Abstract
Background:
A growing body of evidence underscores the beneficial impact of therapeutic drug monitoring (TDM) on the efficacy and cost-effectiveness of anti-tumour necrosis factor (TNF) therapy in patients with inflammatory bowel disease (IBD).
Objectives:
We surveyed clinician attitudes, perceptions and barriers related to TDM in IBD in the Middle East.
Design:
A 15-question survey was distributed through national gastroenterological societies in five Middle Eastern countries (UAE, Saudi Arabia, Kuwait, Lebanon and Egypt).
Methods:
Data on clinician characteristics, demographics, utilization patterns and obstacles related to the adoption of TDM with anti-TNFs were gathered. Logistic regression analysis was used to predict factors influencing the utilization of TDM.
Results:
Among 211 respondents (82% male), 82% were consultants, 8% were physicians with an interest in gastroenterology (GI), and 6% were GI trainees. Of these, 152 met inclusion criteria, treating >5 IBD patients per month and ⩾1 with an anti-TNF per month. TDM was used in clinical practice by 78% (95% CI: 71–85) of respondents. TDM was utilized following the loss of response (LOR) in 93%, for primary non-response (PNR) in 40% and before restarting anti-TNF therapy after a drug holiday in 33% of respondents, while 34% used TDM proactively. No specific factors were associated with the use of TDM. Barriers to TDM use included cost (85%), time lag to results (71%) and lack of insurance reimbursement (65%). Overall knowledge of TDM (70%), interpretation and actioning of results (76%) or awareness of clinical guidelines (57%) were not perceived as barriers. If barriers were removed, 95% would use TDM more frequently; 93% for LOR, 60% for PNR, 50% when restarting after a drug holiday, and 54% would use TDM proactively.
Conclusion:
Most gastroenterologists use TDM for LOR, with cost, time lag and insurance reimbursement being significant barriers. Addressing these barriers would increase the judicious use of reactive and proactive TDM to optimize anti-TNF therapy in IBD.
Plain language summary
Anti-TNF therapies are perhaps the most widely used and available biological therapies for the treatment of inflammatory bowel disease globally even though other agents have been licensed in recent years. The role of therapeutic drug monitoring to optimise outcomes and mitigate against immunogenicity with anti-TNF agents are now being appreciated. Our study investigates clinician attitudes, perceptions, and barriers related to therapeutic drug monitoring (TDM) in the context of anti-tumor necrosis factor (TNF) therapy for inflammatory bowel disease (IBD) through a comprehensive survey distributed from five Middle Eastern countries. Among 211 respondents (82% male), 82% were consultants, 8% physicians with an interest in gastroenterology (GI), and 6% GI trainees. TDM was utilised following loss of response (LOR) in 93%, for primary non-response (PNR) in 40%, and before restarting anti-TNF therapy after a drug holiday by 33% of respondents, while 34% used TDM proactively. No specific factors were associated with the use of TDM. Barriers to TDM use included cost (85%), time lag to result (71%), and lack of insurance reimbursement (65%). Overall knowledge of TDM (70%), interpretation and actioning of results (76%), or awareness of clinical guidelines (57%) were not perceived as barriers. If barriers were removed, 95% would use TDM more frequently; 93% for LOR, 60% for PNR, 50% when restarting after a drug holiday and 54% would use TDM proactively. Most gastroenterologists use TDM for LOR, with cost, time lag, and insurance reimbursement being significant barriers. Addressing these barriers would increase judicious use of reactive and proactive TDM to optimise anti-TNF therapy in IBD.
