Abstract
Inflammatory bowel diseases (IBD), including Crohn’s disease and ulcerative colitis, are chronic immune-mediated diseases with a high incidence and prevalence in Europe. Since these are diseases with associated disability, they require complex management and the availability of high-quality healthcare resources. We focused on the analysis of IBD care in selected countries of Central and Eastern Europe (Croatia, the Czech Republic, Hungary, Moldova, Poland, Romania and Slovakia) targeting the availability and reimbursement of diagnostic and therapeutic modalities, the role of IBD centers and also education and research in IBD. As part of the analysis, we created a questionnaire of 73 statements organized in three topics: (1) diagnostics, follow-up and screening, (2) medications and (3) IBD centers. The questionnaire was filled out by co-authoring IBD experts from individual countries, and then the answers and comments on the questionnaire were analyzed. We identified that despite the financial burden, which still partially persists in the region, the availability of some of the cost-saving tools (calprotectin test, therapeutic drug monitoring) differs among countries, mainly due to variable reimbursement from country to country. In most participating countries, there also remains a lack of dedicated dietary and psychological counseling, which is often replaced by recommendations offered by gastroenterologists. However, there is adequate availability of most of the currently recommended diagnostic methods and therapies in each participating country, as well as the implementation of established IBD centers in the region.
Keywords
Introduction
Inflammatory bowel diseases (IBD), Crohn’s disease (CD) and ulcerative colitis (UC), are chronic immune-mediated inflammatory diseases of the gastrointestinal tract, affecting genetically predisposed individuals, leading to lifelong disabling symptoms which impair the quality of life.1,2 At the same time, due to their increasing prevalence and the financial aspects of their management, IBD also represent a significant socioeconomic burden at the global level. 1
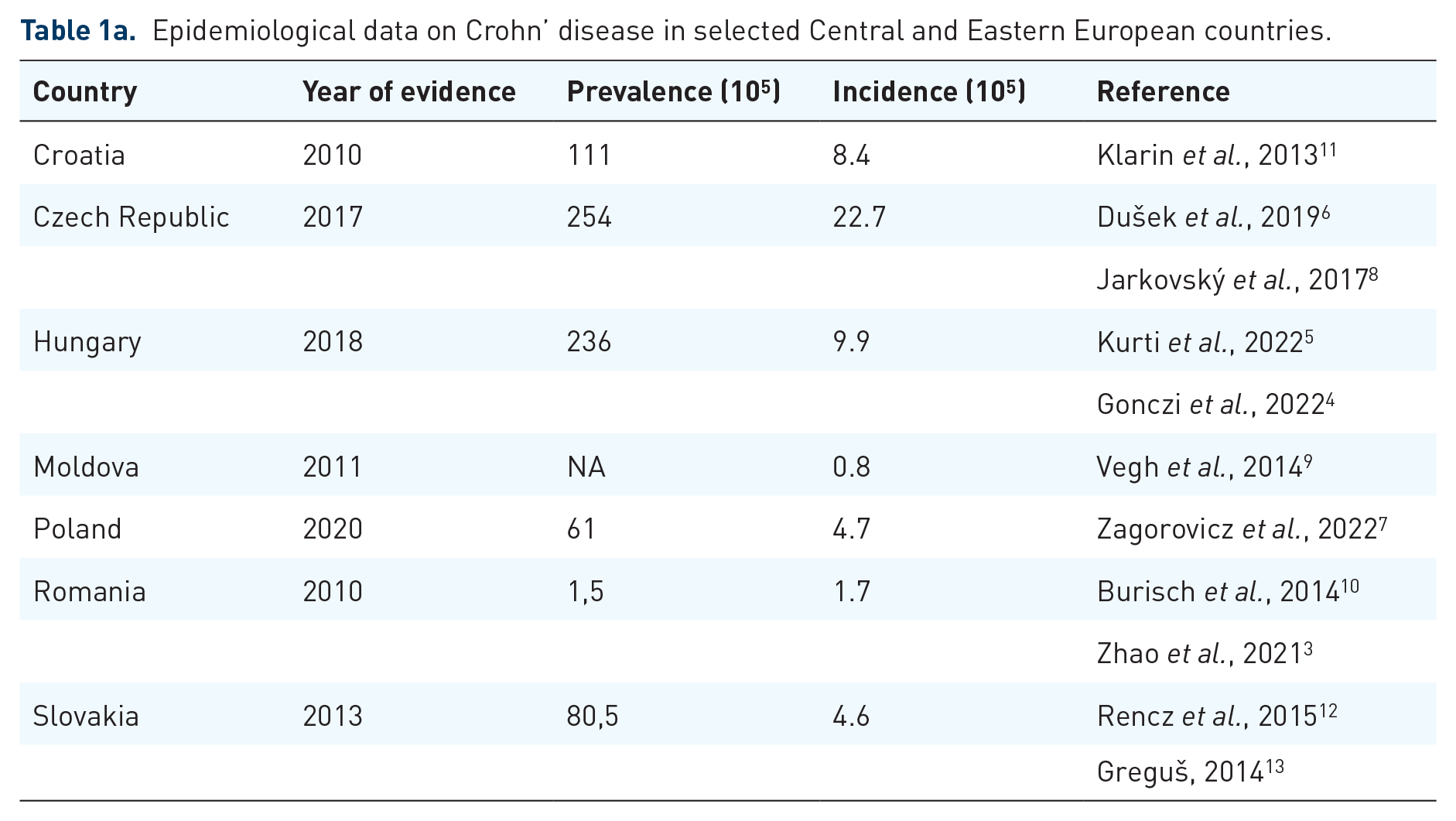
In Europe, 1.3 million patients suffer from IBD, which accounts for a prevalence of 0.2%. In some Western European countries, the prevalence is over 0.3% for both CD (Netherlands: 331 per 100,000 people) and UC (Scotland: 432 per 100,000 people); on the contrary, the lowest prevalence is in South-Eastern European countries. 3 The latest epidemiological data show that in some of the Central European countries the prevalence has already caught up with the West (Czech Republic and Western Hungary) and that the difference between Western and Central European countries has already disappeared.4–6
