Abstract
Immune checkpoint inhibitors (ICIs) have transformed the treatment of patients with advanced cancers. However, the majority of patients do not respond or develop early progressive disease. A substantial number also develop immune-mediated toxicities that may lead to early treatment discontinuation. Gastrointestinal toxicities in the form of diarrhea and colitis are common and may resemble that observed in patients with inflammatory bowel disease (IBD). Alterations in the gut microbiota are thought to play an important role in mediating the intestinal inflammation that is associated with immune-mediated colitis. In this review, the authors’ objective is to provide an overview of the gastrointestinal and hepatic toxicities that can be seen with ICIs and discuss the interactions between gut microbiota and the immune response. The authors also highlight the potential role for fecal microbial transfer (FMT) as an approach to improve therapeutic efficacy and decrease toxicity.
Keywords
Introduction
Immune checkpoint inhibitors (ICIs) are now widely used in the treatment of cancer and have had a significant impact on survival in patients with advanced disease. Inhibition of cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), the programmed cell death protein 1 (PD-1), and its ligand (PD-L1) are now standard of care therapies for multiple tumor types. However, since treatment with ICIs enhances T cell activity and essentially removes the ‘brakes’ from the immune system, these drugs can be associated with serious immune-related toxicities, particularly when used in combination.
Gastrointestinal immune-mediated toxicities, particularly colitis, is one of the more frequently observed and severe toxicities. Published clinical trial data shows that approximately 30% of patients develop clinically significant intestinal inflammation/colitis within the first 16 weeks of treatment with the CTLA-4 inhibitor ipilimumab when used as a monotherapy1–3 and the incidence is greater than 40% with the combination of ipilimumab and the PD-1 inhibitor nivolumab.4,5 Gastrointestinal toxicity with PD-1 inhibitor monotherapy is less common. A recent systematic review of published clinical trials found that while the incidence of diarrhea with anti-CTLA-4 treatment was increased to 30%, only 12% of patients treated with anti-PD-1 developed diarrhea. 6 The incidence of colitis was lower at 5.7% and 0.7% with anti-CTLA-4 versus anti-PD-1 treatment, respectively. However, real-world experience with ICIs has found a considerably higher rate of colitis than has been reported in clinical trials.7–10
However, not all patients treated with ICI experience immune-mediated toxicities such as colitis, and current research is focused on studying the underlying mechanisms for the development of such toxicities. Early interest in the gut microbiota as a potential modulator of ICI efficacy and toxicities was prompted by the observation that treatment with the CTLA-4 inhibitor ipilimumab often resulted in intestinal inflammation as a result of mucosal immune dysregulation.1–3
Recent technological advances have made it possible to study the bacterial communities residing in the gut in greater detail. As a result, the interactions between the gut microbiota and the systemic immune response have become a focus of intense research.
In this review, the authors focus on the role of the gut microbiota in the development of immune-mediated toxicities and compare the clinical and histopathological presentation between ICI-induced colitis with that observed in inflammatory bowel disease (IBD). The authors will summarize what is currently known regarding the association between the gut microbiota and immune-mediated toxicities with a focus on gastrointestinal and hepatic toxicity in patients treated with ICI. Similarities in gut bacterial diversity will be examined in patients with autoimmune conditions such as autoimmune hepatitis and IBD, which includes ulcerative colitis and Crohn’s disease. The authors will also discuss the manipulation of the gut microbiota via fecal microbial transfer (FMT) to treat immune-mediated toxicities.
Clinical and histopathological features of gastrointestinal and hepatic immune-mediated toxicities
The augmentation in antitumor immune responses driven by T cell activation as a result of ICI treatment leads to inflammation in normal tissues. The most common organ systems affected include the gastrointestinal, hepatic, dermatologic, endocrine, and respiratory systems. Specific adverse events as reported in published clinical trials are listed in Table 1. Grading of adverse events reported here is as per the Common Terminology Criteria for Adverse Events, version 4.0.
Common immune-mediated toxicities reported in advanced melanoma patients on clinical trials with immune checkpoint inhibitors.
