Abstract
Latin America (LATAM) is a large region comprising 47 countries and territories. Each one carries a different cultural and historical background, diverse political systems, and a particular approach to healthcare management. There is a lack of high-quality data on the epidemiology of inflammatory bowel diseases (IBD) in this region, including broad and detailed information about the penetration of biological and advanced therapies as treatment strategies. From an IBD perspective, patients experience, in general, fragmentations and inequities in the healthcare systems, with different and usually delayed access to qualified health services. This review explores the barriers to accessing IBD care throughout LATAM. The authors compiled data from multiple sources, such as studies focusing on epidemiology, biological penetration, and surgical rates. In addition, overall access to IBD treatments was assessed through a questionnaire distributed to physicians in LATAM via email and direct messaging to capture local perspectives.
Introduction
Latin America (LATAM) is a large and diverse region. The term does not have a precise definition, but it is commonly used to describe South America, Central America, Mexico, and the islands of the Caribbean. 1 According to the latest Human Development Index (HDI) for countries in LATAM, as included in a recent United Nations Development Program’s Human Development Report, the region comprises countries that fall into four broad categories based on their HDI: very high, high, medium, and low human development. 2 In addition, LATAM is the most unequal region globally, as reported by the United Nations Economic Commission for LATAM and the Caribbean. 3 Inequality in LATAM has deep historical roots and has been reproduced and transmitted through generations. 4
Inflammatory bowel diseases (IBD) comprise ulcerative colitis (UC) and Crohn’s disease (CD), chronic inflammatory disorders of the gastrointestinal tract characterized by periods of active disease and remission of varying duration and severity. 5 Due to their usual early onset, fluctuating disease course, and unpredictable prognosis, IBD is associated with a substantial economic burden on patients and society. A higher IBD prevalence is classically reported in North America and Europe, with prevalence rates reaching 249 and 505 per 100,000 people, respectively. 6 According to the evolution in the epidemiological stages of IBD, it is expected that developing countries are in the emergence stage, during which sporadic cases of IBD are documented, and newly industrialized countries are in the acceleration of incidence stage, during which incidence rises, and prevalence is relatively low.7,8 The exact incidence and prevalence of IBD in LATAM remain unclear due to the granularity of available data; however, an increment in the cumulative prevalence has been observed after a growing incidence. 9
Access to health services is an indicator used to measure a country’s standard of living, as it reflects the efficiency and strength of a healthcare system. 10 IBD is associated with an excessive economic burden, with evidence from the early 2000s indicating that the primary cost drivers of IBD management were surgeries and hospitalizations.11,12 However, more recent analyses pointed out that healthcare costs of IBD have shifted toward medical treatment options, mainly due to the utilization of biological agents.13,14
This narrative review explores potential barriers to access to IBD care throughout LATAM. Due to the lack of country-specific IBD registries assessing the incidence/prevalence of the disease in the entire population, authors compiled data via multiple sources, such as studies focusing on epidemiology, biological penetration, and surgical rates.
Methods
In this article review, relevant publications on Medline/PubMed and SciELO were identified up to August 30, 2022, with the terms: ‘Latin America’, ‘Inflammatory bowel disease’, ‘access’, ‘access to medications’, ‘epidemiology’, ‘health care quality’, ‘evaluation’, ‘high-cost medications’. The authors consulted the websites of the different Ministries of Health from each country in LATAM and the Caribbean, the Pan-American Health Organization, the World Health Organization, and the Organization for the Economic Co-operation and Development, and a search was performed with the same terms. The authors assessed and selected eligible publications in English, Spanish, Portuguese, and French; discrepancies were discussed among all the co-authors.
In addition, a six-item questionnaire on biologics and advanced therapies in IBD was developed using Google Forms. It was distributed to gastroenterologists and IBD specialists in LATAM via email and direct messaging (between one and three physicians per country were attempted to be contacted). The survey was designed to obtain a genuinely regional snapshot from the physicians’ perspective of available therapies, coverage, and overall opinion of medication access. All countries and territories with at least one response were included. This survey is detailed in the supplementary material.