The reason is the significant rise in the incidence of IBD in Central Europe observed in the last years.4,5,7,8 Nevertheless, the incidence of IBD remains the lowest in the Eastern-European countries.3,9,10 Epidemiological data from the participating countries are summarized in Table 1(a) and (b).
Epidemiological data on Crohn’ disease in selected Central and Eastern European countries.
Epidemiological data on ulcerative colitis in selected Central and Eastern European countries.

For patients with IBD, the quality of care provided can have a significant impact on the course of the disease, not only in terms of its complications and survival, but also by affecting their quality of life on a psychological and social level. 14 Due to the growing incidence of IBD in Central and Eastern Europe, the demands on the quality and accessibility of health care provided in this region are increasing, both from a medical and economic point of view. Our aim was to investigate access to IBD care in various Central and Eastern European countries, to have a better insight of potential barriers in IBD care, related to quality and accessibility of diagnostic tools and therapeutic options in this area.
Methods
As part of the literature review, we searched the Medline database (PubMed) for relevant studies published on the topic of IBD with a focus on Central and Eastern Europe. To prepare this work analyzing the barriers in IBD care in the given region, the first two authors (from Slovakia and Romania) were approached, and based on their best judgment, they formed an international working group by contacting IBD-specialized gastroenterologists from tertiary IBD centers from several Central and Eastern countries Europe: Croatia, Czech Republic, Hungary, Moldova and Poland. The two leading authors developed a draft questionnaire aimed at analysis of availability, utilization and reimbursement of diagnostic and screening modalities and currently available medications, as well as analysis of selected critical standards of care points based on the current European Crohn’s Disease and Colitis Organization (ECCO) position paper on quality of care in IBD. 15 After the subsequent comments of the senior authors, the questionnaire was discussed and supplemented with the comments of co-authors from individual countries, creating its final version. The final version of the questionnaire (Supplemental Material) consisted of 73 questions divided into three topics: (1) diagnosis, follow-up and screening, (2) medications and (3) IBD centers. The questionnaire was subsequently completed by co-authors from all participating countries (with the possibility to respond to the statements in the ‘yes/no’ form and in the form of a comment). The completed questionnaires were evaluated and the results were then compiled.
