Abstract
Background:
Patients with HER2-negative locally advanced or unresectable metastatic gastric cancer and gastroesophageal junction (G/GEJ) adenocarcinoma have limited first-line treatment options and a poor prognosis. The GLOW clinical trial showed that zolbetuximab plus capecitabine plus oxaliplatin (CAPOX) significantly prolonged these patients’ overall survival (OS) and progression-free survival (PFS).
Objectives:
This study evaluated the cost-effectiveness of zolbetuximab plus CAPOX as a first-line treatment for HER2-negative locally advanced or unresectable metastatic G/GEJ adenocarcinoma in the United States and China.
Design:
The cost-effective analysis.
Methods:
Based on the GLOW clinical trial data (NCT03653507), we constructed a 10-year Markov model to assess the cost-effectiveness of the zolbetuximab or placebo plus CAPOX treatment regimen. Only direct medical costs were considered. The primary outcomes of the model were quality-adjusted life-years (QALYs) and incremental cost-effectiveness ratios (ICERs). One-way and probabilistic sensitivity analyses were employed to assess the robustness of the model.
Results:
In the United States, zolbetuximab plus CAPOX added 0.24 QALYs and resulted in an incremental cost of $196,791.11 compared with placebo plus CAPOX, which had an ICER of $821,515.65 per QALY gained. For China, the zolbetuximab group gained 0.23 QALYs at an incremental cost of $62,822.69, resulting in an ICER of $273,568.01/QALY. One-way sensitivity analysis revealed that the results were most sensitive to the price of zolbetuximab. Zolbetuximab plus CAPOX had 0% cost-effectiveness at the willingness-to-pay thresholds of $150,000/QALY in the United States and $38,188/QALY in China.
Conclusion:
Zolbetuximab plus CAPOX may be a cost-effective option for patients with locally advanced, unresectable, or metastatic G/GEJ adenocarcinoma when the price of zolbetuximab reduced by 83.37% ($367.7/100 mg) in the United States and 82.25% ($110.8/100 mg) in China.
Keywords
Introduction
Gastric cancer (GC) represents a prevalent malignancy within the digestive tract, ranking fifth in global cancer incidence. In 2020, over a million new cases resulted in approximately 769,000 deaths. GC thus stands as the fourth leading cause of cancer-related mortality worldwide, following lung cancer, colorectal cancer, and liver cancer. 1 Adenocarcinomas comprise over 95% of GC cases, with a notable rise in gastric or gastroesophageal junction (G/GEJ) adenocarcinomas. 2 Due to the nonspecific early symptoms, the majority (80%–90%) of G/GEJ adenocarcinomas are typically diagnosed at an advanced or metastatic stage, indicating that surgical resection is no longer feasible. 3 Patients with locally advanced/metastatic GC have a poor prognosis, with a mere 6% 5-year survival rate. 4
The standard first-line treatment for patients with locally advanced, unresectable, or metastatic G/GEJ adenocarcinoma is to receive platinum–fluoropyrimidine chemotherapy with folinic acid plus 5-fluorouracil and oxaliplatin (FOLFOX) and capecitabine plus oxaliplatin (CAPOX) recognized as standard regimens in Western and Asian countries.5,6 Studies have shown that targeted therapy or immunotherapy in combination with chemotherapy improves overall survival (OS) in patients with GC; trastuzumab is approved for use in approximately 15% of patients with HER2-positive disease.7 –9 Based on the results of the CheckMate 648 trial, 10 nivolumab in combination with chemotherapy is approved for the first-line treatment of advanced or metastatic G/GEJ cancer; however, its efficacy is mainly limited to patients with programmed death ligand 1 (PD-L1) in combination with a positivity score ⩾5. Overall, there remains an unmet need for patients with HER2-negative, locally advanced, unresectable, or mG/GEJ adenocarcinoma.