Epidemiology of IBD in LATAM
Available data indicate remarkable heterogeneity in epidemiology across countries that may be driven by factors such as historical colonization, culture, socioeconomic status, genetic origin, lifestyle, and diet. Similarly to the western world, regions within LATAM that are more developed, industrialized, and exposed to a westernized lifestyle reported a higher prevalence of CD. Better access to healthcare can also influence IBD diagnosis. In general, it has been demonstrated that the prevalence of IBD in LATAM has been expanding after decades of increasing incidence. 15 In addition, a systematic review by Selvaratnam et al. reveals an expressively higher burden of IBD compared to Asian countries. 8
The largest population of patients with IBD is found in Brazil. A recently published study 16 demonstrated data from 212,026 patients with an IBD diagnosis in 2020 (UC: n = 119,700; CD: n = 71,321; IBD-U: n = 21,005) from the public healthcare system. Stable IBD incidence rates were observed from 2012 to 2020. In UC, incidence rates increased significantly, while in CD, the incidence rates dropped significantly in the same period. This study concluded that the incidence of CD was decreasing whereas UC was increasing, leading to stabilization in the incidence of IBD from 2012 to 2020 in Brazil. The cumulative prevalence of IBD has been rising, with 0.1% of Brazilians living with IBD in 2020. 16
Balderramo et al. recently published a prospective study describing the epidemiological profile of patients with IBD enrolled in a registry in Córdoba, Argentina, from 2014 to 2019. A total of 655 patients (561 with UC and 88 with CD) were included. The age-adjusted incidence rates of IBD, UC, and CD were 3.67, 2.99, and 0.68 per 100,000 person-years, respectively, while the IBD prevalence was 68.8/100,000 inhabitants (58.9/100,000 for UC and 9.23/100,000 for CD). 17
Juliao-Baños et al. analyzed 2291 patients with IBD in Colombia, of whom 1813 (79.1%) had UC, 456 (19.9%) had CD, and 22 had unclassified IBD (0.9%). Like in most Latin American countries, there is a predominance of UC over CD in Colombia. 18 Accordingly, the systematic review by Kotze et al. shows that the incidence and prevalence of IBD have been steadily increasing in LATAM, with the UC:CD ratio exceeding 1 in almost all studied regions. 15 Table 1 summarizes relevant data from the studies mentioned above.
Relevant data from epidemiological studies.

–, data not reported; CD, Crohn’s disease; IBD, inflammatory bowel disease; UC, ulcerative colitis.
Latin American studies challenge the results from previous global reviews, which consider the incidence and prevalence of IBD in the region extremely low. Rising prevalence rates and low mortality will likely result in a continued burden of IBD in the region. 19
Biological therapies penetration in LATAM
Penetration of biological therapies in Brazil
A review published in 2019 included 19 studies of patients treated with adalimumab and infliximab in LATAM, of which 8 were Brazilian studies. 20 Those studies described cohorts of patients with IBD; two included CD cases exclusively21,22 and one was only with UC patients. 23 The use of anti-Tumor Necrosis Factor (TNF) in CD ranged from 4.8% in the study by Bechara et al. to 43.4% in the publication by Lima-Martins. 24 On the other hand, the frequency of biological use was lower in UC, ranging from 1.5% in 2015 23 to 5.4% in 2017. 25 Only one study described the penetration of anti-TNF agents in IBD, with 37.93% of patients using biological drugs. 26 Stratifying by anti-TNF type, Arantes et al. reported more patients using adalimumab than infliximab in CD and equal use of both agents in UC. 25 The authors concluded that the penetration of anti-TNF agents in Brazil is comparable to the rest of the world in CD but lower in UC. 19
A recently published study retrospectively analyzed 625 IBD patients (416 CD, 190 UC, 19 IBD-U) in the State of São Paulo, 27 with 54% of the patients on a biologic at the time of data collection. Overall, 63.7% of CD patients were using biologics compared to 34.7% of patients with UC; this study only reported patients using anti-TNF agents.
Due to the large discrepancy in the data, population-based studies are necessary to reveal the actual penetration of biological therapies in the Brazilian population. The studied cohorts, although important, reflect only the local reality of each referral unit and should not be extrapolated to a national level.