Results
Based on the questionnaire survey, we compiled the results from the sections related to IBD care: (1) diagnostics, follow-up and screening, (2) medications and (3) IBD centers. We received answers and comments from representatives of individual countries to all 73 questions.
Diagnostics, follow-up and screening
The results of the survey on availability, use and reimbursement of the main diagnostic, follow-up and screening procedures in IBD centers are summarized in Table 2.
Availability, utilization and reimbursement of diagnostic, follow-up and screening procedures in IBD centers in individual countries.

Reimbursed only in severe cases; therefore, enterography is not performed in all newly diagnosed patients in Moldova.
In the Czech Republic, SBCE is reserved for the indication of high suspicion of CD in patients with negative findings on MRE (same for Slovakia, where a negative findings on upper and lower endoscopy are also required, as well as individual approval by the insurance company).
Patients from Moldova are examined in Romania.
Patients from Poland are examined at the expense of their own or their hospital.
Examination covered by the center or a patient.
Reimbursed only as a part of telemedicine monitoring.
In most countries only available in one specialized center.
Utilization varies between centers (based on preference).
ARM, anorectal manometry; HD, high-definition; IBD, inflammatory bowel disease; IUS, intestinal ultrasound; LTBI, latent tuberculosis infection; MRE, magnetic resonance enterography; SBCE, small bowel capsule enteroscopy; TDM, therapeutic drug monitoring; TPMT, thiopurine methyl-transferase; TRUS, transrectal ultrasound.
Medications
We present the results of the survey on the availability and indications of IBD medications in Table 3. In most participating countries, biological treatment (at least up to the price of biosimilars) is adequately covered by insurance. In Poland, however, treatment must be partly covered by a specific ministerial program due to insufficient insurance coverage. In contrast, in Romania, as the only participating country, the administration of biologics in gastroenterology has a specific reimbursement.
Availability of conventional and advanced IBD medications.

Conventional medications: 5-ASA, azathioprine, methotrexate, 6-mercaptopurine, corticosteroids.
Free choice of the first-line biological medication.
Methotrexate unavailable for IBD indications.
Only available in exceptional cases or at the patient’s expense.
First-line biological treatment option for both indications.
First-line treatment in patients with ulcerative colitis.
First-line treatment in patients with comorbidities.
