Abstract
Behcet’s disease (BD) is a chronic and recurrent systemic vasculitis involving large, medium and small blood vessels as well as arteries and veins. BD with predominant gastrointestinal manifestations is diagnosed as intestinal BD, which is associated with severe complications such as massive gastrointestinal hemorrhage, perforation, and obstruction. Recently, treat-to-target (T2T) strategies have been successfully used in many chronic diseases and been suggested in the management of BD, while there are no related reviews about the global treatment strategy including treatment principles and targets for intestinal BD in detail. Herein, we review the treatment principles from the aspects of departments of Rheumatology and Gastroenterology. In addition, treatment targets of intestinal BD are reviewed from three aspects such as evaluable markers, effective markers and potency-ratio markers. Some definitions and conceptions from inflammatory bowel disease (IBD) bring us reference and enlightenments.
Introduction
Behcet’s disease (BD) is a chronic recurrent systemic vasculitis involving large, medium and small vessels as well as arteries and veins. It was first reported in 1937 by Hulusi Behcet, a Turkish dermatologist. This disease has a high prevalence along the Silk Road, including Japan, Korea, the Middle East, and the Mediterranean region. The most widely accepted criteria include the criteria published by the International Study Group for BD in 1990 and The International Criteria for Behcet’s Disease developed from the collaboration of experts from 27 nations. The sensitivity and specificity of the former are 85 and 96%, respectively, while the latter are 94.8 and 90.5%, respectively.1,2 BD can involve multiple organs, including skin, mucosa, eyes, cardiovascular system, nervous system, gastrointestinal tract, joints, lungs, kidneys, etc. Patients with involvement of important organs usually have poor outcomes.3,4
Gastrointestinal involvement of BD is termed as intestinal Behcet’s disease (intestinal BD). It may cause serious complications including perforation, obstruction, fistula formation and gastrointestinal bleeding in some cases, showing high rates of disability and mortality. The prevalence of gastrointestinal involvement in BD is about 3–60% and it shows regional differences, with approximately 2.8–4.0% in Turkey, India and Saudi Arabia, 10% in China, 38–53% in Japan and 50–60% in the UK. 5 It is noteworthy that intestinal BD can present with or without other organ involvement, with volcano-shaped ulcers in ileocecal area as the typical finding.6,7 Up to now, the diagnosis of intestinal BD mainly depends on clinical manifestations without specific laboratory index. And in a few cases, it is hard to differentiate intestinal BD from other inflammatory gastrointestinal diseases such as Crohn’s disease (CD), intestinal tuberculosis. Japanese and Korean guidelines for intestinal BD have proposed algorithms for definite diagnosis.8,9 Recently, our team also summarized the key points of differential diagnosis among intestinal BD, CD and intestinal tuberculosis in a review about clinical characteristics, diagnosis and evaluation of intestinal BD. 10
The management of intestinal BD is mainly carried out in the Departments of Rheumatology and Gastroenterology, which are complementary to each other. The European League Against Rheumatism (EULAR) updated the recommendations for the management of BD published in 2018, which first proposed that gastrointestinal involvement of BD should be confirmed by endoscopy and/or imaging. 11 Evidence-based Diagnosis and Clinical Practice Guidelines (EDGs) for intestinal BD published by the Japanese Society for Behcet’s Disease (JSBD) in 2020 clearly presented the diagnostic criteria and algorithm. 8 Recently, the Inflammatory Enteropathy Group, Chinese Medical Association (IEGCMA) published a Chinese consensus on diagnosis and treatment of intestinal BD in 2022. 7
Recently, treat-to-target (T2T) strategies have been embraced in many chronic diseases, such as hypertension, diabetes, rheumatoid arthritis, and inflammatory bowel disease (IBD), including ulcerative colitis (UC) and CD. T2T strategies have improved long-term outcomes of related diseases. 12 With a deeper understanding of BD and the development of diagnosis, evaluation and treatment methods, Hatemi proposed for the first time in 2022 to implement a T2T strategy in the management of BD. 13 To date, there is no related reviews about the treatment principles and targets of intestinal BD in detail. Therefore, we review intestinal BD with treatment principles and targets to improve the clinicians’ understanding.
