Abstract
Background:
Despite the established efficacy of ustekinumab (UST) in Crohn’s disease (CD), real-world studies reveal suboptimal treatment persistence and limited endoscopic healing. Current optimization paradigms remain constrained by suboptimal durability and escalating therapeutic demands.
Objectives:
To evaluate the clinical utility and safety of intravenous (IV) ustekinumab maintenance therapy in CD.
Design:
This was a single-center retrospective cohort study.
Methods:
This study included CD patients receiving ⩾2 IV-UST maintenance infusions between June 2020 and October 2023 (N = 234). The protocol featured weight-based induction-equivalent dosing (260–520 mg) at response-guided intervals. The primary endpoint was the corticosteroid-free clinical remission rate at Week 24.
Results:
At 24 weeks, 88.1% (185/210) achieved steroid-free remission with C-reactive protein (CRP) normalization in 90.0% (44/88). Median Harvey-Bradshaw Index decreased from 4 (interquartile range (IQR) 2–5) to 2 (IQR 1–3; p < 0.001). 52-week endoscopic remission (simplified endoscopic score for Crohn’s disease ⩽3) reached 48.7% (56/115), with fecal calprotectin normalization in 33.6% (35/104). Multivariate analysis identified baseline CRP >5 mg/L (adjusted odds ratio (aOR) 3.62, 1.16–11.25), intensive dosing (⩾6 cycles/year; aOR 12.06, 1.99–73.05), and disease duration ⩾1 year (aOR 3.53, 1.08–11.54) as predictors of endoscopic non-remission. Safety analysis demonstrated 44.4% adverse event incidence (104/234) and 3.0% serious adverse events (7/234).
Conclusion:
IV-UST maintenance demonstrates high rates of corticosteroid-free clinical remission and endoscopic healing with manageable safety in CD.
Plain language summary
IV ustekinumab treatment for Crohn’s disease: real-world insights from a hospital
Introduction
Ustekinumab (UST), a fully human monoclonal antibody targeting the interleukin-12/23 p40 subunit, has demonstrated favorable efficacy and safety in the treatment of moderate-to-severe Crohn’s disease (CD). 1 Standard therapy involves an intravenous induction dose (6 mg/kg at week 0) followed by subcutaneous maintenance administration (90 mg every 8 (q8w) or 12 weeks (q12w)).
While clinical trials demonstrate sustained 4-year efficacy with this regimen, real-world observational studies reveal significantly higher treatment refractoriness. Notably, 20%—35% of CD patients exhibit primary non-response (PNR) or secondary loss of response (LOR) in clinical practice.2–4 A nationwide Dutch retrospective study reported progressively declining corticosteroid-free clinical remission rates of 41.4% (6 months), 39% (1 year), and 34% (2 years) under standard UST therapy. 5 Further analysis of 38 CD-focused studies (n = 41) showed modest mucosal healing efficacy, with pooled endoscopic response rates of 61% (95% confidence interval (CI): 50%—72%) and mucosal healing rates of 25% (95% CI: 13%—28%) after 1-year treatment. 6
Emerging evidence supports dose adjustment strategies to overcome therapeutic limitations. In a multicenter inflammatory bowel disease (IBD) cohort (n = 1113), dose escalation (intravenous reinduction or interval shortening) recaptured response in 57% (44/77) of LOR patients, while 40.1% (61/152) of PNR patients achieved delayed clinical response. 7 Notably, a comparative cohort study demonstrated enhanced efficacy with dual intravenous induction (vs standard single-dose), achieving superior rates of clinical remission (88.2%, 45/51), endoscopic response (75.8%, 25/33), and fecal calprotectin (FCP) normalization (70.6%, 36/51). 8 China’s National Volume-Based Procurement policy substantially reduced IV drug costs of UST, while high infusion service accessibility further enhanced feasibility. We therefore positioned IV maintenance as a pragmatic alternative to subcutaneous regimens and conducted a single-center retrospective cohort study to evaluate its real-world clinical utility and safety.