CLDN18.2 is a tight junction protein whose expression is significantly upregulated in most G/GEJ adenocarcinomas. 11 Zolbetuximab is the first CLDN18.2-targeted drug to enter a global phase III clinical trial. Recently, the latest clinical data were released from the GLOW trial (NCT03653507; https://www.clinicaltrials.gov/), 12 a phase III clinical trial evaluating the efficacy and safety of zolbetuximab or placebo plus CAPOX for the treatment of patients with CLDN18.2-positive, HER2-negative locally advanced unresectable, or metastatic G/GEJ adenocarcinoma. The results revealed that zolbetuximab plus CAPOX markedly prolonged the progression-free survival (PFS: 8.21 vs 6.80 months) and OS (14.39 vs 12.16 months) compared to placebo plus CAPOX. Grade ⩾3 treatment-emergent adverse events (AEs) were similar with zolbetuximab and placebo (72.8% vs 69.9%). Thus, the zolbetuximab plus CAPOX regimen seemed to be an attractive first-line option for advanced unresectable or metastatic G/GEJ adenocarcinoma.
Although GLOW has demonstrated that zolbetuximab improves clinical outcomes in patients with GC, its high cost may offset its antitumor efficacy. Currently, zolbetuximab has been applied for marketing in several countries, so it is necessary to conduct an economic assessment to provide some reference for its subsequent pricing decision. Considering differences in the national conditions and medical environments, this study evaluated the cost-effectiveness of zolbetuximab plus CAPOX for the first-line treatment of patients with CLDN18.2-positive, HER2-negative locally advanced unresectable or metastatic G/GEJ adenocarcinomas from third-party payers in the United States and healthcare perspectives in China, representing high- and middle-income regions, respectively.
Methods
The reporting of this study conforms to the CHEERS 2022 (Consolidated Health Economic Evaluation Reporting Standards) statement (Supplemental eTable 1). 13
Patients and intervention
The hypothetical target population for this analysis was patients with HER2-negative, CLDN18.2-positive, locally advanced, unresectable, or mG/GEJ adenocarcinoma who had not received prior systemic treatment, consistent with the patient characteristics of the GLOW trial. 12 Patients received either zolbetuximab 800 mg/m2 (followed by 600 mg/m2) or placebo intravenously on day 1 plus CAPOX (capecitabine 1000 mg/m2 twice daily, days 1–14; and oxaliplatin 130 mg/m2, day 1) in a 3-week cycle for eight cycles. Beginning with cycle 9, patients continued to receive zolbetuximab or placebo plus capecitabine (at the discretion of the investigator) until disease progression, unacceptable toxic effects, withdrawal of consent, or study discontinuation.
All patients received second-line chemotherapy after disease progression. Based on the recommendations of the NCCN Clinical Practice Guidelines in Oncology 14 and systematic treatment information provided by GLOW, 12 we assumed second-line regimens included chemotherapy-based therapies (paclitaxel, docetaxel, or irinotecan monotherapy) and targeted therapies (ramucirumab plus paclitaxel). According to the RAINBOW trial, 15 the median treatment duration of ramucirumab as second-line treatment in patients with advanced GC was 18 weeks. Therefore, the duration of ramucirumab after progression was estimated to be 5 months, with rates of 8.3% in the zolbetuximab arm and 11.1% in the placebo arm. Palliative care was offered to patients whose condition worsened and were in the terminal stage.
Model structure overview
This economic evaluation constructed a Markov model with three health states for an initial decision regarding therapy with zolbetuximab or placebo plus CAPOX. The three mutually exclusive health states were progression-free disease (PFD), progressed disease (PD), and death. 16 As shown in Figure 1, all patients were initially set to a PFD state and either maintained or progressed to the following health state in each cycle. The Markov model operated with a 1-month cycle length and a 10-year time horizon, which can fully cover the whole lifetime of 99.9% of patients. The key outcomes included total cost, quality-adjusted life-years (QALYs), and incremental cost–benefit ratios (ICERs). Model development and data analysis were performed using TreeAge Pro 2022 (TreeAge Software Inc, Williamstown, MA).

Overview of partitioned survival model structure.
