Abstract
Introduction:
Immunomodulators remain fundamental for the medical treatment of Crohn’s disease (CD). Methotrexate (MTX) is widely used as a second-line immunomodulator; however, there is a lack of recent data on MTX monotherapy among the Asian population with CD. Therefore, in this study, we aimed to investigate the tolerability and clinical outcomes of MTX in Korean patients with CD.
Methods:
A retrospective chart review was performed for CD patients treated with MTX monotherapy or in combination with 5-aminosalicylic acid (5-ASA), at the Asan Medical Center, Seoul, South Korea. The tolerability of MTX monotherapy within 6 months was assessed and the clinical effectiveness of MTX was evaluated based on the Crohn’s disease activity index (CDAI).
Results:
In total, 85 patients were included, of which 29 (34.1%) discontinued MTX due to intolerability during the follow-up. Adverse events (AEs) were reported in 41 (48.2%) patients. The most common AE was gastrointestinal disorders (17/41) and only one patient experienced a serious AE, a systemic infection that required hospitalization. Among the 56 patients who tolerated MTX within 6 months, 44 (65.9%) showed a clinical response. Moreover, no factor was significantly associated with intolerability. The administration method was the only factor significantly associated with a response to MTX (p = 0.041). The adjusted odds ratio of parenteral injection compared to oral administration was 5.68 (95% confidence interval (CI), 1.07–30.08).
Conclusion:
In this study, one-third of patients were intolerant to MTX; nonetheless, the response rate was as high as 65.9% among tolerant patients. In addition, no significant factors affected intolerability. In terms of the clinical response, parenteral injection could be better than oral administration.
Introduction
Despite the development of novel biologic agents, conventional immunomodulators such as thiopurines and methotrexate remain fundamental to the medical treatment of inflammatory bowel disease (IBD), including Crohn’s disease (CD).1,2 Immunomodulators are recommended for inducing remission in patients with moderate-to-severe CD and for maintaining a remission in patients with steroid-dependent CD.1–3 In addition, it is recommended that immunomodulators can be used in combination with anti-tumor necrosis factor (TNF) agents to reduce immunogenicity.2,4
Methotrexate (MTX) is an economical and established drug that inhibits folic acid and purine synthesis. 5 Initially, MTX was introduced as a cytotoxic agent; however, it was later found to have an anti-inflammatory effect at low doses. Thus, it was widely introduced for the treatment of various diseases such as rheumatoid arthritis (RA) and psoriasis.6,7 The role of MTX in the treatment of IBD was initially assessed in the 1990s. In 1995, Feagan and colleagues 8 conducted a randomized controlled trial and reported that 25 mg/week of intramuscular MTX was effective in inducing remission in steroid-dependent CD. In 2000, the same research group reported that 15 mg/week of intramuscular MTX was also effective in maintaining CD remission. 9 Oren and colleagues 10 demonstrated that 12.5 mg/week of oral MTX was effective in active CD.
As MTX exhibits more adverse events (AEs) than thiopurines,11,12 MTX is usually considered a second-line immunomodulator for patients intolerant or unresponsive to thiopurines, despite having similar efficacy with remission rates of approximately 40%.11,13–15 Wahed and colleagues 16 reported that MTX achieved a clinical response in 60% of CD patients who were unresponsive or intolerant to thiopurines at 6 months and 17.4% of patients experienced side effects of MTX. Recently, systematic reviews, meta-analyses, and retrospective studies have supported the clinical effectiveness of MTX in CD patients.17,18
As all of the aforementioned studies are from Western countries and most of them were published before 2010, recent data on the role of MTX in CD among Asian populations are lacking, except for small single-center data from China. 19 The incidence and prevalence of IBD in Asian countries has increased recently,20–23 and thus the use of immunomodulators and biologics in Asia is increasing.24,25
Therefore, in this study, we aimed to investigate the clinical role of MTX for treating Korean patients with CD.
