Abstract
Background:
Patients with inflammatory bowel disease (IBD) are at an increased risk of developing Clostridioides difficile infection (CDI). Treatment of CDI in patients with IBD is challenging due to higher failure rates and concomitant IBD activity.
Objectives:
We performed a multicentre cohort study in patients with IBD who received fecal microbiota transplantation (FMT) for recurrent CDI (rCDI), to further investigate factors that influence the clinical outcome and course of both rCDI and IBD.
Design:
This is a multicentre cohort study conducted in five European FMT centres.
Methods:
Adult IBD patients treated with FMT for rCDI were studied. Cure was defined as clinical resolution of diarrhoea or diarrhoea with a negative C. difficile test. The definition of an IBD flare was record based. Long-term follow-up data were collected including new episodes of CDI, IBD flares, infections, hospital admissions, and death.
Results:
In total, 113 IBD patients underwent FMT because of rCDI. Mean age of the patients was 48 years; 64% had ulcerative colitis. Concomitant rCDI was associated with an IBD flare in 54%, of whom 63% had received IBD remission-induction therapy prior to FMT. All FMT procedures were preceded by vancomycin treatment, 40% of patients received FMT via colonoscopy. CDI cure rate was 71%. Long-term follow-up data were available in 90 patients with a median follow-up of 784 days (402–1251). IBD activity decreased in 39% of patients who had active IBD at baseline, whereas an IBD flare occurred in only 5%. During follow-up of up to 2 years, 27% of the patients had infections, 39% were hospitalized, 5% underwent colectomy, and 10% died (median age of these latter patients: 72 years).
Conclusion:
FMT for rCDI in IBD patients is safe and effective, and IBD exacerbation after FMT is infrequent. Further studies should investigate the effects on IBD course following FMT.
Introduction
Clostridioides difficile infection (CDI) incidence is increasing and associated with significant mortality and morbidity worldwide. 1 Risk factors for CDI are patient aged above 60 years, severe underlying illnesses, antimicrobial therapy, hospital stay, and the use of proton pump inhibitors (PPIs).2,3 Patients with inflammatory bowel disease (IBD) are at increased risk of developing CDI. IBD is characterized by a chronic and relapsing course of inflammation requiring long-term immunosuppressive medical therapy and periodic hospitalization. 4
The reduced diversity and perturbed gut microbiota in IBD patients may predispose or affect disease course in CDI. 5 Distinguishing an active episode of CDI from an IBD flare is a clinical challenge because symptoms are overlapping, which is complicated by a higher incidence of asymptomatic colonization in IBD patients. 6 Available microbiological tests lack specificity and sensitivity in IBD patients, 7 and the specific C. difficile pseudomembranes at endoscopy are only present in few patients.8,9
The first episode of a CDI is treated with vancomycin or fidaxomicin, which is also advised for the first CDI recurrence. For a second recurrence of CDI, extended antibiotic courses and fecal microbiota transplantation (FMT) are advised. 10 The cure rate after FMT is up to 94% in patients with recurrent CDI (rCDI),11–13 and comparable results were noted in IBD patients with CDI treated with FMT. 14 Caution has been advised when administering FMT in patients with IBD due to a potential risk to develop an IBD flare after FMT.14,15 In patients with rCDI and a concomitant IBD flare, most physicians would simultaneously initiate antibiotic treatment and remission-induction therapy for the IBD flare. Yet, the optimal timing of FMT in those patients is unknown.
To further investigate the factors that influence the clinical outcome and course of both rCDI and IBD, we performed a study of prospectively registered IBD patients who received FMT for rCDI at one of the five European FMT centres.
Materials and methods
Study population
Adult IBD patients with active disease or IBD in remission with a confirmed diagnosis of Crohn’s disease (CD), ulcerative colitis (UC), or IBD-unclassified treated with FMT for rCDI were included. There were no specific exclusion criteria. All eligible patients provided informed consent at the participating centres at the moment of FMT according to local requirements.
