Abstract
Background:
The impact of gastrointestinal endoscopy on COVID-19 infection remains poorly investigated. We herein performed a systematic review and meta-analysis to evaluate the outcomes of COVID-19 in patients undergoing gastrointestinal endoscopy.
Method:
Ovid Medline, Ovid EMBASE, Ovid the Cochrane Library, and other electronic databases were searched until 30 November 2020 to identify publications with confirmed COVID-19 infection in patients undergoing gastrointestinal endoscopy. The primary outcomes were SARS-CoV-2 transmission, personal protective equipment use, rates of case fatality, complications, and procedural success.
Results:
A total of 18 articles involving 329 patients were included in this systematic review and meta-analysis. The overall basic reproduction rate is 0.37, while the subgroup results from Asia, Europe, and North America are 0.13, 0.44, and 0.33, respectively. The differences in personal protective equipment use between the positive transmission and non-transmission group are mainly in isolation gowns, N95 or equivalent masks, and goggles or face-shields. The rate of case fatality, complication, and procedural success are 0.17 (95% confidence interval = 0.02–0.38), 0.00 (95% confidence interval = 0.00–0.02), and 0.89 (95% confidence interval = 0.50–1.00), respectively. The fatality rate in Europe was the highest (0.23, 95% confidence interval = 0.04–0.50), which is significantly different from other continents (p = 0.034).
Conclusion:
The risk of SARS-CoV-2 transmission within gastrointestinal endoscopy units is considerably low if proper use of personal protective equipment is applied. Similarly, a low fatality and complication rate, as well as a high procedural success rate, indicated that a full recovery of endoscopic units should be considered.
Introduction
The outbreak of the novel coronavirus disease 2019 (COVID-19), caused by SARS-CoV2 infections, has widely spread throughout the world1,2 and has been declared as a pandemic by the World Health Organization (WHO) in March 2020. 3 So far, more than 119 million confirmed COVID-19 cases have been reported, with 2.6 million confirmed death by 17 March 2021. 4 COVID-19 is not only able to cause respiratory illness, but may also lead to gastrointestinal (GI) diseases, for example, enteritis,5,6 pancreatitis, 7 and cholangitis. 8 The potential mechanism might be that SARS-CoV2 binds to the angiotensin-converting enzyme-2 (ACE2) receptors, which are widely expressed in lung, intestine, and liver.9,10 Hence, COVID-19 may have significant impact on the clinical outcomes of GI endoscopic procedures, including disease transmission, rate of fatality, complications, and procedural success.
Up to date, the impact of patients with SARS-CoV-2 infections undergoing GI endoscopy is poorly understood, leading to a controversial discussion. For instance, endoscopic procedures are associated with aerosol generation, which may pose significant risks to health care workers (HCWs) and patients. Here, Repici and colleagues 11 declared that there is a low risk of SARS-CoV-2 transmission within GI endoscopy units with an infection rate of 4.2%. Of note, the burden of SARS-CoV-2-positive patients in their study is significantly low (1 of 802 patients, 0.12%), which compromises the reliability of the conclusion. The SARS-CoV-2 transmission rates within GI endoscopy units remain unknown. 12 Another example is the management of gastrointestinal bleeding (GIB). According to the study by Martin and colleagues, 13 no significant difference in transfusion requirements was observed between the endoscopic therapy group and the expectant therapy group. Conservative management was regarded as a reasonable approach in managing even complex GIB cases. In contrast, Saibeni and colleagues 14 reported on three hospitalized GIB patients who did not receive an endoscopic evaluation, and two of them finally died. The management of endoscopic procedures volume is also a matter of debate. Resumption of endoscopic procedures in a safe setting is of critical importance in regard to mitigate adverse health outcomes caused by the pandemic. 15 It is explorable to finger out the role of procedural volume reduction on restarting endoscopy service. Therefore, a study to assess the outcomes of COVID-19 in patients undergoing GI endoscopic procedures is urgently needed.