Clinical data inputs
The survival data of patients in the zolbetuximab or placebo arm were informed by the results of the GLOW trial and extrapolated using statistical analyses described by Guyot et al. 17 Data points were extracted from the PFS and OS Kaplan–Meier (K–M) curves by using GetData Graph (version 2.26). 18 Then, these data points were used to fit and extrapolate the following parametric survival functions to obtain long-term clinical outcomes, including Gompertz, Exponential, Weibull, Gamma, Log-Logistic, and Log-Normal. 19 The goodness-of-fit was chosen based on the lowest Akaike information criterion, Bayesian information criterion, and visual simulation methods. 16 The areas under the OS curve and the PFS curve indicated the proportion of patients alive and alive with PFD, respectively. The proportions alive and with PD were estimated by the area between the OS and PFS curves. Note that the transition probability of death for patients with PFD was assumed to be the age-specific natural mortality rates in China 20 and the United States. 21 The fitted results are shown in Supplemental eTable 2 and Supplemental eFigure 1, and the final survival functions of the two are shown in Table 1.
GLOW trial clinical data.
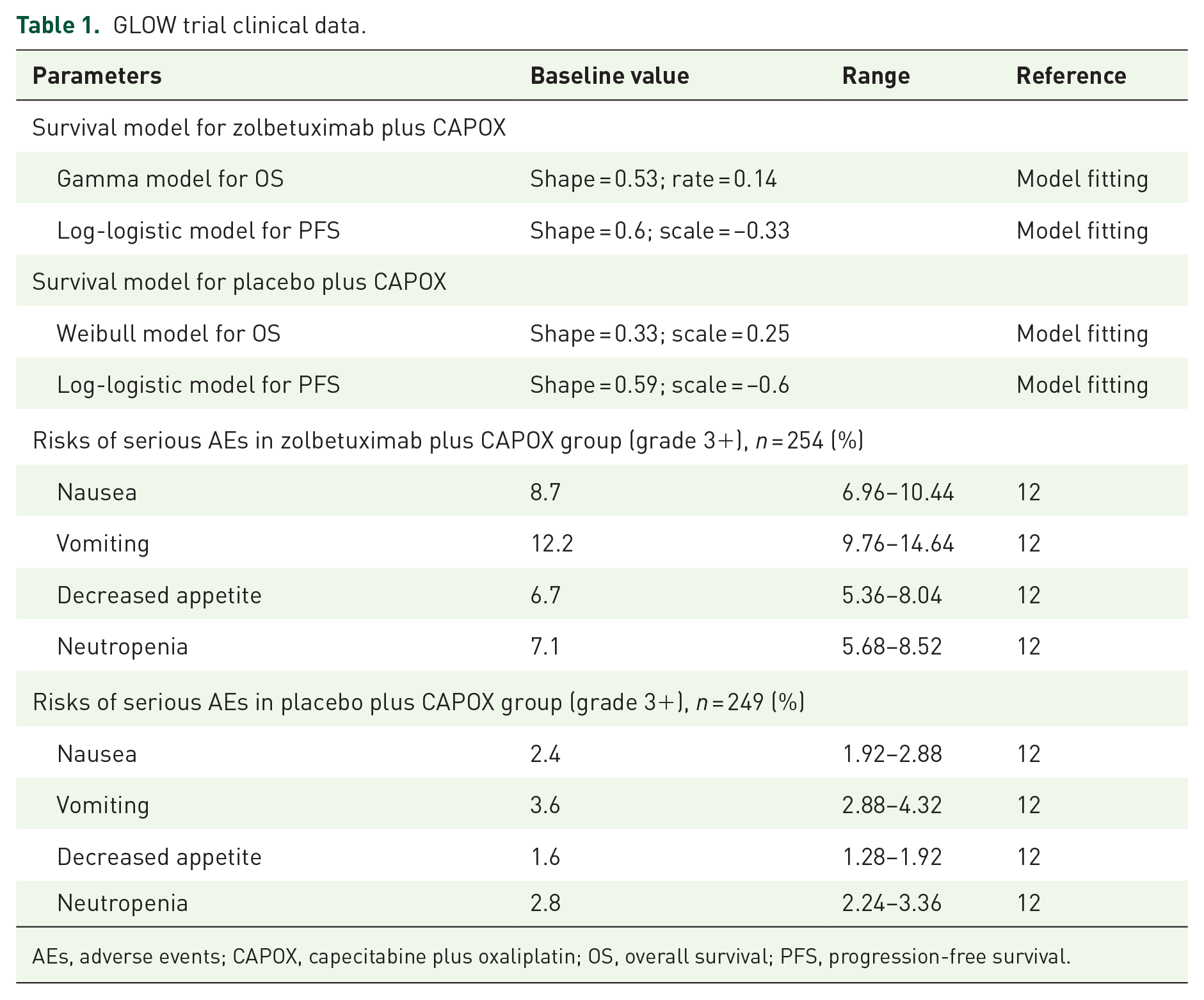
AEs, adverse events; CAPOX, capecitabine plus oxaliplatin; OS, overall survival; PFS, progression-free survival.
