Abstract

Key points
Treatment options for refractory Crohn’s disease are limited.
Immunoglobulin is used in increasing clinical contexts.
Immunoglobulin may be effective in refractory Crohn’s disease.
Immunoglobulin is well-tolerated.
Intravenous and subcutaneous immunoglobulin may have comparable efficacy.
Clinical trials addressing immunoglobulin in inflammatory bowel disease are warranted.
Few modalities exist for inducing and sustaining remission in refractory Crohn’s disease (CD). With current research focused primarily on selective targeting and inhibition of the inflammatory cascade with monoclonal antibodies, we have perhaps lost sight of older yet promising therapies like high-dose immunoglobulin (Ig). While reports of intravenous Ig (IVIg) in steroid-resistant CD date back at least 25 years [Rohr et al. 1987; Wolf et al. 1988; Possoz et al. 1989; Raedler et al. 1988], there are no reports of subcutaneous Ig (SCIg) in refractory CD to our knowledge. We present two novel cases of severe, refractory CD responsive to IVIg with subsequent transition to SCIg and sustained clinical remission. Patient consent was obtained prior to including pictures in this manuscript.
Our first patient is a 40 year-old woman with longstanding penetrating, stricturing CD. She failed monotherapy with tumor necrosis factor-α (TNF-α) inhibitors, but achieved near-remission with hookworm infection. A subsequent flare ultimately necessitated a diverting colostomy, complicated by postoperative peristomal pyoderma gangrenosum (PG) (Figure 1). Dermatology recommended 2 g/kg/month IVIg (Gammunex®) over 4–5 days (total 140 g), extrapolated from prior published experience. By the second day, her PG resolved (Figure 2) and she incidentally noted near-resolution of her abdominal symptoms with solid ostomy output. Due to headaches, her dose was first reduced to 105 g/month administered over 3 days, followed by attempting 5-week intervals. As the approximate half-life of IgG is 21–25 days, intervals above 5 weeks result in significantly lower trough levels and occasional symptom recurrence. She was transitioned to an equivalent dose of SCIg (Hizentra®) divided weekly with headache improvement and continued clinical remission.
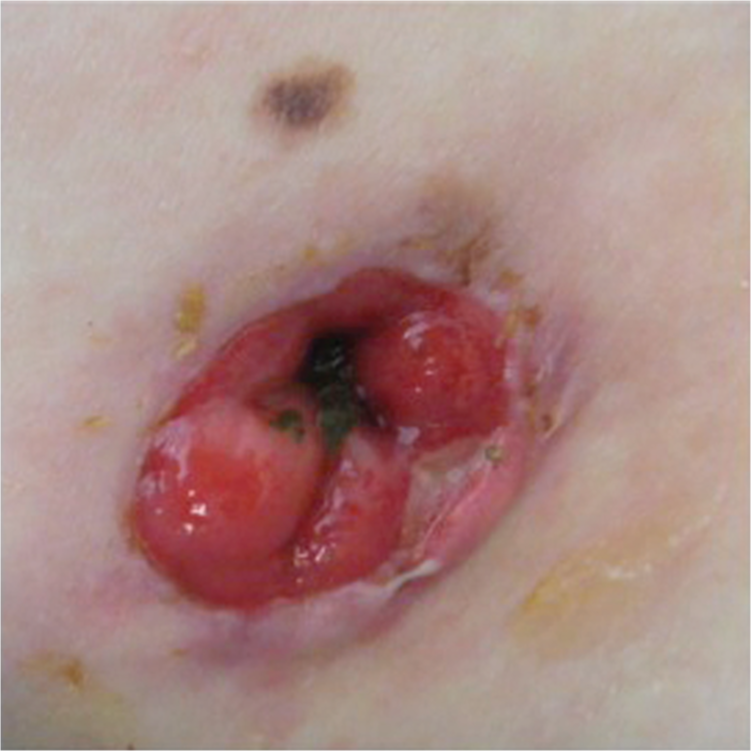
Before intravenous immunoglobulin (IVIg).
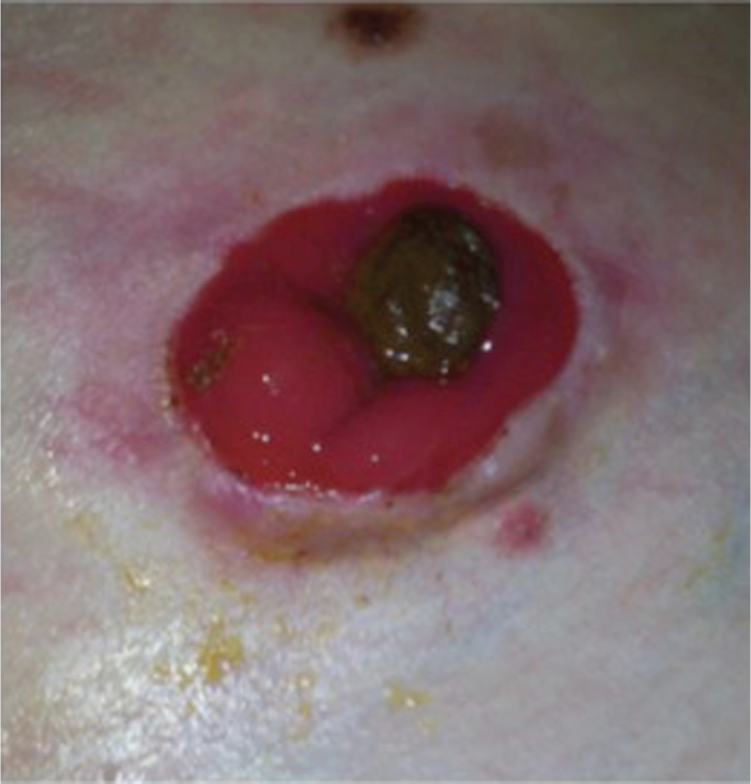
After intravenous immunoglobulin (IVIg).
