Abstract
Background:
Data on the performance of newer biologics in patients with fistulizing Crohn’s disease (CD) are limited.
Objective:
Our study’s objective was to evaluate the response to ustekinumab (UST) and vedolizumab (VDZ) in patients with fistulizing CD.
Design:
Retrospective cohort.
Methods:
We used natural language processing of electronic medical record data to identify a retrospective cohort of individuals with fistulizing CD at a single academic tertiary-care referral center and then performed a chart review. Individuals were eligible for inclusion if a fistula was present at the time of UST or VDZ initiation. Outcomes included medication discontinuation, surgical intervention, development of a new fistula, and fistula closure. Groups were compared with unadjusted analyses and competing risk analyses using multi-state survival models.
Results:
In all, 68 patients were included (48 UST and 20 VDZ). Most patients had one fistula (79%) and had prior anti-tumor necrosis factor-α treatment (98% in UST group, 80% in VDZ group, p = 0.01). VDZ was significantly more likely to be discontinued than UST (p < 0.0001), most frequently due to inadequate clinical response. Those on UST had a longer median time to surgery for CD than those on VDZ (p = 0.008). In those without surgical fistula repair, 79% on UST and 100% on VDZ still had an active fistula at 1 year (p = 0.30).
Conclusion:
In individuals with fistulizing CD, our data suggest that UST has better clinical utility than VDZ based on lower rates of discontinuation, though the sample size is small. These findings highlight the importance of further research on the treatment of perianal fistulizing Crohn’s disease.
Keywords
Introduction
Fistula formation is common in Crohn’s disease (CD), affecting an estimated 12% of CD patients in the United States (US). 1 Fistulas are thought to form from transmural inflammatory infiltration in combination with aberrant tissue remodeling, which causes mesenchymal-to-epithelial cell transformation, nearly always in the setting of bowel wall pressure upstream from a stricture or muscular sphincter. 2 A prior US-based study in Minnesota estimated that 55% of Crohn’s-related fistulas are perianal, 9% are rectovaginal, 6% are enterocutaneous, and the remaining 31% are internal. 1 Risk factors for the formation of fistulas include younger age at diagnosis, male sex, smoking, and ileal involvement at diagnosis. 3 The presence of fistulas is associated with worse quality of life, including higher rates of disability, lower work productivity, and increased sexual dysfunction.4,5
Biologics targeting tumor necrosis factor-α (TNF-α) have been shown in randomized controlled trials to facilitate fistula healing. 6 There are fewer data available regarding the efficacy and effectiveness of ustekinumab (UST) and vedolizumab (VDZ), though some trials suggest that both medications may help with perianal fistula healing.7,8 However, the utility of these medications in patients with other types of fistulas has not been specifically addressed. 9
The objective of our study was to evaluate the real-world effectiveness of treatment with UST and VDZ in patients with CD and an active fistula, most of whom have previously been treated with (and failed) anti-TNF therapy.
Materials and methods
We conducted a retrospective cohort study of all adult patients (age ⩾18 years) treated for CD at the University of Michigan who had a fistula and were started on VDZ or UST between 2014 and 2021. CD was ascertained through chart review. Patients with a fistula were identified using natural language processing of electronic medical records. Patients were eligible for inclusion if they had 14 weeks of follow-up or longer on either medication, and had received more than a single dose of the medication. Patients with prior exposure to both medications were included only for the first medication they tried to reduce the risk of bias. Sample size was determined by inclusion of all eligible individuals within the institution.
Through chart review, we collected demographic data, clinical characteristics of CD including Montreal Classification, 10 prior CD therapies and surgeries, fistula location and type, and extraintestinal manifestations. Chart review was completed in April 2022. Presence of a fistula was verified based on endoscopic findings, clinical examination, and/or imaging from the time of medication initiation and during follow-up. Active fistula was defined as a fistula with drainage on examination or the presence of liquid and/or gas in a fistulous tract on imaging or the presence of active inflammation on imaging surrounding a fistulous tract. Therapeutic data collected included duration of UST or VDZ therapy, concurrent inflammatory bowel disease (IBD)-specific medications, and IBD medication use history. Adverse drug or infusion reactions, Crohn’s-related surgeries (excluding seton placement/fistulotomy and perianal abscess drainage), and discontinuation of UST or VDZ and the reason for discontinuation were also recorded. Primary outcomes of interest were presence of a fistula 1 year after treatment initiation, time to fistula closure, and new fistula formation while on medication. Secondary outcomes were need for surgical management of IBD and treatment discontinuation rate. Fistula closure was defined as patient or clinician report of having no drainage or decrease in size or associated inflammation on imaging. 11 Individuals with missing data at the 1-year time-point were excluded from those analyses. Patients with loss to follow-up were treated as missing after their last documented clinical encounter.