Methods
Study design
This study was conducted in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines (Supplemental Material). 9 This single-center observational study employed a retrospective cohort design to evaluate patients with IBD receiving intravenous ustekinumab (IV-UST) maintenance therapy for ⩾6 months. The study was motivated by IV-UST therapy’s economic advantages in China’s healthcare context:
Two IV doses (¥5062.48 ≈ $693.50 USD) cost 31% less than SC (¥7340.60 ≈ $1005.56 USD).
Three IV doses were cost-equivalent to SC, while SC interval shortening for non-response amplified IV’s cost advantage.
Low infusion fees (¥132.12 RMB ≈ $18.10 USD).
Universal insurance coverage (50%—80% reimbursement) lowered out-of-pocket costs.
Study population
Eligible participants received ⩾2 IV-UST maintenance doses between June 2020 and October 2023 and exhibited active disease at baseline, defined by meeting ⩾1 of the following criteria10,11:
Harvey-Bradshaw Index (HBI) ⩾5
C-reactive protein (CRP) >5 mg/L
FCP >200 μg/g
Simplified endoscopic score for Crohn’s disease (SES-CD) ⩾4
Exclusion criteria included:
Prior interleukin-12/23 inhibitor therapy
Concurrent biological agent usage
Active pregnancy/lactation status
Significant systemic infections.
Treatment protocol
Patients were transitioned from subcutaneous to intravenous maintenance therapy (administered at 4–8 week intervals) based on the following predefined clinical parameters:
Symptomatic relapse (HBI increase ⩾3 from baseline)
Sustained inflammatory burden (CRP, erythrocyte sedimentation rate elevation, or FCP) (after excluding non-IBD etiologies)
Radiological confirmation of active inflammation
Endoscopic disease progression
Extensive intestinal involvement
Suboptimal response to previous therapeutic regimens (conventional therapies, immunosuppressants, anti-TNFα agents).
The intravenous maintenance dose was weight-based and identical to the induction phase (520 mg for patients ⩾85 kg, 390 mg for patients 55 to <85 kg, and 260 mg for patients <55 kg), administered at individual intervals.
Responders achieving sustained clinical remission (HBI <5) and concurrent normalization of key inflammatory biomarkers (CRP ⩽5 mg/L, ESR ⩽20 mm/h, and FCP ⩽250 µg/g) were considered for de-escalation to standard subcutaneous maintenance, underwent protocol-guided de-escalation to standard subcutaneous maintenance. Transition required patient willingness, informed by discussion of comparative efficacy data, convenience, and the higher out-of-pocket cost associated with subcutaneous administration in our setting.
Data source and ethics
Participants were drawn from the Precision Evaluation, Personalized Risk Stratification, and Targeted Intervention in IBD (PRIST) registry—an investigator-initiated, prospective observational cohort established at our tertiary IBD referral center (initiation: July 1, 2019). The study protocol (No. 2023-0004) received ethical approval from the Institutional Review Board of the Second Affiliated Hospital, Zhejiang University School of Medicine. The requirement for written informed consent was waived for this retrospective analysis of anonymized patient data, consistent with institutional regulations, national guidelines, and the Declaration of Helsinki.
Evaluation criteria and study endpoints
Clinical activity was assessed using the HBI, 12 while endoscopic severity was evaluated through SES-CD. 13 Serious adverse events (SAEs) were defined according to International Council for Harmonisation guidelines as events resulting in (1) permanent discontinuation of UST therapy, (2) hospitalization, (3) disease-related surgery, or (4) mortality.
The primary endpoint comprised corticosteroid-free clinical remission at Week 24, defined as HBI ⩽4 points with documented discontinuation of corticosteroid therapy for ⩾30 consecutive days prior to assessment.
Secondary endpoints included the following:
Clinical remission (HBI ⩽4) at Week 24
CRP normalization (<5 mg/L) at Week 24 among patients with baseline CRP ⩾5 mg/L
FCP normalization (⩽200 μg/g) at Week 52 in subjects with baseline FCP >200 μg/g
Endoscopic remission rate (SES-CD ⩽3 points) at Week 52, with exclusion of isolated stricture-related sub-scores.
Safety assessments encompassed systematic monitoring of adverse events (AEs), SAEs, and treatment persistence duration, defined as time from UST initiation to permanent discontinuation.