Methods
Patients
A retrospective chart review was performed for all 269 patients with CD treated with MTX at Asan Medical Center, Seoul, South Korea, from January 2008 to December 2020. Patients aged ⩾ 18 years, who received a diagnosis of CD, and were administered MTX therapy for the first time were included. We excluded patients who were administered MTX as a combination therapy with other IBD medications, such as corticosteroids, thiopurines, and biologics (except for 5-aminosalicylic acid (5-ASA)) and those who were prescribed MTX for reasons other than CD. We collected their demographic data, including sex, age at diagnosis, age at the start of MTX, disease duration from diagnosis to the start of MTX; baseline disease characteristics such as the location, upper gastrointestinal (GI) tract involvement, behavior, perianal modifier, prior bowel resection history, and prior medication history; and details of concomitant 5-ASA medication, laboratory data, MTX dose, and route of administration. We assessed the disease activity using Crohn’s disease activity index (CDAI) score. Each patient’s CDAI score was evaluated at every visit as a routine practice in our center. We collected the patient’s clinical data, including CDAI score, laboratory data, and all AEs observed during the 6 months after the start of MTX monotherapy. Moreover, we collected the maintenance duration of the MTX monotherapy of the study population through long-term follow-up until March 2021.
Outcomes
The primary outcome was tolerability of MTX during the 6 months after the start of MTX monotherapy. Patients who stopped MTX due to AEs or poor compliance within 6 months were classified as the intolerant group. Data about AEs, such as GI disorder, hepatotoxicity, leukopenia, and general weakness, were collected during the 6 months after the start of MTX therapy. The secondary outcomes included the long-term durability of MTX monotherapy, the clinical response at 6 months of MTX therapy, the factors associated with intolerability and response, and the biochemical responses. The clinical response was defined as maintenance of a CDAI score < 150 among patients whose baseline CDAI score was < 150 and the achievement of a clinical remission (CDAI score < 150) or a reduction of the CDAI score ⩾ 100 from the baseline CDAI score among patients whose baseline CDAI score was > 150. Patients who had switched or escalated to biologics due to disease aggravation, but not because of AEs, were classified as the nonresponse group. Clinical factors such as demographic, drug-related, and disease-related factors were analyzed to verify if these were associated with intolerability and response. Biochemical responses were assessed based on the changes in blood markers, including erythrocyte sedimentation rate (ESR), serum C-reactive protein (CRP), and serum albumin.
Statistical analysis
In the descriptive analysis, categorical variables are expressed as a number with percentage. Continuous variables are expressed as median with interquartile range (IQR). To assess the factors associated with tolerability and response, we performed univariate and multivariate logistic regression analyses. The multivariate analyses included variables with p < 0.1 in univariate analysis and were performed using the backward elimination method. The results are presented as the odds ratios (ORs) with 95% confidence intervals (CIs). Linear mixed modeling was used to evaluate the significance of the changes over time in the laboratory values during 6 months of MTX monotherapy. Time was considered a continuous covariate to investigate the trends. Statistical analyses were performed using R statistical package version 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria) and SPSS version 24.0 for Windows (IBM Corp., Armonk, NY, USA).
Ethical considerations
Our study protocol was approved by the Institutional Review Board of Asan Medical Center (IRB No. 2020-1746).
Results
Patients’ characteristics
In total, 85 patients who received MTX monotherapy from 2008 to 2019 were included in this study (Figure 1). The demographic data and disease-related characteristics of the study population are summarized in Table 1. The median age at diagnosis and the start of MTX treatment were 23.0 (IQR: 18.0–30.0) and 31.0 (IQR: 24.0–38.0) years, respectively. The median disease duration was 66.0 (IQR: 24.0–114.0) months. Regarding the administration method, 48 (56.5%) patients received oral administration and 37 (43.5%) patients received parenteral injections, including intramuscular and subcutaneous injections. The median dose of MTX was 15.0 mg/week. Eighty-two (96.5%) patients had a history of prior thiopurine use, and 13 (15.3%) patients had a history of prior biologics use. Furthermore, 70 (82.4%) patients simultaneously received 5-ASA with MTX. The median baseline CDAI score among the total population was 90.7 (IQR: 41.4–159.3).

