Abstract
Background:
Tofacitinib is an oral, small molecule Janus kinase inhibitor for the treatment of ulcerative colitis (UC). Here, we report steroid-free efficacy and safety with tofacitinib among patients with UC who received corticosteroids at baseline of the maintenance study (OCTAVE Sustain).
Methods:
This analysis included patients with clinical response following OCTAVE Induction 1 and 2 who were re-randomized to receive placebo, or tofacitinib 5 or 10 mg twice daily (b.d.), in OCTAVE Sustain for 52 weeks and were receiving corticosteroids at OCTAVE Sustain baseline. Corticosteroid tapering was mandatory during OCTAVE Sustain. Rates of steroid-free remission, endoscopic improvement, and clinical response were assessed, stratified by baseline characteristics. Adverse events (AEs) were stratified by treatment and steroid-free remission status.
Results:
Overall, 289/593 patients had corticosteroid use at OCTAVE Sustain baseline. At week 52, steroid-free remission, endoscopic improvement, and clinical response rates were 10.9%, 11.9%, and 17.8% among patients receiving placebo, 27.7%, 29.7%, and 40.6% among patients receiving tofacitinib 5 mg b.d., and 27.6%, 29.9%, and 43.7% among patients receiving tofacitinib 10 mg b.d., respectively (non-responder imputation; all p < 0.05 tofacitinib 5 or 10 mg b.d. versus placebo). Discontinuations due to AEs were lower among patients with steroid-free remission versus without. AEs of special interest were infrequent.
Conclusion:
For patients with baseline corticosteroid use in OCTAVE Sustain, the odds of achieving steroid-free efficacy endpoints were significantly higher for tofacitinib versus placebo, irrespective of tofacitinib dose. There were no apparent differences in AEs of special interest by steroid-free remission status.
ClinicalTrials.gov: NCT01458574
Introduction
Ulcerative colitis (UC) is a chronic disease characterized by inflammation of the colon and follows a relapsing and remitting or chronic active course. 1 Management of UC requires therapy to both induce and maintain remission, with the goal of maintenance therapy being to achieve long-term, steroid-free remission, defined clinically and endoscopically.2,3 Furthermore, steroid-free remission is considered to be the most important endpoint for patients with UC. 2
While oral systemic corticosteroids are often used as induction therapy for patients with UC, 3 their lack of efficacy as maintenance therapy and risk of adverse events (AEs) mean they are not recommended as a therapeutic option for maintenance of remission.3,4 Moreover, expert guidance from the American College of Gastroenterology recommends a general reduction in corticosteroid use in the management of UC. 3 Despite this, corticosteroids are often utilized inappropriately. 5 A 20-year national population-based study analyzing trends in prescribing for inflammatory bowel disease in the United Kingdom reported significant increases over time in oral corticosteroid use and very prolonged corticosteroid exposure (>6 months). 6 A similar trend has also been described in the United States, where prolonged corticosteroid exposure (⩾120 days of continuous use) increased by 27% over a 7-year period in patients with UC. 7
The AE profile of corticosteroids is well known; immunocompromised patients who receive corticosteroids are at increased risk of serious or opportunistic infections.8,9 Therefore, maintaining remission in the absence of corticosteroids is important in managing patients with inflammatory bowel disease.
Tofacitinib is an oral, small molecule Janus kinase inhibitor for the treatment of UC. The efficacy and safety of tofacitinib have been evaluated in patients with moderately to severely active UC in three phase III studies. 10 Tofacitinib has previously demonstrated significant induction and maintenance efficacy compared with placebo, regardless of baseline oral corticosteroid use.10,11 The rate of steroid-free remission among patients receiving corticosteroids at baseline of OCTAVE Sustain was reported previously in the supplementary information of Sandborn et al.; 10 here, we report additional information to understand the steroid-sparing efficacy and safety of tofacitinib in more detail, including analysis of other steroid-free efficacy endpoints, subgroup analyses, and safety outcomes.
In this post hoc analysis, we evaluated rates of steroid-free efficacy endpoints – including remission, endoscopic improvement, and clinical response – and safety outcomes, among patients with moderately to severely active UC who entered OCTAVE Sustain and were receiving corticosteroids at baseline of that study. Furthermore, steroid-free efficacy outcomes were stratified by prior tumor necrosis factor inhibitor (TNFi) failure status and prior immunosuppressant failure status (both at baseline of OCTAVE Induction 1 and 2), and C-reactive protein (CRP), total Mayo score and corticosteroid dose, at baseline of OCTAVE Sustain.