Statistical analysis
All statistical analyses were performed using IBM SPSS Statistics (version 26.0; IBM Corp, Armonk, NY, USA). Patients with missing endpoint data were excluded from per-protocol analyses. Demographic and baseline disease characteristics were summarized for the entire cohort. Categorical variables were expressed as percentages (n%), while continuous variables were presented as mean ± standard deviation for normally distributed data or median with interquartile range (IQR) for non-normally distributed data, with distribution normality assessed via Shapiro–Wilk testing. Treatment persistence was evaluated through Kaplan–Meier survival analysis with log-rank testing. Group comparisons employed Student’s t-test (normally distributed variables) or Mann–Whitney U test (non-parametric data). Univariate and multivariate logistic regression models were constructed to identify independent predictors of 52-week endoscopic remission. All statistical tests were two-tailed, with p < 0.05 determining statistical significance.
Results
Study population
This retrospective chart review of 688 UST-treated CD patients identified 375 receiving standard IV induction/SC maintenance therapy and 313 eligible for IV-maintenance therapy. As detailed in the Figure 1 flowchart, 234 of the latter group met the final inclusion criteria to comprise the analytic cohort whose comprehensive baseline characteristics are detailed in Table 1.

Flow diagram of the study.
Baseline demographic and clinical characteristics of study participants with inflammatory bowel disease.
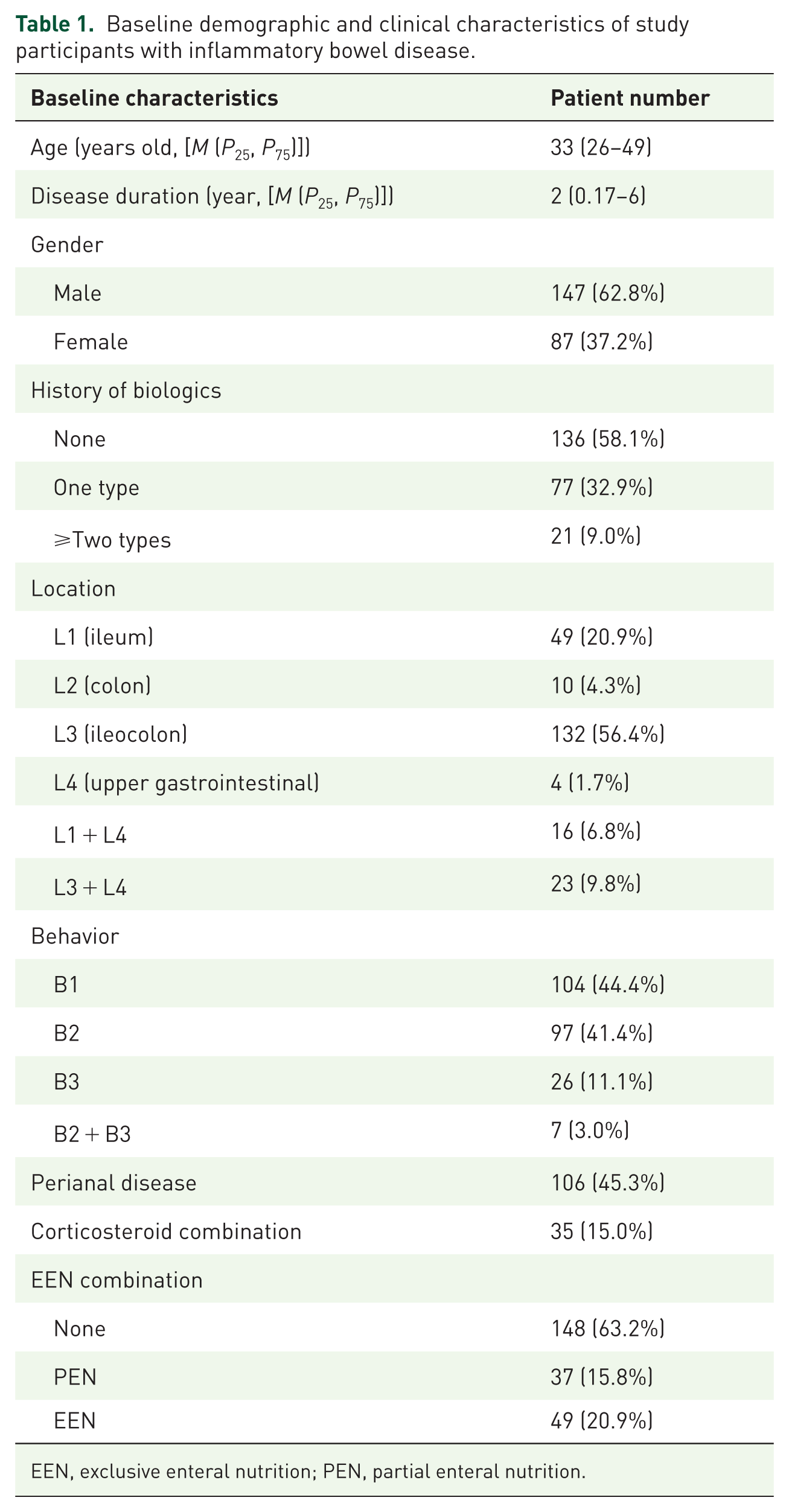
EEN, exclusive enteral nutrition; PEN, partial enteral nutrition.
Demographic profiling revealed a male predominance (62.8%, n = 147) and median age at treatment initiation of 33 years (IQR 26–49). Patients exhibited a median disease duration of 2 years (IQR 0.17–6) prior to UST induction. Disease localization patterns under the Montreal classification showed ileocolonic involvement (L3) as the most frequent presentation (56.4%, n = 132), while non-stricturing/non-penetrating disease behavior (B1) was observed in 44.4% of cases (n = 104). Notably, perianal complications were documented in nearly half the cohort (45.3%, n = 106). Surgical