ASUC, acute severe ulcerative colitis; IBD, inflammatory bowel disease;
IBD centers
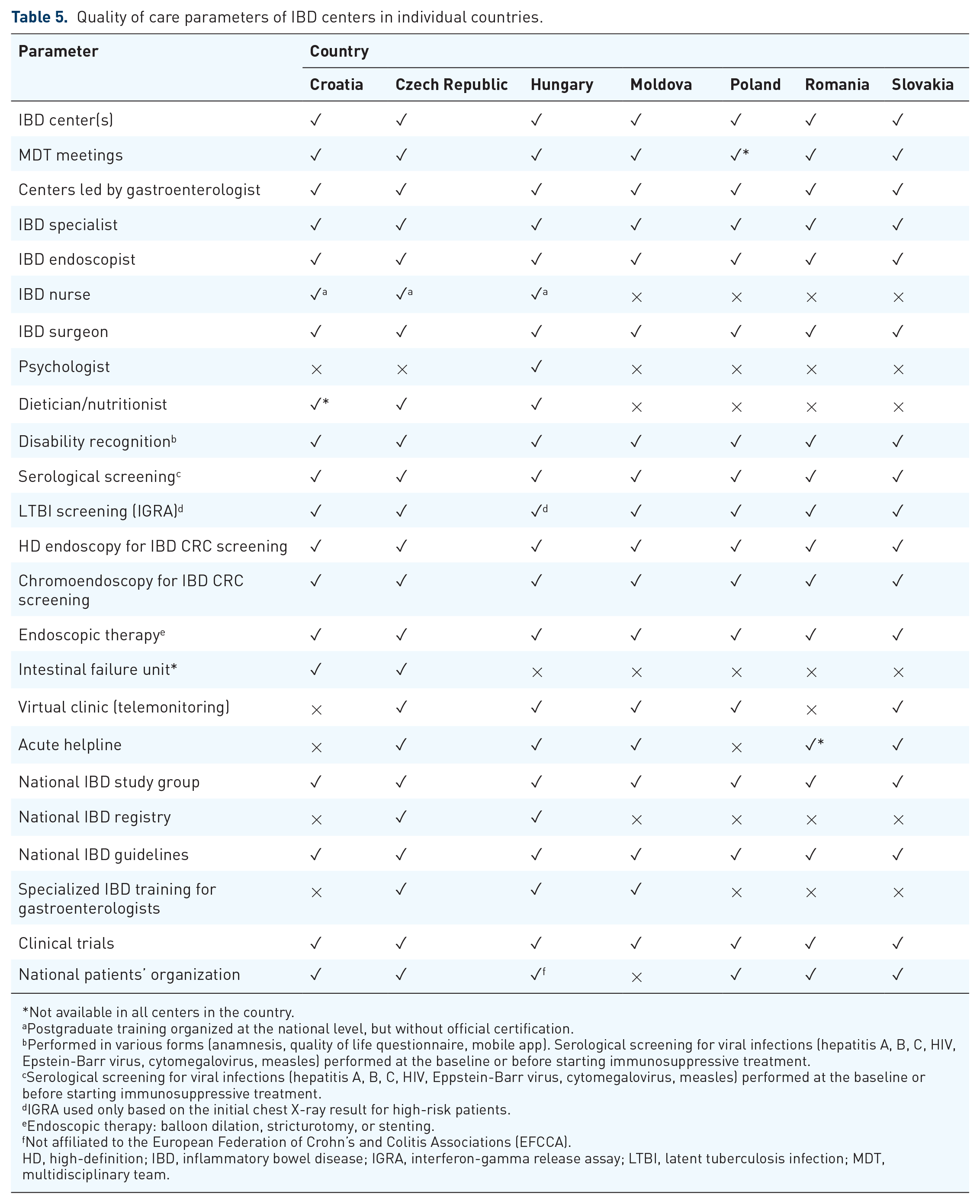
IBD centers are available in all participating countries. Here we present the epidemiological data from the IBD centers (Table 4) and some of their main quality parameters (Table 5). Despite the existence of IBD centers in these countries, IBD patients can also be followed-up by general gastroenterologists. However, patients on biological treatment are centralized exclusively in specialized IBD centers in the Czech Republic, Hungary and Moldova, while administration of biologics by general gastroenterologists is possible in other surveyed countries [only anti-tumor necrosis factor (TNF) biologics in Slovakia].
Epidemiological data from IBD centers from individual countries.

In 2021, 31 centers treated patients with UC and 52 centers treated patients with CD (data from the System and Implementation Analysis Database Platform, BASiW, https://basiw.mz.gov.pl/analizy/zpa/)
N/A, information not available due to the absence of a national IBD database.
CD, Crohn’s disease; IBD, inflammatory bowel disease; UC, ulcerative colitis.
Quality of care parameters of IBD centers in individual countries.
Not available in all centers in the country.
Postgraduate training organized at the national level, but without official certification.