Methods
Patients and study design
Adult patients with moderately to severely active UC (total Mayo score ⩾6 with a rectal bleeding subscore ⩾1 and an endoscopic subscore ⩾2) who had failed or were intolerant to oral corticosteroids, azathioprine, 6-mercaptopurine, infliximab, or adalimumab were eligible to enter the OCTAVE program. 10 OCTAVE Sustain (NCT01458574) was a 52-week, phase III, randomized, double-blind, placebo-controlled trial that evaluated the efficacy and safety of tofacitinib as maintenance therapy for patients with UC. Patients who completed the 8-week OCTAVE Induction 1 and 2 (NCT01465763 and NCT01458951) studies and achieved clinical response (defined as a decrease from induction study baseline total Mayo score of ⩾3 points and ⩾30%, plus a decrease in rectal bleeding subscore of ⩾1 point or an absolute rectal bleeding subscore of 0 or 1) after receiving placebo, or tofacitinib 10 or 15 mg twice daily (b.d.), were eligible to enter OCTAVE Sustain and were re-randomized to receive placebo, tofacitinib 5 mg b.d., or tofacitinib 10 mg b.d. The tofacitinib 15 mg b.d. dose in OCTAVE Induction 1 and 2 was subsequently discontinued following a protocol amendment.
Oral corticosteroids (prednisone-equivalent up to 25 mg/day; budesonide up to 9 mg/day) were permitted during induction studies, provided that the dose remained stable for at least 2 weeks prior to baseline and throughout the induction study period. Corticosteroid tapering was mandatory at the beginning of OCTAVE Sustain. For patients receiving prednisone or equivalent, the tapering protocol was to decrease the daily dose at a rate of 5 mg/week until the dose reached 20 mg/day, then to decrease by 2.5–5 mg/week until the dose reached 10 mg/day, and finally to decrease by 2.5 mg/week until the dose reached 0 mg. For patients receiving budesonide, the tapering protocol was to decrease the daily dose at a rate of 3 mg per 3 weeks until the dose reached 0 mg (made possible due to the availability of formulations from 3 mg). Patients with worsening UC symptoms attributed to corticosteroid tapering were permitted to increase their corticosteroid dosage once (reverting back to previous weeks’ dosage) during the maintenance study and then resume corticosteroid tapering to achieve steroid-free status.
Only patients who received oral corticosteroids at baseline of OCTAVE Sustain are included in these analyses, although one patient had non-oral corticosteroid use at baseline and was included as a protocol deviation. Concomitant immunosuppressant or TNFi therapy was prohibited throughout induction and maintenance studies (immunosuppressant washout period: within 2 weeks of baseline in OCTAVE induction studies; TNFi washout period: within 8 weeks of baseline in OCTAVE induction studies). Immunosuppressants included non-biologic agents, such as azathioprine, 6-mercaptopurine, methotrexate, cyclosporine, and tacrolimus.
The full details of OCTAVE Induction 1 and 2, and OCTAVE Sustain, have been reported previously. 10 This is a post hoc analysis of a parallel-group, randomized clinical trial 10 which followed CONSORT 2010 reporting guidelines. A completed CONSORT checklist for this analysis can be found in the supplementary material. A patient disposition diagram, adapted for patients with corticosteroid use at baseline of OCTAVE Sustain, is shown in Figure S1.
Efficacy assessments
The following steroid-free efficacy endpoints were assessed in OCTAVE Sustain among patients with corticosteroid use at baseline of OCTAVE Sustain: remission, endoscopic improvement, and clinical response (all at week 24, week 52, or sustained at both week 24 and week 52).
Steroid-free was defined as not requiring any treatment with corticosteroids for ⩾4 weeks prior to the visit. Remission was defined as a total Mayo score of ⩽2 with no individual subscore >1, and a rectal bleeding subscore of 0. Endoscopic improvement (defined as mucosal healing in the original OCTAVE Sustain protocol) 10 was defined as a Mayo endoscopic subscore of 0 or 1. Sustained efficacy was defined as having achieved the defined efficacy endpoint at both week 24 and week 52.
All patients with baseline corticosteroid use were further stratified by the following characteristics: prior TNFi failure (yes or no) and prior immunosuppressant failure (yes or no), both at baseline of OCTAVE Induction 1 and 2; and CRP (⩽3 or >3 mg/L), total Mayo score (<3 or ⩾3), and corticosteroid dose (<15 mg/day or ⩾15 mg/day), all at baseline of OCTAVE Sustain. Efficacy outcomes were based on centrally read endoscopic subscores.
Safety assessments
Safety outcomes were assessed in all patients who received corticosteroids at baseline of OCTAVE Sustain. Proportions and incidence rates (IRs; unique patients with events per 100 patient-years of exposure) of AEs, including AEs of special interest, were reported. Safety outcomes were stratified by treatment and by steroid-free remission status at week 52.
Statistical analyses
The full analysis set is defined as all patients randomized to receive placebo, tofacitinib 5 mg b.d., or tofacitinib 10 mg b.d. in OCTAVE Sustain who received corticosteroids at baseline of OCTAVE Sustain. Non-responder imputation (NRI) was applied, meaning patients with missing values were treated as non-responders. Baseline demographics and clinical characteristics were summarized descriptively based on achievement of steroid-free efficacy endpoints at week 24 or week 52.
