Abstract
Background:
With a 15% incidence, KRAS is one of the most common mutations in biliary tract cancer (BTC) and is a poor prognostic factor. Immune checkpoint inhibitors (ICIs) as salvage therapy have modest activity in BTC.
Objectives:
There are limited data on the efficacy of ICIs according to KRAS mutation in BTC. We evaluated the efficacy of ICIs in BTC patients with or without KRAS mutations.
Design:
Retrospective observational study.
Methods:
We conducted molecular profiling in BTC patients who received ICIs as salvage therapy. The expression of programmed death ligand 1 (PD-L1) on tumor cells was assessed using immunohistochemistry. The TruSightTM Oncology 500 assay from Illumina was used as a cancer panel. We analyzed overall survival (OS) and progression-free survival (PFS) of ICI in BTC patients according to KRAS mutation and PD-L1 expression.
Results:
A total of 62 patients were included in this analysis. The median age was 68.0 years; 47 patients (75.8%) received pembrolizumab and 15 (24.2%) received nivolumab as salvage therapy. All patients received gemcitabine plus cisplatin as the frontline therapy, and 53.2% received fluoropyrimidine plus oxaliplatin (FOLFOX) before ICI. The median number of lines of prior chemotherapy was 2.5. The KRAS mutation was found in 13 patients (19.1%), and 28 patients (45.2%) showed 1% or more of tumor cells out of visible tumor cells positive for PD-L1. There was no statistical correlation between KRAS mutation and PD-L1 expression. The median OS and PFS with ICI were 5.6 [interquartile range (IQR): 3.3–8.0] and 3.8 (IQR: 3.0–4.5) months, respectively. There were no statistically significant differences in PFS with ICIs according to KRAS mutation (mutant type versus wild type) and PD-L1 expression (positive versus negative). In subgroup analysis, patients with both KRAS mutation and PD-L1 positivity had longer PFS compared with patients with KRAS mutation and PD-L1 negativity (10.1 versus 2.6 months, p = 0.047). This finding was not shown in patients with wild-type KRAS.
Conclusion:
Our analysis suggested that PD-L1 expression might be a useful biomarker for ICIs in BTC patients with KRAS mutation but not in those with wild-type KRAS.
Introduction
Kirsten rat sarcoma viral oncogene homolog (KRAS) is one of the most common oncogenes among all cancers. KRAS has been thought to be an undruggable target; however, recently, KRAS-targeting agents, which are designed to target the KRAS p.G12C mutation, received approval based on encouraging clinical results. With the start of the success of sotorasib in non-small-cell lung cancer (NSCLC), 1 sotorasib showed improved treatment outcomes in previously treated pancreatic cancer with 21% of objective response and 6.9 months of overall survival (OS). 2 Adagrasib, another agent targeting KRAS p.G12C mutation, also proved clinical efficacy in previously treated colorectal cancer with or without cetuximab. 3
The KRAS protein can activate multiple signaling pathways, including the rapidly accelerated fibrosarcoma, mitogen-activated protein kinase (MEK), extracellular regulated protein kinases signaling pathway, and the phosphoinositide 3-kinase/protein kinase B (Akt)/mammalian target of rapamycin signaling pathway. KRAS pathway activation leads to cell proliferation, differentiation, and migration and inhibits apoptosis, all of which are hallmarks of cancers. 4 During pathway activation, many inflammatory cytokines and chemokines are secreted, facilitating an inflammatory microenvironment and inducing oncogenesis, immune escape, and evasion. 5
Biliary tract cancer (BTC) has a high incidence of KRAS mutation (12.7% 4 ), and KRAS is a poor prognostic factor for BTC. 6 Despite the only 10-month median survival of gemcitabine plus cisplatin (GP), the first-line standard therapy for BTC has not changed for more than 10 years. 7 Recently, TOPAZ-1 trial (Durvalumab or Placebo in Combination With Gemcitabine/Cisplatin in Patients With 1st Line Advanced Biliary Tract Cancer) showed that adding durvalumab to GP increased the median OS to 12.8 months. 8 However, there is few data for the efficacy of immune checkpoint inhibitors (ICIs) according to KRAS mutation in BTC. Herein, we analyzed the efficacy of ICIs according to KRAS mutation in BTC.
Method
Patient selection
This analysis retrospectively included patients who were diagnosed with BTC and received ICI as salvage therapy between March 2020 and August 2022 at our institute. Also, all analyzed patients had available data on programmed death ligand 1 (PD-L1) expression and next-generation sequencing. The following clinicopathologic characteristics were analyzed: age, sex, performance status, primary tumor site, disease status, disease classification, treatment history, and response to ICI.