Performed in various forms (anamnesis, quality of life questionnaire, mobile app). Serological screening for viral infections (hepatitis A, B, C, HIV, Epstein-Barr virus, cytomegalovirus, measles) performed at the baseline or before starting immunosuppressive treatment.
cSerological screening for viral infections (hepatitis A, B, C, HIV, Eppstein-Barr virus, cytomegalovirus, measles) performed at the baseline or before starting immunosuppressive treatment.
IGRA used only based on the initial chest X-ray result for high-risk patients.
Endoscopic therapy: balloon dilation, stricturotomy, or stenting.
Not affiliated to the European Federation of Crohn’s and Colitis Associations (EFCCA).
HD, high-definition; IBD, inflammatory bowel disease; IGRA, interferon-gamma release assay; LTBI, latent tuberculosis infection; MDT, multidisciplinary team.
Discussion
Although all countries participating in the survey are members of the ECCO following guidelines published by this organization,16–19 IBD care may partially differ between individual countries. We therefore aimed to identify standards and potential barriers in IBD care in various countries of Central and Eastern Europe, focusing on the availability and reimbursement of diagnostic and therapeutic modalities, differences in treatment options and strategies, as well as the availability and quality of care provided by IBD centers in these countries, as well as postgraduate IBD education and IBD research. We describe the similarities and differences in these aspects between individual countries and try to analyze and evaluate the current situation to deepen the understanding of some of the regional differences across Europe.
As the quality of health care depends also on current epidemiological data, we identified recently published studies from Poland, Hungary and Czech Republic confirming rapidly increasing incidence and prevalence of IBD in these countries.4–8 However, we also identified the first setback in the unavailability of current epidemiological data from other surveyed countries (Moldova, Romania, Croatia and Slovakia), where the last epidemiological studies were done 8–10 years ago and none of them had a national character.9–12
Diagnostics, follow-up and screening
Imaging modalities
Despite the fact that basic diagnostic modalities [endoscopies, magnetic resonance enterography (MRE)/computed tomography enterography] are available in all participating countries, in Moldova, unlike other countries, MRE is due to financial reasons reimbursed only in acute cases and the examination is therefore not carried out in newly diagnosed patients with CD. This may result in misclassification and suboptimal management of the disease, since in over 50% of patients with CD, performing imaging to evaluate the small intestine involvement led to a change in the therapeutic approach.20,21 The absence of small bowel capsule enteroscopy (SBCE) in the diagnosis of CD in some countries (Moldova and Poland – not reimbursed) can raise issues in patients with involvement of the proximal small intestine, for which SBCE seems superior to other diagnostic tools such as MRE. 22 As for the intestinal ultrasound (IUS), its regular use in IBD centers of almost all participating countries is proof of its increasingly important position in the management of IBD. The IUS has an effective use in the follow-up of IBD, both in the evaluation of disease activity and treatment effectiveness and in the diagnosis of complications (especially contrast-enhanced), but also in tight-control strategies.16,23 IUS may also be a more accessible and cheaper alternative to MRE in the diagnosis and follow-up of small bowel involvement in CD, as it has similar diagnostic accuracy in these cases in expert hands. 16 The centralized availability of transrectal ultrasonography and anorectal manometry for the diagnosis of anorectal disorders (fecal incontinence, defecation dyssynergia, symptoms of functional diseases) is also gratifying, given the increasing data and awareness of anorectal disorders in IBD patients.24,25 However, due to the presumed unavailability of specialists in the region, the use of biofeedback therapy is still lagging, despite its high effectiveness in the treatment of defecation disorders. 25
Fecal calprotectin
Given the potential for non-invasive and cost-effective monitoring of disease activity, the routine availability and full reimbursement of fecal calprotectin (FCP) examination should be standard of care for all IBD patients.26–30 The unavailability of calprotectin examination can worsen disease control and thus increase treatment costs even more, which should be a decisive reason for its routine reimbursement in all countries.31–33
Therapeutic drug monitoring
Therapeutic drug monitoring (TDM) in the management of IBD is the standard of care during anti-TNF treatment based on studies confirming its therapeutic value and cost-effectiveness.34–37 The ECCO guidelines do not establish a clear recommendation for the implementation of TDM in clinical practice, because of insufficient evidence for clinical outcome.18,19 Its use and form of implementation in the monitoring of anti-TNF treatment is therefore still a debated and debatable issue, but the most recent meta-analysis shows significant benefits of proactive monitoring (improved endoscopic remission and response, reduced treatment failure, need of surgery and hospitalization). 38 In the surveyed countries of Central and Eastern Europe, TDM is used, despite the fact that it is not covered by insurance in most countries and centers or patients pay for it at their own expense. The preference of reactive and proactive monitoring here also depends on the choice of the center, but reactive TDM is generally used more frequently.