Flow diagram of the study population.
Baseline characteristics of the study population.

5-ASA, 5-aminosalicylic acid; CDAI, Crohn’s disease activity index; IQR, interquartile range; MTX, methotrexate; UGI, upper gastrointestinal.
Tolerability and AE profile during 6 months of MTX therapy
Among 85 patients, 29 (34.1%) discontinued MTX due to intolerability within 6 months. Out of 29 patients who were intolerant to MTX, 27 and 2 patients discontinued MTX because of AEs and poor compliance, respectively.
In terms of AE, 41/85 (48.2%) experienced AEs during 6 months of MTX therapy (Table 2). Among these patients, only one patient experienced a serious AE, disseminated pulmonary infection with a chemoport insertion site abscess caused by Mycobacterium abscessus that required hospitalization. Hepatotoxicity and general weakness were found in 9 (10.6%) patients, respectively. In addition, leukopenia occurred in 10 (11.8%) patients, and among these, 7 patients experienced grade 1 leukopenia (3000 < WBC ⩽ 4000) and 3 patients experienced grade 2 leukopenia (2000 < WBC ⩽ 3000). Moreover, 6 (7.1%) patients complained of headaches. Other AEs included upper respiratory tract infection, fever, arthralgia, alopecia, and drug eruption.
AEs during 6 months of MTX monotherapy.

AE, adverse event; GI, gastrointestinal; MTX, methotrexate; WBC, white blood cell.
Long-term durability of MTX monotherapy
We collected the long-term maintenance duration data of MTX monotherapy until March 2021. The maintenance duration of all patients is illustrated in Figure 2. The longest follow-up duration of MTX monotherapy was 92 months, whereas the median duration was 11 months. Moreover, 50% and approximately 25% of the patients maintained MTX monotherapy at 11 and 30 months, respectively. During a follow-period of up to 92 months, 61/85 (71.8%) patients discontinued MTX monotherapy due to AEs (35/61, 57.4%), loss of response (21/61, 34.4%), poor compliance (3/61, 4.9%), and pregnancy planning (2/61, 3.3%).

Kaplan-Meier curve of the long-term maintenance duration of MTX monotherapy.
Clinical response to MTX monotherapy
Among the total study population, 25 (29.4%) patients had active disease at baseline and 60 (70.6%) patients were in clinical remission at baseline (the baseline characteristics of the two groups classified according to the underlying disease activity are summarized in Supplementary Table 1). Excluding 29 intolerant patients, the response was assessed in the remaining 15 patients who had active disease and 41 patients who were in clinical remission. Among the 15 patients with initially active disease, 7/15 (46.7%) were responsive to MTX monotherapy after 6 months. Among the 41 patients who were in clinical remission at baseline, 37/41 (90.2%) were responsive to MTX monotherapy (Figure 3). Out of 56 patients in total, 44 (78.6%) were responsive to MTX monotherapy after 6 months. Among the total study population, 44/85 (51.8%) were responsive to MTX monotherapy and 12/85 (14.1%) were unresponsive to MTX monotherapy despite tolerating MTX.

The proportion of patients according to intolerability and response after 6 months of MTX monotherapy.
Factors associated with intolerability and response to MTX
The results of univariate analysis of clinical factors associated with intolerability to MTX within 6 months are summarized in Table 3. No factor in the univariate analysis exhibited a value of p < 0.1. No clinical characteristics including demographic, drug-related, and disease-related factors were significantly associated with intolerability to MTX (a comparison of baseline characteristics between the tolerable group and the intolerable group is presented in Supplementary Table 2).
Univariate analysis of factors associated with intolerability.

5-ASA, 5-aminosalicylic acid; CI, confidence interval; MTX, methotrexate; OR, odds ratio; UGI, upper gastrointestinal.