Participating centres and available data
A multicentre, observational study was conducted with data from five European FMT expertise centres: Leiden University Medical Centre in the Netherlands, Aarhus University Hospital in Denmark, Saint-Antoine Hospital Paris in France, Gemelli University Hospital Rome in Italy, and the Microtrans Registry in Germany. The reporting of this study conforms to the STROBE statement. 16 Most patients were treated in the participating centres itself, except for patients from the Netherlands, who were treated with FMT in hospitals throughout the Netherlands but facilitated by a centralized stool bank; and patients documented in the Microtrans Registry that were treated at different sites in Germany. Data about CDI outcome were collected from all patients and the course of IBD was assessed from treated patients in four out of five expertise centres.
Data collection
In all centres, patients undergoing FMT treatment were registered prospectively. Data about IBD outcome and long-term follow-up were in part collected retrospectively (Supplemental Figure 1).
Data collection was performed by each centre using files of the FMT services and hospital records for the patients. If possible, patients were contacted directly. The following baseline characteristics were collected: age, gender, and the use of PPIs. The following data about the CDI were collected: number of episodes; diagnostics by polymerase chain reaction or toxin enzyme immunoassay; and information about previous treatment with metronidazole, vancomycin, fidaxomicin, or bezlotoxumab. Severe CDI was defined as leukocytes ⩾15 × 109/L and/or a 50% increase in creatinine at baseline. 17 FMT data included the pre-treatment regimen (antibiotics, bowel lavage), total number of FMTs needed per patient, the route of administration of FMT, and the total amount of faeces (grams) used for preparation of the suspensions or capsules that were administered per patient. Data about clinical recurrence and microbiological testing for CDI after FMT were collected at 8–12 weeks after FMT. Long-term follow-up data of CDI recurrence were included if available.
For IBD, information was collected about the diagnosis according to the Montreal classification and the disease duration. Previous and current IBD medication at the moment of FMT and the use of immunosuppressive medication (including corticosteroids and budesonide, immunomodulators and biologicals) was assessed. Both at baseline and 8 weeks after FMT, the presence of an IBD flare was based on information from the treating physician and/or endoscopic scores. In case of a concomitant flare, remission-induction therapy was defined as the use of prednisolone or budesonide at the moment of FMT, or recently initiated antitumor necrosis factor (TNF) treatment (⩽2 months before FMT). Also haemoglobin (mmol/l) and C-reactive protein (mg/l) in the blood and the calprotectin (µg/g) in the faeces were collected at baseline and after 8 weeks.
The long-term follow-up period per patient was calculated from the date of FMT up to 31 December 2020. Long-term follow-up data included information about possibly occurring events and if yes, the number of days after FMT it occurred. Possible occurring events, collected via patient recall or from hospital records, were as follows: a recurrence of CDI, the development of an IBD flare, general infection and antibiotic use, hospital admission, colectomy, and occurrence of death.
Outcome measures
Cure of a CDI was determined 8 weeks after FMT, defined as clinical resolution of diarrhoea or diarrhoea with a negative C. difficile test. A positive C. difficile test and persisting complaints of diarrhoea were defined as no cure. Also the IBD disease activity 8 weeks after an FMT was tracked, based on the judgement of the treating physician.
Statistical analysis
Continuous variables are presented as mean with standard deviation or as median with interquartile range depending on the normality of the underlying distribution. Baseline characteristics were compared using an independent sample-t test, in case of >2 groups a one-way ANOVA test or the Kruskal–Wallis test was used. Paired variables were compared using a paired sample t-test or Wilcoxon signed-rank test. Categorical variables were presented as a total percentage and compared using the chi-square test or Fisher’s exact test in case of <5 persons per group. The two-sided p values ⩽0.05 were considered statistically significant. All data analyses were performed using SPSS, version 25.0.