Penetration of biological therapies in South America
Studies assessing medication use in IBD in Spanish-speaking South America in the biological era are somewhat scarce. In the aforementioned Colombian study, a higher biologic penetration was noted in CD (47.3%) as compared with UC (18.5%) [odds ratio (OR) 3.93; 95% confidence interval (95% CI) 3.14–4.93; p < 0.001]. The most frequently used biologics as first-line therapies were infliximab (54.8%) and adalimumab (56.6%) in UC and CD, respectively. The authors identified factors associated with higher biologic use, including age younger than 40 years, severe and extensive UC, and non-inflammatory behavior in CD. 18
In the prospective cohort study by Balderramo et al. from Córdoba, Argentina, there was a significantly higher biologic use in patients with CD compared with UC (36.4 versus 9.1%, p < 0.001). 17 Lasa et al. assessed treatment adherence in IBD patients in a multicenter, cross-sectional study in seven centers from Argentina. Overall, 447 and 135 patients with UC and CD were included. Biologic use was seen in 24.8% (111/447) and 33.3% (45/135) of the patients with UC and CD, respectively (p = 0.06). 28 Inadequate adherence to biologics was seen in 15.3% of patients (19.5% in UC versus 24% in CD, p = 0.6). The risk of inadequate adherence to biologics was significantly lower than oral 5-aminosalicylates (OR 4.1; 95% CI 2.7–6.3) and thiopurines (OR 1.7; 95% CI 1.1–2.7). 28
In a multicenter, cross-sectional study with a 3-year retrospective data collection period conducted in Argentina, 246 patients with moderate-to-severe IBD (41% with CD and 59% with UC) were included. 29 The cumulative use of biologics during the retrospective period was high (79.2% in CD and 33.8% in UC). However, this might be influenced by the fact that all participating centers were tertiary hospitals and only included patients with moderate-to-severe IBD. 29
Arcucci et al. described a retrospective cohort of 756 pediatric patients with IBD in Argentina (54% with UC, 33% with UC, and 13% with IBD-U). 30 Authors found significantly higher use of anti-TNF agents in patients with CD compared with UC (28.8 versus 11.9%, p < 0.001). Notably, a significant increase in biologic penetration was seen in 2008–2017 compared to before 2007 (36.7 versus 4%, p < 0.001). 30
In a single-center cohort study from Chile, Simian et al. included 716 patients with IBD (71% with UC, 27% with CD, and 2% with IBD-U). There was a significantly higher use of biologics in patients with CD compared to UC (34 versus 7%, p < 0.001). 31
Paredes Méndez et al. included 105 patients with IBD (77% with UC, 23% with CD) in a retrospective study from a single referral center in Peru. Overall, biologic use was described in 33.3% of patients with CD versus 2.4% of patients with UC. 32 Luciano et al. included 238 patients with UC in a cohort study from two centers in Uruguay. Biologic penetration in treatment algorithms was low, as only 1.4% of patients received anti-TNF therapy. 33
Penetration of biological therapies in Mexico, Central America, and the Caribbean
A single hospital-based study from Mexico City from 1987 to 2006 included 848 cases with a reported rate of biological treatment of 1.17%. 34 The same group described the use of biologics in 1.52% of patients in a case-control study, including 132 patients with CD between 1983 and 2013. 35 An epidemiological analysis including 2645 IBD cases from all 32 Mexican states between 2000 and 2017 demonstrated the use of anti-TNF therapy in children with UC and CD of 18.9 and 52.6%, respectively. In adult patients, the proportion of use of biologics was 15.3 and 38.5% in UC and CD, respectively, while 17.6% of elderly patients with UC received an anti-TNF and 24.8% of the patients with CD. 36 These percentages were estimated to be higher, 22% for UC and 76% in CD cases, in an analysis of healthcare databases made by Ciapponi et al. 37
One publication from Costa Rica analyzed a series of cases of patients with CD in a third-level center, where the reported use of biological therapy was 25% in 2009. 38 On the other hand, a publication from the EPILATAM study group reports the use of biologics in 56.4% of 587 patients with IBD in the Caribbean territories (this analysis included patients only from the Dominican Republic, Puerto Rico, and Cuba) between the years 2017 and 2020. 39 According to disease type in the Caribbean, the use of biological therapy, mainly anti-TNFs, was described in 36.8 and 66.8% in UC and CD, respectively. 39
A single center in Cuba reported 80 CD cases between 2011 and 2012 using infliximab in 3.8% of the patients. 40 Finally, data from the IBD registry in the University of Puerto Rico between 1995 and 2007 described the use of anti-TNF therapy in CD in 9.77 and 0.83% of UC cases. 41 Figure 1 shows the map of biological therapy penetration in LATAM.