This systematic review and meta-analysis aimed to report on clinical outcomes of COVID-19 patients who underwent GI endoscopy based on the available data within the identified articles. Here, special emphasis will be put on disease transmission, personal protective equipment (PPE) use, rates of case fatality, complications, and procedural success in COVID-positive patients within endoscopic units. The study may therefore provide evidence-based guidance for clinical decision-making.
Methods
The systematic review and meta-analysis were conducted using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The protocol for the study was registered and approved on PROSPERO (CRD42020225000).
Search strategy
The publications that published on several electronic databases, registries, and guideline, including Ovid Medline (1950-present), Ovid EMBASE (1974- present), Ovid the Cochrane Library of Randomized Trials (1993-present), the World Health Organization International Clinical Trials Platform Search Portal (ICTRP), and ClinicalTrials.gov, were searched on 15 November 2020, using the key terms ‘2019nCoV’, ‘novel corona virus’, ‘COVID19’, ‘betacoronavirus’, ‘SARS-CoV-2’, ‘coronavirus infection’, ‘Wuhan pneumonia’, AND ‘endoscopy’, ‘gastroscopy’, ‘duodenoscopy’, ‘choledochoscopy’, ‘cholangioscopy’, ‘colonoscopy’, ‘rectoscopy’, and ‘proctoscopy’. Detailed retrieval strategy is shown in Supplementary Table 1s.
Study selection and data extraction
Two investigators (X.T. and J.G.) performed the literature screening independently, based on the predefined inclusion and exclusion criteria.
Inclusion criteria were as follows: (1) studies reported data on COVID-19-confirmed patients; (2) these patients underwent GI endoscopy for both diagnosis and therapy; (3) had one of the following outcomes: transmission of SARS-CoV-2, PPE use, fatality, complications, success rate of endoscopic hemostasis; (4) the latest study was included if duplicated studies from the same population were identified; and (5) studies limited to human.
Exclusion criteria include the following: (1) no useful data, for example, outcomes, can be obtained; (2) small sample-size studies (n ⩽ 3) reported COVID-19 fatality, complications, or success rate of endoscopic therapy; (c) no sample-size limit in studies that reported transmission of SARS-CoV-2; (d) duplicated studies; and (e) non-English publications.
In cases of discrepancy and disagreement, the studies were discussed and resolved by consensus, or, if necessary, a third reviewer (D.W.) would be involved. 16
Data extraction was independently conducted by two reviewers (X.T. and J.G.). The following information from each included study was extracted: first author, publication date, country, continent, study design type, number of COVID-19-positive patients, number of total observed patients, endoscopic reasons, ages, sex, and outcomes (transmission, fatality, complications, success rate of endoscopic therapy). Additional data, such as intensive care unit (ICU) admission rate, intervention rate, and upper bleeding rate, was obtained in the studies that reported COVID-19 fatality, complications, or success rate of endoscopic interventions.
Endpoint setting and stratification strategy
Our primary outcomes were transmission of SARS-CoV-2 within GI endoscopic units, PPE use, case fatality rate, complication rate, and procedural success rate in COVID-19-positive patients who have undergone GI endoscopy. The stratified analysis of subgroups in different continents was carried out to determine the differences in outcomes in diverse regions.
Meta-regression and assessment of bias
The univariable meta-regression analysis was performed to predict the impact of patient populations’ characteristics on several outcomes, including rates of fatality, complications, and procedural success. The factors we adopted for the meta-regression analysis involve continents, study period, mean age, male proportion, ICU admission rate, upper GI bleeding rate, and endoscopic intervention rate. Two parameters, that is, continents and study period, are dichotomous data; therefore, studies of North America in continents set and studies started before 11 March 2020 in study period set (the date COVID-19 was announced by WHO to be characterized as a pandemic) were defined as reference studies.