Treatment principles
Severity of disease and previous treatment principles
In the past decade, significant research results on BD have been published enabling a deeper understanding of disease behavior and management. Studies showed that any part of the alimentary tract can be affected in intestinal BD while the ileocecal area was the most frequently involved location. 4 Deep ulcers in intestinal BD were responsible for severe complications such as perforation, gastrointestinal bleeding and obstruction, which indicated that gastrointestinal involvement in BD was a predictive factor for a poor prognosis. 14 For example, Yook et al. 15 in 2013 conducted a retrospective analysis of 124 patients with intestinal BD and found that the incidence of intestinal perforation, fistula, stenosis and abscess was 12.7, 7.6, 7.2, and 3.3%, respectively. Hatemi et al. 6 reviewed the charts of all BD patients in their BD clinic in 2016 and found that 32% patients (N = 60) suffered from acute abdominal pain. 8 of these patients had massive bleeding while the other 11 patients had perforations. Another study in 2017 also showed that 11.2% patients (N = 588) with acute lower gastrointestinal bleeding were identified in patients with intestinal BD. Therefore, it was necessary to prompt timely treatment of intestinal BD to prevent severe complications and to achieve favorable prognosis. 16
In the field of rheumatology, the EULAR recommendations for the management of BD in 2008 and the guidelines for the diagnosis and treatment of BD published by the Rheumatology Society of Chinese Medical Association (RSCMA) in 2011 put forward the treatment principles in detail. The aim of BD treatment is to control symptoms and prevent irreversible organ damage. In addition, multisystem involvement mandates collaboration between different specialties.17,18 In the field of gastroenterology, Japanese gastroenterologists used to have extensively studied intestinal BD. The Japanese consensus statement for the diagnosis and management of intestinal BD was established and updated in 2007 and 2014, which paid more attention to the treatment goals than to the treatment principles. The EDGs for intestinal BD published by the JSBD in 2020 and Chinese consensus on intestinal BD published by the IEGCMA in 2022 also focused on the treatment goals.7,8,19,20
Updated treatment methods and treatment principles
The appropriate adoption of medical therapy was of great importance in intestinal BD as well as proper intervention with surgical management. 17 Conventional medications included 5-aminosalicylic acids, corticosteroids, immunomodulators such as azathioprine, thalidomide, which were capable of inducing and maintaining remission without the need for surgery in some patients. These drugs acted as key components of the standard therapy for intestinal BD according to the Japanese consensus of intestinal BD published in 2007. 9 However, some patients with intestinal BD were refractory to conventional treatment. Park et al. 18 performed a study regarding clinical outcomes after corticosteroid use in patients with moderate-to-severe intestinal BD. A total of 54 patients were enrolled and 46.3% of whom achieved complete remission after 1 month of treatment and 48.1% remained responsive to therapy after 1 year of treatment. Yet it was worth noting that 35.2% of these patients developed corticosteroid dependency and 7.4% received surgery after 1 year. The adoption of biological agents such as anti-tumor necrosis factor (TNF) agents in intestinal BD allowed for effective treatment of these corticosteroid-dependent and other complicated cases. Kinoshita et al. 19 reported that 15 Japanese patients with refractory intestinal BD who showed poor response to corticosteroid and conventional immunosuppressive agents received therapy of an anti-TNF agent (infliximab) at a single center and exhibited significant clinical response with 80% patients at 10 weeks, 64.0% at 12 months, and 50.0% at 24 months. Furthermore, in 2013 Lee et al. 21 conducted a Korean multicenter retrospective study of 28 patients with intestinal BD who received infliximab therapy. They showed that patients at 2, 4, 30, and 54 weeks after treatment presented a clinical response rate of 75.0, 64.3, 50, and 39.1%, and a clinical remission rate of 32.1, 28.6, 46.2, and 39.1%, respectively. It was suggested that infliximab was a well-tolerated and effective therapy for patients with moderate-to-severe intestinal BD. 22
According to these studies, the EULAR and RSCMA updated the guidelines in 2018 and 2021, respectively. The updated treatment principles can be summarized as follows: (1) The key point is to promptly suppress inflammatory exacerbations and prevent recurrences. (2) The prevention of irreversible organ damage and disease progression is the final goal. (3) A multidisciplinary approach and individualized treatment based on patient status, severity of organ involvement, and patient preferences are necessary.11,23 To some extent, it is similar to the principles of treatment for IBD, which aims for early induction of remission and long-term maintenance to prevent relapse. 24
Treatment targets
The treatment principles and targets complement each other and act as global treatment strategy. Up to now, the medical treatment target for intestinal BD is to prompt timely suppression of inflammation and maintenance of disease remission. Here, we elaborate the treatment target from three aspects as follows: evaluable markers, effective markers, and potency-ratio markers. Considering that there is a lack of high-quality research in the field of intestinal BD, some definitions and conceptions are cited from IBD. 12
Evaluable markers: Where should we pick up treatment targets from?