Introduction
The advent of anti-tumour necrosis factor (TNF) therapies has revolutionized the management of inflammatory bowel disease (IBD), enabling better control of immune-related tissue damage and management of long-term sequelae. Evolving paradigms with disease control recognize the importance of achieving mucosal healing and deep remission when possible, and demonstrable effects on reducing corticosteroid use, hospitalization and surgery for IBD.1–3 The concept of ‘treating to target’ developed by the Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE) Committee is widely accepted as the gold standard of care.1,4–6 It emphasizes the composite assessment of clinical symptoms, patient-relevant outcomes and assessment of endoscopic activity aiming for endoscopic remission supported by biomarkers of disease activity. Evolution in goals notwithstanding, our therapeutic armamentarium of biologic and small molecule therapies, (although increasing), is still arguably limited, with cost and wider access to advanced therapies posing unique challenges globally; yet emphasizing the need to select and optimize therapy wisely. Anti-TNF therapies are often recommended as first-line advanced therapy by international guidelines and somewhat reassuringly, the recent approval of biosimilar infliximab (IFX) and adalimumab, at significantly lower cost and comparable immunogenicity, efficacy and safety has improved access to this highly effective therapy.7–12 However, anti-TNF therapies pose clinical challenges.
Primary non-response (PNR) affects up to one-third of IBD patients, while secondary loss of response (SLR) occurs in around 50% of initial responders after 12 months, with another 20% experiencing loss of response (LOR) annually thereafter.13–15 This LOR may be due to low drug levels from immune (anti-drug antibodies) or non-immune clearance mechanisms. Furthermore, it is also well known that after the failure of the first biological treatment, the use of subsequent therapies typically demonstrates the ‘law of diminishing returns’ with successive therapies often being less effective.16–18
Therapeutic drug monitoring (TDM) involves measuring serum trough concentrations and anti-drug antibodies. It has been defined as ‘drug concentration measurement with adjustment of the dose and/or dosing intervals to achieve and maintain serum concentration within a certain therapeutic range to optimize treatment outcomes’.9,19,20 TDM can be either reactive or proactive. In reactive TDM, levels are checked in response to suspected active disease and a dose adjustment is made in response to drug levels.19,21–28 Proactive TDM involves checking serum trough concentrations at predetermined time points regardless of disease activity to prevent sustained low levels leading to a flare or de-escalate therapy in response to supratherapeutic levels.19,29–32 Reactive TDM is supported by international guidelines and widely adopted in routine practice.7–9,19–21 Meanwhile, evidence for proactive TDM to prevent LOR due to low drug levels or de-escalation of combination therapy is growing.19,28–32 A recent Delphi consensus from the UAE supports both reactive and proactive TDM, at the end of induction and at least once during maintenance for responders or when it may influence treatment decisions. 8
The introduction of biosimilars has significantly reduced the cost of anti-TNF therapy, making TDM-based dose optimization a cost-effective option.33,34 Despite the increasing range of therapeutic options for IBD, the progressive nature of the disease, declining response rates associated with multiple drugs and disease duration, underscore the ongoing economic and clinical value of optimization.35,36
Recent studies from the United States, United Kingdom, India and New Zealand have evaluated attitudes, perceptions and barriers to TDM use with anti-TNF therapy.37–40 However, clinician approaches may differ based on access to biologics and healthcare systems in different nations or regions. In the Middle East, where IBD prevalence is increasing, no data on TDM practices are currently available.7,8
We surveyed TDM use in anti-TNF therapy in five participating nations in the Middle East. Our primary aim was to assess factors associated with TDM use (clinician and clinical setting) and identify barriers to its implementation. Our secondary aim was to explore how clinicians would utilize TDM if all barriers were eliminated.
Methods
Study design
A 15-question survey (see Supplemental Appendix 1) was adapted from similar studies conducted in the United Kingdom and India.37,38 Eminent gastroenterologists from five Middle Eastern countries (UAE, Saudi Arabia, Kuwait, Lebanon and Egypt) were approached to be the local leads in their countries and obtain necessary approvals. The approved questionnaire was placed on an online survey tool, and invitations were sent to Consultants and Higher Specialist trainees (Registrar/Fellow) through their membership in national gastroenterology societies between March and August 2021. The invitation included details on the time required for completion and background information, along with a link to the survey (see Supplemental Materials).