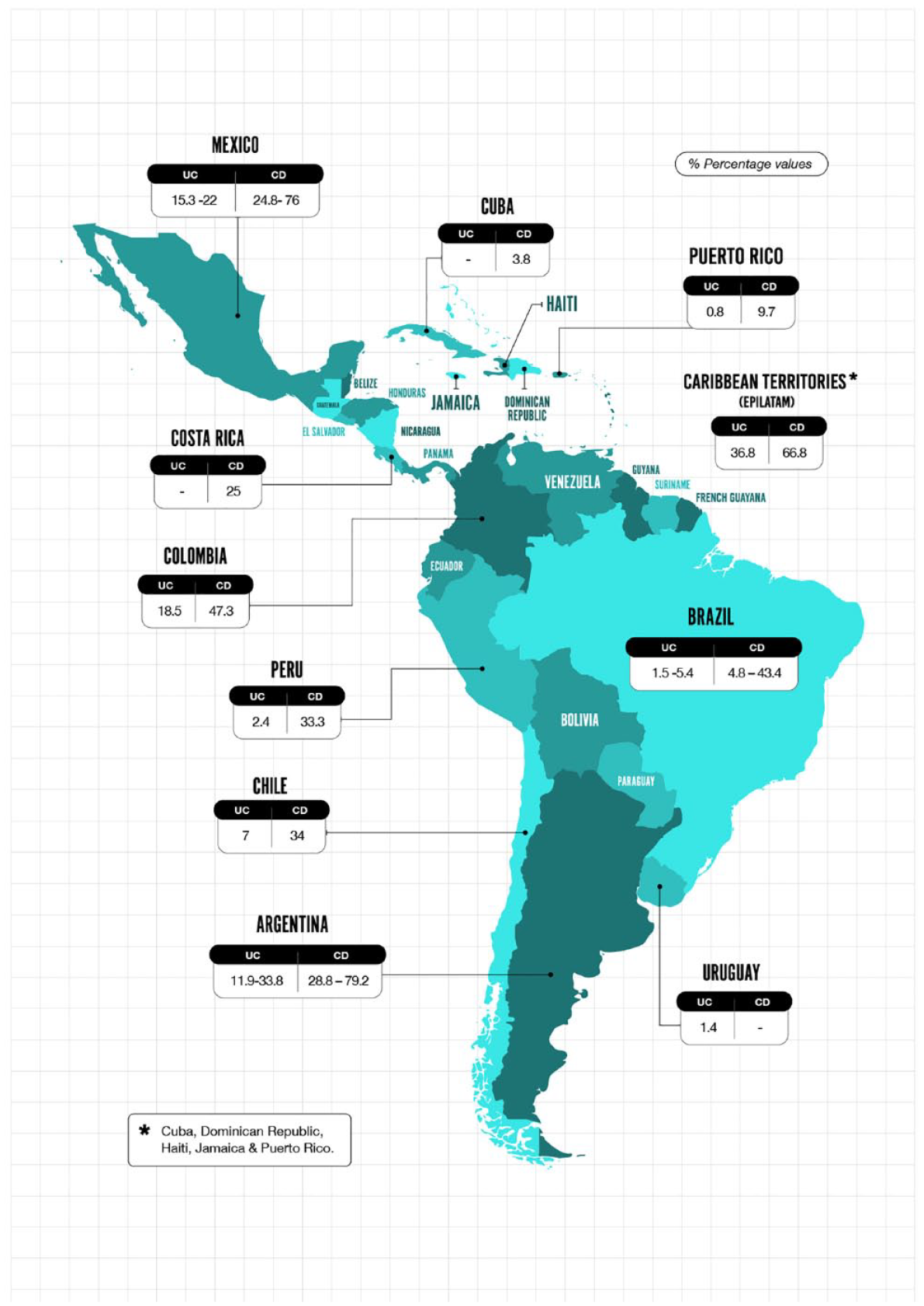
Penetration of biological therapy in LATAM.
Access to biological therapies in LATAM
Access to biological therapies in Brazil
Access to biologics in Brazil is closely related to socioeconomic conditions and geographic regions. Patients in the upper-income class have access to private healthcare, and those with lower incomes usually rely on the public health system. 42 The Unified Health System [Sistema Único de Saúde (SUS)] was created more than three decades ago. 42 All Brazilian residents are eligible for services provided by SUS, the primary source of healthcare for 78% of the population. 43 In 2020, 22% of Brazilians opted for private healthcare. 43
The first biologic approved was infliximab for CD in the early 2000s, and in 2017, two more anti-TNF agents, adalimumab and certolizumab, were incorporated into SUS. Currently, vedolizumab and ustekinumab are not included for CD in SUS, although these medications were approved in Brazil in 2015 and 2017, respectively. 44 Anti-TNF agents are the only available class of agents reimbursed for CD at SUS. In the private sector, biological agents (anti-TNF agents and ustekinumab) have been available for CD since 2017. 45
Regarding UC management, infliximab was incorporated in SUS until 2019. Subsequently, vedolizumab and tofacitinib were included in the public health system. 46 In 2021, biological drugs (golimumab, infliximab, and vedolizumab) were finally included for UC in the private sector. The complete list of IBD drugs available in Brazil is outlined in Table 2.
Access to biologics in LATAM through the perspective of the local IBD physician.

ADA, adalimumab; AF, armed forces; CTZ, certolizumab pegol; GLM, golimumab; HD, health department; IBD, inflammatory bowel disease; IFX, infliximab; LATAM, Latin America; MOH, ministry of health; OZA, ozanimod; P, private; SH, secretariat of health; SS, social security; TOF, tofacitinib; UC, ulcerative colitis; UHS, unified health system; UST, ustekinumab; VDZ, vedolizumab; +, very difficult; ++, somehow difficult; +++, easy; ++++, very easy/no limitations.
