Abstract
Background:
Patients with inflammatory bowel disease (IBD) have an attenuated serologic response to COVID-19 vaccination. It is unclear whether an impaired immune response in vaccinated IBD patients impacts the susceptibility to SARS-CoV-2 infection and occurrence of severe COVID-19.
Objectives:
To evaluate SARS-CoV-2 breakthrough infection rates and the disease course of COVID-19 in vaccinated IBD patients.
Design:
A systematic literature search and meta-analysis was performed.
Data sources and methods:
The search was performed in Embase, Medline, Web of Science Core Collection, Cochrane Central Register of Controlled Trials and CINAHIL. The articles were independently screened and selected by two reviewers. A random-effects model was used to calculate the pooled relative risk for breakthrough infections in vaccinated IBD patients and controls.
Results:
A total of 16 studies were included, with study periods ranging from January 2020 to October 2021 and follow-up time from 3 weeks to 6 months. The breakthrough infection rates range from 0 to 37.4% in vaccinated IBD patients. The disease course of COVID-19 was generally mild, with low hospitalization and mortality rates (0–8.7% and 0–4.3%, respectively). Vaccinated IBD patients had a significantly lower relative risk of breakthrough infection rate compared to unvaccinated controls (risk ratio: 0.07, 95% CI: 0.03–0.18). No difference was observed between IBD patients and non-IBD controls, and between partially and fully vaccinated IBD patients. The impact of immunosuppressive therapy on breakthrough infection rates differs between studies. Most studies showed no impact from immunosuppressive treatment, anti-tumour necrosis factor alpha or corticosteroids and other biologics; one study reported higher rates for patients treated with infliximab versus vedolizumab.
Conclusion:
Vaccination is effective to prevent COVID-19 infections in patients with IBD. Breakthrough infections do occur, but the disease course is generally mild. Available data seem to suggest a declining trend of breakthrough infections during calendar time.
Registration:
The protocol was published in the PROSPERO database (CRD42021292853).
Introduction
Since the start of the coronavirus disease (COVID-19) pandemic caused by SARS-CoV-2 infection, multiple vaccination programmes with both messenger RNA (mRNA) and viral vector vaccines were implemented. Initial concerns on an increased risk of COVID-19 infections and a more severe course for patients with inflammatory bowel disease (IBD) have been refuted.1–3 The overall incidence as well as the incidence of severe COVID-19 infections in patients with IBD is comparable to the general population.1–3
Risk factors for severe COVID-19 in IBD patients comprise a higher age, comorbidity and corticosteroid use. No association between severe COVID-19 and IBD medication, including biologicals, has been observed. 4 On the contrary, a beneficial effect of anti-inflammatory drugs has been suggested. Hypothetically these drugs protect patients from a cytokine storm associated with COVID-19, thereby reducing the expression of angiotensin-converting enzyme 2 receptors necessary for viral entry of COVID-19, which influences both the incidence and severity of COVID-19. 5 Contradictory to this hypothesis, the serologic response to infection was shown to be attenuated in patients with anti-tumour necrosis factor alpha (anti-TNF-α) treatment when compared with vedolizumab .6,7
An impaired immune response following hepatitis A and B, pneumococcal and influenza vaccinations was demonstrated in IBD patients on immunosuppressive therapy.8–11 Given a reduced serologic response following COVID-19 infection, a reduced vaccination response following COVID-19 vaccination might be expected. A recent meta-analysis demonstrated that vaccinated IBD patients had slightly lower seroconversion rates after COVID-19 vaccination than healthy controls. Most importantly, a fast decay in antibody response to vaccination after 4 weeks was demonstrated, which was most pronounced in patients treated with anti-TNF-α, immunomodulators or combination therapy. 12
Although an impaired immune response has been demonstrated following COVID-19 vaccination in IBD patients, it is unclear whether this impacts the rate of (severe) SARS-CoV-2 infections. In this systematic review and meta-analysis, we aim to assess the rate of (severe) SARS-CoV-2 breakthrough infections after vaccination for patients with IBD. In addition, we describe the course of the breakthrough infections after COVID-19 vaccination and evaluate epidemiological and disease-specific risk factors.