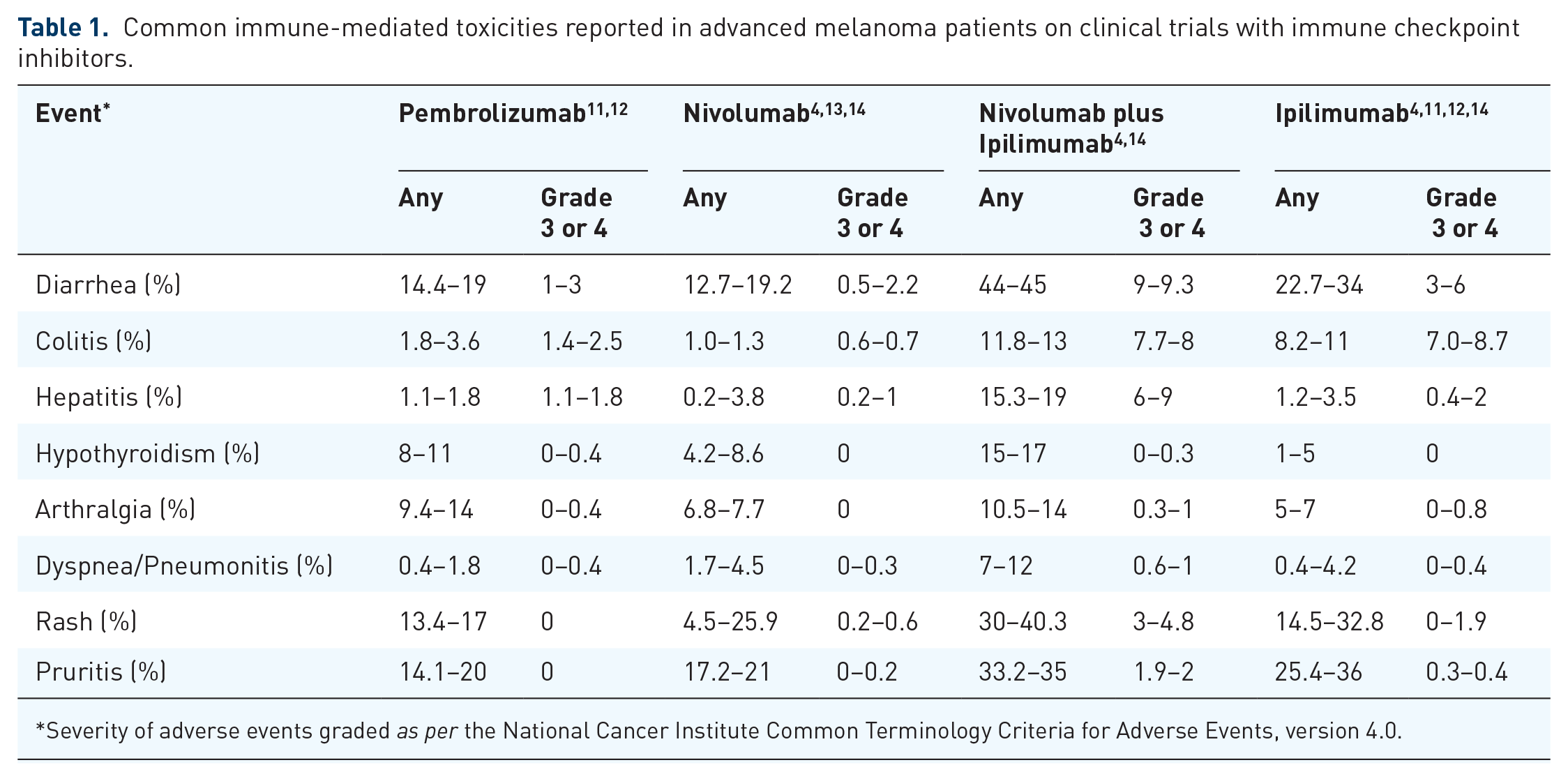
Severity of adverse events graded as per the National Cancer Institute Common Terminology Criteria for Adverse Events, version 4.0.