The factors associated with a response to MTX monotherapy at 6 months are summarized in Table 4. In the univariate analysis, age at diagnosis and the administration method exhibited a value of p < 0.1. In the multivariate analysis, the adjusted OR of age at diagnosis per year was 0.94 (95% CI: 0.88–1.01; p = 0.087), and the adjusted OR of parenteral administration compared to oral administration was 5.68 (95% CI: 1.07–30.08; p = 0.041). The administration method was the only significant factor associated with a response in multivariate analysis (a comparison of the baseline characteristics between the response group and the nonresponse group is presented in Supplementary Table 3).
Univariate and multivariate analysis of factors associated with a response.
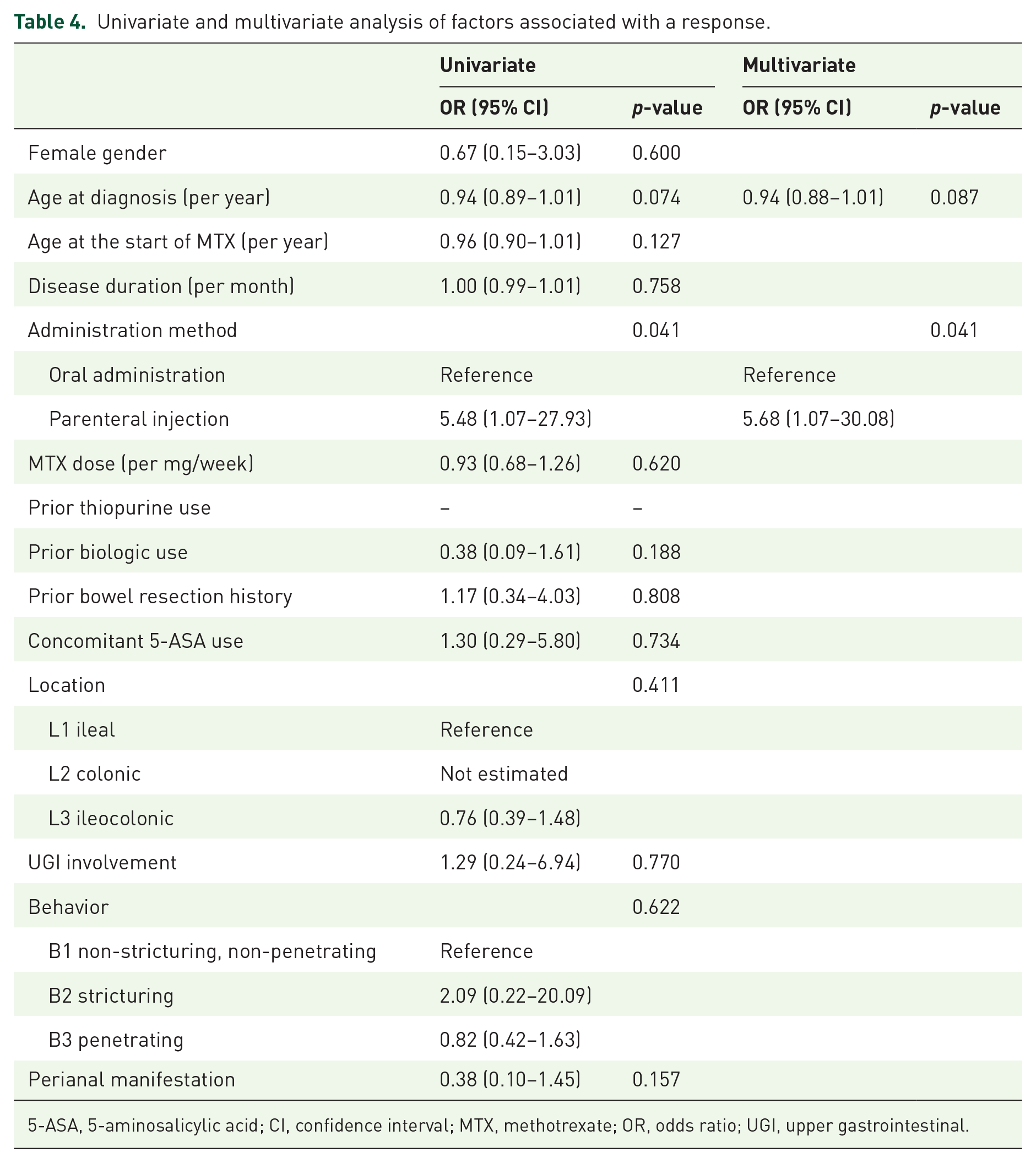
5-ASA, 5-aminosalicylic acid; CI, confidence interval; MTX, methotrexate; OR, odds ratio; UGI, upper gastrointestinal.