Demographic information collected from participants included age, sex, grades (consultant, gastroenterology trainee/registrar, physicians with special interest in GI), number of years in practice since specialist qualification or gastroenterology accreditation (as applicable) and place of work (government hospital, private hospital, private clinic or private individual practice). Additionally, data on the proportion of IBD patients seen in their clinical practice, number of patients with IBD treated personally in a 1-month period and numbers treated with anti-TNF therapy per month were obtained. The participants were asked to indicate their level of agreement or disagreement with potential barriers to using TDM using a Likert five-point scale. Participants treating <5 IBD patients per month and/or having no patients on anti-TNF therapy every month were excluded. The study’s reporting adheres to the Checklist for Reporting Results of Internet E-Surveys (CHERRIES) statement (see Supplemental Appendix 2). 41
Statistical analysis
The data were analysed using R Software Version 3.5.2 (R Foundation for statistical computing,Vienna, Austria). Categorical variables were expressed as frequencies and percentages. Univariate logistic regressions were used to examine associations between available variables and the outcomes of interest (use of TDM and proactive TDM). Associations were reported as p-values and odds ratios with 95% confidence intervals. To determine the independent effects of variables associated with the use of TDM and proactive TDM, a multiple binary logistic regression analysis was planned, including variables with a p-value of <0.1 from the univariate analysis.
Results
Responses were received from 211 participants, of which 152 met inclusion criteria (59 clinicians reported treating less than 5 IBD patients per month and/or having no patients on anti-TNF therapy every month and were therefore excluded from further analysis looking at perspectives on TDM use). Participant inclusion and flow are represented as a flow diagram in Figure 1 and the baseline characteristics of the participants and details are included in Table 1.

Flow diagram showing the inclusion of participants for further analysis.
Eligible participants’ demographic and clinical characteristics.
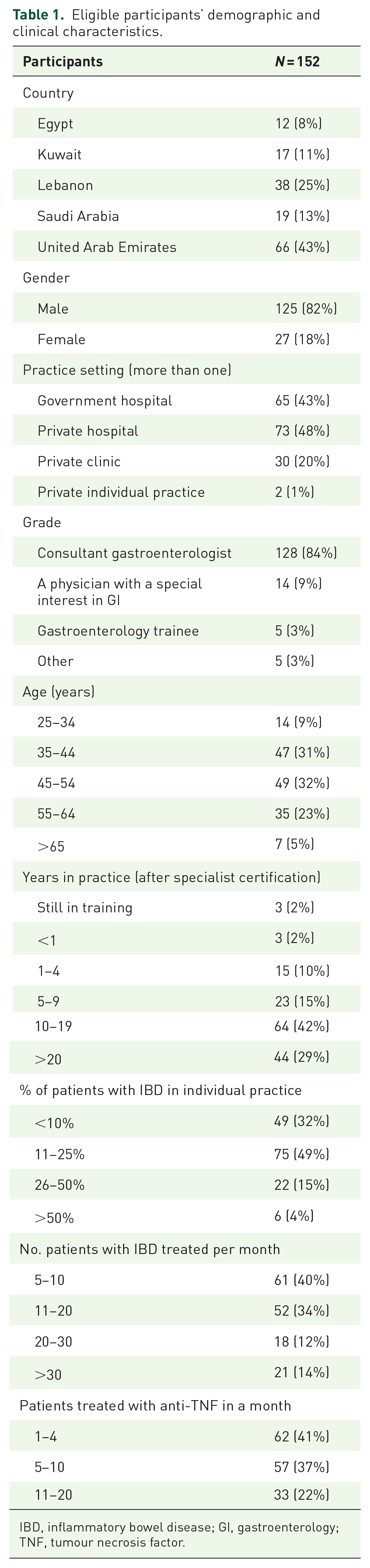
IBD, inflammatory bowel disease; GI, gastroenterology; TNF, tumour necrosis factor.
Practice of TDM
TDM was utilized in clinical practice by 78% (n = 119: Egypt – 6; Kuwait – 15; Lebanon – 28; Saudi Arabia – 13; UAE – 57) of respondents. Of these, 93% (n = 111) used TDM for SLR; 40% (n = 48) for PNR; 33% (n = 39) used it before restarting anti-TNF therapy after a drug holiday; and 34% (n = 41) used TDM proactively (Figure 2). No specific factors were found to be associated with the routine use of TDM (see Table 2). Clinicians using anti-TNF for an average of 11–20 patients per month had 1.26 times higher odds (95% CI: 1.07–1.47) of using proactive TDM compared to clinicians treating fewer patients with anti-TNFs (see Table 3).