Clinical manifestations
In 2011, Cheon et al. 25 first proposed the Disease Activity Index for Intestinal BD (DAIBD) to evaluate the disease activity of intestinal BD. The DAIBD provides a score between 0 and 325 based on an 8-point index that includes symptoms and signs of patients. It classifies disease activity as quiescent (⩽19), mild (20–39), moderate (40–74) and severe (⩾75). The study enrolled a total of 110 patients with intestinal BD, and the results revealed that the DAIBD showed much higher responsiveness than the Crohn’s Disease Activity Index (CDAI) (r = 0.81 versus 0.65, respectively). Park et al. 26 in 2013 reported that DAIBD had predictive value for prognosis and outcome of intestinal BD: ① DAIBD ⩾ 70 was a predictive factor for surgical intervention [Hazard ratio (HR) 5.43; 95% CI 2.65–11.10]; ② DAIBD ⩾ 60 was an independent factor associated with relapse for patients during 5-ASA therapy (HR 2.30; 95% CI 1.10–4.83); ③ DAIBD ⩾ 40 at the time of diagnosis was independently associated with a more severe clinical course (OR 6.20; 95% CI 1.10–33.50). However, some studies have shown that a portion of patients with intestinal BD in clinical remission (DAIBD ⩽ 19) still have endoscopic disease activity. 27
Endoscopy
Endoscopic evaluation of intestinal BD mainly includes gastroscopy, colonoscopy, enteroscopy and capsule endoscopy. According to the guidelines established by the IBD Study Group of the Korean Association of the Study of Intestinal Diseases, the typical ulcerations of intestinal BD were described as few in number (⩽5), large, discrete, and round or oval-shaped ulcerations in the ileocecal area (Figure 1(a)). The other types of ulcers include small aphthous ulcerations to multiple irregularly shaped ulcerations and are defined as atypical ulcerations. 9 A multivariate regression analysis of 167 intestinal BD patients conducted by Lee et al. 27 revealed that the number of intestinal ulcers (⩾2, p = 0.031) and volcano-shaped ulcers (p = 0.001) were predictive factors for severe DAIBD scores and disease activity. Mucosal healing (MH) has been one of the main therapeutic targets stipulated by recent authoritative recommendations and guidelines in IBD and is associated with better long-term outcomes.24,28 The definition of MH currently lacks consensus. A Mayo endoscopic subscore of 0 or 1 or an SES-CD (simple endoscopic score for CD) less than 3 is recognized by most experts as an acceptable measure of MH and has been widely used in UC and CD.29,30 With a deep understanding of intestinal BD, recent studies suggest that MH is a potential predictive factor for the long-term prognosis of intestinal BD (Figure 1(b)). A meta-analysis by Wu et al. 31 in 2020 revealed that MH is associated with a decreased risk of disease recurrence (RR 0.41; 95% CI 0.30–0.57) and surgery (RR 0.33; 95% CI 0.17–0.63) in intestinal BD. However, in clinical practice, there are contraindications for endoscopic examination of some patients, as it is invasive. To date, there are no surrogate noninvasive markers of endoscopy.