For comparison of tofacitinib 5 mg b.d. versus placebo, tofacitinib 10 mg b.d. versus placebo, and tofacitinib 10 mg b.d. versus tofacitinib 5 mg b.d., for steroid-free endpoints of remission, endoscopic improvement, and clinical response, p values were based on logistic regression.
The following explanatory variables were each individually evaluated using logistic regression, with treatment in the model, for association with steroid-free remission or endoscopic improvement at week 52 among patients who received corticosteroids at baseline of OCTAVE Sustain (NRI): prior TNFi failure (yes or no) at baseline of OCTAVE Induction 1 and 2, prior immunosuppressant failure (yes or no) at baseline of OCTAVE Induction 1 and 2, remission status at baseline of OCTAVE Sustain (yes or no), total Mayo score at baseline of OCTAVE Sustain (<3 versus ⩾3), extent of disease (left-sided colitis; extensive colitis/pancolitis; proctosigmoiditis), corticosteroid dose received at baseline of OCTAVE Sustain [<15 mg/day; ⩾15 mg/day; other (defined as those patients with missing doses and patients whose dose could not be converted to prednisone-equivalent dose)], and CRP (⩽3 versus >3 mg/L) at baseline of OCTAVE Sustain. Odds ratios (ORs) and 95% confidence intervals (CIs) from the logistic regression analysis were reported for the association of factors with steroid-free remission or endoscopic improvement at week 52 of OCTAVE Sustain.
These analyses were post hoc for exploratory purposes only, and no multiplicity adjustment was performed.
Ethical approval
All studies were conducted in compliance with the Declaration of Helsinki and the International Conference on Harmonization Good Clinical Practice Guidelines and were approved by the Institutional Review Boards and/or independent ethics committees at each of the investigational centers participating in the studies, or a central Institutional Review Board. All patients provided written informed consent. All authors had access to the study data and reviewed and approved the final manuscript.
Results
Patients
OCTAVE Sustain included 593 patients who had initially received placebo, tofacitinib 10 mg b.d. or tofacitinib 15 mg b.d. (a dose that was subsequently discontinued following a protocol amendment), induction therapy, and had clinical response after 8 weeks of treatment before being re-randomized to receive placebo (N = 198), tofacitinib 5 mg b.d. (N = 198), or tofacitinib 10 mg b.d. (N = 197).
Among all 593 patients entering OCTAVE Sustain, the mean total Mayo score was 3.3 [standard deviation (SD) = 1.8], 30.2% of patients were in remission and 49.7% of patients had endoscopic improvement at baseline of OCTAVE Sustain. Furthermore, 44.7% had prior TNFi failure and 69.6% had prior immunosuppressant failure at baseline of OCTAVE Induction 1 and 2. Overall, 289 (48.7%) patients had corticosteroid use at baseline of OCTAVE Sustain: 277 patients had prednisone use (or equivalent oral systemic corticosteroid); 11 patients had budesonide use; and 1 patient (in the placebo group) had non-oral corticosteroid use at baseline and was included as a protocol deviation. On day 1 of OCTAVE Sustain, the mean corticosteroid dose (prednisone-equivalent) received was 15.8 mg/day (SD = 6.2), 14.9 mg/day (SD = 6.2), and 14.4 mg/day (SD = 6.0) in the placebo, tofacitinib 5 mg b.d., and tofacitinib 10 mg b.d. groups, respectively. Overall, the number of patients in each treatment group who received corticosteroids at baseline of OCTAVE Sustain were 101 (51.0%), 101 (51.0%), and 87 (44.2%), respectively.
Baseline clinical characteristics among patients with and without baseline corticosteroid use
Baseline demographics and clinical characteristics among patients with and without corticosteroid use at baseline of OCTAVE Sustain are shown in Table 1. Across all treatment groups, the proportion of patients with prior TNFi or immunosuppressant failure was slightly higher among patients receiving corticosteroids at baseline of OCTAVE Sustain, compared with those who did not (Table 1).
Baseline demographics and clinical characteristics among patients with and without corticosteroid use at baseline of OCTAVE Sustain (FAS).

b.d., twice daily; BMI, body mass index; CRP, C-reactive protein; FAS, full analysis set; N, number of patients in the specified category with non-missing data; n, number of patients with the specified characteristic within the given category; SD, standard deviation; TNFi, tumor necrosis factor inhibitor.
Data are from baseline of OCTAVE Sustain, unless otherwise specified.
Analysis set includes patients who received tofacitinib 15 mg b.d. in OCTAVE Induction 1 and 2, a dose that was subsequently discontinued following a protocol amendment.
Data at baseline of induction studies.
Baseline demographics and clinical characteristics among patients who received corticosteroids at baseline of OCTAVE Sustain were also stratified by steroid-free remission status at week 24 or week 52 (Table S1).