Results
Baseline patient characteristics
A total of 113 patients with IBD and CDI was included from five European FMT centres (Supplemental Figure 1). Mean patient age was 48.4 years, 54% of the patients were female. Patients were primarily diagnosed with UC (64%). Patients suffered of a mean number of 3.8 CDI episodes. Antibiotic pre-treatment with vancomycin was administered in all but one patient (Table 1). Overall, 40% received FMT via colonoscopy, 27% via nasoduodenal tube, 25% via capsules, 5% via enemas, and 4% via gastroscopy with marked variation between centres. In total, 63% of patients (69/110) received immunosuppressive medication because of IBD, among which 34 (38%) received biologicals (Supplemental Table 1).
The following five centres participated: Leiden University Medical Center (LUMC) in the Netherlands, Aarhus University Hospital in Denmark, Saint-Antoine Hospital Paris in France, Gemelli University Hospital Rome in Italy, and Frankfurt University Hospital in Germany.
Due to missing values, the numbers assessed per clinical variable differ; more detailed information is shown in supplemental Table 1. 5-ASA, 5-aminosalicylic acid; CDI, Clostridioides difficile infection; FMT, fecal microbiota transplantation; IBD, inflammatory bowel disease; IQR, interquartile range; N, number; N.A., not applicable; PPI, proton pump inhibitor; SD, standard deviation; UC, ulcerative colitis; Y, year.
Data on IBD activity at baseline and long-term follow-up were available for 90 patients from four institutions. rCDI was accompanied by activity of IBD in 54% of patients (49/90). In 63% (31/49), concomitant remission-induction therapy was started before FMT was scheduled (prednisolone (n = 28) or anti-TNF (n = 3).
CDI outcomes
In total, 113 patients underwent FMT because of rCDI with faecal preparations (capsules or suspensions) made from ~50 (49.5–55.0) grams of faeces. Eight patients were excluded from the analysis, because the 8-week follow-up was not available for the following reasons: death not related to FMT 16, 23, and 24 days after FMT (n = 3), initiation of palliative care because of lung cancer 43 days after FMT (n = 1), and missing data (n = 4). Thus, assessment of cure at 8 weeks included 105 patients. Three of those 105 patients did not complete the 8-week follow-up because of an surgical ileostomy due to therapy refractory CDI and an IBD flare 4 days after FMT (n = 1), and colectomy 15 and 16 days after FMT because of severe UC (n = 2). Those three patients were included in the analysis and considered as failures, although persisting CDI was not proven. At 8-week post-FMT, 71% of patients (75/105) had a resolution of their diarrhoea or a negative CDI test. Four patients received repeated FMTs in serial, but despite this they had no improvement of their complaints at week 8.
From 90 patients, long-term follow-up was available, with a median of 784 days (402–1251). From one centre, long-term follow-up of treated patients (n = 23) was not available. Sustained cure after FMT occurred in 54 of 86 patients (62.8%).
Factors influencing outcome of FMT treatment for rCDI
From 105 patients, outcome of CDI at 8 weeks after FMT was assessed. Comparing baseline characteristics between patients who were cured (75/105) and patients with a post-FMT recurrence of CDI (30/105), no differences were found. There was no difference between route of administration of FMT (p = 0.412) nor the number of CDI episodes before patients were treated with FMT (p = 0.478). There was no significant association between IBD medication used and the outcome for FMT for CDI.
Also, receiving immunosuppressive medication did not influence outcome with regard to CDI resolution (p = 0.851, Table 2).
Factors of influence on CDI cure at 8 weeks after FMT.