Methods
Search strategy
This systematic review and meta-analysis was conducted according to the Cochrane Handbook for Systematic Reviews of Interventions, 13 in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines. The protocol for this systematic review was published at PROSPERO (CRD42021292853). An expert librarian conducted a literature search on 11 March 2022 in multiple databases such as Embase, Medline, Web of Science Core Collection, Cochrane Central Register of Controlled Trials and CINAHIL (Supplemental Table 1). No restrictions in time, geography or language were made. References in the eligible articles were cross-checked by hand for additional literature.
Study selection
Two authors (NP and LB) independently screened all titles and abstracts. Subsequently, full-text articles (if available) were read for definitive study inclusion. Articles were selected according to the inclusion criteria: (a) observational cohort studies, (b) patients with IBD, (c) vaccination with COVID-19 vaccine, including mRNA and viral vector vaccine, and (d) reported SARS-CoV-2 breakthrough infection rates or disease course. Abstracts which met the inclusion criteria were included. Case reports and systematic reviews were excluded. Discrepancies between reviewers were resolved through consensus with a third author (AV).
Data extraction and quality assessment
Data extraction was independently performed by two authors (NP and LB). The collected data existed of (a) study characteristics, that is, first author name, type of article, year of publication, inclusion time period, follow-up time, study country and sample size; (b) intervention characteristics, that is, type vaccination, number of vaccinations, vaccination interval, follow-up time and prior SARS-CoV-2 infection; (c) epidemiological and disease-specific characteristics, that is, age, sex, ethnicity, body composition, comorbidities, IBD diagnosis and current medication and (d) breakthrough infection rates, symptoms, hospitalization and mortality rates. The quality of the studies was assessed with the Newcastle Ottawa Scale for nonrandomized studies. 14
Outcomes and comparisons
Primary outcome was the SARS-CoV-2 breakthrough infection rate after vaccination in patients with IBD. Breakthrough infection rates were compared with non-IBD controls, partially vaccinated and unvaccinated IBD patients. Furthermore, comparisons were made between IBD subgroups based on IBD type, medical therapy and vaccination type. Secondary outcome was the COVID-19 breakthrough infection severity, including symptomatic disease, hospitalization, intensive care unit (ICU) admission and all-cause mortality.
Data synthesis and analysis