Efficacy
Proportion of patients with baseline corticosteroid use who achieved steroid-free efficacy endpoints
At week 24 or week 52 of OCTAVE Sustain, there was a significant treatment effect for tofacitinib 5 and 10 mg b.d. versus placebo for steroid-free remission (all p < 0.05), endoscopic improvement (all p < 0.01), and clinical response (all p < 0.001) (Figure 1(a)–(c)). Furthermore, there was a significant effect for tofacitinib 10 mg b.d. versus placebo for sustained steroid-free remission (p < 0.05) and for tofacitinib 5 and 10 mg b.d. versus placebo for sustained steroid-free endoscopic improvement (both p < 0.01) and clinical response (both p ⩽ 0.001) (Figure 1(a)–(c)).
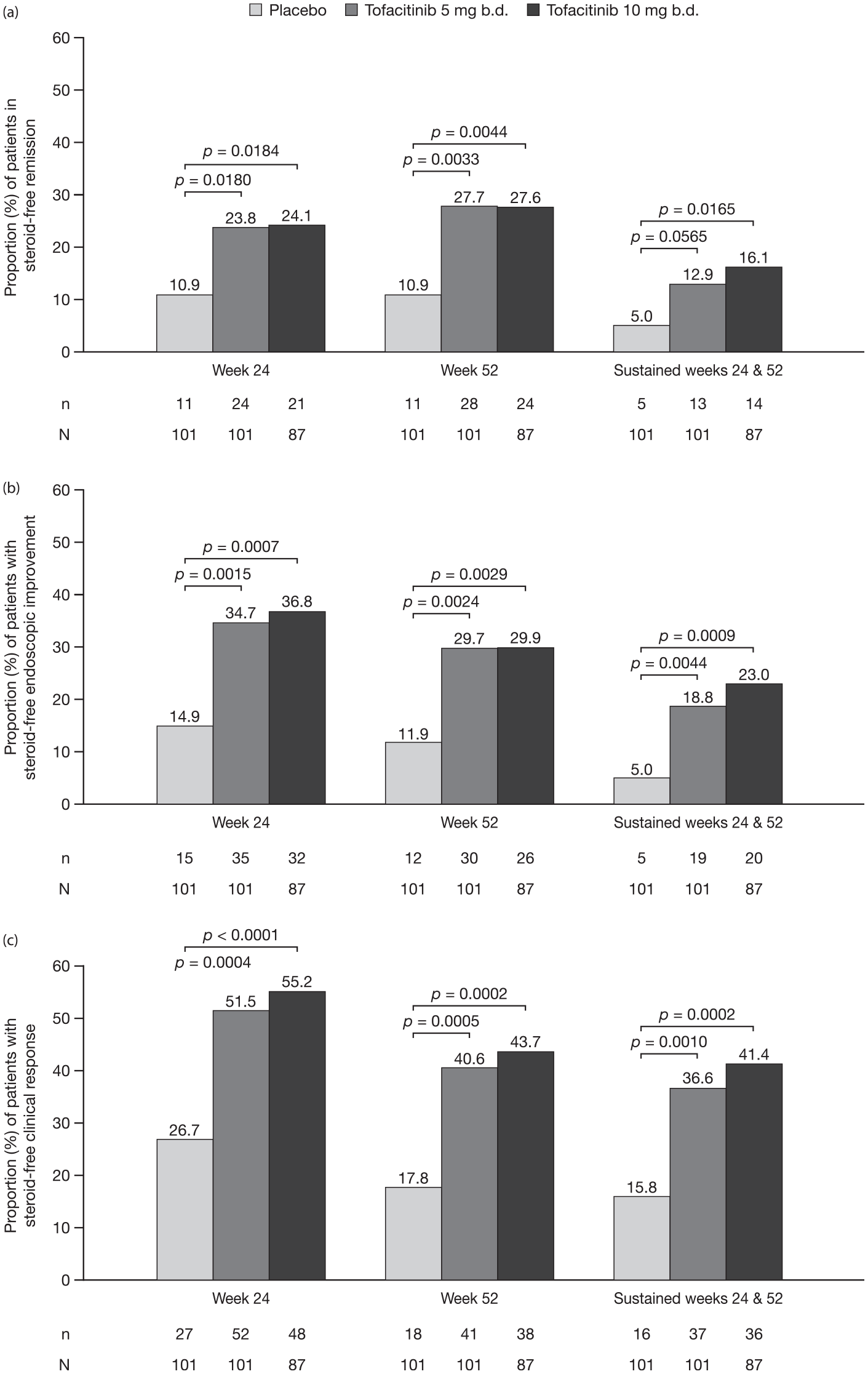
Proportion of patients with baseline corticosteroid use who achieved steroid-free (a) remission, (b) endoscopic improvement, and (c) clinical response at week 24, week 52, and at both time points (sustained) in OCTAVE Sustain (FAS,a NRI).
A similar proportion of tofacitinib-treated patients were in steroid-free remission at week 52, compared with week 24, while fewer tofacitinib-treated patients achieved endoscopic improvement or clinical response at week 52, compared with week 24 (Figure 1(a)–(c)). A lower proportion of patients had sustained steroid-free remission or endoscopic improvement at week 24 and week 52, compared with the proportion of patients with these efficacy outcomes at either week 24 or week 52 (Figure 1(a) and (b)). The proportions of patients with sustained clinical response at week 24 and week 52 were similar to those with clinical response at week 52 (Figure 1(c)).