Visual inspection of funnel plots, Egger’s regression asymmetry test, and Begg’s rank correlation test in each outcome were applied to detect publication bias (p-value < 0.10). 17
Statistical analysis
The pooled proportion data with their corresponding 95% confidence intervals (CIs) were calculated via either a fixed-effects model or random-effects model, using the Freeman–Tukey double arcsine transformation method. The Q test and Higgins I2 static were applied to assess the between-study heterogeneity. Conventionally, an I2 value > 50% or p-value < 0.05 was considered as high heterogeneity. A random-effects model was used when significant heterogeneity was identified; otherwise, a fixed-effects model was preferred. A meta-regression permutation test was applied to calculate the exact p-value. 18 All statistical analyses were conducted with STATA 14.0 (Stata Corporation, College Station, Texas, USA).
Results
Literature search
The initial search of databases identified 452 articles. After duplicate removal, a total of 409 studies were reviewed via title and abstract, which further lowered the number of studies to 366. Another 321 articles were then excluded, because they did not report the confirmed SARS-CoV-2-positive patients who were concurrently undergoing GI endoscopy. Another three studies were removed because the publication was about the same population as another included study. And 24 studies were excluded due to the limited outcomes data available and small sample size. Finally, 18 studies were included in the qualitative synthesis and the meta-analysis. The study selection flow diagram is shown in Figure 1.
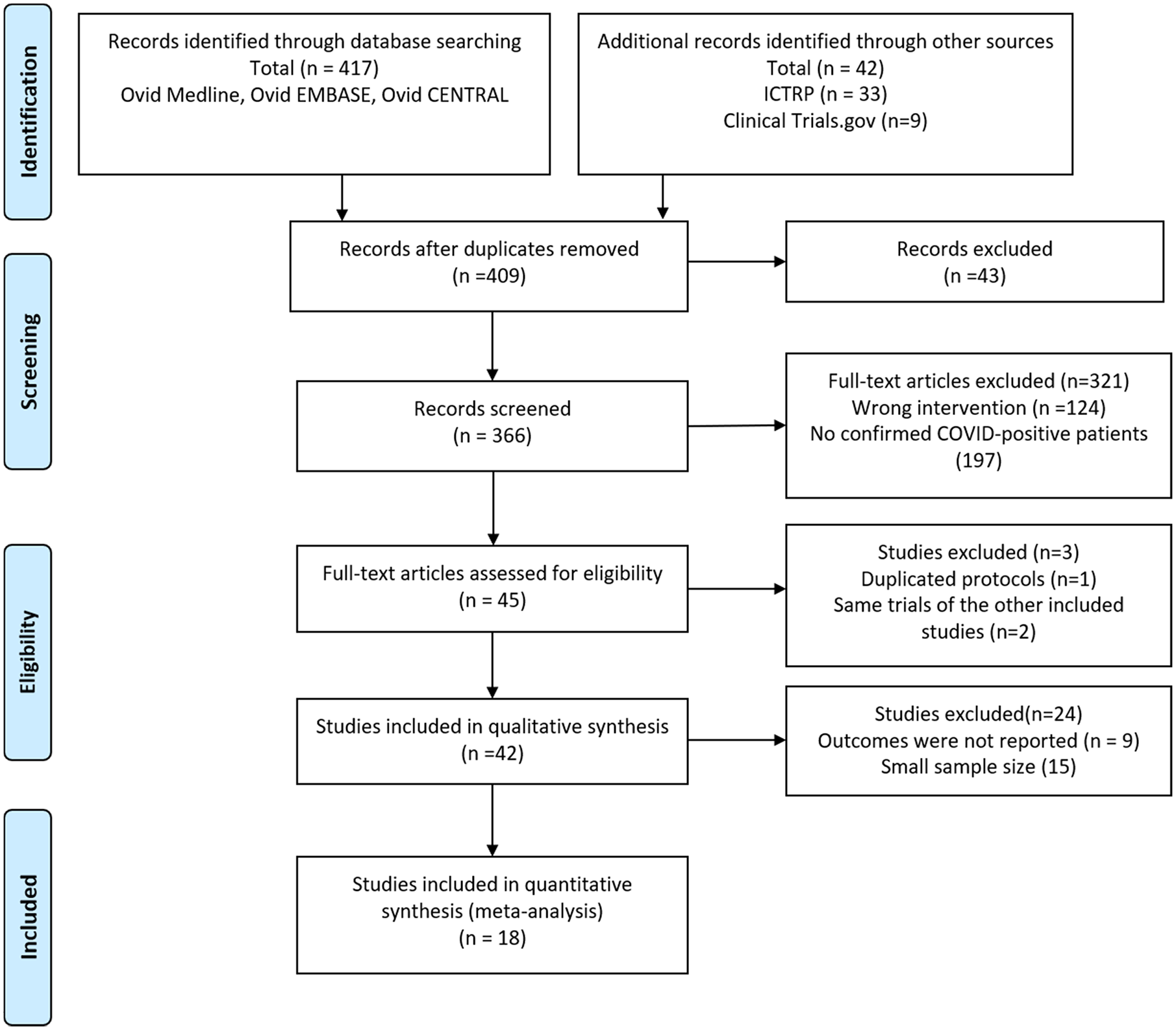
Flow chart of literature search (PRISMA 2009 flow diagram).
Study characteristics
A total of 18 studies involving 329 COVID-positive patients were included in the systematic review and meta-analysis. Baseline characteristics of included studies are listed in Table 1. There are eight studies (n = 150 patients) from Europe, six studies (n = 24 patients) from Asia, and four studies (n = 155 patients) from North America (all from the United States), respectively. No article has yet to be reported from Africa, South America, or Oceania. Three studies are descriptive studies, while 15 studies are other study types, including 2 retrospective cohort studies, 5 cross-sectional studies, and 8 case series or reports. The endoscopic indications involve GIB, emergency endoscopy, and others. The distribution of sex in the reported studies (n = 10) is 160 and 73 for numbers of male and female, respectively. However, there are eight studies which did not report the sex proportion.
