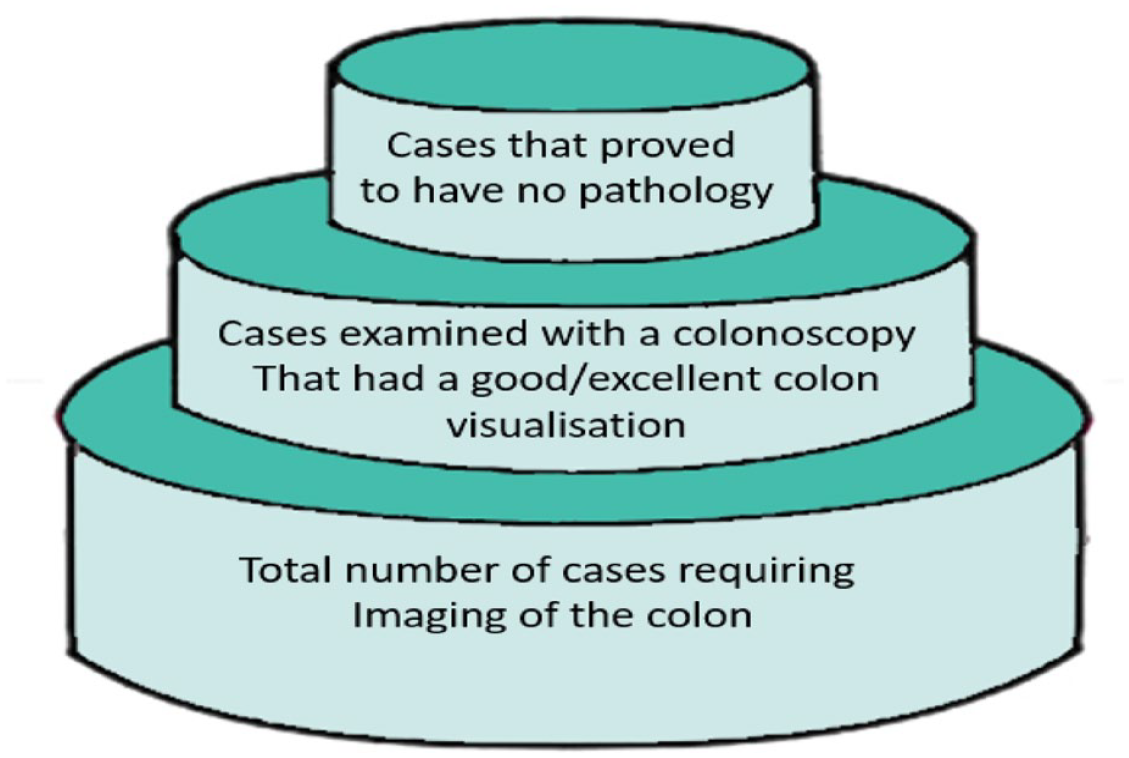
The layer cake theory.
