Abstract
Background:
Various therapeutic strategies are available for the first-line treatment of patients with advanced hepatocellular carcinoma (aHCC). But which approach is the most cost-effective remains uncertain.
Objectives:
This study aims to evaluate the cost-effectiveness of first-line strategies in aHCC patients from the perspective of Chinese and US payers.
Design:
A network meta-analysis (NMA) and cost-effectiveness study.
Data sources and methods:
A NMA was conducted to collect all first-line strategies with aHCC from 1 October 1 2018 until 1 January 2022. The relevant randomized controlled trial literature in PubMed, Embase, and Cochrane Library for the last 3 years were searched. The abstracts of meetings of the American Society of Clinical Oncology, European Society of Medical Oncology, and American Association for Cancer Research were also reviewed. A Markov model that included three states was developed. One-way sensitivity and probabilistic sensitivity analysis were performed to investigate the uncertainty of the economic evaluation. Scenario analysis was conducted to explore the economic benefits of treatment strategies in low-income populations.
Results:
Base-case analysis in China included 1712 patients showed that atezolizumab combined with bevacizumab, sintilimab combined with bevacizumab, lenvatinib (LEVA), and sorafenib (SORA) added 0.46, 1.25, 0.77, and −1.08 quality-adjusted life-years (QALYs), respectively, compared with donafenib, resulting in an incremental cost-effective ratio of $85607.88, $12109.27, and $1651.47 per QALY at a willingness-to-pay (WTP) of $11101.70/QALY. In the United States, only the incremental cost-effectiveness ratios (ICERs) of SORA was higher that were lower than the WTP threshold ($69375/QALY), and LEVA was the most cost-effective strategy with the ICERs were 25022.13/QALY.
Conclusion:
The NMA and cost-effectiveness analysis revealed that LEVA is the favorite choice in the first-line treatment of Chinese aHCC patients and US payers’ perspective when the WTP was $11101.70/QALY in China and $69375.0/QALY in the United States.
Registration:
This study has been registered on the PROSPERO database with the registration number CRD42021286575.
Introduction
Hepatocellular carcinoma (HCC) is the most common tumor type in primary liver cancers, 1 accounting for 75–85% of cases. 2 Most HCC patients are diagnosed at an advanced stage and cannot be surgically removed, 3 which caused a huge social and economic burden. 4 The disease burden of advanced HCC (aHCC) patients in China and the United States has been increasing year by year and showing an upward trend. 1 According to statistics, the cost of health care for treating aHCC is $405 million and is growing at a rate of 5.4% per year in the United States. 5
Nowadays, molecular targeted therapy, immunotherapy, and immune combination therapy have become the main approaches for the treatment of HCC. 6 In China, there are five treatment strategies including lenvatinib (LEVA), atezolizumab combined with bevacizumab (atezo-bev), sintilimab combined with bevacizumab (sinti-bev), sorafenib (SORA), and donafenib (DONA) have become the first-line treatments for aHCC proved by the Chinese National Medical Products Administration (NMPA) and recommended by the guidelines of Chinese society of clinical oncology (CSCO) in 2020. 7 In the United States, SORA, LEVA, nivolumab (NIVO), and atezo-bev are the first-line treatment regimens proved by the U.S. Food and Drug Administration (FDA) and recommended by the National Comprehensive Cancer Network (NCCN). 8 Among them, SORA has always been the standard treatment for aHCC treatment since it was approved by FDA in 2007. 9 Although SORA can bring an obvious survival benefit, the median survival rate of patients who progress to the terminal stage is less than 10%. In 2017, the REFLECT study 10 indicated that LEVA was non-inferior compared with SORA, with the median survival improvement (13.6 months versus12.3 months). Based on this, LEVA became a new choice for first-line clinical therapy recommended by FDA and CSCO guidelines. The IMbrave150 study 11 reported that atezo-bev significantly prolonged patient survival compared to SORA (5.7 months versus 3.2 months). This regimen is the first approved first-line immune combination therapy for aHCC in the world. CheckMate 040 12 and CheckMate 459 13 trials made NIVO the only programmed death 1 inhibitor recommended by the NCCN guideline 14 for first-line treatment of aHCC. The success of IMbrave 150 reveals the arrival of the era of immunotherapy for HCC. ORIENT-32 study 15 confirmed that sinti-bev prolonged progression-free survival (PFS; 4.5 months versus 2.8 months) in Chinese aHCC patients compared with SORA. The ZGDH3 study 16 proved that DONA became the first drug since 2007 to achieve superior overall survival (OS) compared with SORA on 30 June 2021. As early as 9 June 2021, DONA was officially approved by NMPA, becoming a new option for the first-line treatment of aHCC patients.