history analysis identified prior intestinal resection in 33.3% of patients (n = 78). Concomitant corticosteroid use at baseline was recorded in 15.0% of participants (n = 35), while nutritional support through exclusive enteral nutrition (EEN) had been implemented in 20.9% of cases (n = 49). Treatment history stratification demonstrated that 58.1% of the cohort (n = 136) were biologic-naïve, with 32.9% (n = 77) having prior exposure to one biologic agent and 9.0% (n = 21) exhibiting refractory disease with failure of ⩾2 biologic therapies.
Clinical efficacy assessment
The 24-week treatment course was completed by 229 patients, with 19 excluded due to incomplete HBI documentation at endpoint evaluation. Among the 210 analyzable cases, steroid-free clinical remission was attained in 88.1% (185/210), while 90.0% (189/210) sustained clinical remission through week 24. Biochemical assessments demonstrated CRP normalization in 90.0% (44/88) of tested patients. Marked reductions in inflammatory biomarkers were observed: median CRP levels decreased from 3.20 mg/L (IQR 1.10–10.55) at baseline to 1.87 mg/L (IQR 0.7–5.4) post-treatment (p < 0.01), paralleled by a decline in median HBI scores from 4 (IQR 2–5) to 2 (IQR 1–3) at week 24 (p < 0.001).
Extended follow-up at 52 weeks (n = 144) revealed endoscopic remission according to SES-CD criteria in 48.7% (56/115) of evaluable patients, with concurrent normalization of FCP levels in 33.6% (35/104) of cases.
Univariate analysis (Table 2) identified no statistically significant differences in baseline demographics, Montreal classification profiles, treatment histories, or concomitant medication use between responders and non-responders. However, multiple parameters demonstrated significant predictive value: elevated baseline CRP (5.81 vs 3.05 mg/L, p = 0.039), persistent CRP elevation at week 24 (3.50 vs 1.39 mg/L, p < 0.001), higher week-24 HBI scores (median 2 vs 1, p = 0.049), and frequent intravenous UST administration (>5 annual infusions; 59.32% vs 35.71%, p = 0.011).
Comparative analysis of patients with endoscopic remission versus non-remission.

CRP, C-reactive protein; EEN, exclusive enteral nutrition; FCP, fecal calprotectin; HBI, Harvey-Bradshaw Index; UST, ustekinumab.