Costs and utility inputs
Only direct medical costs were analyzed in the model, including costs of drug, routine follow-up, costs for management of AEs, and costs for palliative care. Costs in the Chinese perspective were converted to the US dollar using an average exchange rate of US dollars in 2023 ($1 = RMB 7.0467) to enhance the manuscript’s readability for an international audience. The price of zolbetuximab has not been published in any global market. To determine the cost of zolbetuximab, we selected the median price of four targeted drugs recommended by the NCCN 14 for the treatment of GC: trastuzumab, nivolumab, pembrolizumab, and ramucirumab. We also performed an uncertainty analysis on the price of zolbetuximab, using the maximum and minimum values of the above drugs as the range of variation to improve the accuracy of the analysis.
Chinese drug prices were obtained from Yaozhi.com, utilizing the median value of all awarded bid prices for 2023. 22 The US drug costs were obtained from the Medicare Part B drug average sales price, as provided by the Centers for Medicare & Medicaid Services. 23 Average body parameters were employed (United States: weight 70 kg; body surface area 2.1/m2; China: weight 65 kg; body surface area 1.72/m2)24,25 to calculate drug dosages. AEs management costs accounted for grade ⩾3 adverse reactions with significantly different rates (⩾4% difference) between treatment arms in the GLOW trial 12 (see Table 1). Costs associated with AE management, follow-up, and palliative care were derived from previously published studies.26 –30 All costs were adjusted to 2023 using the Medical-Care Inflation data set in Tom’s Inflation Calculator. 31
Utility values for PFD and PD were obtained from published studies, with values of 0.797 and 0.577, respectively. 32 Concurrently, our study also considered the disutility resulting from significant AEs, assuming occurrence during the first cycle.33 –35 Both costs and utility were adjusted at a discount rate of 3% 36 and 5% 37 in the United States and China, respectively. The willingness-to-pay (WTP) threshold for the United States was $150,000, as suggested by Sanders et al. 36 According to the China Guidelines for Pharmacoeconomic Evaluations, 37 China’s WTP was set to three times its GDP per capita. China’s GDP per capita in 2023 was US $12,729.3, and the WTP was US $38,188.
The inputs considered in the model are listed in Table 2.22 –30,32 –37
Model inputs parameters in China and the United States.

AEs, adverse events; PD, progressed disease; PFD, progression-free disease.
Sensitivity analysis
To evaluate the robustness of the results, we conducted one-way and probabilistic sensitivity analyses. In the one-way sensitivity analysis, the unit price of zolbetuximab varies depending on the price range of trastuzumab, nivolumab, pembrolizumab, and ramucirumab. The estimated range of other parameters was based on either the published literature or assuming a 20% change from the base-case value (Table 2). The results were presented in the form of tornado diagrams.
In the probabilistic sensitivity analysis, 1000 iterations of Monte Carlo simulation were generated with key parameters being sampled simultaneously from the specified distrubutions as shown in Table 2. Results were presented as cost-effectiveness acceptability curves and incremental cost-effectiveness scatter plots, indicating the probability that each treatment choice was cost-effective at the WTP threshold.
Results
Base-case analysis
The results of base-case analysis for the United States and China are shown in Table 3. For the United States, in comparison with placebo arm, zolbetuximab plus CAPOX treatment provided an additional 0.24 QALYs, with an incremental cost of $196,791.11, which was associated with an ICER of $821,515.65/QALY. For China, the incremental effect of the zolbetuximab arm was 0.23 QALYs compared with the placebo arm, and the incremental cost was $62,822.69, leading to the ICER of $273,568.01/QALY.
The results of the base-case analysis.