UST and VDZ group outcomes were compared in crude analyses with Wilcoxon rank-sum test, Fisher’s exact test, and Pearson’s chi-squared test. Time to fistula closure, new fistula development, and medication discontinuation were visualized using Kaplan–Meier estimates and analyzed as crude comparisons with log-rank tests. Adjusted group differences in time to fistula closure and time to new fistula development were estimated with multi-state survival models to account for the competing risk of medication discontinuation using the {survival} package in R. 12 Individual follow-up time was defined as time from medication start until an outcome of interest or until most recent gastroenterology follow-up. All analyses were performed with R [R Core Team (2021). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria]. All tests were two-tailed with a significance level of p < 0.05. Patient data were de-identified prior to analysis. The study has been reported according to the STROBE statement (checklist provided as Supplemental Material). 13
Results
In all, 68 patients met criteria for inclusion. Of these, 48 received UST and 20 received VDZ (Table 1). Individuals who had exposure to both medications were only included for the first medication taken for more than one dose. Overall, 46% of patients were female and 87% were white. Median disease duration at UST or VDZ initiation was 18 years, and at UST or VDZ initiation the median age was 38 years. Most had only one fistula (79%). Perianal fistulas were the most common type (54%). Most fistulas had been first identified within the year prior to starting therapy, with a median time from identification to therapy of 274 days (interquartile range: 60–897 days). There were no significant differences in demographics or CD-related features between the groups (Table 1).
Baseline characteristics of individuals with CD and an active fistula started on UST or VDZ.

n (%); median (interquartile range).
Pearson’s chi-squared test; Fisher’s exact test; Wilcoxon rank-sum test.
Missing for n = 2.
CD, Crohn’s disease; UST, ustekinumab; VDZ, vedolizumab.
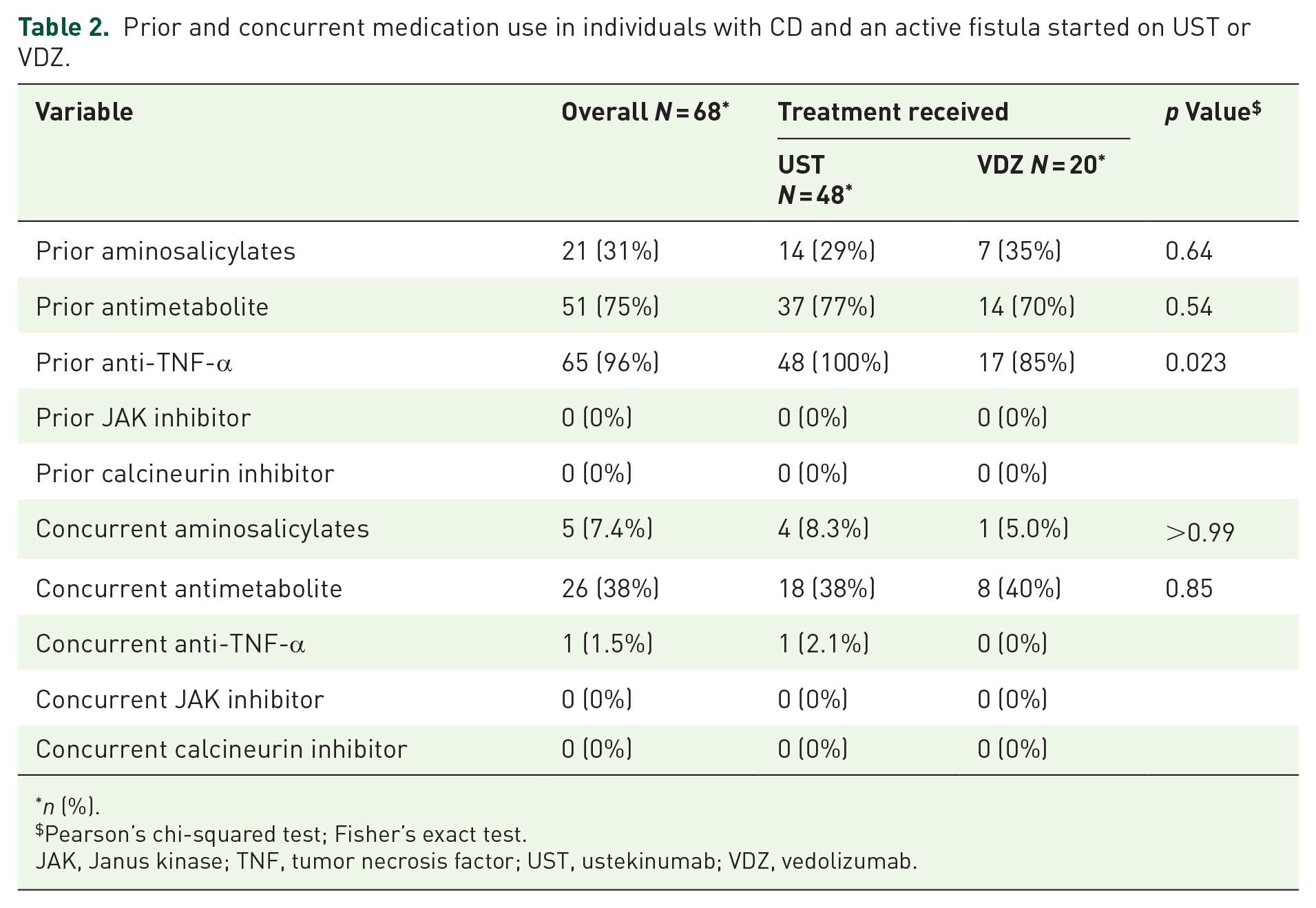
Significantly more patients started on UST had received prior anti-TNF treatment than those started on VDZ (100% in UST group, 85% in VDZ group, p = 0.02, Table 2). There were no other significant differences in prior medication therapy or prior surgery for IBD between the groups (Tables 1 and 2). Overall, around 40% of patients in both groups were on concurrent antimetabolite therapy, such as azathioprine or methotrexate with no significant difference between the groups (p = 0.8). No patients underwent stem-cell therapy for fistula treatment during the study period. Antibiotic use was inconsistently reported and unable to be reliably ascertained for inclusion.
Prior and concurrent medication use in individuals with CD and an active fistula started on UST or VDZ.
n (%).
Pearson’s chi-squared test; Fisher’s exact test.
JAK, Janus kinase; TNF, tumor necrosis factor; UST, ustekinumab; VDZ, vedolizumab.
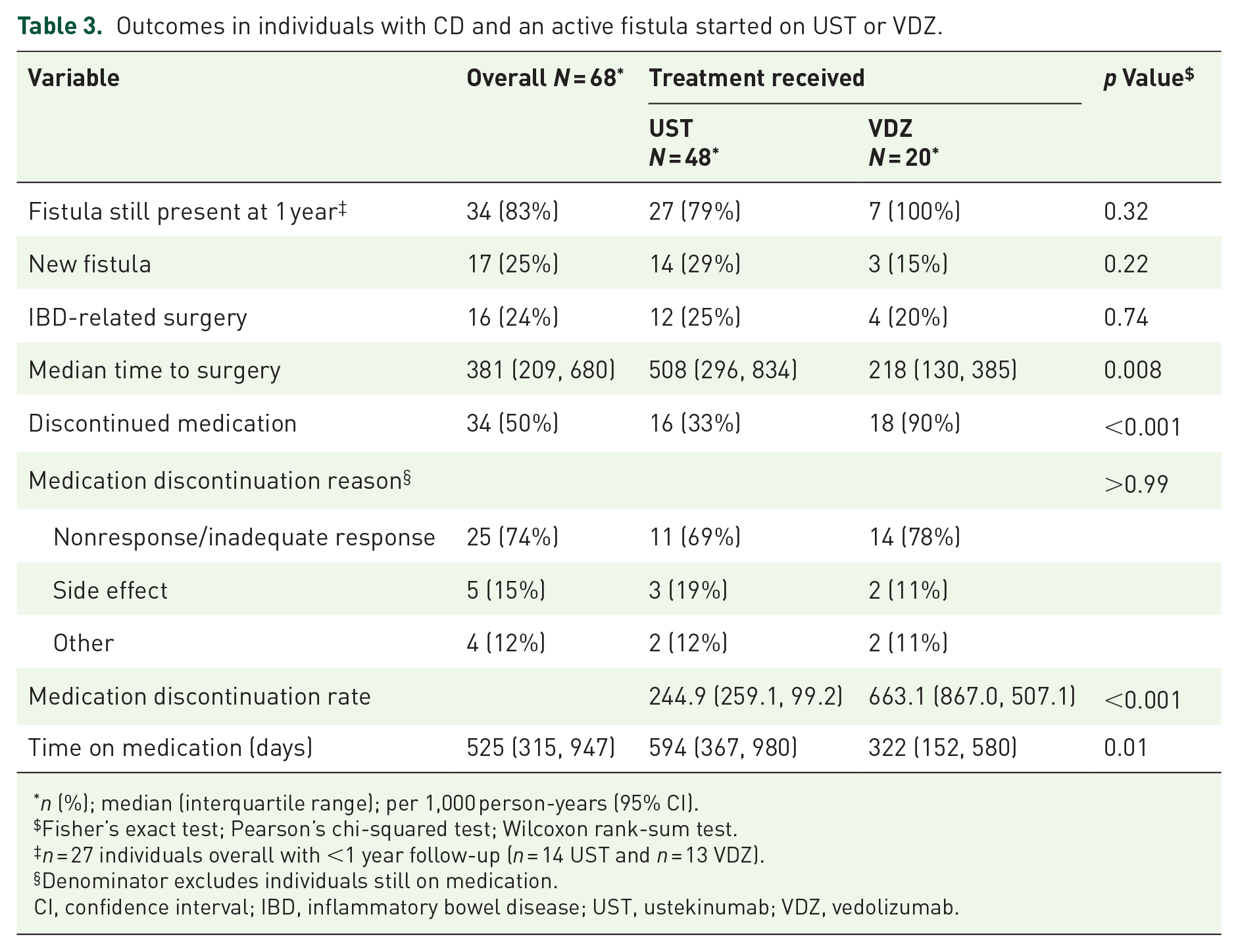