A meta-analysis on the breakthrough infection rates of fully vaccinated IBD patients in comparison with vaccinated non-IBD controls, unvaccinated and partially vaccinated IBD patients was performed. We used a random-effects model and the Mantel-Haenszel method. Continuity correction of 0.5 was used in studies with zero cell frequencies. To assess heterogeneity, DerSimonian-Laird estimator was used for τ 2 . We critically appraised the available data for other outcome measures and did not perform a meta-analysis for other outcome measurements due to lack of data. A two-sided p-value <0.05 was considered statistically significant.
Results
Eligible studies
The initial literature search revealed 663 articles potentially eligible for inclusion (Figure 1). After full-text screening, 16 articles were included in the systematic review. No additional articles were found through citation search.
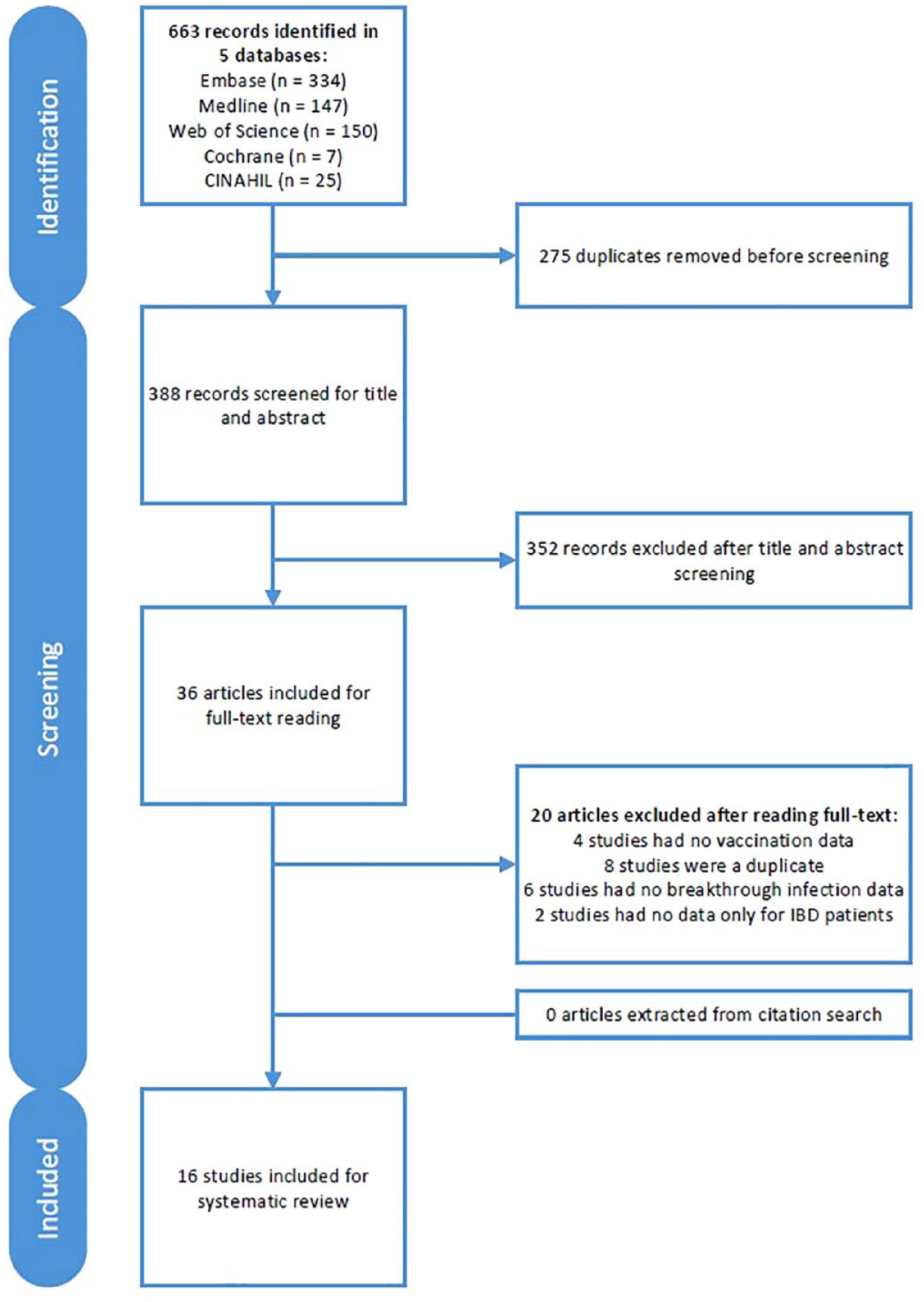
Flowchart of the study selection process according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines.
Quality assessment
Details of the quality assessment are shown in Supplemental Table 2. Eleven articles comprised a control group, including healthy controls (n = 1), 15 non-IBD patients (n = 4),16–19 unvaccinated IBD patients (n = 3),20–22 IBD patients after a SARS-CoV-2 infection (n = 1), 23 irritable bowel syndrome (IBS) patients (n = 1) 24 and an unspecified control group (n = 1). 25 Eight studies were of high quality (7–9 stars)16–21,23,26 and eight studies were of moderate quality (4–6 stars)15,22,24,25,27–30 due to no selection of a control group,26–30 no selection of a matched control group,15,22,24,25 no demonstration that patients did not have a SARs-CoV-2 infection at the start of the study22,24,25,29,30 or unclear follow-up time. 24
Study characteristics
The characteristics of the included articles are summarized in Table 1. Included studies were performed between January 2020 and October 2021 with a follow-up time ranging from 30 days to 6 months. All articles included patients which were fully vaccinated,15–30 three articles included an additional subgroup of patients who were partially vaccinated20,21,30 and none of the articles included patients who received three or more vaccinations. Fully vaccinated is defined as either after a second dose in a two-dose series (e.g. Pfizer-BioNTech; Moderna ) or after a first dose in a single-dose series (e.g. Janssen/Johnson & Johnson). 31 In 14 available studies, 36,109 patients and 41,624 controls received the mRNA vaccine with mRNA-1273 (Moderna)17,21–25,27,28,30 or BNT162b2 (Pfizer-BioNTech).15–17,19–25,27–30 In four studies, 315 patients and 95 controls received the adenovirus vector vaccine with Ad26.CoV2.S (Janssen, Johnson & Johnson),22–24,30 ChAdOx1 nCoV-19 (Oxford-AstraZeneca),22,24 GAM-COVID-Vac (Sputnik) 22 or Ad5-nCoV-S (CanSino). 22 In one study, six patients received the inactivated SARS-CoV-2 vaccine with CoronaVac (Sinovac). 22 Four studies (patients: n = 13,030; controls: n = 11,676) did not specify the type of vaccination.17,18,25,26
Baseline characteristics of the included studies.