Interestingly, Vilela et al. published the results of a survey done on 286 Brazilian physicians regarding the perception of access to advanced IBD therapies. For CD, 70.4% of the physicians responded that they have difficulty accessing biologics, and 72.3% reported that this problem also was observed with conventional therapies for CD treatment. For UC, the difficulties accessing biological drugs were perceived even higher (95.1%), while limitations were reported by 57.7% of the physicians regarding other treatment options. 45
The first biosimilar of infliximab was approved in Brazil in 2015, and currently, there are three infliximab and three adalimumab biosimilars approved for IBD treatment. Biosimilars have an essential role in mitigating inequality in Brazil; their increased uptake will likely result in improved access to biological therapies due to price competition and cost savings. 47
Access to biological therapies in South America
Chile
In Chile, the healthcare system is divided into public and private sectors. Since 2015, the Ricarte Soto Law (N° 20.850) has granted universal access to high-cost diagnostics and therapeutics in both public and private sectors. CD and UC have been contemplated in this law since 2017 and 2019, respectively.48,49 Treating severe CD with infliximab and adalimumab is fully covered when failure to conventional therapies has been documented. 50 Similarly, in patients with moderate-to-severe UC, treatment with infliximab, adalimumab, and golimumab is fully reimbursed after failure to steroids and/or immunomodulators. 51 Although holding regulatory approval, access to non-anti-TNF advanced therapies is still challenging. In the private sector, health insurance might cover between 70 and 90% of the total cost of treatment, with the rest as an out-of-pocket expense to the patients. 52
Another considerable challenge in Chile is access to IBD specialists with experience in management with biologics. As of 2019, 85% of CD patients in biologics under the Ricarte Soto Law were from the Metropolitan Region, and only 15% were patients from all other regions of the country. Although this may indicate differences in prevalence across areas, it might likely highlight that specialized IBD care is concentrated in the big urban centers. 49
Uruguay
In Uruguay, the National Resources Fund is a public organization created in 1980 and regulated by Law N° 16.343, which finances highly specialized medical procedures and high-cost medications. 53 The National Resources Fund fully covers anti-TNF biologics infliximab or adalimumab for patients with moderate-to-severe IBD who are refractory or intolerant to conventional therapies.54,55 Additionally, golimumab is available for patients with UC. 55 Patients with extraintestinal manifestations may access anti-TNF therapy and patients with chronic pouchitis refractory to conventional therapy.54,55 Access to anti-TNF therapy is expedited if the coverage criteria are met, and continuity of treatments is the norm. However, a survey revealed that 59% of patients with IBD had difficulty accessing anti-TNF therapy. 56
The fund does not cover non-anti-TNF advanced therapies, and access to these other mechanisms, despite having regulatory approval, is extremely difficult. Patients may need to go through legal processes involving a writ of protection to reimburse the cost of these medications.
Argentina
In Argentina, the healthcare system is broadly divided into public, private health insurers, and social security, and each of these systems is atomized into several subsystems. 57 The Superintendence of Health Services oversees the Unique Reimbursement System (SUR), financing medical conditions of low incidence, high economic impact, and long-term treatment.
The SUR is part of a broader fund allocation, the Redistribution Solidarity Fund (FSR). 58 Under the SUR, patients with moderate-to-severe IBD refractory or intolerant to conventional therapies can access anti-TNF therapy (infliximab, adalimumab, and certolizumab pegol for CD, and infliximab, adalimumab, and golimumab for UC). 59
As in other Latin American systems, access to non-anti-TNF advanced therapies is challenging and usually reserved in failure, intolerance, or contraindication to anti-TNFs. Each subsystem will bear the cost of biologics not contemplated in the SUR (e.g., non-anti-TNF). Patients usually encounter substantial barriers to accessing these medications. Several laws are in place for the integral protection of people with disabilities; under these, full reimbursement of drugs is granted. 60 Not infrequently, IBD patients who meet specific criteria can obtain a certificate of disability that guarantees full access to high-cost medications.
Access to biological therapies in Mexico, Central America, and the Caribbean
Overall, the total expenditure on managing IBD in the region is unknown. 61 At large, there are no well-established and effective strategies for price negotiations for biological and advanced therapies, partly due to the separation of the demands of each country and the lack of information between them as buyers. Even with biosimilars in the region, the economic effort to access these medications is still higher than in developed countries such as Spain or the United States.62,63
Mexico
The healthcare system in Mexico is divided into private and public sectors. However, the public system is subdivided into multiple institutions of social security and the Secretariat of Health (SOH), the latter in charge of the population’s attention without any health coverage.64,65
Current biologics and advanced therapies for IBD in Mexico include anti-TNF therapies (infliximab, adalimumab, infliximab biosimilar, certolizumab pegol, and golimumab), vedolizumab, ustekinumab, and tofacitinib. There is no standardization for access to high-cost medications even though a national fund was created for this purpose. 65 The private and SOH may cover justifiable cases with any available approved therapies, whereas the social security system currently only covers anti-TNF drugs for IBD.