In unadjusted analyses of individuals who remained on therapy for at least 1 year and did not have surgical fistula repair, 79% in the UST group (n = 27) and 100% in the VDZ group (n = 7) still had an active fistula at 1 year (Table 3). Of the seven individuals in the UST group with fistula closure, four had closure documented by clinical examination and three had radiographic evidence of closure. Four of the individuals with fistula healing on UST had perianal fistulas, two had enteroenteric fistulas, and one had an enterocutaneous fistula. Statistically, the difference in likelihood of closure between the groups was not significant (Table 3, Fisher’s exact test p = 0.3) and the time to fistula closure (unadjusted Poisson p = 0.35) was not statistically significant. The rate of new fistula formation was 18/100 person-years (P-Y) in the UST group [95% confidence interval (CI): 11–29 per 100 P-Y] and 12/100 P-Y in the VDZ group (95% CI: 4–34 per 100 P-Y) with no significant difference between groups (Figure 1, unadjusted Poisson p = 0.78, while the multi-state model controlling for the competing risk of medication discontinuation had p = 0.28).
Outcomes in individuals with CD and an active fistula started on UST or VDZ.
n (%); median (interquartile range); per 1,000 person-years (95% CI).
Fisher’s exact test; Pearson’s chi-squared test; Wilcoxon rank-sum test.
n = 27 individuals overall with <1 year follow-up (n = 14 UST and n = 13 VDZ).
Denominator excludes individuals still on medication.
CI, confidence interval; IBD, inflammatory bowel disease; UST, ustekinumab; VDZ, vedolizumab.