Although several studies have been exploring the mechanism of action of aHCC drugs and clinical trials, relevant economic studies are scarce and limited to the comparison of two drugs. 17 The research of Chi-Leung et al. 18 indicated that atezo-bev is cost-effective versus SORA if all aHCC patients are assumed to be cured. Hongfu et al. 19 conducted an economic study of LEVA versus SORA revealed that LEVA has economic benefits under the threshold of three times the per capita GDP from the perspective of the Chinese health system. However, there is no previous research that directly compares different first-line treatment options for aHCC. The conflict between cost and effectiveness may limit physicians and decision-makers to make rational choices about the best treatment.
Therefore, this study conducted a systematic review, network meta-analysis (NMA), and cost-effectiveness analysis to evaluate the costs and healthcare outcomes of first-line treatment strategies from the perspective of the US payers and Chinese aHCC patients.
Methods
This study followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement 20 and was compliant with the 2022 Comprehensive Health Economic Assessment Reporting Standards (CHEERS 2022). 21 This study has been registered on the PROSPERO database with the registration number CRD42021286575.
Network meta-analysis
Study strategy
Two investigators independently extracted baseline data for patients in the study. The relevant randomized controlled trial (RCT) literature in PubMed, Embase, and Cochrane Library for the last 3 years were searched. And the abstracts of meetings of the American Society of Clinical Oncology (ASCO), European Society of Medical Oncology (EMSO), and American Association for Cancer Research were also reviewed from 1 October 2018 until 1 January 2022. Only the most recent data were retained for the results of the same trial. The detailed search strategy is found in Supplemental eFile 1.
Statistical analysis
Analysis was performed using the Gemtc package of the R software, version 4.0.2.
For time-event variables such as PFS and OS, the hazard ratio (HR) and the 95% confidence interval (CI) were used as the effect size; for dichotomous variables such as objective response rate and incidence of adverse reactions, the odds ratio (OR) was used as the effect size. Calculate I2 to assess the overall heterogeneity of the model. If there was no significant heterogeneity (I2 < 50% or p < 0.1), a fixed-effects model was used, otherwise a random-effects model was used. The adjusted HR and OR values are both calculated using a Bayesian method embedded in the following formula (using HR as an example) 22 : ln(HR) = [ln(UL − HR) + ln(LL − HR)]/2; seln(HR) = [ln(UL − HR) − ln(LL − HR)]/(1.96 × 2). Quality assessment of included RCTs was assessed according to the Cochrane recommendations using RevMan, version 5.4.
Cost-effectiveness
Model structure
A three-state Markov model was developed based on the disease progression of aHCC: PFS, progressed disease (PD), and death state (Supplemental eFigure 1). The hypothetical target population for this analysis was assumed to be consistent with the patient characteristics of RCT. Five treatment options were evaluated: atezo-bev, sinti-bev, LEVA, DONA, and SORA in Chinese patients, and four therapies in the United States: atezo-bev, LEVA, NIVO, and SORA.
The period of the model is 1 month, which is consistent with the drug treatment. The time horizon of the model was 10 years as the survival rate at 10 years was less than 5%. Patients started in this model at the state of PFS and can be transferred to other states or remain in their current state. The patients were assumed to have an average weight of 65 kg and an age of 60. The transition probability from PFS status to death is 7.18‰ 23 in China and 8.98‰ 24 in the United States of natural mortality in 2020. The discount rate for costs and health outcomes was set to be 5% (0–8%) 25 in China and 3% (0–5%) in the United States. 26
Clinical data
The PFS and OS curve data of the RCTs were extracted using GetData Graph Digitizer (version 2.26) for individual data reconstruction. Exponential, Weibull, Logistic, Lognormal, and Loglogistic distributions were selected to refit the individual data and reconstruct the Kaplan–Meier curves, respectively. The optimally fitted distributions were judged according to the Akaike information criterion and the survival function parameters were obtained. Additional details concerning model fitting are given in Supplemental eTables 1 and 2 and in Supplemental eFigures 2 and 3.