Medications
Conventional medications: when and how do we use them?
Despite the fact that aminosalicylates are not indicated in the treatment of CD, their prescription for this indication is very high. In Europe, aminosalicylates are used by up to 60% of patients with CD, while in Eastern Europe it is up to 90%. 39 According to some studies, there is a group of CD patients with mild course, that may benefit from aminosalicylates monotherapy and do not require a short-term therapeutic step-up.40,41 However, unlike thiopurines, the benefit in the prevention of colorectal cancer (CRC) is uncertain.42,43 The reason for the massive prescription of aminosalicylates for CD in Eastern Europe was probably associated with the poorer availability of biologics in this region in the last two decades.
Biologics and small molecules: availability, reimbursement and therapeutic strategies
While in the past the main healthcare costs for IBD patients were driven by hospitalization and surgeries, today it is biological therapy that accounts for 48–73% of costs for a patient having a 5-year diagnosis of IBD in Europe. 44 Data from the Epi-IBD study showed that between 2010 and 2015, patients in Western European countries were treated with biologics twice as often as in Eastern European countries. 39 Nevertheless, no difference in outcomes such as disease progression, hospitalizations or surgeries, was observed between the two regions. Due to the current adequate availability of diagnostic and advanced therapeutic options in the countries of Central and Eastern Europe, we do not anticipate the future emergence of significant interregional differences within Europe, but rather their disappearance.
As the pool of biologics approved for the treatment of IBD has expanded based on the latest ECCO guidelines, additional drug cost challenges arise. Certain restrictions apply in the surveyed countries, mainly in terms of limiting indications and financial coverage of treatment. A step forward, related to the costs of and access to biologic was brought about by the introduction of anti-TNF biosimilars, which are currently readily available and reimbursed in all of the countries participating in the survey. However, the availability of treatment options could increase even more with the arrival of biosimilars of non-anti-TNF drugs (ustekinumab, vedolizumab, tofacitinib) and also with new drugs currently arriving on the market (upadacitinib, ozanimod, risankizumab and others). In this effervescent context of medicines, it is essential to keep up with the clinical availability of already approved medicines, together with the possibility of free choice of first-line biological treatment, which is still a challenge in some countries of Central and Eastern Europe, as expressed according to the participating countries. Moreover, the relatively long time between the approval of a new drug by the European Medicines Agency and its implementation into clinical practice remains a stumbling block in the availability of new drugs in the region (up to 24 months in the surveyed countries).
IBD centers: quality of care and limitations
Ensuring adequate IBD care should include the existence of expert IBD centers that should meet several quality indicators: specialized staff availability, multidisciplinary team (MDT) collaboration in a form of regular MDT meetings, availability of an infusion facility for parenteral drugs, available outpatient clinic or acute helpline and telemonitoring. 15 IBD centers operating in this way are widely present in the region of Central and Eastern Europe (Table 4). Detailed analysis of their functioning from the international perspective is very difficult and may be subject to quality assessment biases. We therefore decided to select objectively evaluable parameters and focus on their comparison between individual countries.
The first obvious difference between the individual countries is the centralization of the care of IBD patients on biological therapy. It is subject to the Czech Republic, Hungary and Poland, which are also countries with a functional national register of IBD patients on biological treatment. Centralization of care can be beneficial in terms of quality of care for more challenging patients, tracking epidemiological trends and conducting studies at the national level, and also in easily obtaining centralized data related to treatment and patient outcomes.