Costa Rica
There is access in the country to tofacitinib, ustekinumab, and most anti-TNF therapies (excluding certolizumab pegol). There is currently no local access to vedolizumab. Patients with private insurance can get coverage for any available treatments. However, the market share of private insurance is still very narrow in Costa Rica, particularly for high-cost diseases.66,67 The public healthcare system is administered only by the Caja Costarricense de Seguro Social (CCSS), with a coverage of 94.9% of the population.68,69
This public system guarantees access to essential therapies included in the official national list of medications. All the current biologics and advanced treatments for IBD are considered high-cost medications and, thus, are not part of this list. However, physicians can submit applications to a central committee for approval of IBD therapies on a case-by-case basis. 70 This process is slow and carries a high rate of denials, opening a judicial pathway through the Supreme Court of Costa Rica that patients can choose to pursue, with an approval rate of 79%, increasing the overall care costs in the country. 71
Dominican Republic
The country has private and public health coverages; however, the High-Cost Department of the Ministry of Health manages all the approvals, coverage, and negotiations with the pharmaceutical industry to buy medications for IBD and other high-cost diseases. 72
The Dominican Republic has the same biologics and advanced therapies for IBD as Costa Rica. The approval process allows patients to access the treatments prescribed by their gastroenterologists; however, this is still a centralized, slow, and case-by-case methodology that deals with occasional medication shortages, representing a challenge for patients and physicians.73,74
IBD surgery in LATAM
With the approval and overspread use of biological agents, combined with more current treatment strategies, surgical rates seem to decrease globally. 75 This reduction can be considered a natural consequence of the availability of new agents (biologics with different mechanisms of action and small molecules) and modern therapeutic strategies, such as the treat-to-target approach and early treatment, in patients with shorter disease duration. Despite available data coming from western countries’ referral centers, if surgical rates are reduced in population-based studies in newly industrialized countries, this still needs some reflection, mainly in LATAM.
Initial reports were based on small single-center studies. For example, a Puerto Rican study described surgical rates in two very different periods (1966–1973 and 2002–2008). 76 Authors reported an 87.9% increase in surgery for IBD in their center in the second study period. This finding can be interpreted as their development as a national tertiary referral IBD center (when more cases are referred) and as an association with the increase in the incidence and cumulative prevalence of both CD and UC in the country and the Caribbean region.
A recent systematic review which included cohort descriptions and population-based studies from LATAM and the Caribbean, described interesting results. 9 Of 61 included publications, 38 reported surgical rates, with most studies not demonstrating temporal trends but simple descriptions as a snapshot. Overall, the authors concluded that surgical rates may have reduced over time in different countries based on studies performed in different periods. In Brazil, a reduction could be observed from 1980–1999 (CD: 57.8%, UC: 21.9%) to 2016–2017 (CD: 31.7%, UC: 5.8%). The same was observed in Peru for CD, a drop from 70.5% in 1990–2010 to 50.0% in 2004–2014.
On the other hand, surgery rates for UC remained stable in many regions: in Peru, from 6.9% in 2001–2003 to 6.2% in 2004–2014, and in Uruguay, from 8.3% in 1951–2003 to 10.5% in 1985–2015. The authors additionally observed that the reduction in the surgical rate for CD was possibly associated with the increased use of TNF inhibitors in the continent over the last two decades. Interestingly, due to the later approval of these agents for UC, an increase in the use of anti-TNFs could not be observed. That was speculated as a possible reason why surgical rates did not decrease in that systematic review. 9
An ecological study with 47,699 hospitalizations due to IBD, performed exclusively in the public system in Brazil, demonstrated the tendency toward reduction in surgical rates at a national level. 77 During 11 fiscal years, IBD-related surgical procedures decreased from 8.8% in 2005 to 5.7% in 2015. Surgical rates were higher in CD as compared to UC. However, these data should be interpreted with caution, as the number of surgeries captured was extremely low for a continental country such as Brazil (445 procedures in 2005 and 235 in 2015), probably associated with methodological issues in data collection. More interestingly, most surgical procedures were performed in the southern and southeastern regions, more urbanized and developed areas. If this is associated with referral bias, as more tertiary referral centers are in these areas, this still needs to be determined.
Magro et al. analyzed CD-related main surgical procedures performed in 9 years in the Brazilian national public health system in relation to medical treatment. 78 Interestingly, authors observed that from a total of 1181 operations, rates were stable in patients using azathioprine [Average Annual Percent Change (AAPC) 1.34; CI −8.41 to 12.12; p = 0.797] and adalimumab (AAPC −1.64; CI −13.84 to 12.29; p = 0.807). On the other hand, surgical rates significantly decreased from 33.3 to 4.5% during the study period in patients who used infliximab (AAPC −17.05; CI −28.19 to −4.17; p = 0.011).