Characteristics of included studies.

GIB, gastrointestinal bleeding; NR, not reported; SD, standard deviation; UGI, upper gastrointestinal.
Outcomes of COVID-19 patients who have undergone GI endoscopy
Transmission of SARS-CoV-2 and PPE use within GI endoscopy units
Fifteen studies with a total of 122 COVID-positive patients reported the potential COVID-19 transmission rate, as shown in Table 2.11,19,20,22–24,26–34 Three studies11,19,32 reported potential transmission of SARS-CoV-2 within GI endoscopy units, for a total of 46 HCWs or patients. The total basic reproduction rate (R0) equals 0.37, while the subgroup results from Asia, Europe, and America are 0.13, 0.44, and 0.33, respectively. Although the transmission routes of most cases remain unknown, transmission via close contact with COVID-positive patients is the most common reason for traceable cases (6 out of 9 cases, 66.7%; see details in Figure 2(a)).
The systematic review of COVID-19 transmissions within GI endoscopy units.

GI, gastrointestinal.

The reported COVID-19-positive cases transmitted within GI endoscopic unit and the presumed reasons (a), and the personal protective equipment (PPE) use presented in seven studies which reported no SARS-CoV-2 transmission within GI endoscopic unit. (b) Multiple reason refers to the combination of close contact and nonoccupational transmission.
The effect of PPE use on the transmission of SARS-CoV-2 within GI endoscopy units is evaluated by comparing the differences of PPE use between the studies that reported no transmission and the studies that reported positive transmissions. Eight studies20,27,29–34 were finally included for the analysis of PPE use in GI endoscopy units. Seven studies20,27,29–31,33,34 claimed that no transmission of SARS-CoV-2 was found in their GI endoscopy unit, whereas one study 32 demonstrated successful screening of 3 COVID-positive transmitted cases. As is shown in Figure 2(b), the frequencies of PPE use in two group sets (no transmission group and positive transmissions group) were displayed. Three kinds of PPE were deemed to be the most important equipment for protecting HCWs from corona virus disease, including isolation gowns, N95 or equivalent masks, and goggles or face-shields.