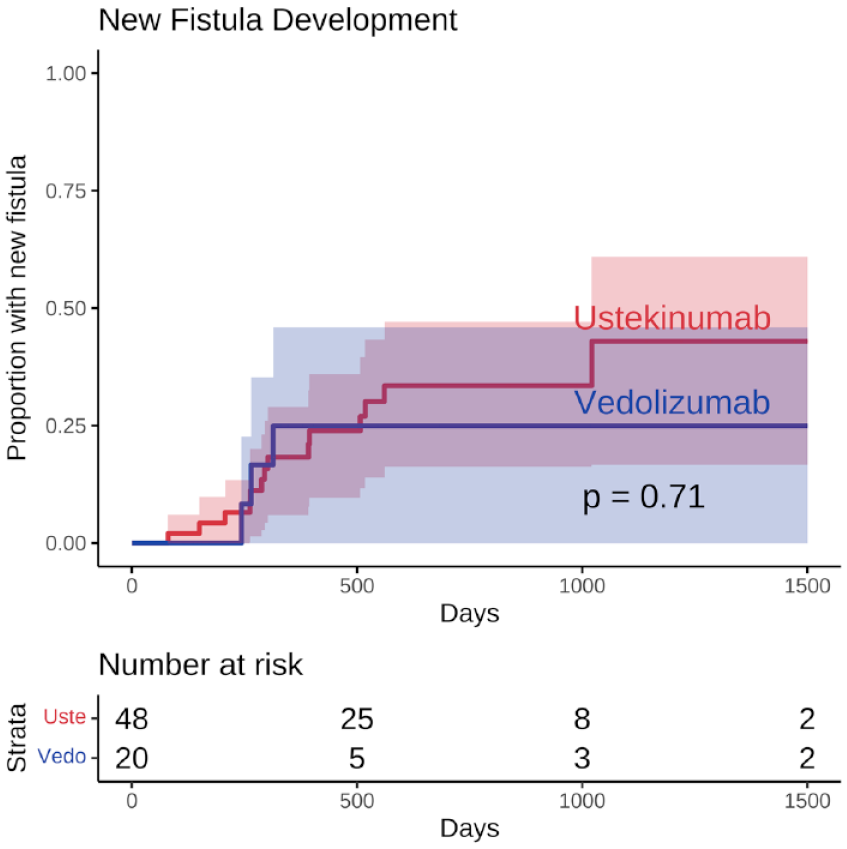
Kaplan–Meier curve for time to new fistula development in individuals with CD and an active fistula started on UST or VDZ. Log-rank test p values for unadjusted comparisons.
Overall, 12 patients in the UST group and four in the VDZ group had surgery while on therapy (Table 3). We found that individuals on UST had a rate of surgical CD management of 16/100 P-Y (95% CI: 9–26 per 100 P-Y), which was similar to that people on VDZ, who had a rate of 20/100 P-Y (95% CI: 8–49 per 100 P-Y). However, individuals in the UST group and a significantly longer median time to surgery (Table 3, p = 0.008), though the mean time to surgery was not significantly different (Figure 2). Of the 12 individuals who had surgery while on UST, five had resections with ostomy creation, and seven had intestinal resections without ostomy creation. In contrast, all four individuals who had surgery while on VDZ had a resection with ostomy creation.

Kaplan–Meier curve for time to IBD-related surgery in individuals with CD and an active fistula started on UST or VDZ. Log-rank test p values for unadjusted comparisons.
Median time on UST was 594 days and on VDZ was 322 days (Table 3, p = 0.01).
Adjusted multi-state modeling confirmed VDZ was significantly more likely to be discontinued than UST (Figure 3, p < 0.0001). The most frequent reason for discontinuation was lack of adequate overall clinical response.