Costs and utilities
Only direct medical costs were analyzed based on the perspective of Chinese aHCC patients and the US payers’ perspective. The medicine prices in China mostly came from the median of the latest Chinese medicine bid price announced by YAOZH (www.yaozhi.com), and sourced from the Red book online and published literature in the United States. If the drug is reimbursed by medical insurance, it will be calculated at 60%.
The adverse event (grade ⩾3) reported in this study were mainly from the reported RCTs. The cost of adverse reaction treatment was derived from published literature and consultation with clinical specialists. The costs for hospitalization, computed tomography, and magnetic resonance imaging were obtained from the medical service price documents published by Chinese public hospitals and published literature.
Direct non-medical costs were ignored because they are difficult to measure accurately. In addition, this study assumed equal indirect costs to patients for the five treatment options and was not included in the model calculation. All cost values in China were converted at the first half of the 2021 exchange rate (1 USD = 6.47 RMB).
Effectiveness
Cost, quality-adjusted life-year (QALY), and incremental cost-effectiveness ratios (ICERs) were the main output evaluation results. In this study, the willingness-to-pay (WTP) threshold was set at 1 time the per capita GDP, which is $11101.70 in China and $69375 in the United States.
Statistical analysis
In this study, the GetData Graph Digitizer version 2.24 software was used to plot the survival curve. SurvHE package in R software (4.1.2) simulates survival curves. TreeAge Pro 2020 (TreeAge Software) was used to complete the construction of the model.
Sensitivity analysis
One-way sensitivity analysis and probability sensitivity analysis (PSA) were presented to reflect the impact of the uncertainty in the model inputs of this evaluation. The range of variation and distribution of the parameters are shown in Table 1. In case of lack of upper and lower limit values, the calculation was based on ±20% of the parameters. The second-order Monte Carlo simulation was used to randomly simulate the sampling for 1000 times, and the results were represented by ICER scatter plots and cost-effectiveness acceptable curves.
Model inputs.

AIC, Akaike information criterion; CT, computed tomography; DONA, donafenib; MRI, magnetic resonance imaging; NA, not applicable; NIVO, nivolumab; NMA, network meta-analysis; OS, overall survival; PD, progressed disease; PFS, progression-free survival; SORA, sorafenib.
Scenario analysis
Sintilimab, atezolizumab, and LEVA have charitable drug donation programs in China for low-income patients, so this study set up a scenario analysis. Assume that the patient is fully eligible for charitable drug donations. The WTP is set to $1216.20 for low-income people in China in 2021.
Results
Network meta-analysis
In the meta-analysis, four RCTs containing a total of 1712 patients in China and three RCTs in the United States were included. The basic characteristics of the included literature are shown in Supplemental eTables 3 and 4. Network plot of evidence was reported in Supplemental eFigure 4. Risk bias was assessed in seven aspects for each literature included in the analysis. The results of the risk bias assessment are proposed in Supplemental eFigure 5. It can be seen that the bias in the included literature was mainly found in the blinded approach.
The surface under the cumulative ranking results (Supplemental eTable 5) indicated that atezo-bev showed the best OS benefit, LEVA achieved the best PFS and second-best OS benefit, and DONA had the best safety profile in China. Atezo-bev achieved the best PFS and OS and LEVA had the best safety profile in the United States. HRs for NMA are indicated in Supplemental eTables 6 and 7.
Cost-effectiveness analysis
Base-case analysis
Base-case results in Table 2 suggested that in China, the QALY of atezo-bev, sinti-bev, LEVA, and SORA treatment groups were more effective than DONA (9.23, 10.02, 9.55, 7.69 QALYs), which increased by 0.46, 1.25, 0.77, and −1.08 QALYs, respectively. The ICERs of atezo-bev and sinti-bev groups were $85607.88/QALY and $12109.27/QALY, both of which were greater than WTP. LEVA is the most cost-effective regimen.
Summary of the cost ($) and outcome results in base-case analysis.