Immunohistochemistry of PD-L1
The expression of PD-L1 on tumor cells was assessed using immunohistochemistry (IHC). Tumor samples obtained through endoscopic biopsy, percutaneous needle biopsy, or surgical resection at initial diagnosis or progression were used. Tissue sections were freshly cut into 4-μm sections, mounted on Fisherbrand Superfrost Plus Microscope Slides (Thermo Fisher Scientific, Waltham, MA, USA), and dried at 60°C for an hour. IHC staining was carried out on a Dako Autostainer Link 48 system (Agilent Technologies, Santa Clara, CA, USA) using a Dako PD-L1 IHC 22C3 PharmDx kit (Agilent Technologies, Santa Clara, CA, USA) with an EnVision FLEX visualization system. Then the samples were counterstained with hematoxylin according to the manufacturer’s instructions. PD-L1 expression was considered positive if it was observed in 1% or more of tumor cells out of visible tumor cells. 9
TruSightTM oncology 500 assay
Forty (40) ng of DNA was quantified with the Qubit dsDNA HS Assay (Thermo Fisher Scientific, Waltham, MA, USA) on the Qubit 2.0 Fluorometer (Thermo Fisher Scientific, Waltham, MA, USA) and then sheared using a Covaris E220 Focused-ultrasonicator (Woburn, MA, USA) and the 8 microTUBE–50 Strip AFA Fiber V2 following the manufacturer’s instructions. The treatment time was optimized for formalin-fixed paraffin-embedded material. The treatment settings were as follows: peak incident power (W): 75; duty factor: 15%; cycles per burst: 500; treatment time (s): 360; temperature (°C): 7; water level: 6. For DNA library preparation and enrichment, the TruSightTM Oncology 500 Kit (Illumina Inc., San Diego, CA, USA) was used following the manufacturer’s instructions. Post-enriched libraries were quantified, pooled, and sequenced on a NextSeq 500 (Illumina Inc., San Diego, CA, USA). The quality of the NextSeq 500 (Illumina) sequencing runs was assessed with the Illumina Sequencing Analysis Viewer (Illumina Inc., San Diego, CA, USA). Sequencing data were analyzed with the TruSight Oncology 500 Local App Version 1.3.0.39 (Illumina Inc., San Diego, CA, USA). The TruSightTM Oncology 500 is a comprehensive tumor profiling assay designed to identify known and emerging tumor biomarkers, including small variants, splice variants, and fusions. Importantly, the TruSightTM Oncology 500 measures tumor mutational burden (TMB) and microsatellite instability (MSI), features that are potential key biomarkers for immunotherapy. TMB was reported as mutations per megabase (Mb) sequenced, and high TMB was defined as more than 10 mutations per Mb (⩾10 Mut/Mb).
Statistical analysis
The clinical features and treatment outcomes were analyzed, and categorical variables were evaluated using the chi-square test and Fisher’s exact test. OS was defined as the time from the first day of ICI to death from any cause, and living patients were censored at the time of analysis. Progression-free survival (PFS) was defined as the time from the first day of ICI to the date of confirmed progressive disease according to RECIST v1.1. We analyzed OS and PFS of ICI in BTC patients according to PD-L1 expression and KRAS mutation. Survival was estimated based on Kaplan–Meier curves and compared using a log-rank test. p Values from two-sided statistical tests were considered statistically significant at p < 0.05. Survival analyses were performed using IBM PASW version 24.0 software (SPSS Inc., Chicago, IL, USA).
We have followed TRIPOD guidelines. The study was conducted and reported according to the TRIPOD (Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis) statement. 10
Results
Clinical features
A total of 62 BTC patients were included in this analysis. In all, 38 (61.3%) patients were male, and the median age was 68.0 [interquartile range (IQR): 59.3–73.7] years. The primary site was intrahepatic in 31 (50.0%) patients, extrahepatic in 21 (33.9%), and gallbladder in 10 (16.1%). Overall, 55 (88.7%) patients had metastatic disease, and 7 (11.3%) had locally advanced disease when they were considered in a palliative setting. Pembrolizumab was administered as a salvage therapy in 47 patients (75.8%), and nivolumab was given in 15 (24.2%). All patients received GP as the frontline therapy, and 53.2% received fluoropyrimidine plus oxaliplatin (FOLFOX) before ICIs. The median number of lines of prior chemotherapy was 2.5. TMB-high tumors were found in 9 (14.5%) patients and TMB-low tumors in 47 (75.8%) patients. Only one (1.6%) patient had an MSI-H tumor (Table 1).
Clinicopathologic characteristics of the patients.