Case fatality rate in COVID-positive patients undergoing GI endoscopy
Five studies21,24–27 with a total of 183 COVID-positive subjects reported the case fatality rate in confirmed COVID-19 patients undergoing GI endoscopy (Figure 3(a)). Random-effects meta-analysis demonstrates that the total case fatality rate is 0.17 (95% CI = 0.02–0.38). Subgroup analysis among different continents shows significant differences between populations in Europe compared with North America. The case fatality rate in Europe [0.23 (95% CI = 0.04–0.50)] is significantly higher than the one reported from North America [0.05 (95% CI = 0.01–0.10)]. No studies from Asia reported the case fatality rate in COVID-positive patients undergoing GI endoscopy. Furthermore, high heterogeneity across all studies was observed (I2 = 84.46%).

(a) Forest plot of case fatality rate, (b) forest plot of complication rate, and (c) forest plot of success rate in endoscopic hemostasis.
Complication rate in COVID-positive patients who have undergone GI endoscopy
Four studies13,24–27 with a total of 68 COVID-positive patients pointed out the complication rate in confirmed COVID-19 patients who have undergone GI endoscopy (Figure 3(b)). The pooled complication rate is 0.00 (95% CI = 0.00–0.02), and there is no statistically significant difference between the European group and the North American Group (p-value = 0.90) using a fixed analysis. Only one study is available from North American for the subgroup analysis. Again, no Asian study reported the complication rate in the investigated population. There is no significant heterogeneity between included studies (I2 = 0.00%).
Procedural success rate in COVID-positive patients who have undergone GI endoscopy
In total, 41 subjects from 3 studies13,25,27 are included in the quantitative analysis of procedural success rate (Figure 3(c)). Only the outcome data from endoscopic hemostasis procedures can be obtained. The pooled procedural success rate equals to 0.89 (95% CI = 0.50–1.00) using random-effects models (I2 = 71.79%). Subgroup analysis shows no significant difference is observed between subgroups (p-value = 0.05).
Meta-regression
A univariate meta-regression analysis was conducted to evaluate the effects of populations’ characteristics on each outcome. The variables that potentially influence outcomes, such as continents, study periods, mean age, male proportion, ICU admission rate, upper GI bleeding rate, and endoscopic intervention rate, were included for the meta-regression analysis. The results of case fatality rate, complication rate, and procedural success rate show that none of the included factors is significantly different to affect Case Fatality Rate (CFR) (Table 3). However, ICU admission rate is a variable that contributes to the complication rate of COVID-positive patients undergoing GI endoscopy (p-value = 0.041, SE = 0.006).
Meta-regression of case fatality rate, complication rate, and endoscopic hemostasis.