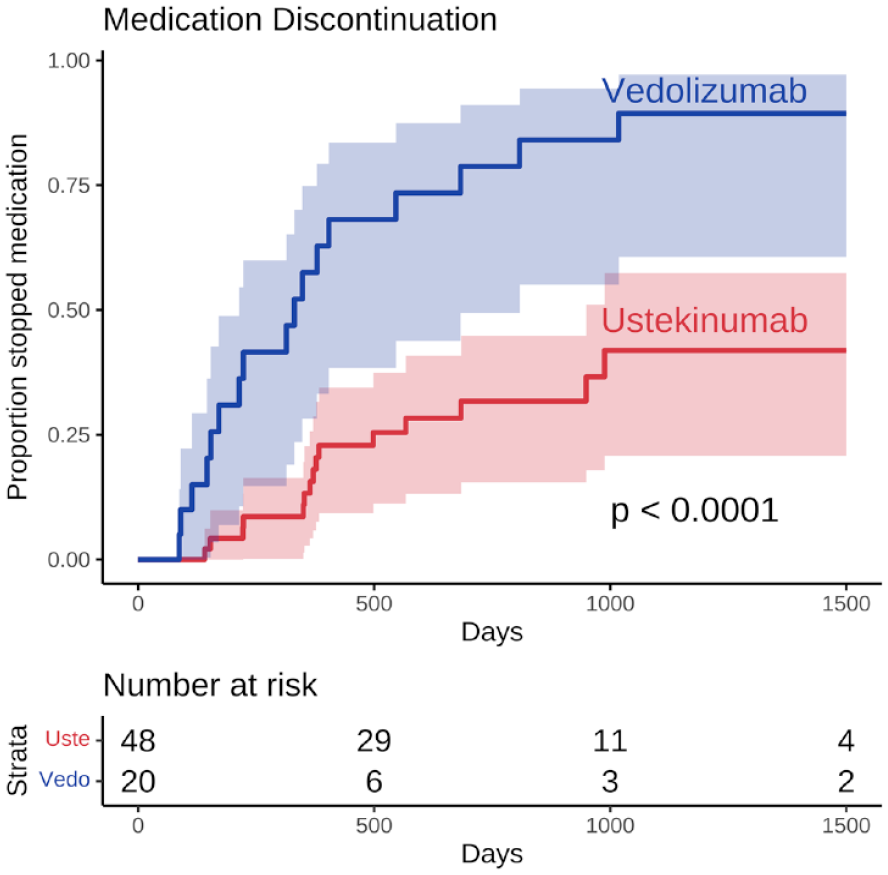
Kaplan–Meier curve for time to medication discontinuation in individuals with CD and an active fistula started on UST or VDZ. Log-rank test p values for unadjusted comparisons.
Discussion
In this single-center study of the real-world effectiveness of VDZ and UST for individuals with fistulizing CD, modeling suggests that the clinical utility of UST may be better than VDZ in patients with an active fistula, though the sample size is small. The UST group was significantly more likely to remain on the medication and had significantly longer median time until surgery. There was no significant difference between VDZ and UST in fistula healing or new fistula formation, though these analyses were limited by the high rate of VDZ discontinuation.
While not a head-to-head trial, our study has the advantage of comparing outcomes in the same clinical population. Our finding that that individuals with an active fistula at the time of medication initiation were more likely to discontinue VDZ than UST is an important indicator of clinical effectiveness that is consistent with some of existing literature. In our study, 90% of individuals on VDZ stopped this medication. This is similar to observational VDZ data in individuals with penetrating disease, which reported a 68% discontinuation rate in patients with perianal disease. 14 Even in a trial setting, penetrating disease is associated with relatively high discontinuation rates, with the ENTERPRISE study reporting 30% of individuals with a perianal fistula prematurely discontinuing VDZ despite enrollment in a clinical trial. 8 By contrast, rates of UST discontinuation in our study and others of individuals with fistulas are around 30–35%. 15 The major factor behind medication discontinuation in our study was lack of adequate clinical response. Both medications have favorable side effect profiles, which limit the need to stop because of medication intolerance.