ECOG, Eastern Cooperative Oncology Group; GP, gemcitabine + cisplatin; IQR: interquartile range; MSI, microsatellite instability; MT, mutant type; TMB, tumor mutation burden; WT, wild type.
KRAS mutation and PD-L1 expression
The KRAS mutation was found in 13 patients (19.1%). The types of mutations were as follows: G12A in 2 (15.4%) patients, G12C in 1 (7.7%), G12D in 3 (23.1%), G12R in 1 (7.7%), G12V in 2 (15.4%), G13D in 1 (7.7%), Q61H in 1 (7.7%), Q61R in 1 (7.7%), and R68S in 1 (7.7%). We summarized the clinical features of patients with KRAS mutation in Figure 1. In these patients, PD-L1 positivity was detected in 28 (45.2%), showing no statistical correlation between KRAS mutation and PD-L1 expression (p = 0.589, Table 2).

Summary of the clinicopathologic characteristics of patients with KRAS mutation.
PD-L1 status and KRAS status.

MT, mutant type; PD-L1, programmed death ligand 1; WT, wild type.
Treatment outcomes according to PD-1 expression, KRAS mutation, and TMB
The median OS and PFS with ICI were 5.6 (IQR: 3.3–8.0) and 3.8 (IQR: 3.0–4.5) months, respectively. Between mutant and wild-type KRAS, there were no statistically significant differences in OS and PFS with ICIs (OS; 5.0 versus 5.6 months, p = 0.775, PFS; 4.0 versus 3.8 months, p = 0.724, Figure 2(a)). The median OS and PFS between patients with PD-L1 positivity and those with PD-L1 negativity were not statistically different (OS; 6.1 versus 5.2 months, p = 0.812, PFS; 3.6 versus 4.1 months, p = 0.785, Figure 2(b)). However, in patients with KRAS mutation, those who were also PD-L1 positive had longer PFS than patients who were PD-L1 negative (10.1 versus 2.8 months, p = 0.047) (Figure 3(a)). This finding was not observed in patients with wild-type KRAS (Figure 3(b)). The tumor responses to ICI are presented in Tables 3 to 5.
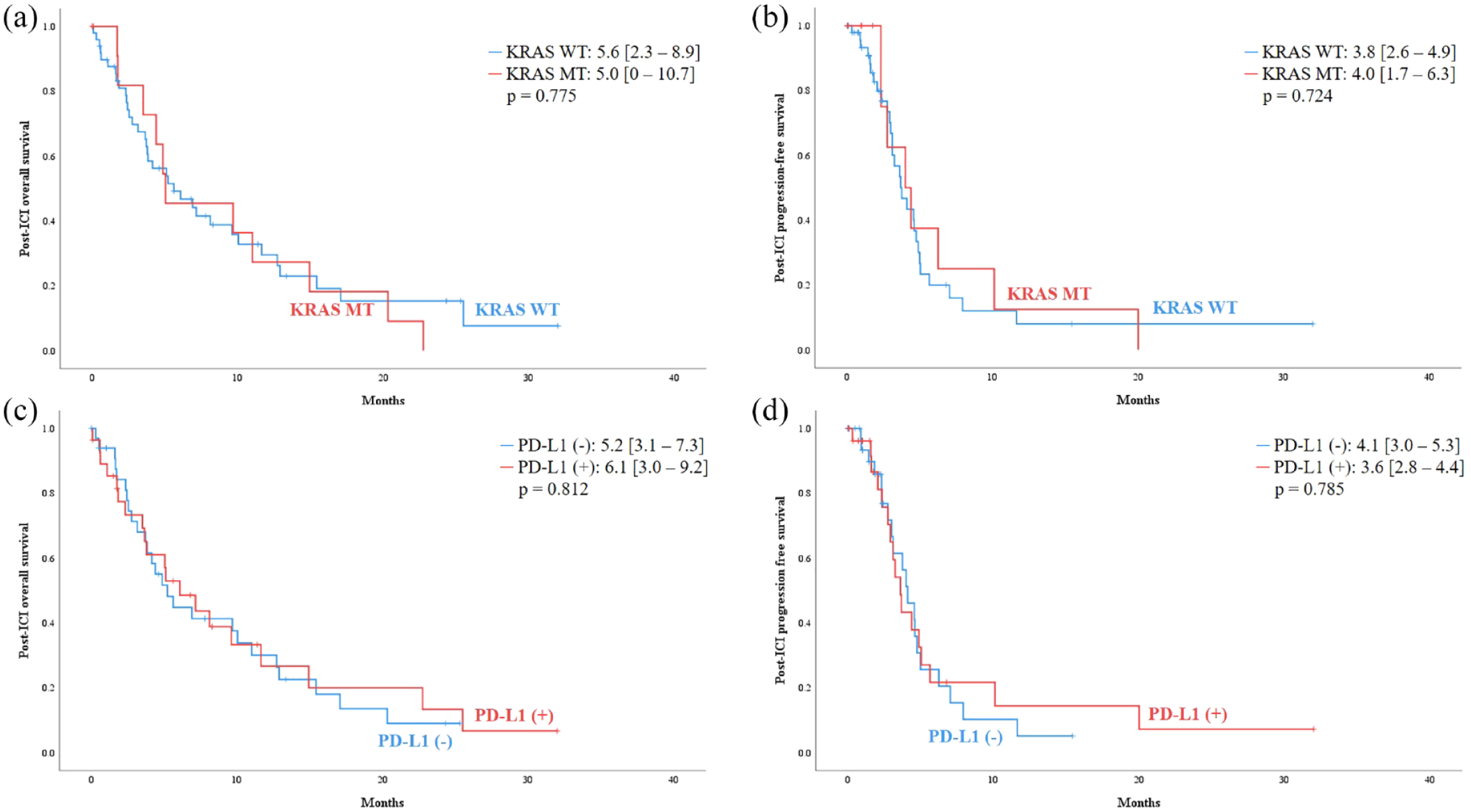
(a) OS and (b) PFS of patients with KRAS wild-type or mutation-type tumors. (c) OS and (d) PFS of patients with positive or negative PD-L1 expression.
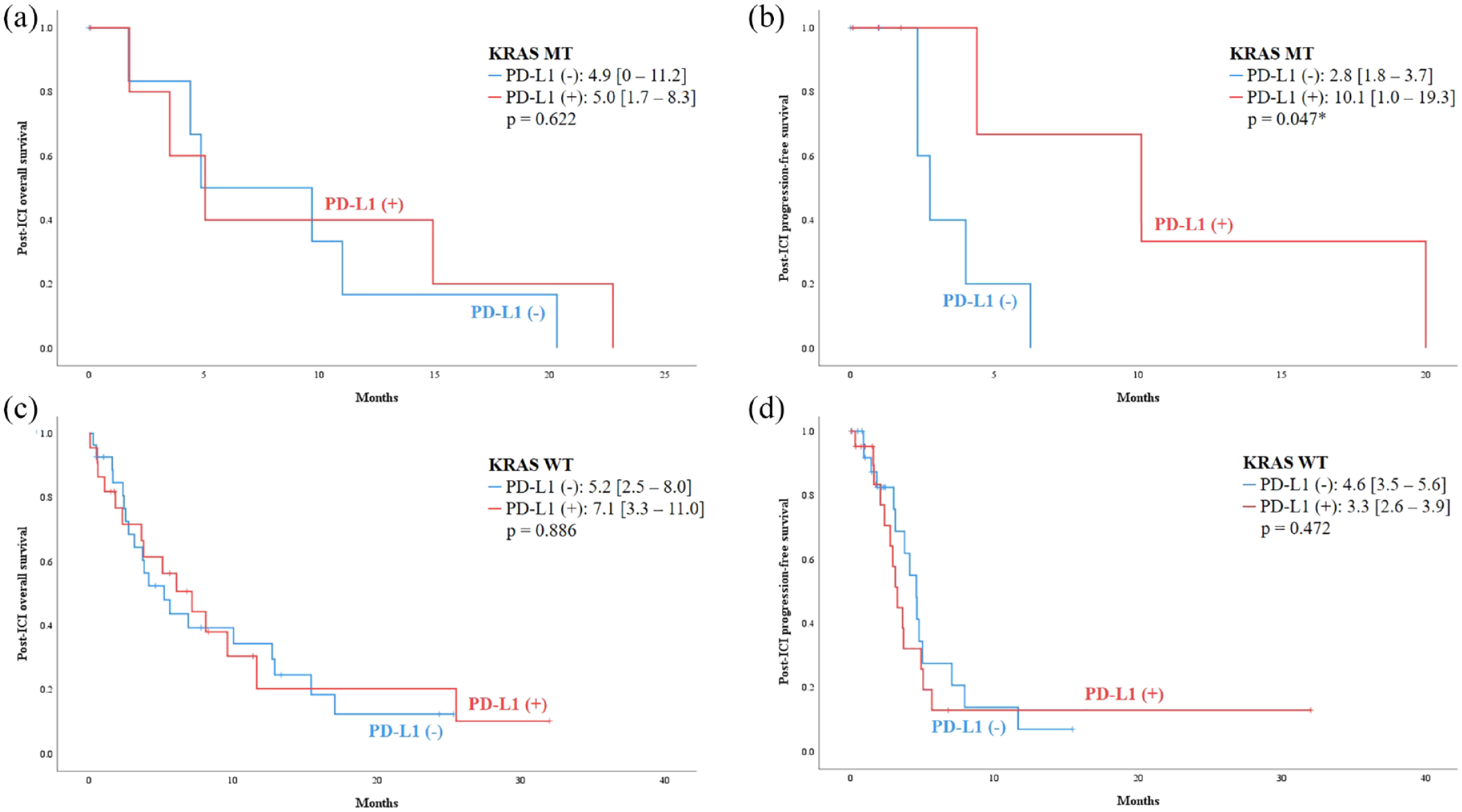
Subgroup analysis. (a) OS and (b) PFS of patients with KRAS mutation type according to PD-L1 expression. (c) OS and (d) PFS of patients with KRAS wild type according to PD-L1 expression.