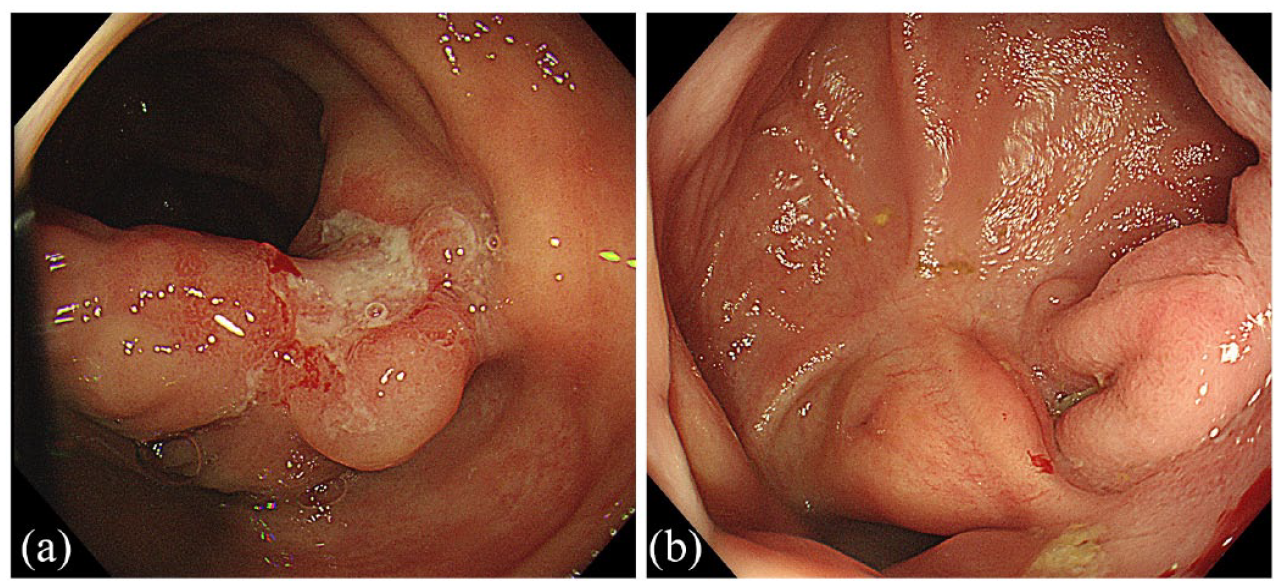
Endoscopic findings of intestinal Behcet’s disease. (a) Typical ulcer in ileocecal area before treatment; (b) Mucosal healing of ulcer in ileocecal area after treatment.
Radiology
The radiologic evaluation of intestinal BD is mainly based on computed tomography enterography (CTE). On CTE, intestinal BD commonly manifests as geographic, relatively large, and deep penetrating ulcers with bowel wall thickening and mural hyperenhancement in the ileocecal region. Occasionally, intestinal BD manifests as a cecal mass or an aneurysmal dilatation of the terminal ileum masquerading as a malignant tumor. Patients with gastrointestinal complications such as intestinal perforation, fistula, stenosis and abscess may have corresponding imaging findings (Figure 2). 32 Recently, magnetic resonance enterography and bowel ultrasonography have also been increasingly used in the diagnosis and treatment of chronic intestinal inflammatory disease. 33 One study by Peker et al. 34 revealed that a polypoid pattern and homogeneous mural enhancement were the findings seen more frequently in BD (p < 0.001) than in CD (p = 0.003). Another study by Ma et al. 35 in 2021 showed that focal lesions (OR 0.16, 95% CI: 0.04–0.56) and large ulcers (OR 0.06; 95% CI: 0.01–0.55) were independent predictors of intestinal BD over CD in bowel ultrasonography. Transmural healing (TH) refers to healing of the full-thickness bowel wall under radiology. At present, there is no internationally recognized definition, and most studies use bowel wall thickness (usually 3 mm or less as the critical value) for evaluation. 33 There were more significant research results for TH in CD than in UC: ① A study of 218 CD patients who completed a 2-year treatment course with anti-TNF-α agents showed that TH was associated with longer intervals until clinical relapse (HR 0.87, p = 0.01), hospitalization (HR 0.88, p = 0.002) and surgery (HR 0.94, p = 0.008) than MH. 36 ② There is controversy regarding the correlation between TH and MH in CD. In two studies of CD patients (N = 66 and N = 90), a good correlation was observed between MH and TH (k = 0.63, p < 0.001; k = 0.64, p < 0.001).37,38 Another study of CD patients (N = 214) revealed that TH and MH are associated with improved long-term outcomes in CD, but there was poor correlation between TH and MH. 39 ③ An update on the Selecting Therapeutic Targets in IBD (STRIDE-II) conducted by the International Organization for the Study of IBD in 2021 recommended that TH should be used as an adjunct to MH to represent a deeper level of healing in CD. 12 We speculate that this may be related to the pathophysiological mechanism of CD as full-thickness inflammation.