Type of vaccination percentage is calculated on the number of vaccinated patients and controls. The type of vaccination is specified as CanSino, Ad5-nCoV-S; Johnson & Johnson, Ad26.CoV2.S; Moderna, mRNA-1273; Oxford-AstraZeneca, ChAdOx1 nCoV-19; Pfizer-BioNTech, BNT162b2 mRNA; Sinovac, CoronaVac; Sputnik, Gam-COVID-Vac.
5-ASA, 5-aminosalicylic acid; ADA, adalimumab; anti-TNF-α, anti-tumour necrosis factor alpha; AVV, adeno viral vector vaccine; AZA, azathioprine; CD, Crohn’s disease; CTZ, certolizumab; IBD-U, inflammatory bowel disease undefined; IBS, irritable bowel syndrome; IFX, infliximab; IL12/23-i, interleukin 12/23 inhibitor; IM, immunomodulatory; IPAA, ileal pouch–anal anastomosis; IQR, interquartile range; JAK-i, Janus kinase inhibitor; mRNA, mRNA vaccines; MTX, methotrexate; N, number; p-d, person-days; SD, standard deviation; TOFA, tofacitinib; UC, ulcerative colitis; UST, ustekinumab; VDZ, vedolizumab; y, year.
Breakthrough infections
Fifteen studies assessed the breakthrough infection rate. The majority of studies reported breakthrough infection rates below 6% within the study follow-up time, although rates vary between 0% and 37.4% (Figure 2).15–21,23–30 A calculated pooled breakthrough infection rate was not possible due to differences in follow-up time and period of included studies. Seven studies compared the breakthrough SARS-CoV-2 infection rate between IBD patients and non-IBD controls. Meta-analysis of these studies showed a risk ratio (RR) of 1.01 (95% CI: 0.92–1.10) (Figure 3).15–19,24,25 Furthermore, the pooled relative risk of breakthrough infections in vaccinated IBD patients was significantly lower than the risk of infection in unvaccinated IBD patients (RR: 0.07, 95% CI: 0.03–0.18, Figure 4). Heterogeneity was significantly present (I2 = 83%, p < 0.01).19–21 No differences were found between fully and partially vaccinated IBD patients (RR: 0.67, 95% CI: 0.38–1.18, Figure 5).20,21,30
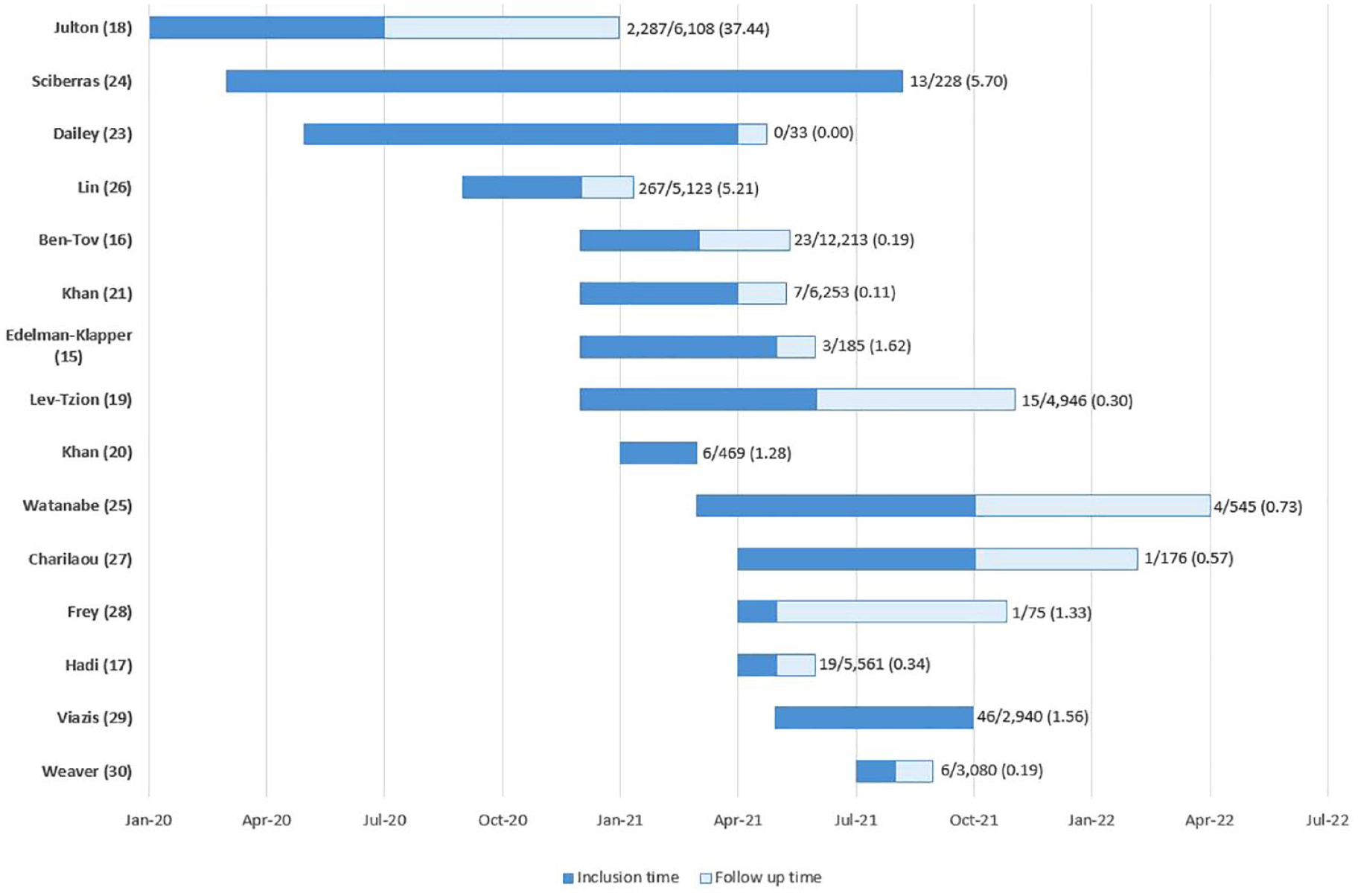
SARS-CoV-2 breakthrough infection rates in inflammatory bowel disease (IBD) patients within the study follow-up time.

Meta-analysis of breakthrough SARS-CoV-2 infections in patients with IBD versus a control group (i.e. healthy control group, non-IBD group and IBS group).

Meta-analysis of SARS-CoV-2 infections in vaccinated and unvaccinated inflammatory bowel disease (IBD) patients.

Meta-analysis of SARS-CoV-2 infections in vaccinated and partially vaccinated inflammatory bowel disease (IBD) patients.