Current use of TDM: pooled data for all countries and country-wise data.
Univariate logistic regression analysis of variables associated with the use of therapeutic drug monitoring.
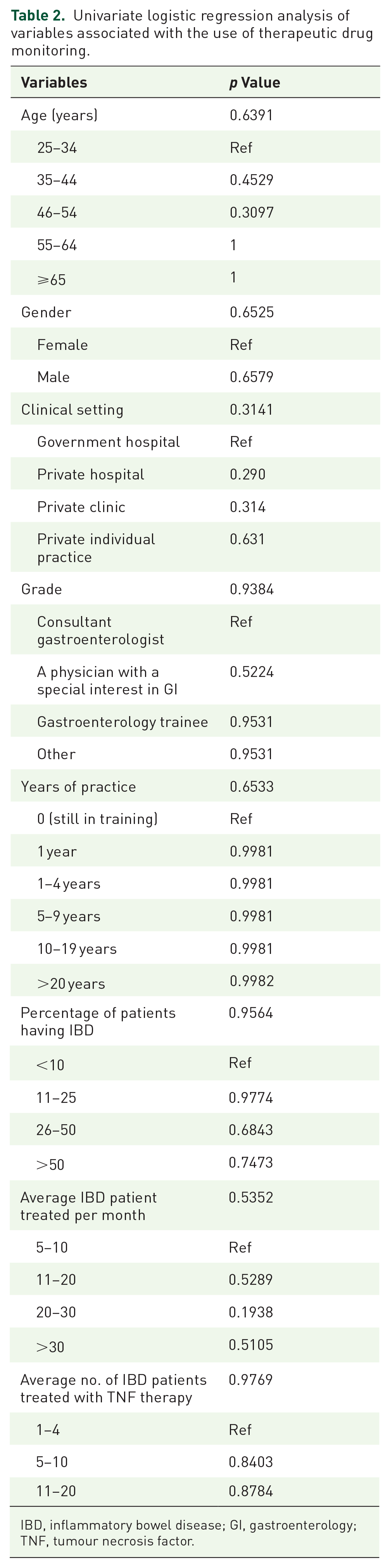
IBD, inflammatory bowel disease; GI, gastroenterology; TNF, tumour necrosis factor.
Univariate logistic regression analysis of variables associated with the use of proactive therapeutic drug monitoring.
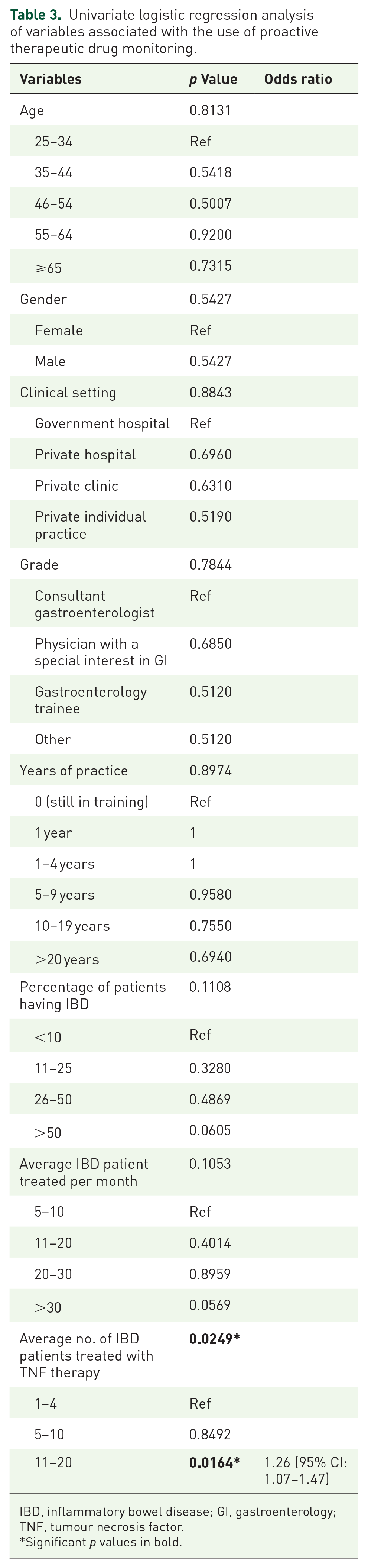
IBD, inflammatory bowel disease; GI, gastroenterology; TNF, tumour necrosis factor.