Multivariate regression modeling (Figure 2) established four independent predictors of endoscopic non-remission: baseline CRP >5 mg/L (adjusted odds ratio (aOR) 3.62, 95% CI 1.16–11.25), intensive IV dosing regimen (⩾6 annual cycles; aOR 12.06, 1.99–73.05), and prolonged disease duration (⩾1 year; aOR 3.53, 1.08–11.54).

Independent predictors of endoscopic non-remission at week 52: multivariable logistic regression analysis.
Safety analysis
The median annual IV UST administration frequency was 5.28 doses (IQR 3.48–6.60), with a treatment persistence rate of 92.3% (216/234) at study conclusion (Figure 3). Discontinuation occurred in 18 patients (7.7%), primarily due to LOR (n = 11), surgical intervention (n = 5), varicella infection (n = 1), and one mortality case from diffuse peritonitis.
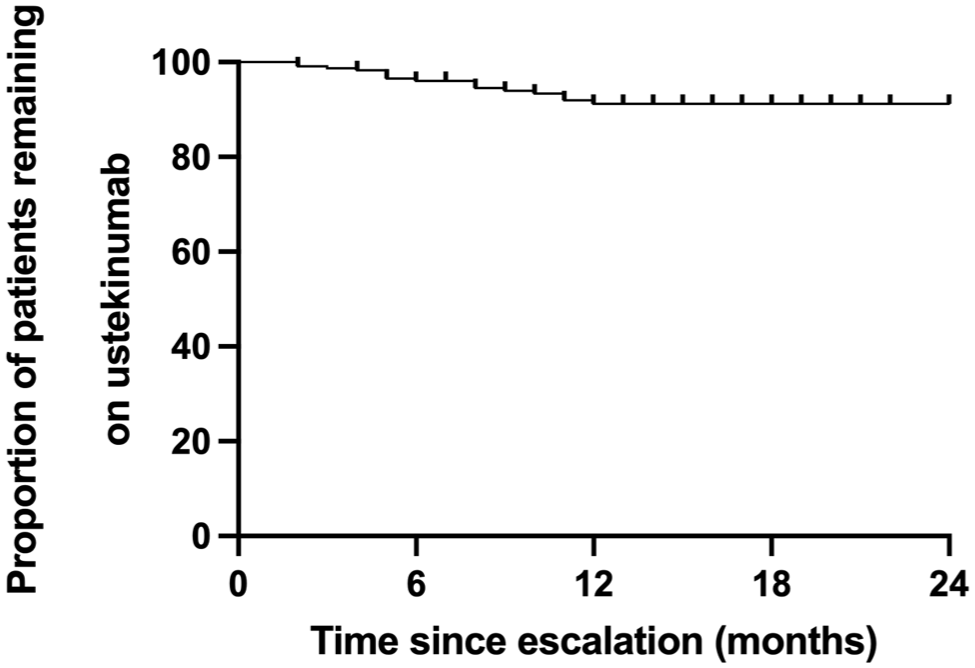
Treatment persistence of protocolized ustekinumab maintenance therapy: Kaplan–Meier analysis over 52-week follow-up.
AEs were reported in 44.4% of the cohort (n = 104), with fatigue (n = 31), musculoskeletal symptoms (n = 25), injection-site reactions (n = 22), influenza-like illness (n = 19), alopecia (n = 14), and hepatic dysfunction (n = 9) constituting the most frequent manifestations. Additional AEs included transient urinary symptoms, cephalalgia, pyrexia, chest discomfort, gastrointestinal disturbances, and hematological abnormalities.
A total of 3.0% (7/234) SAEs were documented, comprising one mortality, two intestinal obstruction-related surgeries, one perforation-related surgery, one gastrointestinal bleeding (GIB), one Clostridioides difficile (C. diff) infection, and one varicella infection. The fatal case involved an elderly male with recurrent disease who discontinued regular follow-ups; he developed acute bowel perforation requiring emergency surgery and succumbed to hemorrhagic shock postoperatively. Both obstruction surgeries involved patients deferring pre-planned resections until recurrence during UST therapy. The perforation case occurred in a patient with prior conservative management of perforations, undergoing elective surgery during UST-induced remission. The GIB case manifested solely as occult blood positivity in a refractory CD patient subsequently switched to Upadacitinib. The C. diff infection emerged after 6 months of UST combined with azathioprine/corticosteroids in severe CD. Varicella infection occurred post-UST initiation with concomitant thalidomide, resolving completely without recurrence after treatment resumption.