Proportion of patients with baseline corticosteroid use who achieved steroid-free efficacy endpoints, stratified by prior TNFi failure
Among patients with no prior TNFi failure, a higher proportion of tofacitinib-treated patients were in steroid-free remission at week 24 or week 52, and sustained steroid-free remission at week 24 and week 52 versus placebo-treated patients (Figure 2(a)).

Proportion of patients with baseline corticosteroid use who achieved steroid-free remission at week 24, week 52, and at both time points (sustained) in OCTAVE Sustain, stratified by (a) prior TNFi failure status, (b) prior immunosuppressant failure status, and (c) CRP at baseline of OCTAVE Sustain (FAS,a NRI).
In general, a lower proportion of tofacitinib-treated patients with prior TNFi failure achieved steroid-free remission versus those who had not previously failed TNFi, regardless of tofacitinib dose received (Figure 2(a)).
In general, similar trends were observed for the proportion of patients with steroid-free endoscopic improvement and clinical response with and without prior TNFi failure (Figures S2a and S3a).
Proportion of patients with baseline corticosteroid use who achieved steroid-free efficacy endpoints, stratified by prior immunosuppressant failure
A higher proportion of tofacitinib-treated patients achieved sustained steroid-free remission, and steroid-free remission, endoscopic improvement, and clinical response at week 24 or week 52 versus placebo-treated patients, regardless of prior immunosuppressant failure (Figure 2(b); Figures S2b and S3b).
Proportion of patients with baseline corticosteroid use who achieved steroid-free efficacy endpoints, stratified by CRP at baseline of OCTAVE Sustain
Among patients with baseline corticosteroid use and CRP ⩽3 mg/L at baseline of OCTAVE Sustain, higher proportions of tofacitinib-treated patients achieved steroid-free remission, endoscopic improvement, and clinical response at week 24, week 52, and at both time points versus placebo-treated patients, regardless of tofacitinib dose (Figure 2(c); Figures S2c and S3c).
Proportion of patients with baseline corticosteroid use who achieved steroid-free efficacy endpoints, stratified by Mayo score or corticosteroid dose at baseline of OCTAVE Sustain
A higher proportion of patients with Mayo score <3 at baseline of OCTAVE Sustain achieved remission and endoscopic improvement, but not clinical response, at week 24, week 52, and at both time points versus patients with Mayo score ⩾3 (Figures S2d, S3d and S4a). The proportions of patients with baseline corticosteroid use who achieved steroid-free remission and endoscopic improvement, but not clinical response, were generally similar between categories (<15 mg/day versus ⩾15 mg/day) when stratified by corticosteroid dose at baseline of OCTAVE Sustain (Figures S2e, S3e, and S4b).
Association between explanatory variables and achievement of steroid-free remission or endoscopic improvement at week 52
In the univariate logistic regression analysis, having prior immunosuppressant failure was significantly associated with lower odds of achieving steroid-free remission or endoscopic improvement at week 52 of OCTAVE Sustain (Figure 3). Furthermore, lower CRP (⩽3 versus >3 mg/L) at baseline of OCTAVE Sustain was significantly associated with higher odds of achieving steroid-free remission or endoscopic improvement at week 52 of OCTAVE Sustain. There was insufficient evidence to conclude that prior TNFi failure or extent of disease (both at baseline of OCTAVE Induction 1 and 2), remission status, total Mayo score (<3 versus ⩾ 3), or corticosteroid dose (<15 mg/day versus ⩾15 mg/day and other versus ⩾15 mg/day) (all at baseline of OCTAVE Sustain) were associated with achieving steroid-free remission or endoscopic improvement at week 52 (Figure 3).

Univariate logistic regression results for assessment of association of baseline factors with (a) steroid-free remission and (b) steroid-free endoscopic improvement at week 52 of OCTAVE Sustain (FAS,a NRI).
Safety
A summary of safety stratified by steroid-free remission status at week 52 is presented in Table 2.
Summary of safety and AEs in patients who received corticosteroids at baseline of OCTAVE Sustain, by week 52 steroid-free remission status and treatment group.