Our second patient is a 32 year-old gentleman with longstanding fibrostenosing CD who remained symptomatic despite five bowel resections, 6-mercaptopurine, the US Food and Drug Administration (FDA) approved TNF-α inhibitors (infliximab, adalimumab and certolizumab) and integrin inhibitor (natalizumab), the non-FDA approved interleukin (IL)-23/IL-12 inhibitor (ustekinumab), as well as nontraditional therapies (hookworm infection, naltrexone and bupropion). With 2 g/kg/month IVIg (Flebogamma®) divided over 4–5 days, he reported marked improvement by the second day. Unfortunately, his symptoms recurred while foregoing IVIg due to infusion-related headaches. Despite premedication, slower infusion rate and a lower osmole formulation (Gammunex®), his headaches persisted. Similar to our first case, his headaches improved with transition to SCIg (Hizentra®) and he regained clinical remission.
Ig’s mechanism of action is likely multifactorial given its successful application to an increasingly broad spectrum of diseases. Fundamentally, its mechanism involves replacement and/or immunomodulation, a distinction that is limited at best in CD. Variable production of Ig and even common variable immunodeficiency itself has been implicated in CD; Ig may serve as primary replacement in such patients. Its immunomodulator role is more nebulous, with several postulated mechanisms ranging from Fc-receptor blockade to B-cell anergy to a generalized anti-inflammatory effect. Although clearly multifactorial, reduced T-cell proliferation and trafficking seems the most pathophysiologically sound mechanism in CD. IVIg-mediated reduced trafficking is also described in ulcerative colitis [Levine et al. 1992].
Most literature of IVIg in CD is anecdotal, with no controlled trials to date. The lack of interstudy concordance with respect to threshold for use, dosing and concurrent therapies, among others, expectedly limits generalizability [Kobrynski, 2012]. There is no experience with SCIg in CD. Controlled studies comparing IVIg and SCIg predominantly involve low-dose administration (400–600 mg/kg/month) for primary immunodeficiency; notably, similar serum IgG levels and clinical response (defined by rate of serious infection and/or hospitalization) is consistently reported [Abolhassani et al. 2012; Chapel et al. 2000]. Evidence for high-dose SCIg (2 g/kg/month), however, is unfortunately limited to case reports. Danieli and colleagues’ cohort of seven patients with inflammatory myositis successfully maintained on high-dose SCIg [Danieli et al. 2011] shares company with only a handful of other immune-mediated neurologic and dermatologic case reports [Köller et al. 2006; Misbah et al. 2011]. Extrapolating from pharmacokinetic data suggesting comparable serum IgG levels between IVIg and SCIg [Danieli et al. 2011], sustained clinical response with transition to SCIg seems plausible. Of note, we used a 1:1 conversion when transitioning from IVIg to SCIg, as practiced in Europe, as opposed to 137% of the monthly IVIg dose recommended in the US [Gardulf et al. 2006].
Importantly, Ig is well-tolerated with the incidence of adverse effects estimated around 0.3–0.5% with SCIg and 5% with IVIg [Gardulf, 2007; Chapel et al. 2000]. Patient satisfaction and quality of life are also higher with SCIg [Berger et al. 2010]. Although SCIg obviates the need for venous access, multiple subcutaneous sites are needed for administering higher doses; newer formulations may mitigate this somewhat.
Given Ig’s promising safety profile, its limited clinical use and lack of well-powered studies conceivably reflects its cost and off-label categorization. To date, 13 IVIg formulations and 5 SCIg formulations (10%-Gammagard®, 10%-Gamunex-C®, 10%-Gammaked®, 16%-Vivaglobin® and 20%-Hizentra®) [NLM DailyMed, 29 July 2013] are approved in the US, with alternate brands available in other countries. Associated costs for each brand extend beyond the retail price and include storage, shelf-life, preparation and administration, among other considerations [Mark, 2011]. The unit price of SCIg is comparable to IVIg, but its overall cost is lower as it obviates infusion center visits and nursing administration. IV Gammunex® 2 g/kg/month administered over 3 days at our infusion center is approximately $55,000, while Hizentra® 2 g/kg/month divided weekly is approximately $20,000 for a 70-kg patient. Comparatively, induction doses of TNF-α and integrin inhibitors range from $7,000–12,000 per infusion. Literature regarding the cost of Ig is expanding and will undoubtedly evolve as new formulations are approved and older ones are improved. Despite its off-label categorization, we did not experience untoward difficulty providing Ig therapy for our two patients.
In conclusion, IVIg and SCIg are potentially effective salvage therapies with an encouraging safety profile. Although reports of IVIg therapy for refractory CD date back almost three decades, we are the first to present the successful transition to high-dose SCIg. Controlled trials with objective data such as inflammatory markers and endoscopic evaluation are certainly necessary before Ig approaches the ranks of the currently approved therapies for refractory CD. Additionally, extraintestinal manifestations like pyoderma gangrenosum [Meyer et al. 2006] and ‘priming’ with prior monoclonal antibody therapy [Sandborn et al. 2008] merit exploration as predictors of Ig responsiveness and identifying appropriate candidates.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