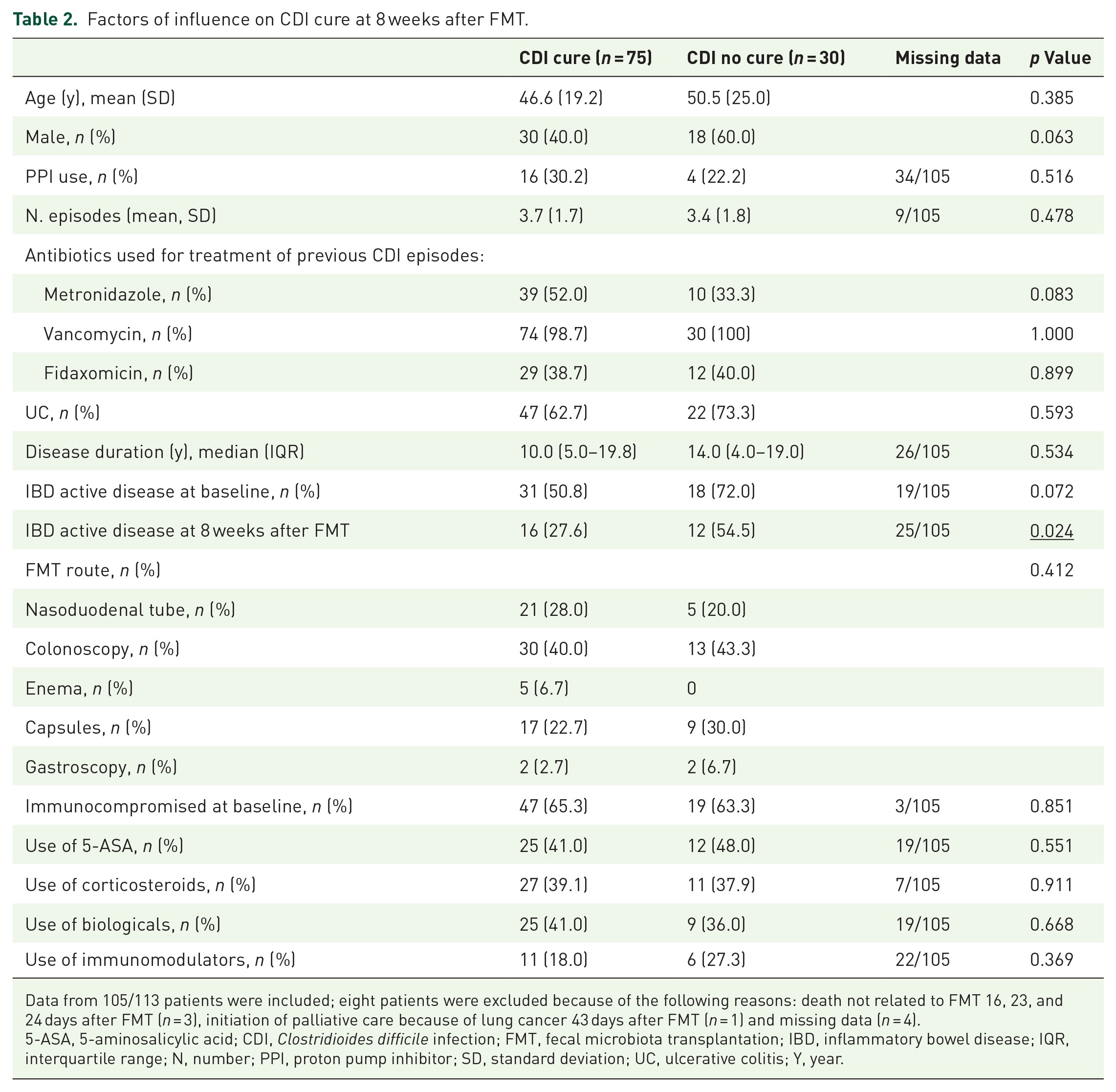
Data from 105/113 patients were included; eight patients were excluded because of the following reasons: death not related to FMT 16, 23, and 24 days after FMT (n = 3), initiation of palliative care because of lung cancer 43 days after FMT (n = 1) and missing data (n = 4).
5-ASA, 5-aminosalicylic acid; CDI, Clostridioides difficile infection; FMT, fecal microbiota transplantation; IBD, inflammatory bowel disease; IQR, interquartile range; N, number; PPI, proton pump inhibitor; SD, standard deviation; UC, ulcerative colitis; Y, year.