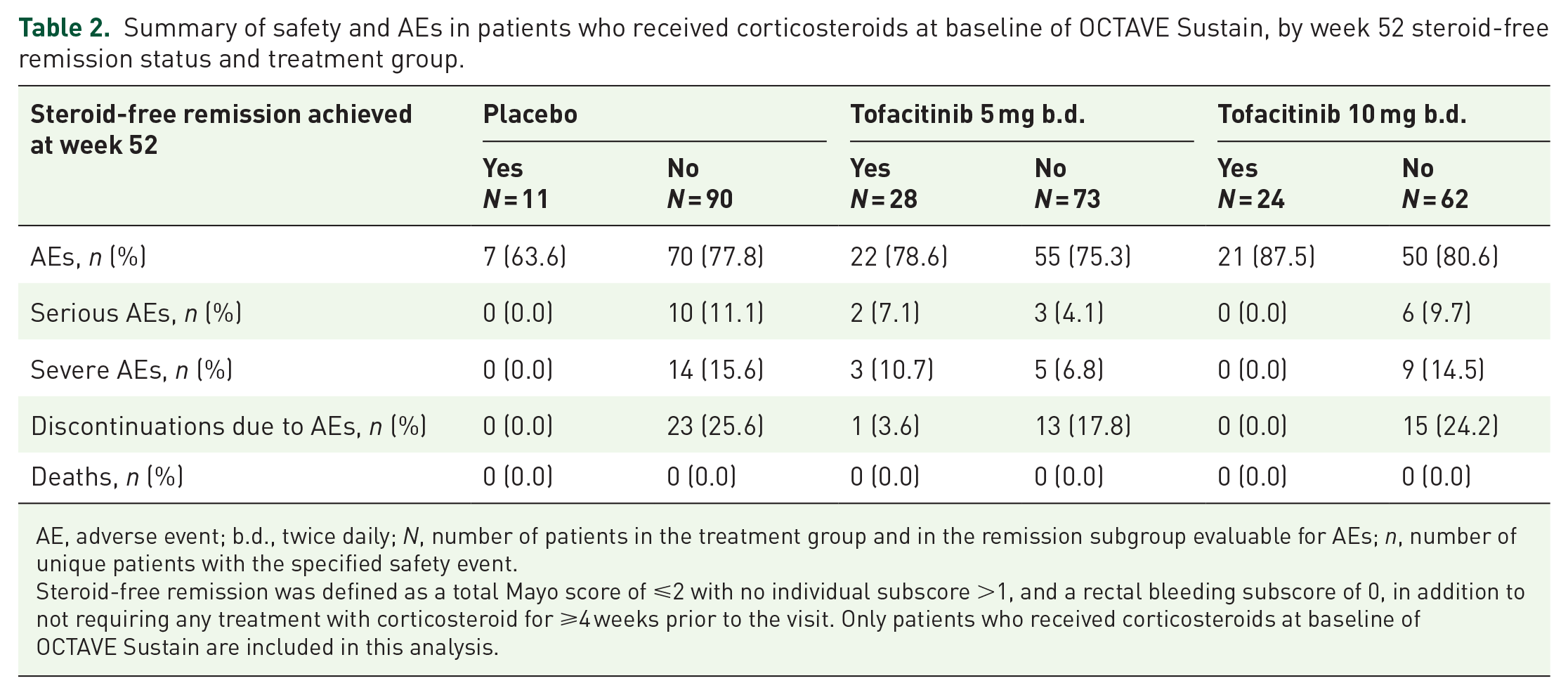
AE, adverse event; b.d., twice daily; N, number of patients in the treatment group and in the remission subgroup evaluable for AEs; n, number of unique patients with the specified safety event.
Steroid-free remission was defined as a total Mayo score of ⩽2 with no individual subscore >1, and a rectal bleeding subscore of 0, in addition to not requiring any treatment with corticosteroid for ⩾4 weeks prior to the visit. Only patients who received corticosteroids at baseline of OCTAVE Sustain are included in this analysis.
The proportions of tofacitinib-treated patients with AEs were similar among patients with and without steroid-free remission at week 52 and were similar across treatment groups (Table 2). The most frequent serious or severe AE among placebo-treated patients who did not achieve steroid-free remission at week 52 was exacerbation or worsening of UC. Serious or severe AEs that occurred in patients who received tofacitinib and achieved steroid-free remission were chest pain, myocardial infarction, gastrointestinal bleeding, influenza, pneumonia, lumbar discopathy, and muscular pain (all in the tofacitinib 5 mg b.d. group). The proportion of patients who discontinued tofacitinib therapy due to AEs was lower among patients who achieved steroid-free remission at week 52, compared with patients who failed to achieve this endpoint (Table 2).
Among patients who received corticosteroids at baseline of OCTAVE Sustain, no deaths, malignancies (excluding non-melanoma skin cancer), or deep vein thrombosis events occurred in any treatment group. All other AEs of special interest are detailed in Table 3.
Summary of AEs of special interest in patients who received corticosteroids at baseline of OCTAVE Sustain, by week 52 steroid-free remission status and treatment group.

AE, adverse event; b.d., twice daily; CI, confidence interval; IR, incidence rate (unique patients with events per 100 PY of exposure); MACE, major adverse cardiovascular events; N, number of patients in the treatment group; n, number of unique patients with the specified safety event; NMSC, non-melanoma skin cancer; PE, pulmonary embolism; PY, patient-years.
Steroid-free remission was defined as a total Mayo score of ⩽2 with no individual subscore >1, and a rectal bleeding subscore of 0, in addition to not requiring any treatment with corticosteroid for ⩾4 weeks prior to the visit. Only patients who received corticosteroids at baseline of OCTAVE Sustain are included in this analysis.