Significant p values in bold.
The main barriers to TDM use reported by the respondents were cost (85%); time lag in receiving results (72%), and not being reimbursed by insurance (65%). Respondents mostly disagreed or strongly disagreed that uncertainty about availability (43%), lack of overall knowledge of TDM (70%), lack of knowledge regarding how to interpret and what to do with results of TDM (76%), TDM is cumbersome and/or time-consuming (45%), perceived lack of an evidence base for TDM use (66%) and lack of awareness of clinical guidelines (57%) are barriers to its use (Figure 3).

Barriers to TDM: the test is expensive (a); uncertainty about availability in my practice (b); lack of overall knowledge of TDM (c); lack of knowledge on how to interpret and what to do with the results of TDM (d); time lag from serum sampling to results of TDM (e); TDM is cumbersome and/or time-consuming (f); lack of good evidence-based medicine of the usefulness of TDM in IBD (g); lack of clinical guidelines recommending the use of TDM (h); not reimbursed by insurance (i).
In the absence of any barriers to TDM use, 145 out of the 152 respondents expressed a willingness to use TDM more frequently. Among this group, 93% would opt for TDM for SLR, 60% for PNR, 50% when resuming treatment after a drug holiday and 54% would proactively check it (see Figure 4). Specifically, 63% of these respondents would perform proactive TDM at least once a year, while the remaining 37% would rely on clinical judgement for periodic assessments if all barriers were eliminated.
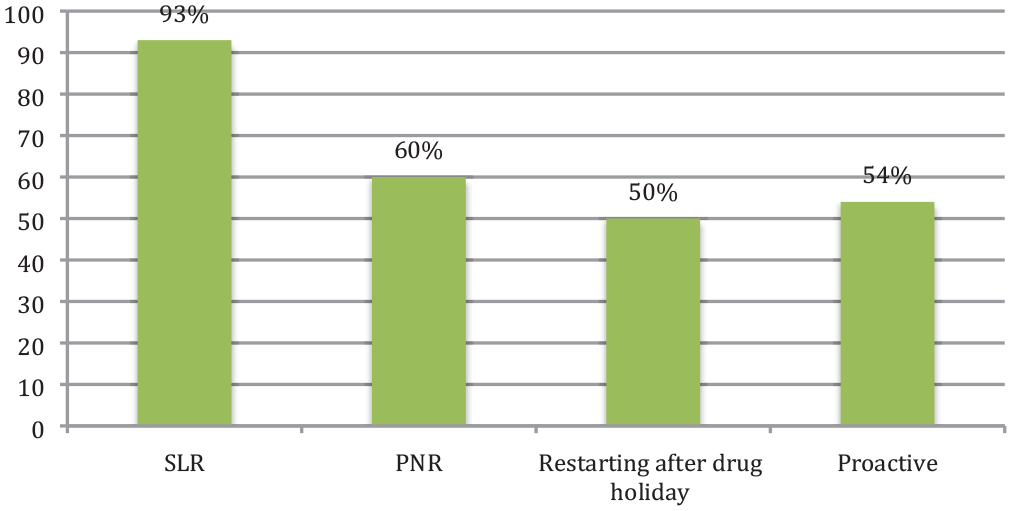
If barriers to TDM are removed.