The incidence of gastrointestinal toxicity is generally higher and more severe in patients treated with CTLA-4 inhibitors when compared with patients treated with PD-1 inhibitors shown in Table 1. The median time to onset for diarrhea also differs between CTLA-4 and PD-1 inhibition, with ipilimumab-induced diarrhea generally occurring 5–8 weeks after treatment initiation compared with 3–6 months after PD-1 inhibitor treatment.3,15,16
CTLA-4 inhibition leads to a more similar presentation to IBD in terms of clinical severity when compared with the gastrointestinal toxicities that may be observed with PD-1 inhibition. Colitis, which exists as diarrhea associated with abdominal pain, rectal bleeding or mucus, or with large bowel inflammation on imaging, is seen in both IBD and in patients treated with ICI.
Although ipilimumab-induced colitis and IBD may share some similar clinical features, they have distinct histopathologies. In both cases often a pattern of patchy areas of inflammation is observed in the intestinal mucosa along with a lymphocytic infiltrate. 1 With ipilimumab-induced colitis, there is usually involvement of the descending colon. Endoscopic assessment may be normal or range from mild colitis to severe inflammatory changes including: exudates, granularity, erythema, loss of vascularity, and erosions/ulcerations.1,17 A dense, predominantly lymphocytic infiltrate may be seen with neutrophilic inflammation. Granulomas, which are associated with Crohn’s disease, are not observed in ICI-mediated colitis 1 and increased crypt apoptosis along with crypt atrophy/dropout, which may be seen in recurrent ICI colitis, is rare in IBD.16,18,19
There are also differences in the serologic markers of inflammation that have been observed between patients with IBD and those with ipilimumab-induced colitis. In a study that included assessment of serologic markers typical of IBD, there were distinct features identified in ipilimumab-treated patients. 1 The pattern of antibody positivity with the presence of both anti-Saccharomyces antibody (ASCA) and perinuclear-staining antineutrophil cytoplastic antibody (p-ANCA) was unique to ipilimumab-treated patients. ASCA or p-ANCA positivity, which is highly predictive for IBD, 20 were found in 50% of ipilimumab-treated patients that had no gastrointestinal immune-mediated toxicities. The fluctuations in antibody titers observed in ipilimumab-treated patients differed from the stability of these titers generally observed in patients with Crohn’s disease. 21
Comparison of anti-PD-1 and anti-CTLA-4-associated colitis has revealed similar histopathologic features including increased crypt epithelial cell apoptosis, crypt atrophy/dropout, and lymphocytic colitis. 17 However, in contrast with anti-CTLA-4 colitis, with anti-PD-1 colitis, there are usually no features of chronic mucosal injury including neutrophilic inflammation and neutrophilic crypt abscesses. 22
It is important to note here that, although there are many similarities between IBD and the gastrointestinal toxicities observed with ICIs, both IBD and ICI-induced colitis encompass a heterogeneous group of disorders. There is significant variability observed in both disease severity and the course of the disease within IBD and ICI-induced colitis. The distinct histopathologic and serologic findings in ipilimumab-induced colitis compared with IBD colitis strongly suggests that there are likely to be important differences in the underlying mechanisms for the development in gastrointestinal immune-mediated toxicities in these patients.