No deaths, malignancies (excluding NMSC) or deep vein thrombosis events occurred in any treatment group.
Events that occurred >28 days after the last dose of study drug were excluded.
Excludes tuberculosis and herpes zoster with two adjacent dermatomes.
All events, including those outside the 28-day risk period, were included.
Adjudicated events.
Among all patients with corticosteroid use at baseline of OCTAVE Sustain who received tofacitinib 10 mg b.d. maintenance therapy, with or without steroid-free remission at week 52, herpes zoster IRs were 13.74; 95% CI 2.38–40.14 and 8.96; 95% CI 1.85–26.18, respectively. Corresponding IRs of opportunistic infection (including herpes zoster opportunistic infection; excluding tuberculosis and herpes zoster with two adjacent dermatomes) were 4.25; 95% CI 0.11–23.69 and 5.90; 95% CI 0.71–21.30 (Table 3). No events of serious herpes zoster were reported in OCTAVE Sustain. 10
Discussion
Tofacitinib has previously demonstrated significant induction and maintenance efficacy compared with placebo, regardless of baseline oral corticosteroid use.10,11 This study presents post hoc analyses of the OCTAVE Sustain maintenance study assessing the rates of steroid-free remission, endoscopic improvement, and clinical response for tofacitinib versus placebo among patients with moderately to severely active UC who were receiving corticosteroids at baseline of OCTAVE Sustain.
Among patients with corticosteroid use at baseline of OCTAVE Sustain who received tofacitinib 5 or 10 mg b.d., a greater proportion achieved steroid-free remission, endoscopic improvement, and clinical response at week 24 or week 52, compared with patients who received placebo. Rates were generally similar between patients who received tofacitinib 5 and 10 mg b.d.; therefore, among patients receiving corticosteroids at baseline of OCTAVE Sustain, tofacitinib dose did not appear to impact achievement of steroid-free remission, endoscopic improvement, and clinical response with tofacitinib maintenance therapy.
Logistic regression analyses demonstrated that prior immunosuppressant failure was significantly associated with a lower likelihood of achieving steroid-free remission or endoscopic improvement at week 52, and lower CRP (⩽3 versus >3 mg/L) at baseline of OCTAVE Sustain was significantly associated with a higher likelihood of achieving steroid-free remission or endoscopic improvement at week 52. Other baseline factors, including corticosteroid dose, remission status, total Mayo score (all at baseline of OCTAVE Sustain), prior TNFi failure, or extent of disease (at baseline of OCTAVE Induction 1 and 2), were not significantly associated with steroid-free remission or endoscopic improvement at week 52. CRP is a marker of systemic inflammation that has been shown to correlate with the extent of UC at diagnosis, 12 and high CRP is a risk factor for poor outcomes, including colectomy; 13 therefore, lower CRP at baseline of OCTAVE Sustain may indicate less inflammation following completion of OCTAVE Induction 1 and 2. Prior immunosuppressant failure as a risk factor may identify patients with more refractory disease, whereby systemic immune modulation with immunosuppressants or Janus kinase inhibitors may fail to completely resolve inflammation. In contrast, prior TNFi failure was not identified as a significant risk factor in these analyses, despite it often being considered a marker of more refractory disease.
While there was insufficient evidence from the logistic regression analysis to conclude that prior TNFi failure was associated with steroid-free remission or endoscopic improvement at week 52, prior TNFi failure had a non-significant (p = 0.0560) trend toward a lower likelihood of achieving steroid-free remission. Furthermore, among patients who entered the tofacitinib maintenance study, OCTAVE Sustain, and were receiving corticosteroids at baseline, those with prior TNFi failure had generally lower rates of steroid-free remission, endoscopic improvement, and clinical response at week 52, compared with no prior TNFi failure.
Patients who have failed TNFi therapy may be considered more refractory to UC treatment than those who have not.14,15 Tofacitinib is generally effective among patients who have previously failed TNFi therapy; steroid-free remission was previously reported in patients who were in remission at baseline of OCTAVE Sustain, regardless of prior TNFi failure status. 16 It should be noted that in the current study, which reports steroid-free remission among all patients with corticosteroid use at baseline of OCTAVE Sustain, only 28.0% of patients included in the analysis were in remission at baseline.