Discussion
This is the first survey of attitudes and barriers to TDM use from the Middle East. The majority (78% of our respondents) reported utilizing TDM for anti-TNF therapy in IBD. Although slightly lower compared to studies from the United States, United Kingdom and New Zealand, which found 90%, 97% and 93% TDM use, respectively, only 20% of respondents in an Indian survey utilized TDM.37–40
Among those who used TDM, 93% used it ‘reactively’ for SLR, and 40% used it for PNR. Similar variations in figures for indication of use were found in other countries (96% and 72% for SLR and PNR in the United Kingdom, 87% and 66% in the United States, and 89% and 74% in India, respectively). The study in New Zealand reported that 87% of the participants performed TDM for PNR and/or LOR.37–40
Unlike other studies, no specific factors were found to be associated with the use of TDM in the Middle East. In the United Kingdom, clinicians with a larger volume of IBD patients (>50% of their practice), working in a teaching hospital and practicing for >20 years were independent factors associated with TDM use. Similarly, in the United States, clinicians with a larger anti-TNF cohort demonstrated increased usage. In India, having between 11% and 25% of a practice made up of IBD patients and seeing/treating a higher number of IBD patients/month were associated with TDM usage.37,38,40
In our present study (from the Middle East), most clinicians had a relatively smaller proportion of IBD patients in their practice (49% had 11–25% and 32% had <10% of their practice consisting of IBD patients). Similarly, in India, 76% had 11–25% of their practice consisting of IBD patients and 23% had <10%. Conversely, in the United Kingdom, many clinicians had much larger proportions of IBD patients in clinics with 45% of clinicians having >50% of their clinics comprising IBD patients.37,38
In India and the Middle East, the clinic setting is broadly differentiated by government versus private hospital-based practice, whereas in the United Kingdom, there are district general and teaching hospitals, the latter being more likely to have specialist IBD clinics and are often located in larger cities. In the United Kingdom study, clinicians working in teaching hospitals favoured TDM. 37 In India, smaller (tier 2) cities demonstrated higher uptake. This suggests that in the United Kingdom, patients in teaching hospitals, where clinicians have higher exposure, are more likely to access TDM to assess and optimize anti-TNF therapy. Whereas in India, clinicians may be inclined to more frequent follow-up and TDM use when working in more manageable environments (lower overall population of patients and lower burden of IBD). 38 There were no such factors identified in the Middle East which may suggest a more standardized approach across all clinical settings. It will be interesting to note how this practice evolves with the rapid rise in the incidence of IBD in the Indian subcontinent and the Middle East. 42
A third (34%) of participants in the present study reported proactive TDM use as compared to 37% in the United States and 54% in the United Kingdom, respectively.37,40 By contrast, India and New Zealand had lower rates of proactive TDM use, with 5% and 13%, respectively.37–40 While the UAE guidelines, a recent UAE Delphi consensus on TDM in IBD and other international guidelines from Europe, the United Kingdom and the United States recommend reactive TDM, there is increasing evidence to support proactive drug monitoring.7–12,29,31,32,43 Post hoc analysis of randomized controlled trials (RCTs) and exposure–outcome relationship data from proactive studies demonstrate higher post-induction and maintenance anti-TNF drug levels are associated with more favourable therapeutic outcomes.44–46 In the trough concentration adapted infliximab treatment (TAXIT) RCT, proactive TDM was linked to less frequent occurrences of undetectable IFX concentrations and a reduced risk of relapse. 29 Proactive TDM is asserting its relevance in induction for more severely active patients with higher drug clearance, to de-escalate the dose in well-selected patients in deep remission and as an alternative to combination therapy with an immunomodulator when clinically appropriate.29,47–51
We identified the main barriers to TDM which were cost (85%), the time lag in receiving results (72%) and lack of insurance reimbursement (65%). Interestingly, similar barriers of time lag and cost were identified in the United Kingdom, United States and India. However, there were variations in perceptions regarding the significance of clinical guidelines as a barrier. While the 2020 UK survey highlighted the absence of clinical guidelines as a concern, this was not echoed in the Indian survey.37,38,40 By contrast, in the Middle East survey, respondents disagreed or strongly disagreed that clinical guidelines (57%), lack of overall knowledge (70%) and perceived lack of evidence (66%) were barriers to TDM use. It is worth noting that although TDM is now integrated into numerous international guidelines, it still represents a relatively recent addition to the standard of care.7,12
Although drug monitoring and potential dose increase may be unappealing to insurance companies, 52 TDM is cost-effective by reducing the time patients spend on ineffectual treatment.53–55 Furthermore, it is now well established that switching from one to another biologic may be associated with attrition in response to subsequent agents. 56 At present, several factors contribute to the lag time for TDM. Dosing schedules and lab turnover can result in a delay of weeks before a drug can be optimized. Point-of-care testing and dashboard-driven prediction models may address this barrier.19,57
Interestingly, although most clinicians were already utilizing TDM for SLR, the removal of barriers would lead virtually all of them to use it. However, despite the removal of barriers, a sizeable (40%) proportion of respondents would still not employ TDM for PNR. The First UAE IBD consensus guidelines, along with many other international guidelines, recommend that LOR should first be managed by dose optimization guided by the measurement of serum levels.7–12 Evidence demonstrates that drug optimization can be successful with both PNR and SLR.15,20,43,58
A major strength of our study is the involvement of respondents from multiple Middle Eastern countries, providing a broader perspective on TDM utilization and barriers in the region. The survey was adapted from studies conducted in the United Kingdom and India, with inputs from eminent gastroenterologists from each participating country in the Middle East involved as local leads.37,38 This ensures that cultural or contextual differences specific to the Middle East have been addressed in the questionnaire. The study also offers valuable comparisons with TDM practices and barriers reported in the United Kingdom, United States and India, contributing to a broader understanding of the subject.