IBD outcomes
From four centres, including a total of 90 patients, information about the IBD status at the moment of FMT was available. At the moment of FMT, 54% of patients (49/90) had IBD activity concomitant with the CDI episode for which FMT was performed, whereas IBD was in remission in 46% of patients (41/90) at the time of FMT.
From seven patients, data on activity of IBD at 8 weeks were lacking because death not related to FMT (n = 3), palliative care (n = 1), or missing data (n = 3). At 8 weeks after FMT, 63% (52/83) had no signs of IBD activity, 34% (28/83) had active IBD after 8 weeks, and 4% (3/83) was operated, due to therapy refractory IBD 4 days after FMT (ileostoma), and 15 and 16 days after FMT (colectomy).
Of the 41 patients with reported IBD in remission before FMT, 5% (2/41) had active IBD at 8 weeks after FMT. Endoscopy was not performed in those patients before FMT, and pre-existent activity cannot be excluded. Interestingly, of the 49 patients with active disease at baseline, 38.8% (19/49) had an improvement of their IBD complaints after FMT, which may also be related to remission-induction therapy that was initiated before FMT. In 75% (62/83) of the patients, FMT performed for rCDI did not influence IBD disease activity after FMT; 80% (33/41) was still in remission, and 59% (29/49) still had active IBD or were operated due to severe disease activity (n = 3) during the 8 weeks following FMT.
Interestingly, only 27.6% (16/58) of the CDI-cured patients still had activity of IBD at 8 weeks, whereas 54.5% (12/22) of patients that were not cured of CDI had activity of IBD at 8 weeks post-FMT (p = 0.024, Table 2).
From 82 of 90 patients, follow-up data about IBD outcomes beyond 8 weeks with a median follow-up after FMT of 492 days (206–859) were available. From eight patients, follow-up data about IBD outcomes beyond 8 weeks were lacking because of colectomy (n = 3), death (n = 3), or palliative care (n = 1) within 8 weeks and missing data (n = 1)). An IBD flare occurred in 32% (26/82) later than 8 weeks after FMT, with a median time of 231 days (166–428) after the FMT. Of these 26 patients, 10 patients did not have an IBD flare at the moment of FMT. Because the IBD flare in these patients occurred >8 weeks after FMT, these flares were considered to reflect the relapsing and remitting course of IBD, and not likely caused by FMT.
Safety
Long-term follow-up data were available in 90 patients with a median follow-up of 784 days (402–1251). During follow-up, four (5%) patients underwent a colectomy of which two patients had their surgery 15 and 16 days after the FMT. In those two patients, disease activity at baseline was severe and repeated FMTs were offered. One more patient was operated and received an ileostomy 4 days after FMT because of severe UC, which was already going on before FMT.
In total, 10% (9/90) of the patients died during follow-up, with a median time of 274 days (23.5–669.0) after FMT (Table 3). The median age of these patients was 72 years (62.0–76.5). Furthermore, 27% (22/83) developed an infection that required treatment with antibiotics, after a median time of 162 days (21–560) after the FMT, and 39% (33/85) was admitted to the hospital after a median time of 213 days (55–516) after FMT. Reasons for hospital admission were as follows: infection (n = 14), IBD flare (n = 9), and other reasons (n = 10).
Overview of events during long-term follow-up data.

CMV, cytomegalovirus; DVT, deep vein thrombosis; FMT, fecal microbiota transplantation; IBD, inflammatory bowel disease; n, number; STEMI, ST-elevation myocardial infarction.
Discussion
In this European multicentre cohort study of FMT provided for rCDI in patients with IBD, we found an overall rCDI resolution rate after 8 weeks of 71% following one FMT and a very low risk of FMT-induced IBD flare.