Our analysis of IBD centers thus represents an evaluation of the level of care provided especially to demanding patients in the given countries, while the level of care for a large group of patients followed by general gastroenterologists remains unclear.
The role of MDTs
Because patients’ particular disease profile can highly differ among individuals, the options for treatment choice and patients’ management can vary among various types of clinicians; therefore, the multidisciplinary approach is important for reaching consensus regarding patient management and contributing to standardizing IBD care. This kind of approach involves various types of specialists in IBD care: gastroenterologist, nurse, surgeon, radiologist, pathologist, dietitian, psychologist, etc. Together with patient empowerment through dedicated organizations and MDT, the approach to IBD management can lead to improving IBD care. 45 The implementation of MDTs dedicated to IBD patients could be useful in several clinical settings, such as surgery-related decisions, determining the appropriate timing or selection of medications, therapeutic approach for patients experiencing extraintestinal manifestations, offering nutritional, psychosocial support and the transition from pediatric to adult clinical care. 46 IBD centers in all participating countries cover most of the essential specialties. The limitation remains, in almost all countries, the unavailability of integrated dietary and psychological counseling.
The limited availability of dietitians within the MDT can be an important drawback, considering their role in assessing nutritional status, measuring dietary intake and offering personalized recommendations on oral/enteral or parenteral nutritional support in both, inpatient and outpatient care. The European Society for Clinical Nutrition and Metabolism (ESPEN) guidelines underline the importance of the active contribution of a dedicated dietician or nutritionist as part of the MDT, offering individual recommendations to each IBD patient. 47 The availability of a dedicated dietician was reported only in Croatia, Hungary and the Czech Republic, while in the other participating countries the nutritional aspects of IBD care are covered by the gastroenterologists, during regular visits for disease evaluation. However, due to the need to cover several aspects during the visit, counseling provided by a gastroenterologist can be significantly limited by the specialist’s time availability and thus the insufficient depth of the consultation and subsequent follow-up. Therefore, it is important that specialized clinical nutrition guidelines, such as the ESPEN guidelines and the International Organization for the Study of Inflammatory Bowel Disease dietary guidance, offer the scientific background and support to gastroenterologists to take this role in the absence of a dedicated nutritionist. 47
Specialized psychological support is needed to improve identifying and managing psychological distress among IBD patients, to minimize its negative impact on patients’ care. This should especially include addressing concerns of patients who are newly diagnosed, such as disease impact, intimacy issues, stigma, but also to promptly identify and optimally manage psychological burden associated with this chronic disease. 48 In the scenario of the unavailability of a specialized psychologist, this position is taken over by a doctor or a nurse, who, however, are often unable to replace the patient’s need for professional advice. The exception in the region is Hungary already offering integrated psychological assistance for IBD patients in the IBD centers. However, there are several initiatives to promote psychological support for IBD patients, involving psychologists, clinicians and the patients’ association, as is the case of the Romanian IBD patients’ association, which has launched a dedicated support program.
The availability of intestinal failure units is also gaining importance, since there are reports on increasing numbers of home parenteral nutrition (HPN) patients in the context of IBD. 49 However, availability of intestinal failure units was limited in most of the countries, except Croatia and the Czech Republic, that have intestinal failure units in place. Considering the epidemiological trends, we assume that the region will gradually see an increase in patients requiring HPN, and the need for specialized care, including intestinal failure units, will gradually be optimized.