Discussion
Our study, representing the largest cohort (N = 234) to date investigating IV-UST maintenance for CD, demonstrated promising efficacy with 88.1% corticosteroid-free clinical remission at 24 weeks and 48.7% endoscopic remission at 52 weeks, alongside manageable safety profiles (44.4% overall AEs; 3.0% SAEs). These outcomes gain particular significance when contextualized with real-world therapeutic challenges. Contemporary evidence highlights suboptimal treatment persistence with UST regimens, reflected in a 52-week continuation rate of merely 62.9%, 2 while current predominant optimization strategies—including interval shortening and intravenous reinduction—demonstrate limited efficacy, achieving corticosteroid-free clinical remission in 40% of patients and endoscopic remission in 29%. 14 Furthermore, 24.8% of patients undergoing dose intensification required secondary escalation, with no subsequent dose reduction observed during follow-up. 15 Notably, the purported benefits of two-dose induction in severe CD 8 are potentially confounded by concomitant EEN regimens. This evidence landscape underscores the potential clinical value of our protocol in addressing documented therapeutic challenges.
This strategy leverages key pharmacokinetic advantages of intravenous administration: weight-based IV dosing achieves significantly higher serum concentrations than subcutaneous regimens, as demonstrated in the UNITI-1 trial, where median week 8 levels were threefold greater with IV therapy (6.4 vs 2.1 µg/mL)—a difference directly correlated with superior clinical remission, endoscopic improvement, and CRP reduction. 1 Despite substantially higher drug exposure relative to standard regimens, the safety profile of intravenous maintenance remained manageable, with AE rates consistent with expectations for biologic therapy in moderate-to-severe CD. This observation takes on added significance given the cohort’s significant baseline inflammatory burden—an established contributor to treatment-related complications—by supporting the feasibility of sustained high-exposure therapy. Among cases experiencing SAEs, severe baseline inflammatory burden and suboptimal treatment adherence emerged as predominant risk factors, suggesting UST’s safety profile is driven more by disease activity than administration route.
Several limitations warrant consideration. First, the single-center retrospective nature precludes comparative efficacy assessment against subcutaneous regimens due to the absence of a randomized control group. Second, the restricted follow-up duration necessitates longer-term evaluation to establish sustained efficacy and safety outcomes. Third, retrospective ascertainment of AEs via medical records and telephone follow-up may introduce omissions and recall bias. Finally, pharmacokinetic modeling correlating UST through concentrations with clinical endpoints was not performed. Future prospective studies should validate exposure–response relationships, implement protocolized safety monitoring, and extend follow-up duration for intravenous maintenance therapy.
Conclusion
In conclusion, this study supports the clinical feasibility of intravenous UST maintenance therapy for CD, demonstrating substantial rates of corticosteroid-free clinical remission and endoscopic healing with manageable safety. These outcomes provide a foundation for prospective evaluation of intravenous maintenance strategies in biologic-refractory CD.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251375356 – Supplemental material for Intravenous ustekinumab maintenance in Crohn’s disease: a single-center retrospective cohort study
Supplemental material, sj-docx-1-tag-10.1177_17562848251375356 for Intravenous ustekinumab maintenance in Crohn’s disease: a single-center retrospective cohort study by Yuting Wang, Shiyuan Lu, Wen Hu, Shuyan Li and Yan Chen in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We thank Dr Xiaoying Wang, Dingting Xu, Hanyun Zhang, Qiao Yu, and Song Xu from the Department of Gastroenterology, the Second Affiliated Hospital, who helped in the clinical management and follow-up of the patients. We thank Dr Zexin Chen, from the Second Affiliated Hospital, who assisted in the statistical procedure.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