Biochemical response during MTX monotherapy
The blood inflammatory markers, ESR and serum CRP, and serum albumin revealed significant changes during 6 months of MTX monotherapy (Figure 4). ESR and serum CRP were significantly decreased (for ESR, ptrend = 0.01; for serum CRP, ptrend = 0.03). The median level of ESR at baseline and week 24 was 21 mm/h (95% CI: 12.0–38.5) and 11 mm/h (95% CI, 3.75–23.75), respectively. Moreover, the median level of serum CRP at baseline and at week 24 was 0.6 mg/dL (95% CI: 0.2–1.3) and 0.29 mg/dL (95% CI: 0.13–0.69), respectively. Serum albumin revealed a significantly increasing trend over 6 months (ptrend = 0.028). Furthermore, the median level of serum albumin at baseline and week 24 was 3.9 g/dL (95% CI: 3.6–4.1) and 4.1 g/dL (95% CI: 3.8–4.3), respectively.

The changes in biochemical markers during 6 months of MTX monotherapy. Erythrocyte sedimentation rate (ESR) (a), serum C-reactive protein (CRP) (b), and serum albumin (c).
Discussion
Recently, novel drugs for CD treatment are gaining immense attention, particularly after the emergence of biologics; however, immunomodulators, which are old drugs compared to biologics, are still used in the medical treatment of CD as monotherapy or in combination therapy with biologics. Despite being economical and widely used, recent data on the real clinical role of MTX among CD patients are lacking. This study revealed that MTX is intolerable in about one-third of patients but can be clinically and biochemically effective in tolerant patients when used as a monotherapy.
One of the most important concerns regarding MTX is its tolerability. During 6 months of MTX therapy, 48.2% of patients experienced certain AEs, 31.8% of patients discontinued MTX due to AEs, but only one patient experienced serious AE in this study. In the randomized study by Feagan and colleagues, 8 17% of patients discontinued MTX within 16 weeks due to AEs. The incidences of AEs ranged from 37% to 45%, but, serious AEs were rarely observed in previous retrospective studies.9,12,18 Discontinuation of MTX due to AEs within 6 months occurred in 7.4%, 11.4%, 12.2%, 16%, and 17% of patients in previous studies, respectively.8,12,18,19,26 In addition, discontinuation of MTX increased gradually over time, and 29.6%–32% of patients discontinued MTX within 24 months.12,19 The incidences of AEs from previous studies are consistent with our finding, but discontinuation of MTX was more frequent in our patients. As only one serious AE was reported, we presume that the patients in this study discontinued MTX due to minor AEs.
Among all AEs, the most common AE in previous studies was GI disorders, including nausea and vomiting, which occurred in 15.2%–22.2% patients, even up to 40%.9,12,18,19,27 Subsequently, general weakness and hepatotoxicity were reported in 8.2%–16% and 6.78%–10% patients, respectively.8,9,12,18,19,27,28 This AE profile is consistent with that of the present study, as general weakness and hepatoxicity occurred in 10.6% of patients, respectively. The incidence of leukopenia was higher in our study population. The previously reported incidence of leukopenia during MTX treatment ranged from 1% to 7%.12,18,27,29–31 Leukopenia occurred in 11.8% of our study population; of 10 patients with leukopenia, 7 (8.2%) had grade 1 leukopenia with little clinical significance; the other 3 (3.5%) patients who had grade 2 leukopenia can be considered clinically significant.