Radiological findings of intestinal Behcet’s disease with complications. (a) Wall thickening of ileocecal area with luminal stenosis and mural hyperenhancement; (b) Intestinal- abdominal fistula and mural abscess.
Histology
The main techniques to obtain pathology of intestinal BD include surgical resection and endoscopic biopsy. It has been shown in typical surgical pathology results that vasculitis, such as fibrinoid necrosis and inflammatory cell infiltration of small blood vessels in the bowel and mesentery, could be found (Figure 3(a)). It is relatively hard to obtain evidence of vasculitis in endoscopic biopsy, as the material is usually from the mucosal layer of pathological tissue (Figure 3(b)).23,40 Histological healing (HH) refers to the histopathology that indicates the improvement of inflammation and the repair of damaged structures. The research results of HH in UC were similar to those of TH in CD, which showed that HH may be superior to MH in long-term remission and cancer prevention.41–43 HH is also recommended as an adjunct to MH in UC in the STRIDE-II consensus. 12 In my opinion, this may be related to the fact that the pathophysiological mechanism of UC is superficial intestinal inflammation, which makes it easier to see bowel wall inflammation in the mucosal samples obtained by endoscopic biopsy.

Pathological findings of tissue specimens in ileocecal area of intestinal Behcet’s disease. (a) (from right hemicolectomy): Ulceration of ileocecal region and abscess formation with thickening of vascular wall and swelling of endometrium; (b) (from colonoscopy biopsy): Nonspecific mucosal inflammation and cryptitis.
Biomarkers
Erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) are important biomarkers for evaluating disease activity in intestinal BD despite the lack of specific factors to date. Bai et al. 44 conducted a retrospective study of patients with intestinal BD (N = 29) in 2007 and revealed that ESR and CRP increased significantly during disease activity (92.3 and 92.0%). Two studies by Jung YS et al.45,46 in 2011 and 2012 revealed that a CRP level ⩾ 0.4 mg/dL was an independent predictive factor for postoperative recurrence and reoperation, while a CRP level ⩾ 1.5 mg/dL was associated with a poor response to 5-ASA therapy. The important role of fecal calprotectin (FC) in the evaluation of chronic intestinal inflammatory diseases such as IBD has been proven in previous studies.47,48 In recent years, increasingly more studies have shown that FC is helpful in the diagnosis of intestinal BD and associated with disease activity.49–51 Further research is needed for large-scale clinical application in consideration of the variable cutoff values, false positive by drug interference and storage at room temperature. 52 In addition, FC may be a potential noninvasive biomarker of MH and is correlated with TH and HH in IBD according to some research, and it is also worth further attention and research in intestinal BD.53–56
Effective markers: How should we pick up effective treatment targets?
According to the characteristics of the above evaluable marker and relevant research, the EDGs for intestinal BD by the JSBD in 2020 and the RSCMA guidelines for BD in 2021 updated the treatment targets that clinical remission and the normalization of CRP are the initial targets, and MH was recommended as a further target, which was supported by the Chinese consensus on intestinal BD published by the IEGCMA in 2022.7,8,23 TH and HH, which indicated deeper remission, were not included in the guidelines as treatment targets due to a lack of relevant research in intestinal BD. We speculate that TH may be more promising in the further study of intestinal BD than HH, considering that intestinal BD is more similar to CD in its clinical symptoms, imaging findings and endoscopic characteristics than UC. 13 It is worth noting that some studies in IBD revealed that the combination of evaluable markers (e.g. CRP and FC, CDAI and FC) may be promising treatment targets according to the effectiveness of disease-activity evaluation.57,58 This new idea is worthy of reference for intestinal BD. In clinical practice, some patients with vasculitis, such as BD, may suffer from irreversible damage to the involved organs and blood vessels, such as coronary artery stenosis and ischemia, pulmonary hypertension, renal insufficiency and so on, while inflammation is well controlled. Moreover, it is well known that ischemia of blood-supply mesenteric vessels is the cause of ischemic bowel disease and one of the causes of refractory duodenal ulcer. 59 Therefore, we suggest that future studies should focus on whether the definition of MH in intestinal BD needs to be adjusted or modified.