CAPOX, capecitabine plus oxaliplatin; ICER, incremental cost-effectiveness ratio; P + C, placebo + CAPOX; QALYs, quality-adjusted life-years; Zol + C, zolbetuximab plus CAPOX.
Sensitivity analysis
One-way sensitivity analysis revealed that the cost of zolbetuximab, the utility of PFD, and the body surface area were associated with model outcomes of both the United States and China. The remaining parameters, such as the discount rate and proportion of ramucirumab, had only moderate or low associations with the outcome. Nevertheless, none of the variables could reduce the ICER values below the WTP thresholds for the United States and China (Figure 2).

Tornado diagram for one-way sensitivity analyses in the United States (a) and China (b).
The results of the probabilistic sensitivity analysis are shown in Figure 3 and Supplemental eFigure 2. The cost-effectiveness acceptability curve at a WTP threshold of $150,000/QALY showed a nearly 0% probability of zolbetuximab plus CAPOX (compared to placebo plus CAPOX) as a cost-effective strategy in the United States. Similarly, for patients in China, the probability that zolbetuximab plus CAPOX is a cost-effective option at the WTP threshold of $38,188 is 0%.

Cost-effectiveness acceptability curve for the United States and China.
Given the significant influence of the cost of zolbetuximab on ICER, we repeatedly calculated the acceptable probability of zolbetuximab by continuously reducing the price of zolbetuximab, as shown in Table 4 and Figure 4. For zolbetuximab plus CAPOX treatment to be cost-effective, that is, acceptable with a probability of >50%, the assumed price of zolbetuximab needed to be reduced by 83.37% in the United States and 82.25% in China.
Summary of univariable and probabilistic sensitivity analysis.

Compared with placebo plus CAPOX.
ICER, incremental cost-effectiveness ratios; QALYs, quality-adjusted life-years.

Cost-effectiveness acceptability curve of different zolbetuximab cost in the United States and the China.
Discussion
Zolbetuximab has garnered significant attention for treating advanced CLDN18.2-positive, HER2-negative G/GEJ adenocarcinoma. A recent meta-analysis demonstrated that zolbetuximab plus chemotherapy significantly improved PFS (hazard ratio (HR) 0.64; 95% confidence interval (CI) 0.49–0.84; p < 0.01) and OS (HR 0.64; 95% CI 0.49–0.84; p < 0.01) compared to chemotherapy alone. 38 These clinical benefits suggest that zolbetuximab holds promise for patients with advanced CLDN18.2-positive GC/GEJ cancer. However, the rising costs of new cancer therapies have created a significant challenge for healthcare system sustainability. As highlighted in previous studies,39,40 a comprehensive cost-effectiveness analysis is essential to inform policy decisions and guide the optimal allocation of healthcare resources. Therefore, it is necessary to evaluate the economics of zolbetuximab use. Although zolbetuximab is not yet available on the market, our analysis provides critical insights for healthcare decision-makers and could serve as a reference for pricing discussions. Given the different healthcare environments, we conducted economic evaluations from the perspectives of the U.S. and Chinese healthcare systems.
Our study found that the ICER comparing zolbetuximab with placebo was $196,791.11 per QALY gained in the United States and $62,822.69 per QALY in China, both exceeding the respective WTP thresholds of $150,000/QALY in the United States and $38,1880/QALY in China. According to cost data, in the United States, costs for drugs, management of AEs, palliative care, etc., are several times higher than those in China. That revealed why the ICER in the United States was more significant than in China—and further illustrated the differences between developed and developing countries’ national conditions and medical environments.
One-way sensitivity analysis showed that the cost of zolbetuximab, the utility value of PFD, and body surface area significantly affected the model results but could not reduce the ICER below WTP in both the United States and China, confirming the robustness of our model. Probability sensitivity analysis showed that the probability of zolbetuximab plus CAPOX being economical at the WTP thresholds of $150,000 and $38,188 was 0%. Zolbetuximab plus CAPOX would only be cost-effective if the price of zolbetuximab (100 mg) were reduced to $367.7 in the United States and $110.8 in China.