The liver is another commonly involved organ in ICI-mediated toxicity, although hepatic toxicity is less frequently observed than luminal gastrointestinal toxicity. Hepatic toxicity is usually observed as asymptomatic elevations in transaminases (raised alanine aminotransferase or aspartate aminotransferase). In cases of ipilimumab-induced liver injury, common histopathologic features include panlobular hepatitis with a mixed inflammatory cell infiltrate, composed of lymphocytes along with lobular macrophages, usually arranged in a prominent sinusoidal distribution. 17 With PD-1 inhibition, the pattern of hepatotoxicity usually includes predominantly lobular hepatitis with mild portal inflammation.23,24 Plasma cell infiltration, which is a hallmark of autoimmune liver disease, is rare in ICI-induced liver toxicity. 17
Although the clinical and histopathological features of the gastrointestinal and hepatic toxicities that may occur secondary to ICI therapy have been well characterized, the exact mechanisms by which such immune-mediated toxicities occur is still largely unknown. It is also unknown whether there are predisposing factors that can identify patients at high risk for colitis. One such factor may involve the composition of the gut microbiome, which includes commensal gut bacteria, and other microorganisms including fungi, viruses, and parasites. Recent research has found that the gut microbiota plays an important role in the response to anticancer treatment. 25
The role of the gut microbiota in ICI response and toxicity
Gut bacteria have been shown to be an important mediator of ICI toxicity and efficacy through their effects on the immune system26–28 and their interaction with host factors that influence antitumor immunity.29,30 Mouse models and observational studies in ICI recipients indicate that the interaction between specific bacteria and the host influences ICI response and toxicity.31–36 Gut microbiome analysis in patients with metastatic melanoma,34,35 lung, 36 and genitourinary 36 malignancies has identified bacteria that are associated with responsiveness, toxicity, or both, in humans (Table 2). The gut microbiome is, therefore, emerging as a novel diagnostic and therapeutic target in ICI recipients.
Gut bacteria and association with a response, toxicity, or both, with immune checkpoint inhibitors.
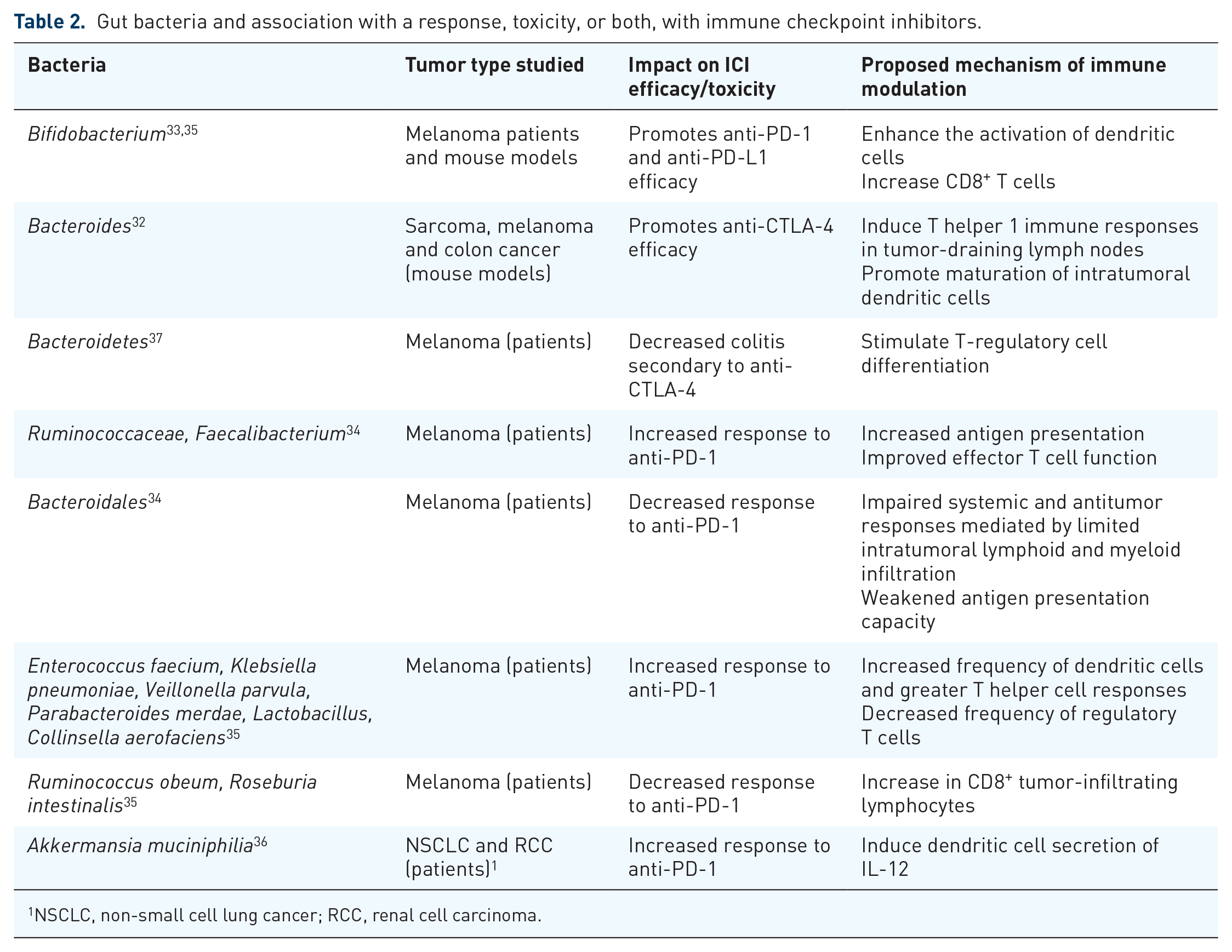
NSCLC, non-small cell lung cancer; RCC, renal cell carcinoma.