The serious AE requiring hospitalization that occurred in one patient was pulmonary and extrapulmonary infection caused by nontuberculous Mycobacteria (NTM). The patient developed a chemoport insertion site abscess and active disseminated pulmonary infection due to NTM after 12 weeks of MTX therapy. There are previous case reports of NTM infection in RA patients treated with low-dose MTX.32,33 Notably, MTX use was not significantly associated with NTM infection among RA patients,33,34 and CD itself and active disease status can cause immunologic disorders, 35 so it is unclear whether MTX is directly associated with the development of NTM infections. Therefore, close monitoring of opportunistic infections among CD patients under immunomodulators is necessary.
There was no clinical factor associated with intolerability in this study. Vasudevan and colleagues 12 explored the tolerability and discontinuation of immunomodulators among patients with IBD and found that no clinical factor, except smoking, was associated with the discontinuation of immunomodulators. Low dose and oral administration of MTX revealed lower rates of discontinuation, but both were statistically insignificant. Among patients with RA treated with low-dose MTX, the administration method was independent of AEs and tolerability.36,37 These are consistent with our study results. Oral administration may be preferred for fear of intolerability; however, in practice, oral administration is not beneficial.
Apart from the short-term tolerability of MTX, our results for the long-term maintenance duration of MTX monotherapy are disappointing. Nearly 50% of the patients discontinued MTX monotherapy at 11 months, and two-thirds discontinued at 30 months. In the retrospective cohort study by Vasudevan and colleagues, 12 withdrawal from MTX increased over time from 16% at 6 months to 26% and 32% at 12 and 24 months, respectively, and the median time for discontinuation was 7.2 months. As their study included UC patients and allowed for combination therapy with biologics, this result may have to be interpreted differently from our study results.
In this study, MTX monotherapy was effective in 78.6% of tolerable patients and 51.8% of the whole study population. When classified according to the baseline disease activity, MTX was effective in 28.0% of the patients who had initially active disease and in 61.7% of the patients who were in clinical remission at baseline. In the randomized controlled study by Feagan and colleagues, 8 39.4% of the patients achieved a clinical remission after 16 weeks of intramuscular MTX monotherapy. Wahed and colleagues 16 revealed that a clinical response was achieved in approximately 60% of CD patients, who were unresponsive or intolerant to azathioprine/mercaptopurine, within 6 months of MTX therapy. In a retrospective study comparing MTX and thiopurine, the clinical remission rate at week 16 was 68.6% among patients treated with MTX. 38 Furthermore, 62.9% and 48.1% of patients achieved a clinical response and clinical remission after 12 months of MTX monotherapy in a single-center experience in China. 19 Among patients refractory to anti-TNF agents, the short-term clinical response and remission rates were 60% and 30.9%, respectively. 18 The clinical response rate in the present study was similar to or slightly lower than that of previous studies. While previous studies included a subset of patients with CD, this study included a heterogeneous population of CD patients and revealed the short-term effectiveness of MTX therapy in this population, especially for the maintenance of clinical remission.
According to our results, the administration method was the only significant factor associated with a response, and parenteral injection was more strongly associated with a response than oral administration. In randomized controlled studies that proved the effectiveness of MTX monotherapy compared to placebo, MTX was administered intramuscularly.8,9 In other randomized studies in which MTX was administered orally, MTX was not superior to placebo.10,39 Moreover, in retrospective studies revealing the effectiveness of MTX, most of the study population was administered MTX parenterally.28,38 Thus, the current guidelines recommend MTX monotherapy to be administered parenterally for maintaining remission in steroid-dependent CD patients.1,2 Oral administration of MTX is widely used in clinical practice because it is convenient. However, the rationale for the effectiveness of oral administration of MTX among CD patients is weak. To achieve a better response to MTX therapy, parenteral injection is preferable.