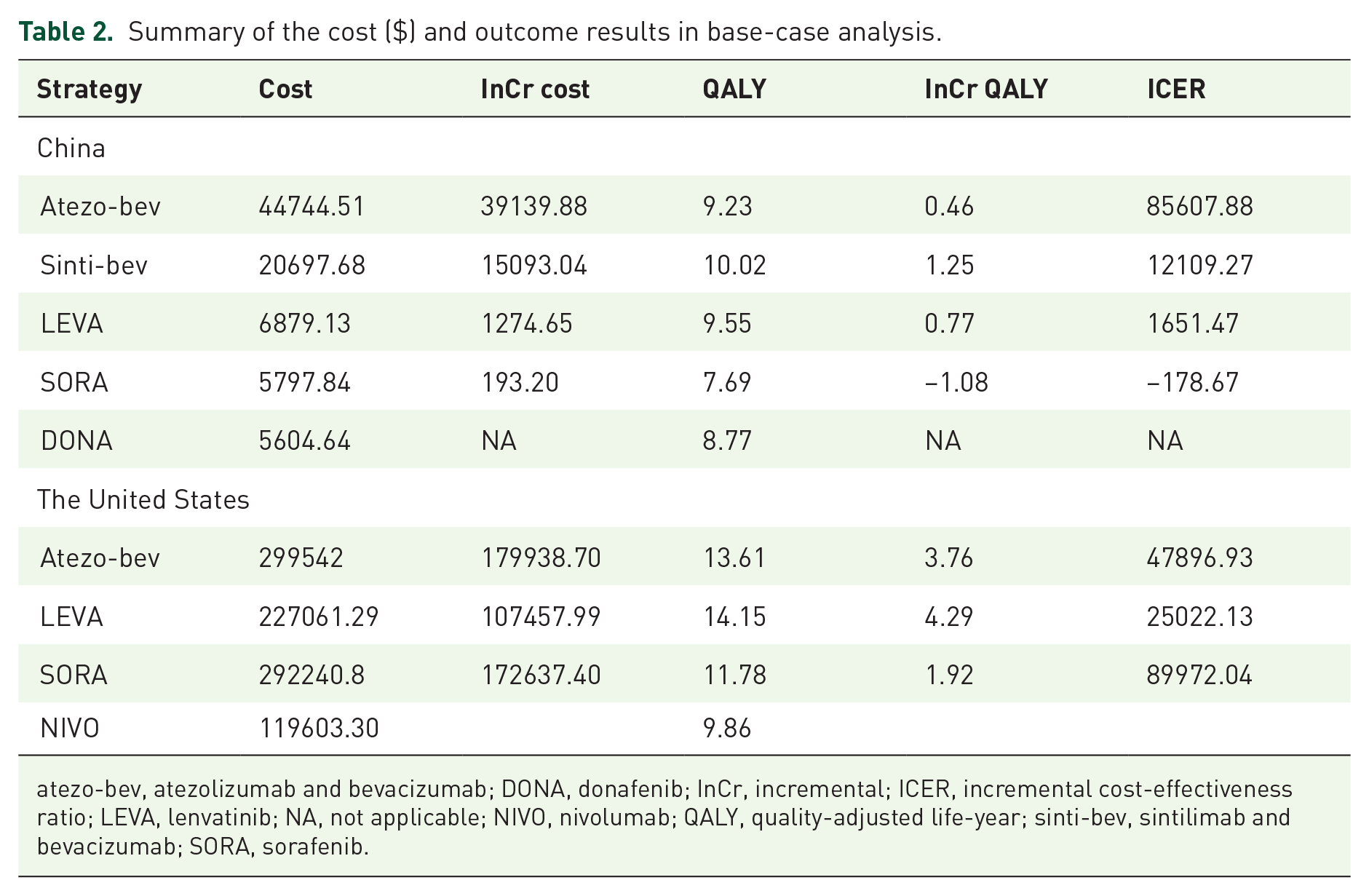
atezo-bev, atezolizumab and bevacizumab; DONA, donafenib; InCr, incremental; ICER, incremental cost-effectiveness ratio; LEVA, lenvatinib; NA, not applicable; NIVO, nivolumab; QALY, quality-adjusted life-year; sinti-bev, sintilimab and bevacizumab; SORA, sorafenib.
In the United States, compared with the NIVO strategy, the mean incremental costs and QALYs of atezo-bev, LEVA and SORA group were $179938.70 and 3.76, $107457.99 and 4.29, $172637.40 and 1.92. The ICERs were $47896.93/QALY, $25022.13/QALY, and $89972.04/QALY. Given the WTP of $69375.0/QALY, ICERs of LEVA and atezo-bev were within the thresholds, and LEVA was more cost-effective than atezo-bev.