The gut microbiota and intestinal inflammation
It is currently unclear why treatment with ICIs leads to inflammation in the gut and colitis in only a subset of patients. One potential mechanism is activation of the immune system by the trillions of ‘foreign’ microbiota that inhabit the gut. This theory is supported by findings that show that IBD patients have imbalances in their commensal gut bacteria, known as dysbiosis. 38 Although it is unclear whether intestinal inflammation itself leads to gut dysbiosis, or whether dysbiosis is the cause of colitis in IBD patients, perturbations in the intestinal bacterial populations have been correlated with colitis. 39 The dysbiosis seen in the intestines of IBD patients can resemble that of cancer patients at increased risk of developing immune-mediated colitis, with a decrease in bacterial diversity correlated with an increased risk of colitis in both of these groups of patients. 37
There is a growing quantity of work that suggests particular bacterial populations in the gut may be associated with the risk of colitis. For example, an early study involving a small number of metastatic melanoma patients showed increased representation of bacteria belonging to the Bacteroidetes phyla in patients at lower risk of developing colitis secondary to CTLA-4 blockade. 37 Bacteroidetes is one of the major phyla comprising the gut microbiota, and members of the Bacteroidetes phylum are thought to have an immunomodulatory role limiting inflammation by stimulating T-regulatory cell differentiation.40,41 However, the broad recognition of gut microbes by the immune system is not likely to be dependent on specific bacteria and, therefore, it may be that it is the diversity of gut commensals that impacts the development of ICI-induced colitis. Further research is needed to better understand why, if there is broad recognition of gut microbes, only a minority of patients develop colitis and other immune-mediated toxicities. A decrease in bacterial abundance and diversity has also been associated with other forms of autoimmune disease besides colitis, including in a mouse model of autoimmune hepatitis, 42 mouse and human studies of multiple sclerosis,43,44 and rheumatoid arthritis.22,45 This link between gut dysbiosis and systemic autoimmune diseases suggests that the gut bacteria may impact immune cell function at distant sites.
The presence of ‘colitis protective’ bacterial species in the gut may not necessarily have a negative impact on the therapeutic efficacy of ICI blockade. In a study by Vetizou and colleagues melanoma mouse models treated with the combination of Burkholderia cepcia and Bacteroides fragilis, which were associated with response to CTLA-4 inhibition, had reduced histopathological evidence of colitis induced by CTLA-4 blockade, but still maintained evidence of therapeutic response. 32 However, in this study the mice did not develop overt colitis, but only histological signs of mild inflammation. It is unclear how this mouse model relates to ICI-induced colitis observed in humans. In another study Bifidobacterium, which has been associated with anti-PD-L1 efficacy in melanoma mouse models, 33 in combination with other bacteria including Lactobacillus has also been shown to be protective against colitis through modulation of cytokine production and strengthening of gut barrier function.46,47
Relationship between alterations in commensal gut microbiota and intestinal inflammation
In IBD patients a broad pattern of alterations in commensal bacteria has been observed, including decreased abundances of bacterial taxa within the phyla Firmicutes and Bacteroides and increases in Gammaproteobacteria48,49 This dysbiosis may then lead to increased inflammation in the gut through the activation of T helper 1 (Th1) and T helper 17 (Th17) cells. 27 It is possible that such T cell activation may result in damage to the protective mucus layer within the intestinal epithelium, enabling pathogenic bacteria and other microbes to remain in the intestinal tissues. The bacteria can then secrete antigens resulting in sustained inflammation.