We acknowledge some limitations of our work. Despite the involvement of a wide spectrum of clinicians, we recognize the possibility of selection bias that occurs in most survey-based studies. Participants who chose to respond might have different attitudes or experiences compared to non-participants. While the study covered five Middle East countries, we were unable to include the entire region. Therefore, the findings might not fully represent the entire Middle East population. Furthermore, the exclusion of participants treating fewer than five IBD patients per month and those not using anti-TNF therapy may limit the generalizability of the findings to all gastroenterologists in the region. Additionally, the study was conducted in a specific timeframe (March to August 2021), and we recognize that the practice of IBD as indeed TDM will evolve and we hope, to improve following this study with improved understanding and wider acceptance.
In conclusion, we found that while TDM is widely used by most clinicians in the Middle East for SLR, its adoption for PNR is comparatively lower. Significant barriers, such as cost, time lag and lack of insurance reimbursement, hinder widespread implementation of TDM. If these barriers were eliminated, more clinicians would likely adopt TDM; yet, a considerable proportion may still refrain from using it post-drug holidays and proactively. The potential integration of point-of-care testing and lower-cost assays could persuade clinicians to use TDM more often and in varied scenarios as described. Our study does highlight a need for improved adherence to international guidelines, suggesting the importance of educational initiatives and broader dissemination of guidelines to increase awareness. With the prevalence of IBD on the rise in the Middle East, optimizing the use of anti-TNF therapies through personalized dosing based on patient metabolism and disease will prove crucial to enhance outcomes and cost-effectiveness for both healthcare institutions and individuals living with IBD.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848241230902 – Supplemental material for Attitudes, perceptions and barriers in implementing therapeutic drug monitoring for anti-TNFs in inflammatory bowel disease: a survey from the Middle East
Supplemental material, sj-pdf-1-tag-10.1177_17562848241230902 for Attitudes, perceptions and barriers in implementing therapeutic drug monitoring for anti-TNFs in inflammatory bowel disease: a survey from the Middle East by Gaurav B. Nigam, Kelly Chatten, Ala Sharara, Talal Al-Taweel, Othman Alharbi, Hussein Elamin, Sameer Al Awadhi, Vito Annese and Jimmy K. Limdi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pdf-2-tag-10.1177_17562848241230902 – Supplemental material for Attitudes, perceptions and barriers in implementing therapeutic drug monitoring for anti-TNFs in inflammatory bowel disease: a survey from the Middle East
Supplemental material, sj-pdf-2-tag-10.1177_17562848241230902 for Attitudes, perceptions and barriers in implementing therapeutic drug monitoring for anti-TNFs in inflammatory bowel disease: a survey from the Middle East by Gaurav B. Nigam, Kelly Chatten, Ala Sharara, Talal Al-Taweel, Othman Alharbi, Hussein Elamin, Sameer Al Awadhi, Vito Annese and Jimmy K. Limdi in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