Sensitivity analysis results
Figure 1(a) shows that the conclusion is most sensitive to the cost, but changes do not affect the conclusion. The increase in the cost of sinti-bev and TX made it less economically beneficial (Figure 1(b)). Figure 1(c) indicated that the top factor that have the greatest impact on the conclusion are the cost of LEVA treatment. A univariate sensitivity analysis of the cost of LEVA showed that with the price increase, when the cost of LEVA exceeds $1357.87 (242.10%), LEVA is no longer economical relative to DONA in China.

Tornado diagrams of univariable sensitivity analyses in China.
The results in the United States were substantially sensitive to the discount rate, the cost of LEVA, the utility of PD in the comparison of atezo-bev versus NIVO, LEVA versus NIVO, and SORA versus NIVO (Figure 2). Changes in these parameters did not affect the conclusion.

Tornado diagrams of univariable sensitivity analyses in the United States.
PSA results in Supplemental eFigure 6 showed that the scatter distribution was concentrated and the model was robust. The probability that the sinti-bev regimen being cost-effective was 30.9% at the WTP threshold of $11101.70 per QALY in China.
The acceptability of DONA is higher than LEVA when the WTP is less than $20,00 (Figure 3(a)). The probability of LEVA being cost-effective was 95% at WTP of 11191.70/QALY. The cost-effectiveness acceptability curve showed that the probability of sinti-bev being cost-effective increased from 0.1% to 48% when the threshold value ranged from $11101.70/QALY to $28242.0/QALY. The probability of LEVA was the optimal treatment at the WTP of $29425.0/QALY and $69375.0/QALY in the United States (Figure 3(b)), compared with other competing therapies.

Cost-effectiveness acceptability curves in China (a) and in the United States (b).
Scenario analysis
Table 3 shows that the ICER of atezo-bev, sinti-bev, and LEVA in low-income people was greater than WTP ($1216.20), and the ICER of SORA was negative, so DONA was the most cost-effective therapy under WTP in Chinese low-income patients.
Summary of the cost ($) and outcome results in base-case analysis of low-income patients.

atezo-bev, atezolizumab and bevacizumab; DONA, donafenib; InCr, incremental; ICER, incremental cost-effectiveness ratio; LEVA, lenvatinib; NA, not applicable; QALY, quality-adjusted life-year; sinti-bev, sintilimab and bevacizumab; SORA, sorafenib.
Discussion
This study conducted a NMA as a basic analysis and 10 years Markov model from the perspective of Chinese aHCC patients and US payers. The results of the study showed that LEVA was the most economical treatment when the WTP threshold was 1 time the national per capita GDP in China and the United States. Univariate sensitivity analysis suggested that the increase in LEVA price may make it lose its economic benefits in China. Scenario analysis showed that DONA was the most cost-effective option in low-income populations.
The sensitivity analysis suggested that an increase in the price of LEVA may make it no longer economically beneficial in China. In fact, with the official implementation of the new version of the National Medical Insurance Catalogue on 1 March 2021, LEVA has been substantially reduced in price and entered into medical insurance, greatly reducing the financial burden of patients. In addition, the patent of LEVA in China will expire on 19 October 2021. Some generic drugs have been approved for marketing at present, and the treatment cost of patients will be greatly reduced. Therefore, LEVA may still be the most economical treatment option in the future for Chinese patients.
This finding is consistent with that of John et al. 30 who compared LEVA and SORA based on the perspective of Canadian health care. The research indicated that LEVA dominated SORA in the base-case analysis but would no longer be the dominant strategy if the cost of SORA was reduced by 57%. However, the cost of SORA does not influence our results. The results of Giuliani J et al. 35 were also consistent with this study. The study assessed the cost-effectiveness of LEVA and atezo-bev as first-line therapy for aHCC. Including results from 1455 patients, LEVA is a cost-effective treatment for first-line aHCC, combining the pharmacological cost of the drug with a measure of efficacy represented by PFS. Hongfu et al. 36 conducted a Markov in Chinese patients confirmed the cost-effectiveness of LEVA. The reason for the cost-effectiveness of LEVA may be related to monotherapy. In addition, it may be related to the mechanism of action of LEVA. LEVA is a small molecule inhibitor of VEGFR 1–3, fibroblast growth factor receptor 1–4, PDGF Rα, KIT, and RET.32,36
The superiority of LEVA in patients with HCC in the United States has been recognized by the FDA. 37 Although one-third of the patients was enrolled in non-Asian regions, the REFLECT study was not designed to assess efficacy in any country or region, and differences in subgroup analyses may be related to other location factors. The conclusions can therefore be extrapolated to US patients.