Impact of gut microbiota on immune-mediated toxicities at distant sites
There are several potential mechanisms where an increase in gut permeability may result in the translocation of bacteria, immune cells, and microbial products through the circulation to distant sites to influence immune-mediated toxicities (Figure 1).

Proposed mechanisms accounting for effects of the gut microbiota on immune-mediated toxicities at distant sites. (a) Increased gut permeability after immune checkpoint inhibitor- (ICI) induced inflammation can lead to the translocation of pathogenic bacteria into the bloodstream. These bacteria can then modulate the systemic immune response by priming gut derived T helper 17 cells. These activated cells can then travel to distant organs via draining lymph nodes and cause immune-mediated toxicities; (b) Bacterial products may leak through the gut epithelium due to ICI-mediated damage to the intestinal mucosa. These bacterial metabolites and toxins can then travel through the bloodstream to modulate systemic immunity at distant sites; (c) Disruption to the gut epithelium resulting from ICI-mediated inflammation can release self-antigens which cross-react with bacterial antigens recognized by T cells. These activated T cells primed against self-antigens can then travel to distant sites causing an immune attack on distant organs. Figure created with BioRender.
Pathogenic Th17 cells that are activated in the gut could migrate to distant sites through draining lymph nodes of target organs and contribute to toxicities in other organs such as the thyroid, liver, and lungs. This possibility is supported by the observation that Th17 cells that are bacteria-specific and primed in lymph nodes draining the intestine can be found in other lymph nodes and the spleen. 50 Such primed immune cells can circulate systemically and play a role in immune responses at distant sites. 51
Gut microbiota can also affect immune responses through modulation of antigen-presenting cells such as dendritic cells (DCs). In the study by Sivan and colleagues 33 DCs isolated from mouse models treated with Bifidobacterium were found to have enhanced the presentation of melanoma-derived antigens to T cells, stimulating T cell proliferation and IFN-γ production.
An alternative mechanism that has been proposed for the impact of the gut microbiota on the efficacy and toxicity of ICI at distant sites involves specific bacterial products including metabolites and toxins translocating from the gut epithelium into the circulation. These bacterial products can travel to distant organs and cause inflammation. For example, several earlier studies have observed an important role for microbial-derived short-chain fatty acids (SCFAs) on immune cell regulation, particularly that of regulatory T cells.52–54 SCFAs inhibit histone deacetylase activity and promote an anti-inflammatory cell phenotype, critical for maintaining immune homeostasis. 55
Another proposed mechanism for the association between the gut microbiota and the efficacy/toxicity of ICIs involves molecular mimicry between specific commensals/pathogenic organisms and tumor neoantigens. 56 In the paper by Vetizou and colleagues 32 the therapeutic efficacy of CTLA-4 blockade was restored in mouse models via gavage with B. fragilis, immunization with B. fragilis polysaccharides, or by adoptive transfer of B. fragilis specific T cells. This observation suggests a cross-reactivity between microbial and tumor antigens recognized by the same T-cell receptor. This cross-reactivity may also occur between microbial antigens, and self-antigens and lead to immune-mediated toxicities. In the case of colitis, specific gut microbes may be responsible for causing intestinal cell injury and releasing self-antigens. These antigens may then generate immune responses that cross-react with additional tissue-specific self-antigens.