There are certain limitations to this study. First is the possibility of confounding factors and selection bias since this was a retrospective single-center study. Second, a majority of the study population was prescribed 5-ASA along with MTX, so the outcome of this study might not be considered to be the outcome of true “monotherapy”. However, current guidelines recommend against the use of 5-ASA for induction and maintenance of remission in patients with CD, because 5-ASA was not superior to placebo in previous studies.1,2 Thus, the role of 5-ASA is insignificant. Third, endoscopic and radiologic data were not available during the short-term follow-up for most patients. Hence, it was impossible to evaluate the effectiveness of MTX in mucosal healing. Fourth, analysis of the change in fecal calprotectin, which is one of the important parameters while evaluating responses in CD, could not be performed due to a lack of follow-up data. Despite the absence of such data, CDAI is a valuable and practical parameter widely used in the real medical field, so the results of this study using CDAI are worthwhile. Fifth, considering the biochemical response, not all patients underwent laboratory tests every 4 weeks; therefore, the number of patients evaluated each time was different. Finally, as patients who discontinued MTX monotherapy were excluded, the possibility of selection bias in evaluating the biochemical response cannot be ignored.
Despite these limitations, this is the first study to evaluate the utility of MTX among adult CD patients using a meaningful number of samples in Asia. Our study confirmed that despite the intolerability in one-third of the patients, MTX can be considered a treatment option for Asian CD patients with little safety concern because serious AEs were extremely rare. A better response can be expected from parenteral injection than oral administration, as recommended by the current guidelines.
Supplemental Material
sj-docx-1-tag-10.1177_17562848211043017 – Supplemental material for Short-term tolerability and effectiveness of methotrexate monotherapy in adult patients with Crohn’s disease: a retrospective study
Supplemental material, sj-docx-1-tag-10.1177_17562848211043017 for Short-term tolerability and effectiveness of methotrexate monotherapy in adult patients with Crohn’s disease: a retrospective study by Hee Seung Hong, Kyuwon Kim, Kyunghwan Oh, Jae Yong Lee, Seung Wook Hong, Jin Hwa Park, Sung Wook Hwang, Dong-Hoon Yang, Byong Duk Ye, Jeong-Sik Byeon, Seung-Jae Myung, Suk-Kyun Yang and Sang Hyoung Park in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848211043017 – Supplemental material for Short-term tolerability and effectiveness of methotrexate monotherapy in adult patients with Crohn’s disease: a retrospective study
Supplemental material, sj-docx-2-tag-10.1177_17562848211043017 for Short-term tolerability and effectiveness of methotrexate monotherapy in adult patients with Crohn’s disease: a retrospective study by Hee Seung Hong, Kyuwon Kim, Kyunghwan Oh, Jae Yong Lee, Seung Wook Hong, Jin Hwa Park, Sung Wook Hwang, Dong-Hoon Yang, Byong Duk Ye, Jeong-Sik Byeon, Seung-Jae Myung, Suk-Kyun Yang and Sang Hyoung Park in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848211043017 – Supplemental material for Short-term tolerability and effectiveness of methotrexate monotherapy in adult patients with Crohn’s disease: a retrospective study
Supplemental material, sj-docx-3-tag-10.1177_17562848211043017 for Short-term tolerability and effectiveness of methotrexate monotherapy in adult patients with Crohn’s disease: a retrospective study by Hee Seung Hong, Kyuwon Kim, Kyunghwan Oh, Jae Yong Lee, Seung Wook Hong, Jin Hwa Park, Sung Wook Hwang, Dong-Hoon Yang, Byong Duk Ye, Jeong-Sik Byeon, Seung-Jae Myung, Suk-Kyun Yang and Sang Hyoung Park in Therapeutic Advances in Gastroenterology
Footnotes
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