Treatment of CDI in patients with IBD is challenging, because both CDI and IBD impact on each other’s pathogenesis. 18 The resolution rate reported in this study is slightly lower than those in patients with rCDI but without IBD. Still, our observations on CDI in IBD are particularly encouraging because no other reasonable alternative treatment options exist. Importantly, FMT appears safe in patients with IBD and CDI, and exacerbations induced by FMT were very rare. In our cohort, two-thirds were judged as having no activity of IBD (remission) at time of rCDI. After FMT treatment, two of these patients had a flare of IBD. Endoscopy was not performed prior to FMT to confirm remission of IBD, and activity may have been present before FMT and being masked by concomitant rCDI. The low number of IBD flares encountered in our cohort contrasts with previous studies suggesting exacerbations of IBD after FMT in up to 22% of patients. 19
IBD predisposes to both infection and colonization with C. difficile, probably because of the disturbed microbiota associated with IBD. There are several difficulties related to assessing and treating patients with concomitant IBD and CDI. First, it may be difficult to interpret a positive C. difficile stool test, as it may point to carriership without clinical consequences, carriership maintaining disease activity, or diarrhoea associated with an active CDI. In general, endoscopy is readily performed in those patients. In case of disease activity of IBD, both C. difficile and IBD activity require treatment. Unfortunately, antibiotic treatment of CDI in patients with IBD appears less effective, and the course of IBD may be more severe in the presence of CDI. 7 In our cohort, most patients with rCDI and active IBD were treated with prednisolone before FMT was performed. This strategy appears safe, without reported serious adverse events related to FMT. However, three patients in our cohort required colectomy or surgical ileostomy because of ongoing severe IBD activity, which underlines the need for continuous treatment of IBD during treatment of CDI. Interestingly, patients without disease activity at 8 weeks after FMT were more likely to be cured by FMT compared with those with disease activity at 8 weeks after FMT, emphasizing the need for treatment of IBD activity concomitantly with treatment of CDI. Although controlled trials are lacking, we propose that rCDI in patients with concomitant IBD activity should be treated with remission-induction therapy (either prednisolone or biologicals) in combination with prolonged antibiotic therapy (vancomycin or fidaxomicin). A reasonable suggestion for a treatment algorithm is presented in Figure 1.

Proposed treatment algorithm FMT in IBD patients.
Despite attempts to standardize FMT in Europe,20,21 protocols for FMT treatment still differ between centres. However, baseline patient characteristics were similar between centres, suggesting that the participating centres apply common criteria for selection of patients with IBD and CDI for FMT. All centres generally pre-treat patients with vancomycin. Different routes of installation illustrate local differences. All options were effective in this study, and future studies could identify preferred routes in patients with IBD.22,23 For now, the choice for the delivery route of FMT should be based mainly on patient characteristics and local preferences. 21
FMT is an established treatment approach for treatment of rCDI or severe CDI, but also appears a promising treatment strategy in a subset of patients with active UC 24 and is being studied for treatment in CD. 25 While a single FMT in rCDI patients generally suffices to achieve CDI resolution, it seems that repeated administration of FMT is required to improve clinical outcomes in patients with IBD. 26 However, in CDI patients without prompt improvement after FMT, early repeat FMT seems warranted as well. This approach is also required in a subset of patients with severe and antibiotic refractory CDI. In our cohort, eight patients received two FMTs as part of the treatment of rCDI. This illustrates that patients with IBD after FMT require close monitoring for response. Although FMT may positively influence the course of IBD in a subset of patients, IBD activity remained unchanged after one FMT in most of our patients. Although improvement of IBD disease activity after FMT was reported in 39% of our patients with active disease at baseline, this seems most likely explained by remission-induction therapy initiated before FMT. Although this cannot be excluded, it seems unlikely that a single FMT contributed to this effect in our cohort. Future studies may elucidate whether resolution of CDI in IBD may positively influence the course of IBD in those patients.