Subgroup analyses in one study revealed no significant difference in breakthrough infection rates between Pfizer-BioNTech and Moderna. 21 One comparative study assessed the risk of breakthrough infections per IBD subtype compared to healthy controls. According to this study, both patients with CD and UC were not at increased risk of breakthrough infections (p = 0.055 and p = 0.310, respectively).
When comparing the IBD subtypes, patients with CD showed higher infection rates after >7 days (HR: 3.56; 95% CI: 1.29–9.83; p = 0.01) and >14 days (HR: 3.38; 95% CI: 1.07–10.64; p = 0.04) when compared to patients with UC. 16 In addition, one study showed that patients with infliximab had higher rates of breakthrough infections than patients with vedolizumab (p = 0.0039), 26 whereas other studies showed no difference between immunosuppressive treatment (immunomodulators and biologics) versus no treatment (p = 0.45), 16 between anti-TNF-α/corticosteroids versus without anti-TNF-α/corticosteroids (p = 0.25) and between anti-TNF-α/corticosteroids versus other biologics (p = 1.0). 19
COVID-19 disease course
The majority of patients reported mild constitutional and respiratory symptoms, including fatigue, anosmia/ageusia, fever, cough, myalgia, hoarse voice, confusion and chest pains.16,20,26,29,30 The rates for hospitalization (0–8.7%), severe SARS-CoV-2 infections (0.05–3.4%), ICU admission or mechanical ventilation (0) and all-cause mortality (0–4.3%) were low (Table 2).16,20–22,26,29,30 One article defined severe SARS-CoV-2 infections as COVID-19-related hospitalization or death, and no differences were found for severe SARS-CoV-2 infection and all-cause mortality rates between fully vaccinated and unvaccinated IBD patients (p = 0.18 and p = 0.11, respectively). 21 Another article defined severe SARS-CoV-2 infection as ICU admission, mechanical ventilation and/or death, and it showed that all IBD patients with a severe SARS-CoV-2 infection used combination therapy and were significantly older (mean age 59 years versus 39 years, p = 0.03) . Higher hospitalization rates were observed in patients on combination therapy (15.8%) compared to biological (2.0%) or immunomodulatory monotherapy (0%) (p = 0.08), and hospitalized patients were significantly older (mean age 53 years versus 39 years, p = 0.04) . Vaccine type showed no difference in disease severity and hospitalization rates. 22
Secondary outcomes.