Treatment response according to KRAS status.

MT, mutant type; WT, wild type.
Treatment response according to PD-L1 status.

PD-L1, programmed death ligand 1.
Treatment response according to KRAS and PD-L1 status.

MT, mutant type; PD-L1, programmed death ligand 1; WT, wild type.
There were no statistically significant differences in OS and PFS between TMB-high and TMB-low tumors (OS; 10.3 versus 4.5 months, p = 0.235, PFS; 5.7 versus 4.5 months, p = 0.333).
Discussion
Among the 62 BTC patients herein, the KRAS mutation was found in 14 (22.6%), and PD-L1 positivity was detected in 28 (45.2%). There was no statistical correlation between KRAS mutation and PD-L1 expression. In patients with PD-L1 expression, those who also had KRAS mutation showed longer PFS with ICIs compared with patients with PD-L1 expression only. However, this difference was not observed in patients with wild-type KRAS. This finding suggests that PD-L1 expression might be a novel predictive marker for ICIs, especially in BTC patients with KRAS mutation.
PD-L1 expression has been used as a potential predictive biomarker for ICI. Nonetheless, negative PD-L1 expression does not mean that patients would not respond to ICI. We observed that PD-L1 status did not affect survival including OS or PFS of patients. Similarly, in a phase 2 study of nivolumab in refractory BTC patients, PD-L1 expression was not correlated with survival. 9 The TOPAZ-1 trial showed similar efficacy between PD-L1-positive and PD-L1-negative patients. 8
There are some data showing a correlation between KRAS mutation and PD-L1 expression. In NSCLC, several studies have reported significantly higher PD-L1 expression in NSCLC patients with KRAS mutation,11–15 and the mechanisms are being researched.16,17 It is not revealed much in BTC; however, some studies showed that KRAS mutation induces the expression of PD-L1.18,19 Our data did not show any correlation between KRAS and PD-L1 in BTC patients; However, as this study only included small number of patients, further studies would be needed.
In this analysis, patients with both KRAS mutation and PD-L1 expression had better PFS than patients with PD-L1 expression and wild-type KRAS. Some studies have suggested KRAS mutation as a potential biomarker for ICI use. A meta-analysis reported that ICI as salvage therapy improved OS in NSCLC patients with KRAS mutation but not in NSCLC patients with wild-type KRAS.20,21 Currently, combination treatments with KRAS inhibitors are being tried, such as those with agents targeting related singling pathways such as RTK, SHP2, SOS1, or MEK. 4 As KRAS mutation fosters an immunosuppressed tumor microenvironment (TME), KRAS inhibitors change the TME from immunosuppressed to immunoreactive.5,22 Therefore, a combination of KRAS inhibitors and ICI should be tried.
In conclusion, we observed that KRAS mutation and PD-L1 expression did not predict the efficacy of ICI in patients with BTC. However, PD-L1 expression might be a useful biomarker of ICI use in BTC patients with KRAS mutation but not in those with wild-type KRAS. Furthermore, we suggest that treatment with ICI plus anti-KRAS therapy might be valuable in BTC. However, as this study only included a small number of patients, further studies would be needed.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231170484 – Supplemental material for The efficacy of immune checkpoint inhibitors in biliary tract cancer with KRAS mutation
Supplemental material, sj-docx-1-tag-10.1177_17562848231170484 for The efficacy of immune checkpoint inhibitors in biliary tract cancer with KRAS mutation by Sun Young Jeong, Jung Yong Hong, Joon Oh Park, Young Suk Park, Ho Yeong Lim, Jae Yeon Jang, Youngkyung Jeon and Seung Tae Kim in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