An additional important finding of our study was the median time to surgery and differences in surgery type. The median time to surgery was significantly longer in the UST group compared to VDZ. Though the number of patients who underwent surgery while on these medications was small, there was a trend in the need for ostomy creation between the two groups, with all individuals in the VDZ group having surgical resections with ostomy creation and slightly less than half of people in the UST group having surgeries with ostomy creation. This suggests that the UST group may have had better mucosal healing that was amenable to anastomosis and improved distal disease, decreasing the need for diversion. Indeed, prospective registry data and retrospective observational data have shown that in individuals with CD are more likely to achieve and remain in remission on UST,16,17 and a recent meta-analysis shows that UST is more effective than VDZ for maintenance in people with anti-TNF-refractory CD. 18
In comparison with other data on UST and fistulizing CD, our study includes more granular detail but found similar results. The licensing trials for UST included individuals with penetrating CD, but in the initial publications, outcomes in this subgroup were not reported.7,19 Pooled post-hoc data on fistula healing from the CERTIFI, UNITI-1, and UNITI-2 trials of UST versus placebo showed that among individuals with active fistulas, 24–27% on UST had fistula resolution at 8 weeks across all dosage groups versus 14% in placebo group, but it was not statistically significant. 20 A prospective registry-based study in the Netherlands reported perianal fistula resolution in 35.7% of patients on UST at 24 weeks. 15 A French study of individuals with perianal CD found 33% of patients with setons at initiation of UST successfully had them removed while on the medication. 21 A meta-analysis by Attauabi et al. summarizing observational and post-hoc randomized controlled trial data on UST and active perianal fistulizing CD estimated the pooled rate of fistula closure to be 16.7% at 52 weeks. 22 We found that of individuals on UST for 1 year, 21% had fistula closure, close to the efficacy reported in randomized trials and other observational studies.
Like UST, there are sparse randomized controlled trial data on the efficacy of VDZ for fistula healing and few observational studies, most of which are limited to perianal fistulas. In our cohort, none of the individuals who continued on VDZ for a year had fistula healing. This is more consistent with the low rate seen in the observational data and may be related to our cohort’s high rate of discontinuation of VDZ for lack of clinical effect. A post-hoc analysis of data from the GEMINI 2 trial showed a trend toward better fistula healing on VDZ maintenance than placebo (28% with fistula closure versus 11% at week 14), though it was not statistically significant. 23 A separate post-hoc analysis of the ENTERPRISE study comparing two VDZ regimens reported 43% of patients had perianal fistula closure at week 30. 8 Observational data from France showed that among individuals with a seton who were started on VDZ, 15% had successful removal of the seton while on therapy. 14
Only a minority of patients in our study were on a second immunosuppressive agent, which limits our ability to assess the specific outcomes in that subgroup, and nearly all had prior exposure to anti-TNF therapy. Given the high rate of prior biologic treatment, we may have a more treatment-resistant group of patients, which may have limited their ability to achieve adequate clinical response and have led to the formation of more chronic fistulas. However, despite this, there were still patients who had fistula healing on UST in our study. We did not see statistically significant differences in fistula healing or new fistula formation between the different groups. This is likely to be due to a combination of the small sample size and the high rate of VDZ discontinuation. It is also possible that medication selection bias due to unmeasured clinical variables could conceal a difference between the groups. There could be bias in patient selection for the second-line biologic therapy (after anti-TNF) for fistulizing disease, as UST was selected 2.5 times more often than VDZ. This might suggest that UST was preferred in more severe fistulizing disease.
Strengths of our study include selection of patients with all types of fistulas, close follow-up in a single academic medical system, and prescribing patterns that represent real-world use rather than trial protocols. Limitations include the retrospective design, use of a single-center academic cohort that may not be broadly generalizable, lack of standardized measurement of fistula drainage and activity across patients, lack of a consistent protocol for monitoring clinical response, inability to control for provider choice in selection of medication, small sample size that was further diminished because of high rates of medication discontinuation, and variable follow-up time.
Conclusion
In conclusion, patients with an active fistula were more likely to stay on UST than VDZ and were able to wait longer until CD surgery. There was a trend toward more fistula healing in the UST group, though this outcome was not significant. These findings highlight the importance of further research in this challenging complication, including head-to-head trials to compare second-line biologics after anti-TNF for fistulizing disease, and the development of novel therapeutics to treat chronic fistulas.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221148254 – Supplemental material for Vedolizumab more likely to be discontinued than ustekinumab in anti-TNF-experienced patients with fistulizing Crohn’s disease
Supplemental material, sj-docx-1-tag-10.1177_17562848221148254 for Vedolizumab more likely to be discontinued than ustekinumab in anti-TNF-experienced patients with fistulizing Crohn’s disease by Kira L. Newman, Laura A. Johnson, Ryan W. Stidham and Peter D. R. Higgins in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