Sequential treatment after failure of first-line therapy for HCC is also a matter of consideration. According to CSCO guidelines, 7 regorafenib and cabozantinib can be used as second-line drug options after SORA treatment failure/intolerance. The study by Francesco et al. 38 suggested that cabozantinib can be prescribed as a third-line rather than second-line agent in clinical practice [median OS:12.1 months (95% CI: 9.4–14.8)]. SORA followed by sequential regorafenib improves survival benefit in patients with advanced HCC [median survival: 10.6 months (95% CI 9.1–12.1)].39,40
The increase in WTP value may have an impact on the study conclusions in China. Sensitivity analysis showed that the acceptability of sinti-bev increased gradually with the increase in WTP. Actually, with the improvement of the Chinese national economic level, the WTP value has increased year by year. When it is greater than $28242.0, sinti-bev is cost-effective.
The results of this study could extrapolate to developing countries with a level of economic development and affordability comparable to that of China. However, due to the unbalanced economic development in China, the per capita GDP of each province varies greatly. For example, the per capita GDP difference between Beijing and Gansu can be as high as $20646.21 ($25910.35 versus $5264.14). Therefore, in the economically developed areas of Beijing and Shanghai, the WTP thresholds are $25910.35 and $24680.83, respectively, 23 and sinti-bev can be selected as a more economical alternative to LEVA.
The conclusions of this study can provide a reference for medical insurance payment decisions. In China, pharmacoeconomic evaluation has now become important evidence to support national health insurance negotiations. Economic evaluation of first-line treatment strategies for HCC patients is helpful to provide decision-making basis for clinical rational drug use, and provide reference for the update of the medical insurance catalogue. In addition, the budget impact analysis of drugs for HCC treatment is also expected in the future to investigate the impact of the introduction of new drugs on health insurance reimbursement.
This study has several advantages: First, this study is the first economic evaluation of multiple first-line treatments comparing China and the United States. The included patient populations were all from RCTs, and a NMA was performed on the Chinese subgroup. The fitted survival data are better matched the characteristics of the population and reduce the bias of the evaluation results due to demographic differences. Second, this study compared the efficacy and safety of the treatment regimens as a control group through a NMA. And the HR data were supplemented for Markov model analysis. Third, a situational analysis was set up to explore the differences in the treatment of aHCC among the four treatment strategies in low-income and general patients. It provided an evidence-based reference for Chinese medical and health policymakers.
Our research has several limitations. First, this study obtained the parameters related to the Markov model by fitting the PFS and OS curves reported in the RCT, which are different from the real world and may lead to model uncertainty. However, the survival curve and the fitting curve of the clinical trial were matched and found to be highly consistent, so the effect on the results was small. Second, this study did not consider that adverse reactions will cause the utility value of patients with PFS and PD status to decrease, which will cause a certain bias in the research results. Third, this study assumes that the second-line treatment costs are the same after disease progression, but in reality, there will be different drug options according to the individual situation of the patient. Finally, this study only included the treatment cost of serious adverse drug reactions with an adverse reaction variation greater than 3%, which may lead to discrepancies between the results and the actual results. However, the results of univariate sensitivity analysis showed that the incidence of adverse reactions and treatment costs had little effect on the results, so the conclusions of the study would not be reversed.
Conclusions
The findings of this NMA and economic evaluation suggest that compared with other therapies, LEVA was the most economically effective first-line treatment strategy for aHCC in China and the United States.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221140662 – Supplemental material for First-line treatments for advanced hepatocellular carcinoma: a network meta-analysis and cost-effectiveness analysis in China and the United States
Supplemental material, sj-docx-1-tag-10.1177_17562848221140662 for First-line treatments for advanced hepatocellular carcinoma: a network meta-analysis and cost-effectiveness analysis in China and the United States by Ke-Xin Sun, Shan-Shan Cao, Feng-Hao Shi, Yue Guan, Meng Tang, Mei-na Zhao, Yu-Fan Jian, Bin Cui, Zhi-Yan Li, Jing-Wen Wang, Feng Yu and Yi Ding in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