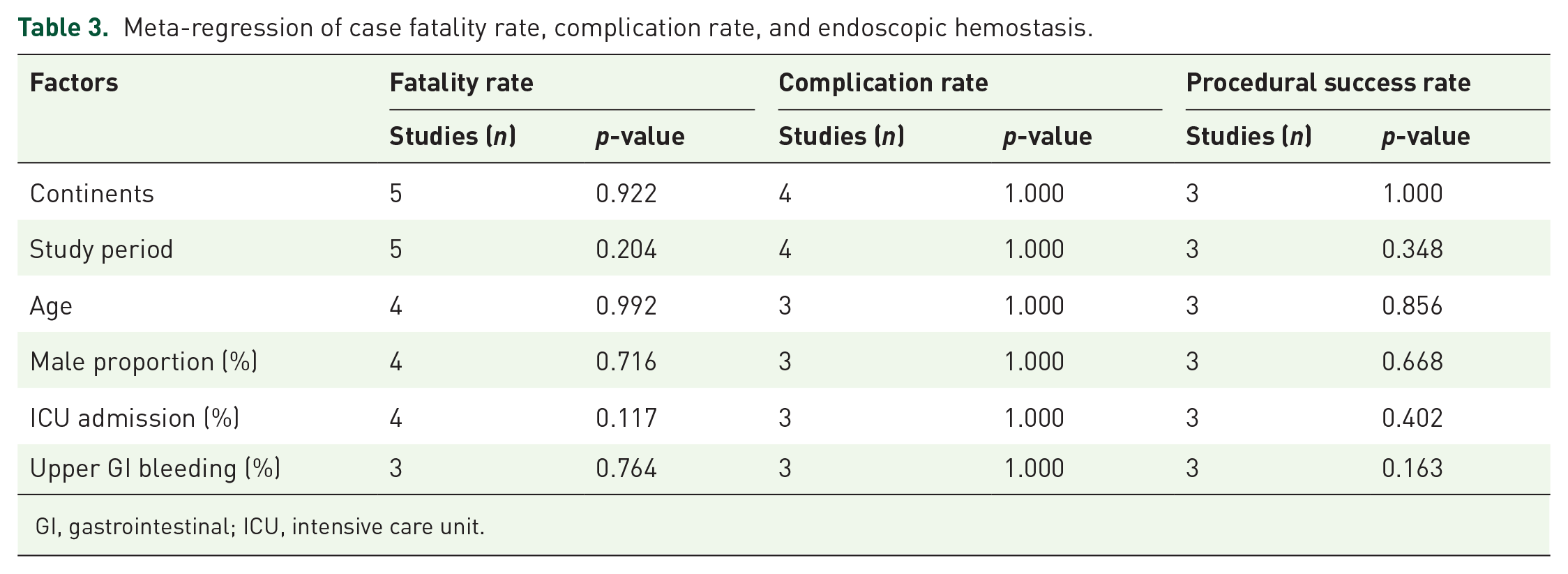
GI, gastrointestinal; ICU, intensive care unit.
Assessment of publication bias
Visual inspection of funnel plots and Egger’s plots are applied to assess the publication bias. The funnel plot and Egger’s plots of studies on each outcome is illustrated in Supplementary Figure 1s and Figure 2s, respectively. No evidence of obvious asymmetry is found in the funnel plots. The statistical results of both Egger’s and Begg’s test support there is no publication bias in our study (Egger’s test: p-value = 0.109 for case fatality rate, p-value = 1.000 for complication rate, and p-value = 0.758 for procedural success rate; Begg’s test: p-value = 0.462 for case fatality rate, p-value = 1.000 for complication rate, and p-value = 1.000 for procedural success rate).
Discussion
We conduct the first systematic review and meta-analysis to explore the clinical outcomes of COVID-19 patients within GI endoscopy units, including 329 patients from 18 studies worldwide. Overall, the basic reproduction rate (R0) for SARS-CoV-2 transmission within GI endoscopic units was 0.37, which is significantly lower than the reported R0 with a range from 2 to 4. 35 The rate of case fatality, complications, and procedural success among SARS-CoV-2-infected patients undergoing GI endoscopy was 0.17 (95% CI = 0.04–0.35), 0.00 (95% CI = 0.00–0.02), and 0.89 (95% CI = 0.50–1.00), respectively. These results imply that under the sufficient supply of PPE, GI endoscopy is a safe and efficient approach for the diagnosis and treatment of digestive diseases in COVID-19-positive patients. Emergency endoscopy should be fully considered irrespective of SARS-CoV2 infection status. In addition, elective GI endoscopy is supposed to return to pre-pandemic volumes after the COVID-19 pandemic has declined.