In combination, these observations provide potential mechanisms by which gut microbes may impact the systemic immune response and thus the development of immune-mediated toxicities at distant sites. Importantly, there are a number of systemic diseases such as rheumatologic conditions, obesity, allergic and infectious diseases in which disruption of the integrity of the intestinal epithelial barrier has been associated with disease pathogenesis. 57
Therapeutic manipulation of gut microbiota for refractory colitis
A common treatment for ICI-associated colitis is immune suppression with corticosteroids or drugs targeting tumor necrosis factor-α (TNF-α),16,58 both of which have significant adverse effects. Therefore, there has been interest in developing other treatment approaches for ICI-induced colitis. One approach that has gained interest is manipulation of the gut microbiota through FMT which has been used in the treatment of refractory Clostridium difficile-associated colitis and in IBD.59–61
More recently, FMT has been used to treat refractory colitis secondary to ICI in two patients with persistent symptoms despite corticosteroids, an anti-TNF-α agent (infliximab) and an anti-integrin agent (vedolizumab). 62 In this study, fecal microbes obtained from a single healthy donor were transplanted into two patients resulting in complete resolution of clinical symptoms and reduced inflammation on endoscopic evaluation. Baseline microbiota differed in these two patients, as can be expected, due to the multiple factors that may influence the diversity of the gut microbiota including diet and ethnicity. After FMT, there was a higher abundance of bacteria considered to be protective against ICI-associated colitis including Bacteroides. 37 Analysis of immune infiltrates in the colonic mucosa post-FMT showed a reduction in CD8⁺ T cells and an increase in CD4⁺ FoxP3⁺ T cells, suggesting a potential mechanism by which FMT may reduce ICI-associated intestinal inflammation. However, it is important to note here that the use of FMT occurred shortly after the administration of dual immunosuppressive agents in both patients. In IBD, such agents are known to take weeks to take effect. In this study, in which only two patients were treated and there was no control treatment, the observed effect of FMT may simply have been a result of the delayed efficacy of the prior immunosuppressive therapies. Future studies should be carried out with upfront use of FMT in patients with colitis refractory to steroids without the prior use of additional, more potent immunosuppressive therapies to identify the true impact of FMT on immune-mediated colitis.
Studies examining the impact of the gut microbiota on colitis to date have involved a small number of patients with a variety of different cancers and prior anticancer therapies and, therefore, the exact role of the gut microbiota is unclear. Larger studies are required to clarify the role that the gut microbiota may play in modulating the immune response and toxicity with checkpoint blockade.
An important question is whether the use of FMT to treat colitis could result in decreased antitumor immune responses and decrease the efficacy of ICI therapy. Results of FMT in mouse models in the study by Vetizou and colleagues suggests a selective immune-modulatory effect for certain bacteria. They found that the antitumor effect of B. fragilis did not lead to an increase in colitis, but rather led to a decrease. 32 This selective effect could not be replicated by treatment with the antibiotic vancomycin, an antibiotic mainly active against Gram⁺ bacteria including Clostridiales. However, the impact of vancomycin on the human microbiota is not yet well characterized. The mechanisms by which this antibiotic may enhance the antitumor effects of CTLA-4 inhibition, but worsen colitis induced by ipilimumab, needs to be further investigated. A recent study examining the impact of vancomycin on the gut microbiota showed that treatment with vancomycin actually led to an increase in pathogenic bacteria such as Proteobacteria, at the expense of beneficial commensal bacteria, such as Bacteroidetes. 63 In the future, large prospective studies need to be carried out to determine the optimal way to manipulate the gut microbiota, whether it is through FMT, antibiotics, probiotics, or defined bacterial consortia. Ultimately it will be important to establish a defined bacterial consortium that is a scalable and safe intervention to alter the gut microbiota in favor of improved efficacy and decreased toxicity.
Conclusion
In this review, the authors have highlighted the common toxicities that may occur with ICI and similarities between ICI-associated colitis and hepatitis and those resulting from autoimmune disorders including IBD and autoimmune hepatitis. The authors have provided an overview of the role that the gut microbiota may play in ICI efficacy and toxicity and discussed the potential for manipulation of the gut microbiota to improve clinical outcomes. Future studies are needed to understand the impact of environmental factors including diet, ethnic background, and medications, as well as tumor characteristics including primary tumor type and prior anticancer treatments (chemotherapy, surgery and, radiation) on the gut microbiota.
Footnotes
Funding
This work was support by grants to R.C.P. from The University of Toronto, Division of Medical Oncology, Strategic Innovation Grant, and to A.M. from the Canadian Cancer Society (grant 703185) and Canadian Institute of Health Research (grant 144628).
Conflict of interest statement
The authors declare that there is no conflict of interest.