Although the price of zolbetuximab is unknown, previous research on its cost-effectiveness in treating advanced unresectable or metastatic G/GEJ adenocarcinoma, based on the SPOTLIGHT trial, 41 provides some insights. Huang et al. 42 reported an ICER of $185,353.28/QALY for zolbetuximab plus mFOLFOX6 compared to mFOLFOX6, exceeding China’s WTP threshold, which is consistent with our findings. However, Huang et al.’s study had limitations, including using nivolumab’s price as a reference for zolbetuximab. This may be inappropriate, as nivolumab likely has a higher benefit in specific indications, leading to a higher price. Moreover, conducting cost-effectiveness evaluations solely from a developing country’s perspective may underestimate the cost-effectiveness of zolbetuximab in broader contexts.
To our knowledge, this study is the first to assess the cost-effectiveness of zolbetuximab plus CAPOX as a first-line treatment for patients with CLDN18.2-positive, HER2-negative advanced G/GEJ adenocarcinoma. Zolbetuximab is currently under regulatory review for marketing approval in China and the United States, and our results provide an economic reference for post-marketing price negotiations. Considering the U.S. and Chinese healthcare systems, this study highlights the differences between high- and middle-income countries regarding national conditions and healthcare environments. The above results indicated that, at the currently assumed prices, zolbetuximab plus CAPOX is not a cost-effective first-line treatment strategy for patients with CLDN18.2-positive, HER2-negative advanced G/GEJ adenocarcinoma in the United States and China. However, the actual price of zolbetuximab is still unknown, so our findings should serve as a reference for future price negotiations rather than a basis for restricting its use.
In our study, the relatively small clinical benefit between the two treatment groups (1.41 months for PFS and 2.23 months for OS) also contributes to the limited cost-effectiveness. Expensive antineoplastic drugs are also critical in making treatment regimens less economical. As our previous study on atezolizumab plus bevacizumab and chemotherapy (CBA) versus bevacizumab and chemotherapy (CB) for U.S. patients with cervical cancer demonstrated, price reductions of up to 56.6% for atezolizumab are necessary for CBA to become cost-effective. 43 Beyond price reductions, other approaches—such as optimizing dosing regimens or targeting specific subgroups—can also enhance cost-effectiveness. This is further supported by Cao et al. 44 ’s findings that nivolumab plus ipilimumab was more cost-effective in treating advanced esophageal squamous cell carcinoma in patients with PD-L1 expression ⩾1% (subgroup). Moreover, the WTP threshold and other related medical costs (such as examination fees and AE management) also affect the cost-effectiveness of treatment regimens. Therefore, in actual clinical practice, improving the cost-effectiveness of treatment strategies requires a comprehensive consideration of multiple factors.
There are several limitations in the analysis. First and most importantly, due to the lack of a price for zolbetuximab, we referred to the median cost of several other targeted drugs for GC, which makes our results unconvincing. However, the results of the sensitivity analyses suggest that a substantial adjustment in the price of zolbetuximab would not change our conclusions. Second, much of the cost data used in the study were taken from the published literature and may differ from the actual data; we adjusted the cost data to 2023 prices, thereby increasing the study’s credibility. Finally, due to the lack of detailed information on subsequent anticancer regimens, we assumed postprogression treatment based on information provided by GLOW and NCCN recommendations. This assumption may differ slightly from actual treatment in the real world.
Conclusion
Zolbetuximab plus CAPOX may be a cost-effective option for patients with locally advanced, unresectable, or metastatic G/GEJ adenocarcinoma when the price of zolbetuximab reduced by 83.37% ($367.7/100 mg) in the United States and 82.25% ($110.8/100 mg) in China. These findings may provide some economic guidance for postmarketing price negotiations for zolbetuximab.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848241297052 – Supplemental material for First-line treatment with zolbetuximab plus CAPOX for ClDN18.2-positive gastric or gastroesophageal junction adenocarcinoma: a cost-effectiveness analysis
Supplemental material, sj-pdf-1-tag-10.1177_17562848241297052 for First-line treatment with zolbetuximab plus CAPOX for ClDN18.2-positive gastric or gastroesophageal junction adenocarcinoma: a cost-effectiveness analysis by Jianying Lei, Jiahao Zhang, Caicong You, Wu Fu, Maobai Liu and Na Li in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