As seen, scarce data regarding surgery rates in IBD in LATAM are available. There is still room for more population-based studies to include this important variable to check if current therapies are really leading to a reduction in surgery rates in the continent.
Areas for research opportunities
Clinical research has been neglected in LATAM for decades, not only in IBD but in medicine overall. The lack of funding and protected time for research, combined with generally low salaries or service fees, lead Latin American physicians to have an eminently clinical workload. However, over recent years, significant efforts have been made to energize IBD research throughout the continent.
IBD is now recognized as a global disease, with an alarming rise in incidence and prevalence in developing countries. LATAM is no exception, with available data showing a steady increase in incidence. 79 Nevertheless, epidemiological data on IBD in LATAM are somewhat heterogeneous, and high-quality studies are scarce. 9 In this context, more large population-based registries are needed to fully disentangle gaps in the knowledge of epidemiology and the natural history of IBD in this region. This information could lead to the creation of local IBD-specific healthcare policies, directing recourses and planning toward managing these high-burden diseases.
Latin American patients have been historically underrepresented in IBD clinical trials. A systematic review of 2019 by Selvaratnam et al. showed that the IBD population of South America seems to have a unique disease phenotype, likely with a predominance of colonic CD and pancolonic UC. Also, UC was more common than CD. 8 Knowing the effectiveness of interventions in this specific population is paramount in a region that struggles with access to care and allocation of resources. Moreover, pharmacoeconomic studies assessing different interventions will become even more relevant in an area with prominent economic constraints. 80
There is a paucity of international multicenter studies in LATAM, as collaborations between institutions across countries have been rare. With a new push in clinical research in IBD in the region, these collaborative studies are becoming more common. An example, the SURGICROHN-LATAM study was a multicenter study that evaluated surgical complications in patients with CD undergoing early versus late ileocolonic resections in 10 tertiary centers from Argentina, Brazil, Chile, and Colombia. 81 Strategies that promote international research collaboration can be fostered through a partnership between local IBD study groups. A list of the most important IBD study groups in LATAM is found in Table 3. Pan-American Crohn’s and Colitis Organization (PANCCO) is an international organization founded in 2014, comprising IBD experts from Latin American countries and Canada. Since its foundation, PANCCO has been implementing strategies to improve IBD’s academic and scientific consolidation among the country members. Over the past years, the organization has been promoting the education of healthcare professionals in the field of IBD by developing national and international scientific and educational activities and encouraging national and international IBD research efforts. Additionally, it is worth mentioning the creation and publication of guidelines adapted to managing IBD patients from LATAM.82–85
The most important IBD study groups.
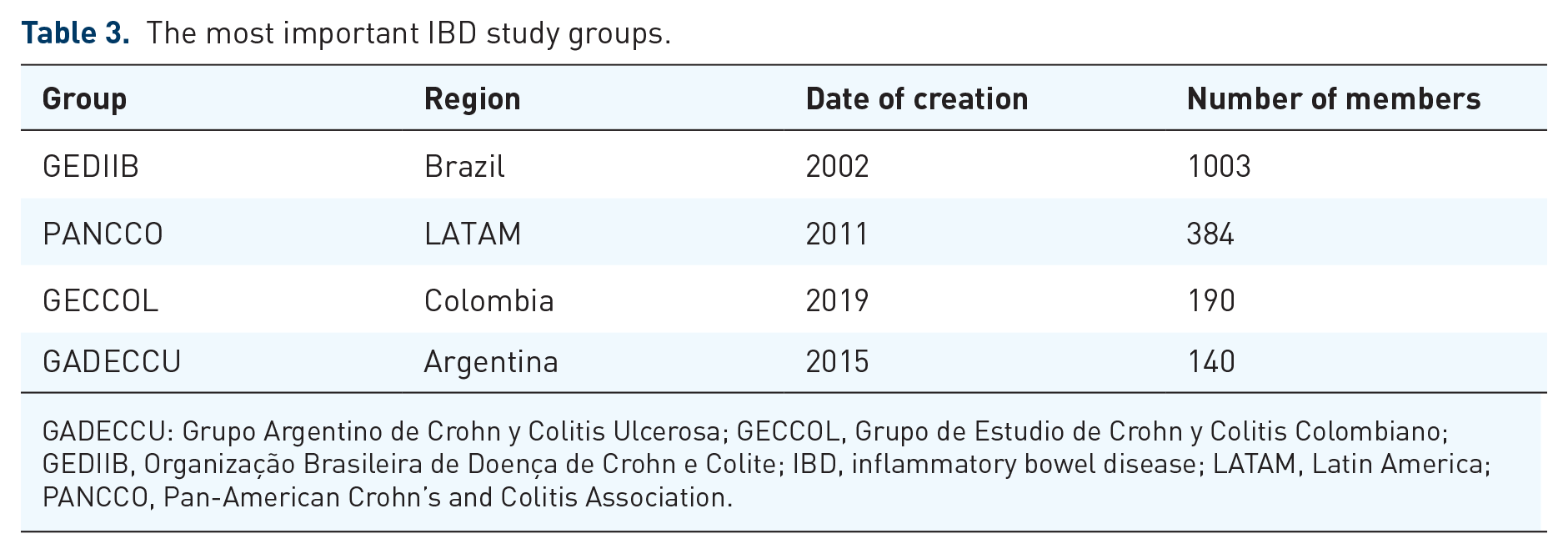
GADECCU: Grupo Argentino de Crohn y Colitis Ulcerosa; GECCOL, Grupo de Estudio de Crohn y Colitis Colombiano; GEDIIB, Organização Brasileira de Doença de Crohn e Colite; IBD, inflammatory bowel disease; LATAM, Latin America; PANCCO, Pan-American Crohn’s and Colitis Association.