Potency-ratio markers: Why should we weigh the pros and cons of different treatment targets?
The concept ‘the deeper the remission, the better the outcomes’ has been recognized by an increasing number of clinicians engaged in chronic intestinal inflammatory diseases. 12 However, immunosuppressive drugs such as glucocorticoids, traditional immunosuppressants, and biological agents have their related adverse drug reactions, which may be life-threatening in serious conditions. In addition, some drugs are relatively expensive.60–62 Therefore, seeking deeper remission may be associated with more adverse reactions and costs, especially for patients with advanced age, immunosuppressed conditions, underlying diseases, high risk of treatment and poor economic status. Consequently, the EDG for intestinal BD in 2020 by the JSBD emphasized that adverse events due to enhanced treatment must be comprehensively considered before upgrading the treatment target. 8 In addition, a simple algorithm for the treatment goals in UC and CD suggested by the STRIDE-II consensus is worthy of reference for clinicians engaged in the field of intestinal BD. The key points are summarized as follows: ① It is emphasized that it is necessary to balance the benefits and risks before adjusting the treatment targets; ② the treatment targets are stratified according to time, which means that remission of clinical symptoms and biomarkers presents as early and initial goals, while deep remission acts as a late and final goal; ③ the treatment targets are individualized based on the disease characteristics such as MH and HH for UC and MH and TH for CD. 12
Key points to be made clear
What’s more, there were several key points which should be made clear in our review. It was listed in a table form as follows (Table 1): ① Application: as we all knew, intestinal BD was gastrointestinal involvement of BD while BD was an autoimmune disease with multiple organ involvement. For patients with BD, skin, mucosa, and joint involvement could cause impairment of quality of life but do not cause permanent damage whereas untreated eye, vascular, nervous system, and gastrointestinal system involvement could cause serious damage and even death. 63 And it was worth noting for clinicians in disease assessment that the treatment targets mentioned in our review applied to gastrointestinal assessment of BD after treatment especially presenting mainly with gastrointestinal involvement. The treatment targets for BD needed to be multidimensional as it was a systemic vasculitis which can affect multiple organs with different course and prognostic importance 12 ; ② Reference: in consideration of lack of high-quality research in the field of intestinal BD and clinical similarity between intestinal BD and IBD to some extent, we reviewed the treatment targets of intestinal BD by mentioning several definitions and conceptions in IBD for partial reference. However, it was also worth noting that intestinal BD and IBD were clearly different kinds of diseases. The former was the gastrointestinal manifestations of chronic recurrent systemic vasculitis while the latter was a disease of nonspecific chronic inflammation confined to the intestine with different which could have extraintestinal manifestations during the clinical course.40,64 Therefore, as we mentioned above, further studies should be conducted to explore whether the definitions and conceptions from IBD needed to be adjusted or modified in intestinal BD; ③ Significance: we reviewed the treatment principles and targets of intestinal BD, aiming to improve clinicians’ global understanding of the disease treatment. Certainly, the concrete medical treatment plans in intestinal BD such as reduction strategies of corticosteroid, advantages and disadvantages of different drugs, and application of biologic agents and conventional immunosuppressive agents were of the same importance which needs further discussion in the future.65,66
Key points to be made clear.

BD, Behcet’s disease.
Conclusion
In summary, BD is a systemic vasculitis with multiple organ involvement. For intestinal BD namely BD with predominant gastrointestinal manifestations, we review the global treatment strategy with treatment principles and targets. The key points of the treatment principles of intestinal BD focus on prompt suppression of inflammatory exacerbations and prevention of recurrences to prevent irreversible organ damage and disease progression. As for treatment targets in intestinal BD, further seeking MH is expected to have a better prognosis after achieving remission of clinical symptoms and normalization of CRP. Subsequent multicenter, prospective and large-sample-size studies are needed for further exploration of treatment targets, such as noninvasive markers and whether TH and HH can improve the long-term prognosis of intestinal BD.