We noted that the cross-study heterogeneity is considerably high. Most cases were reported from Europe with a higher fatality rate, complication rate, and lower procedural success rate. Although the subgroup and meta-regression analysis were performed, the source of heterogeneity could not be fully explained. Here, we assume that multiple reasons may lead to the high heterogeneity among different studies. First, the inclusion criteria of GI endoscopy for COVID-positive patients vary significantly between the studies included. The indications for endoscopy could be emergency endoscopy, GIB, dealing with the complications of nutrition tube (e.g. dislocation or clogging), which is also influenced by local epidemic conditions, implemented policies, and medical supplies. Second, there are no large-scale data of COVID-19 infections patients within GI endoscopy units owing to the reduction of endoscopy volume to minimize the transmission of SARS-CoV-2. The baseline characteristics of included patients, including age, sex proportion, and ICU admission rate, are therefore quite different. Third, as the continuous mutation of SARS-CoV-2, the individual genotypes of virus may also contribute to the differences in clinical outcomes. Recently, SARS-CoV-2 variant B.1.1.7, initially detected in the United Kingdom, was proved to be associated with a higher transmission risk, severities, and fatality of the disease. 36
Our study systematically reviewed the available data on transmission of SARS-CoV-2 within GI endoscopy units and provided solid evidence for a considerable low risk of SARS-CoV2 infections for both HCWs and patients. Almost 50% of COVID-19-positive patients have shown detectable virus RNA in their fecal samples.37,38 Therefore, there are considerable concerns arising about the potential fecal-oral or fecal-aerosol transmission, which imposes GI endoscopic experts to implement strict precautions, such as a high threshold for admitting endoscopy, to minimize the infection risk during GI endoscopy, especially for aerosol-generating procedures (AGP). However, our results demonstrated there is very limited transmission of SARS-CoV-2 in GI endoscopic units if proper PPE was applied. Close contact is still the predominant transmission route. A proper PPE should at least include isolation gowns, N95 or equivalent masks, and goggles or face-shields. Of note, the study by Wang and colleagues demonstrated that wearing a surgical mask during GI endoscopy is insufficient for preventing transmission of SARS-CoV-2, 32 which is consistent with other studies. 39 Interestingly, Kim and colleagues 34 reported about a patient with unknown COVID-infection at time of examination who wore a surgical mask in a GI endoscopy unit: no patients in the recovery room nor HCWs were infected. This case report suggested that precautions from patients’ side could also provide an efficient approach to lower the risk of COVID transmission, but the possibility of individual transmission capability should also be taken into account.
In our results, the fatality rate of COVID-19 patients undergoing GI endoscopy (0.17, 95% CI = 0.04–0.35) seems to be relatively high, compared with the CFR around 2% that had been reported by the WHO. 4 However, nearly all cases that had been included in the investigation of CFR were from hospitalized populations, part of them are even from ICUs. This phenomenon is mostly assignable to a high threshold of endoscopy admission that bases on expert consensus. 40 The mortality of in-hospital patients with COVID-19 was reported at 28–39% and even higher in patients with comorbidities.41–45 These data significantly exceed the CFR in our study. In addition, those patients with hemodynamic instability, emergent bleeding, obstructive jaundice, and so on are more likely to be referred to endoscopic units. 46 These symptoms normally accompany with high risk of fatality. Several studies also revealed that most cases did not die of endoscopy-related incidents but due to SARS-CoV2 infections.21,25–27 Hence, we believe the high CFR of COVID-19 patients within GI endoscopy units is dependent of the characteristic baseline of included population. Hence, we strongly believe that the endoscopy-related CFR of COVID-19 patients who receive GI endoscopy is significantly low.
When analyzing complications, the systematic review and meta-analysis demonstrate a low complication rate in COVID-19 patients undergoing GI endoscopy (0.00, 95% CI = 0.00–0.02). In the current publications, there are no major complications associated with GI endoscopic procedures. The low complication rates, in some extent, indirectly support our hypothesis of low endoscopy-related CFR. In regard to endoscopic intervention, only the data involving endoscopic hemostasis can be obtained; the success rate of endoscopic hemostasis was therefore analyzed based on three studies.13,25,27 The endoscopic hemostasis procedure shows a high success rate with a pooled value of 89% (95% CI = 50%–100%). The main interventions for hemostasis are endoscopic clipping, hemostatic injection, rectal packing, cautery, and so on.