Discussion
The epidemiological evolution of IBD can be stratified into four stages: emergence, acceleration in incidence, compounding prevalence, and prevalence equilibrium. 7 Most Latin American countries are developing and newly industrialized, so most regions will likely fit into the second and third epidemiological stages. Few studies have described the epidemiology of IBD in LATAM at a population-based level. Nonetheless, the recent research by Balderramo et al. 17 along with the most robust study derived from a public health database in Brazil, 16 clearly demonstrates that most of the region is progressively evolving toward the stability of incidence and increasing prevalence. The current number of IBD patients in the continent is possibly underestimated due to this lack of high-quality population-based studies. More epidemiological research in the region is warranted.
Biologic penetration in therapeutic algorithms in LATAM is variable. The apparent reason for this variation is the local implications of heterogeneity between countries and healthcare systems. A previous systematic review demonstrated that the use of biologics in CD varied from 1.51% in Mexico up to 46.9% in Colombia. 20 However, more recent data from Argentina reported cumulative use of biologics as high as 79.2% in CD patients. 18 Even though this could have been influenced by the fact that all participating centers were tertiary hospitals, the wide heterogeneity in biologic penetration calls attention to potential inequity regarding access to treatment.
Regarding surgical rates over time, scarce and granular data suggested a trend toward reducing surgery over time in LATAM. However, the only population-based study reported very low rates of surgery, which may reflect collection bias. 78 Moreover, whether the increase in biological use influences surgical rates remains to be elucidated.
This review also evaluated overall access to IBD treatments and approvals through a six-item questionnaire designed to capture perceptions of Latin American physicians (see Table 2). It was possible to obtain responses from 25 out of 47 countries and territories. Most of the non-respondents (18/22) were territories from the Caribbean region.
Anti-TNF therapies are available in the included countries and territories, except Belize. At least one non-anti-TNF advanced treatment is available except in Cuba, Bolivia, Venezuela, and Paraguay.
The perception of overall access to therapies was classified as very difficult, somehow difficult, easy, and very easy/no limitations. Only Aruba’s physicians evaluated their country as having very easy access to medications, and responses from Brazil, Colombia, and Bermuda ranked access to medications as easy. The remaining regions were classified as having very difficult or somehow difficult access to IBD treatments.
Although this survey has many inherent methodological limitations (mainly selection bias of respondents), we found that treatment practices are still highly diverse, in general, without local expert guidelines for management and may reflect substantial inequalities in access to biologicals in LATAM.
Conclusion
With IBD considered a global disease, knowledge of specific local characteristics for epidemiology, access to healthcare, treatment options, and different outcomes in particular regions are essential to improve patient care. Increasing incidence rates and improvement of care in CD and UC in Latin American countries over the last decades may result in a massive cumulative prevalence rate of IBD patients in the years to come. This may impact future health resource planning specifically targeted to CD and UC.
Despite intrinsic differences in access to biological agents, anti-TNFs are currently available in most countries. Despite being accessible in fewer countries, newer biologics and advanced therapies with different mechanisms of action represent a challenge throughout IBD care in LATAM. This region has obvious limitations regarding options for IBD patients compared to other more developed areas.
More data on surgical rates are also needed to identify a possible link to better disease control. Despite all limitations observed in healthcare systems in Latin American countries, IBD-related care seems to be improving over the last decades. Combined research throughout the countries is needed to position better the specific challenges in IBD care at an international level.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231167953 – Supplemental material for IBD barriers across the continents: a continent-specific analysis: Latin America
Supplemental material, sj-docx-1-tag-10.1177_17562848231167953 for IBD barriers across the continents: a continent-specific analysis: Latin America by Natália Sousa Freitas Queiroz, Camilla de Almeida Martins, Abel Botelho Quaresma, Pablo A. Olivera Sendra, Kenneth Ernest-Suarez and Paulo Gustavo Kotze in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