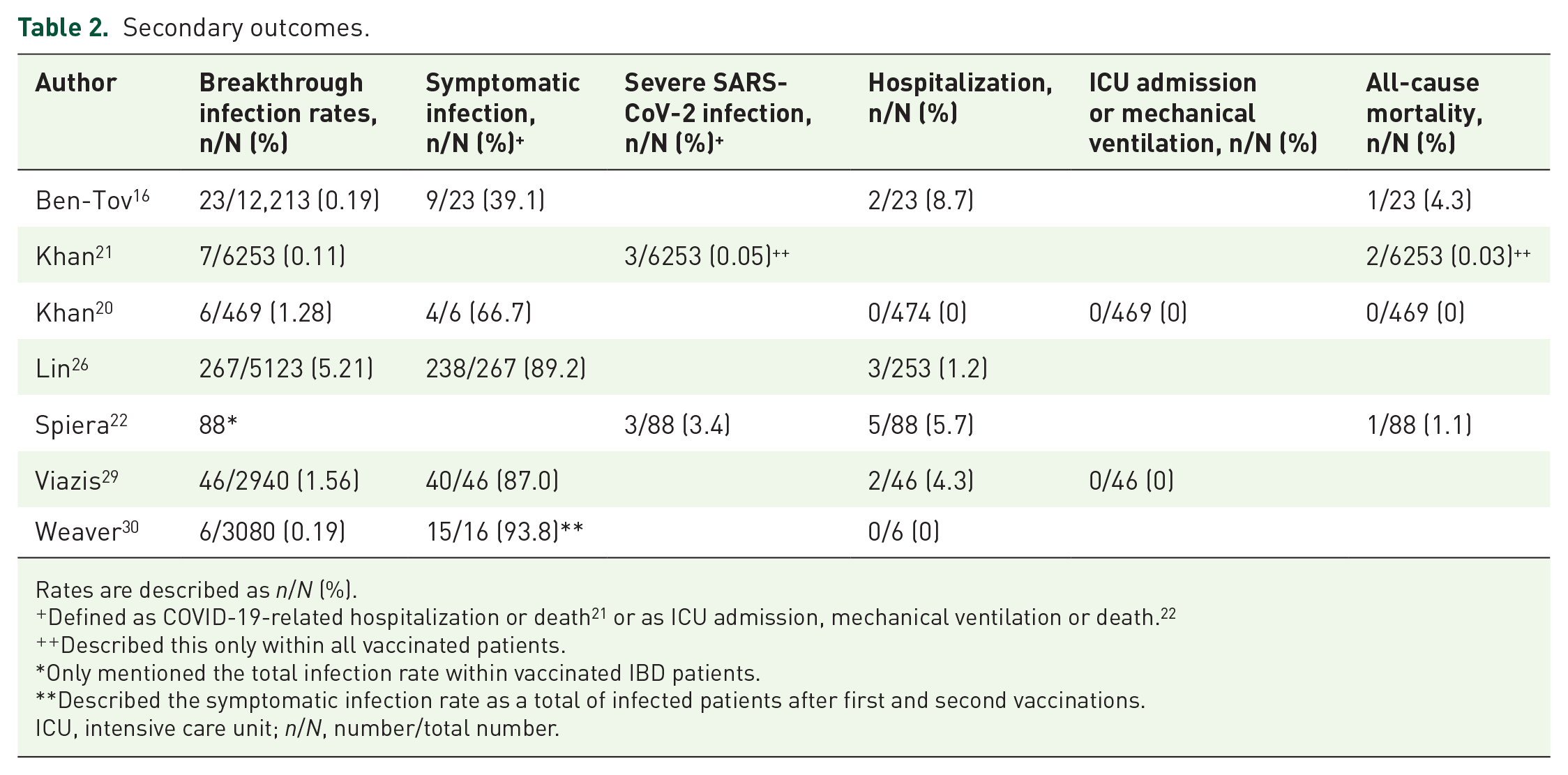
Rates are described as n/N (%).