It has been reported that the risk of death from SARS-CoV2 infections in COVID-19 patients who have undergone treatment for other comorbidities, such as cancer, was overestimated. 47 A recent meta-analysis and a large European cohort study pointed out that insufficient cancer treatments play a role in the fatality and severity of COVID-19.48,49 Our results proved a low risk of transmission, endoscopy-related case fatality, complication, and procedural failure in COVID-19 patients who received GI endoscopy. The endoscopic diagnosis and treatments for comorbidities (e.g. cancer), apart from SARS-CoV-2 infections, are highly recommended to be continued during the era of epidemics.
A few methodological limitations existed in this study. First, most included publications are small-sample case series or cohort studies. Second, the inclusion and exclusion criteria of admitting endoscopy for COVID-19 patients are different in various endoscopic units due to medical supplies, local anti-epidemic policy, and healthcare burden. Finally, the overall heterogeneity is significantly high and could not be fully explained by subgroup and meta-regression analysis, which probably undermines the reliability of our conclusion. However, few cases of data can be recorded because of the rigorous restriction of expert consensus. This systematic review and meta-analysis comprehensively collected and analyzed the clinical outcomes of COVID-19 patients receiving GI endoscopy, which can guide the clinical practices for reopening GI endoscopy.
Conclusion
Many endoscopy units recently resume routine procedures to catch up on the postponed cases during COVID-19 pandemics. The clinical outcomes of COVID-19 patients receiving endoscopy are of significant importance. Our study demonstrates a low risk of SARS-CoV-2 transmission within GI endoscopy. The analysis of the PPE use suggests appropriate equipment was recommended to include isolation gowns, N95 or equivalent masks, and goggles or face-shields. In addition, a low rate of fatality, complication, and a high procedural success rate exhibit the need to lift the restriction for admission of GI endoscopy. Our results may provide evidence-based recommendations for the resumption of GI endoscopy.
Supplemental Material
sj-docx-3-tag-10.1177_17562848211042185 – Supplemental material for Systematic review and meta-analysis of clinical outcomes of COVID-19 patients undergoing gastrointestinal endoscopy
Supplemental material, sj-docx-3-tag-10.1177_17562848211042185 for Systematic review and meta-analysis of clinical outcomes of COVID-19 patients undergoing gastrointestinal endoscopy by Xiangzhou Tan, Jianping Guo, Zihua Chen, Alfred Königsrainer and Dörte Wichmann in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-1-tag-10.1177_17562848211042185 – Supplemental material for Systematic review and meta-analysis of clinical outcomes of COVID-19 patients undergoing gastrointestinal endoscopy
Supplemental material, sj-tif-1-tag-10.1177_17562848211042185 for Systematic review and meta-analysis of clinical outcomes of COVID-19 patients undergoing gastrointestinal endoscopy by Xiangzhou Tan, Jianping Guo, Zihua Chen, Alfred Königsrainer and Dörte Wichmann in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-2-tag-10.1177_17562848211042185 – Supplemental material for Systematic review and meta-analysis of clinical outcomes of COVID-19 patients undergoing gastrointestinal endoscopy
Supplemental material, sj-tif-2-tag-10.1177_17562848211042185 for Systematic review and meta-analysis of clinical outcomes of COVID-19 patients undergoing gastrointestinal endoscopy by Xiangzhou Tan, Jianping Guo, Zihua Chen, Alfred Königsrainer and Dörte Wichmann in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We acknowledge support by Open Access Publishing Fund of University of Tübingen.
Author contributions
XT, ZC, and DW contributed to concept and design; XT and JG contributed to acquisition and interpretation of data; XT and DW contributed to drafting of the manuscript; DW, AK, and ZC contributed to critical revision of the manuscript; and all authors approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (81201904 and 81974386) and the program of China Scholarships Council (201806370236).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