IBD endoscopy
Both UC and CD affecting the colon are associated with an increased risk of CRC; therefore, the CRC screening strategy for this group of patients is guided by specific recommendations. Based on European Society of Gastrointestinal Endoscopy guidelines from 2019, it is recommended that patients with long-standing colitis should undergo neoplasia surveillance by pancolonoscopy with the routine use of chromoendoscopy. 50 According to the results of our survey, high-definition (HD) endoscopy is available and regularly used in the detection of dysplasia at least in IBD centers in all participating countries of our survey, with a clear preference for virtual chromoendoscopy over dye-enhanced chromoendoscopy. It should also be mentioned that in all surveyed countries, IBD endoscopies, including surveillance colonoscopies, are also performed by general gastroenterologists. Although, based on the Spanish study, there was no significant difference in dysplasia detection between expert and non-expert endoscopists (18.5% versus 13,1%), 51 the availability of HD endoscopy and chromoendoscopy may be lower among general gastroenterologists in our region due to the financial burden. Therefore, the question arises whether IBD surveillance colonoscopies in this high-risk patient group should be performed outside IBD centers.
IBD centers are led by gastroenterologists. What is the role of a nurse in our region?
The presence of IBD nurse positions has been included to a greater extent to represent standard of care within specialized IBD clinics and implementing a service with IBD nurses has been reported to improve the outcomes of IBD care.52,53 However, their value in the acute hospital setting is still questioned in the context of available IBD centers with dedicated gastroenterologists. It has been debated whether the addition of IBD nurses improves outcomes, since most data are based on short-follow-up studies after implementation.54,55 The positive outcomes of the addition of IBD nurse were supported by one study with a longer follow-up period (8 years) of the IBD nurse position in a well-established setting, by reducing hospital admissions, providing advice and evaluation through virtual clinic (phone and email). 56 In all participating countries, IBD centers are led by an IBD specialist and the nurse is mainly responsible for direct patient care, documentation and administration. Due to the above-discussed lack of psychological and dietary counseling in the region, it is often the nurses who partially participate in patient education and often psychological support as well. At the same time, in most centers, they are responsible for the virtual clinic, where they provide patients with basic advice and are thus often the first line in their care. Adequate education should therefore be an essential part of their specialization. In our region, the concept of IBD nurses is only starting to spread slowly, and postgraduate training organized at the national level is only available in a few countries and that without official certification.
IBD education and research
In the United States, as part of the IBD specialization, it is possible to complete a dedicated IBD training, entitled IBD fellowship, which is a specialized 1- and 2-year combined clinical and research training program for gastroenterologists. In Europe, such structured programs do not exist, and doctors-in-training are dependent on only a few weeks of experience during internships in university hospitals. Short-term IBD trainings are organized in this way in some of the participating countries to this survey. However, 2- to 3-month internships mediated by ECCO are available in Europe (only for a few participants a year). As for IBD study groups, they are available and organized with the support of national societies in most countries. Their role should consist in the unification of procedures at the national level, the preparation of national recommendations and the organization of scientific work, including the management of the IBD registry. However, national registries of IBD patients on biologics are only available in the Czech Republic and Hungary.
Conclusion
Regarding the quality parameters we focused on, we identified that IBD care is in accordance with international guidelines in most of the participating countries from Central and Eastern Europe, including the presence of established IBD centers, but mainly with limited access to expert dietary and psychological counseling. Although there is evidence that updated epidemiological data, available patient registries as well as diagnostic and monitoring tools (FCP and TDM) not only increase the quality of the provided health care, but also reduce the financial burden in the IBD care, there is suboptimal availability of some of these tools in most countries of Central and Eastern Europe, although currently without indications of impact on disease outcomes. The financial burden appears to be the main setback in the availability of non-essential diagnostic and monitoring methods and of newer therapeutic agents in some of the countries.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231174290 – Supplemental material for Barriers in inflammatory bowel disease care in Central and Eastern Europe: a region-specific analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848231174290 for Barriers in inflammatory bowel disease care in Central and Eastern Europe: a region-specific analysis by Michal Prokopič, Georgiana Gilca-Blanariux, Peter Lietava, Anca Trifan, Anna Pietrzak, Agata Ladic, Marko Brinar, Svetlana Turcan, Tamás Molnár, Peter Bánovčin and Milan Lukáš in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