Defined as COVID-19-related hospitalization or death 21 or as ICU admission, mechanical ventilation or death. 22
Described this only within all vaccinated patients.
Only mentioned the total infection rate within vaccinated IBD patients.
Described the symptomatic infection rate as a total of infected patients after first and second vaccinations.
ICU, intensive care unit; n/N, number/total number.
Discussion
This systematic review and meta-analysis provides an overview of SARS-CoV-2 breakthrough infection rates and COVID-19 disease severity following COVID-19 vaccinations. Breakthrough infections after COVID-19 vaccination do occur in fully vaccinated IBD patients, though at a similar rate as compared to non-IBD controls. Data suggest a declining trend of breakthrough infections during calendar time. Furthermore, the relative risk of breakthrough infections in vaccinated IBD was lower than the risk of infection in unvaccinated IBD patients. The course of breakthrough infections is generally mild, and hospitalization and mortality rates are in line with the rates in non-IBD patients. Potential risk factors associated with a severe course of breakthrough infections include combination therapy with both anti-TNF and an immunomodulator and older age at infection.
In this study, contradictory results were found regarding the impact of immunosuppressive therapy on the COVID-19 breakthrough infection rate in IBD patients. One article showed a negative impact of infliximab compared with vedolizumab, whereas other studies showed no impact of immunosuppressive treatment, anti-TNF-α or corticosteroids and other biologics. A higher rate of breakthrough infections may be explained by a combination of (a) lower seroconversion rates following COVID-19 vaccination in patients using steroids or combination therapy with anti-TNF-α and an immunomodulator, and (b) a faster antibody decay in patients treated with anti-TNF-α or immunomodulators. 12 Although one report showed a significantly lower geometric mean titre in patients with a confirmed breakthrough infection, 26 no cut-off values have been determined to identify patients at increased risk. This underlines the importance to advice full vaccination and revaccination for these specific subgroups.
The strength of this study: this is the first systematic review and meta-analysis which focused on breakthrough infection rates and the severity of COVID-19 infections. A comprehensive overview of breakthrough infection rates and performed multiple comparative subgroup analyses are provided. Nevertheless, some limitations should be taken into consideration. First, seasonal fluctuations in SARS-CoV-2 infection rates did not allow us to calculate a pooled breakthrough infection rate given differences in follow-up time and period of included studies. 32 Second, we were not able to perform more detailed subgroup analysis due to limited studies. Third, most articles included the mRNA vaccine (Pfizer or Moderna) questioning the generalizability of our results to the viral vector vaccinations (Johnson and AstraZeneca). Currently, the booster vaccination is introduced worldwide, and in this review, no studies reported the breakthrough infection rates after third or fourth vaccination.
In conclusion, this review and meta-analysis showed that vaccination is useful, considering the breakthrough infection rates in vaccinated IBD patients are significantly lower than the risk of infection in unvaccinated IBD patients. Although breakthrough infections do occur, severe SARS-CoV-2 infections, hospitalization, ICU admission or mechanical ventilation and all-cause mortality rates are low. Available data seem to suggest a declining trend of breakthrough infections during calendar time. The effect of IBD medication on the rate of breakthrough COVID-19 infections and disease course requires further elucidation.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231174295 – Supplemental material for SARS-CoV-2 breakthrough infections after COVID-19 vaccination in patients with inflammatory bowel disease: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848231174295 for SARS-CoV-2 breakthrough infections after COVID-19 vaccination in patients with inflammatory bowel disease: a systematic review and meta-analysis by Natasja van de Pol, Qiuwei Pan, Lauranne A. A. P. Derikx, Linda Bakker, C. Janneke van der Woude and Annemarie C. de Vries in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We would like to thank W. M. Bramer, biomedical information specialist, for conducting the literature search.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