Immunosuppressive medication is the cornerstone of IBD treatment and was used by 63% of patients in our cohort. This did not affect outcome of FMT for CDI, and FMT seems safe in patients with immunosuppressive medication provided that the donor screening and storage of samples is performed according to consensus guidelines.20,23 Although FMT appears safe in immunocompromised patients, 21 the comorbidity and vulnerability of patients with IBD and rCDI require consideration. Recently, Zhao et al. provided an updated review of the burden of IBD in general by discussing the latest population-based studies, including data about the disease course, hospitalization, and mortality rate. This review showed that hospitalization rates in IBD patients were still high. 27 This was similar in our cohort, in which 39% of IBD patients with CDI were admitted to the hospital for various reasons and 27% of them suffered from an infection that required antibiotic treatment during long-term follow-up. Of note, 10% of these patients with IBD and CDI died during follow-up. Despite the fact that patients who died had a median age of 72 years, this mortality rate seems relatively high and points to the frailty of this specific group of patients with IBD suffering from rCDI. However, FMT appears safe in patients with IBD and CDI, and IBD exacerbation after FMT is infrequent. Osman et al. underlined the favourable short-term safety profile of FMT as treatment for CDI in a large real-life cohort with more than 5000 patients. They observed only one case of an IBD flare in a patient with a background of uncontrolled UC on biologics and immunomodulators. Of note, it is not entirely clear how many patients with IBD were included in this study. 28 Long-term follow-up safety data of FMT treated patients are more sparse. In a controlled study of 84 rCDI patients, the long-term effect of FMT treatment was compared with the outcome of antibiotic treated patients. With an average follow-up of 3.8 years, there was no increased risk of severe diseases such as cancer and autoimmune diseases in FMT-treated patients compared to the control group. 29 Although progress has been made and FMT for rCDI in IBD patients seems to be safe and an acceptable treatment, larger prospective studies and (international) registries are required to gain more insights into long-term follow-up data for these patients.30,31
Limitations apply to this study. First, the focus of the FMT centres that contributed patients to this study was treatment of rCDI. Data about the course of IBD were lacking in a subset of patients and not collected in a standardized way. Therefore, the presence of IBD activity was assessed based on information from the treating physician and not always based on the endoscopy scores. Furthermore, patients were not treated in a controlled clinical study, but as part of standard patient care. Consequently, follow-up was less stringent, explaining the missing data from a subset of patients. The latter limitation may, however, also be considered a strength, as our study population represents a real-life patient cohort with data collected by several established stool banks in Europe. In fact, this is the first international multicentre study assessing the effects of rCDI in patients with IBD, with a prolonged follow-up period of up to 2 years, providing insights into the recurrence of CDI after >8 weeks after FMT, and pointing to the vulnerability of this specific group of patients.
In conclusion, FMT in patients with IBD and rCDI appears safe and moderately effective. Treatment should be directed against both activity of IBD if present and rCDI. Concomitant IBD activity, comorbidities, and the use of immunosuppressive treatment call for careful treatment planning and monitoring during follow-up.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231156285 – Supplemental material for Short- and long-term follow-up after fecal microbiota transplantation as treatment for recurrent Clostridioides difficile infection in patients with inflammatory bowel disease
Supplemental material, sj-docx-1-tag-10.1177_17562848231156285 for Short- and long-term follow-up after fecal microbiota transplantation as treatment for recurrent Clostridioides difficile infection in patients with inflammatory bowel disease by Emilie (E.) van Lingen, Simon (S. M. D.) Baunwall, Simone (S. C.) Lieberknecht, Nicolas (N.) Benech, Gianluca (G.) Ianiro, Harry (H.) Sokol, Alessandro (A.) Gasbarrini, Giovanni (G.) Cammarota, Marcel (M. K.) Eriksen, Andrea (A. E.) van der Meulen-de Jong, Elizabeth (E. M.) Terveer, Hein (H. W.) Verspaget, Maria (M.) Vehreschild, Christian (C. L.) Hvas and Josbert (J. J.) Keller in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