Recent observations have been made in real-world studies of the effectiveness of tofacitinib, which suggest steroid-free remission can be achieved among patients receiving tofacitinib maintenance therapy in a real-world setting. In a retrospective observational cohort study of 134 patients with UC with and without concomitant corticosteroids at treatment initiation, steroid-free remission (defined as partial Mayo score ⩽1) was observed in 44% of patients at week 26, and was unaffected by prior biologic exposure status. 17 In a real-world French cohort study of 38 patients with UC, refractory to both TNFi and vedolizumab, 34% of patients were in steroid-free remission at week 48. 18 Due to the invasive nature of endoscopy, real-world studies often assess the endpoint of ‘clinical’ remission (based on partial Mayo score) rather than the more stringent endpoint of remission based on total Mayo score assessed in randomized controlled trials such as the OCTAVE trials; therefore, direct comparisons between randomized controlled trials and real-world studies cannot be made. Furthermore, it is important to note that the patients described in these real-world studies did not all receive corticosteroids at baseline, and the majority had prior failure and/or exposure to biologic therapy.17,18
While the proportions of patients experiencing AEs overall were generally similar between patients with and without steroid-free remission at week 52 in all treatment groups, serious and severe AEs were less frequent among patients who achieved steroid-free remission, compared with those who did not, in the placebo and tofacitinib 10 mg b.d. groups. Serious and severe AEs included worsening UC, which was the most frequently occurring serious AE reported in the tofacitinib UC clinical program. 19 Moreover, discontinuations due to AEs were also less frequent among patients who achieved steroid-free remission, compared with those who did not.
In the tofacitinib UC clinical program, serious infections were more frequent among patients who received tofacitinib versus placebo as induction therapy; however, rates were comparable between treatment groups in the Maintenance Cohort and between tofacitinib dose groups in the Overall Cohort. 20 In these post hoc analyses, there were no events of serious infections among patients who achieved steroid-free remission. This is not unexpected, as prolonged systemic corticosteroid use is associated with increased susceptibility to infection among patients with UC. 4
Tofacitinib is a known risk factor for herpes zoster among patients with UC. 19 Although the sample size was small, IRs of herpes zoster and opportunistic infections were higher among patients in the tofacitinib 10 mg b.d. group receiving corticosteroids at baseline of OCTAVE Sustain (IR = 13.74; 95% CI = 2.38–40.14 and IR = 4.25; 95% CI = 0.11–23.69, respectively), compared with IRs reported in the Overall Cohort in the tofacitinib UC clinical program (IR = 4.1; 95% CI = 3.1–5.2 and IR = 1.3; 95% CI = 0.8–2.0, respectively). 19
Among patients with inflammatory bowel disease, corticosteroid use in combination with other immunomodulatory and biologic therapies has been reported to be associated with opportunistic infections. 21 Prior immunosuppressant use was not associated with the development of herpes zoster or opportunistic infections in the tofacitinib UC clinical program. 20 The use of corticosteroids is a well-established risk factor for herpes zoster in patients with inflammatory bowel disease;22–24 therefore, rapid reduction or elimination of corticosteroids is key to minimizing the risk of herpes zoster and opportunistic infections in patients initiating immunomodulatory therapies such as tofacitinib.
A key limitation of this study is the post hoc nature of the analyses, and the study was not designed to evaluate rates of steroid-free remission among specified subgroups of patients. These analyses are for exploratory purposes only and no multiplicity adjustment was performed. Endpoints were evaluated at only two time points during OCTAVE Sustain. There are currently no comparative studies with any other oral therapeutic treatment for UC. Furthermore, data interpretation is limited by small patient numbers. Nevertheless, data are presented from a randomized, placebo-controlled study including endoscopic assessment at both visits, week 24, and week 52.
Overall, these results suggest that a substantial proportion of patients who were receiving corticosteroids at baseline of OCTAVE Sustain achieved steroid-free remission, endoscopic improvement, and clinical response with tofacitinib maintenance therapy.
The findings of this study support and add to a previous analysis of patients with baseline corticosteroid use in OCTAVE Sustain. 10 Patients who achieved steroid-free remission were less likely to discontinue therapy than patients who failed to achieve steroid-free remission. Patients in the tofacitinib 10 mg b.d. group receiving corticosteroids at baseline of OCTAVE Sustain had higher IRs of herpes zoster and opportunistic infections versus the Overall Cohort in the tofacitinib UC clinical program. For patients with baseline corticosteroid use in OCTAVE Sustain, the odds of achieving steroid-free efficacy endpoints were significantly higher for tofacitinib 5 or 10 mg b.d., compared with placebo. Prior TNFi failure status had only a limited effect, which was not associated with significantly lower odds of achieving steroid-free remission at week 52. There were no apparent differences in AEs of special interest by steroid-free remission status.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221090834 – Supplemental material for Corticosteroid-free efficacy and safety outcomes in patients receiving tofacitinib in the OCTAVE Sustain maintenance study
Supplemental material, sj-docx-1-tag-10.1177_17562848221090834 for Corticosteroid-free efficacy and safety outcomes in patients receiving tofacitinib in the OCTAVE Sustain maintenance study by Stephan R. Vavricka, Thomas Greuter, Benjamin L. Cohen, Walter Reinisch, Flavio Steinwurz, Marc Fellmann, Xiang Guo, Nervin Lawendy, Jerome Paulissen and Laurent Peyrin-Biroulet in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848221090834 – Supplemental material for Corticosteroid-free efficacy and safety outcomes in patients receiving tofacitinib in the OCTAVE Sustain maintenance study
Supplemental material, sj-docx-2-tag-10.1177_17562848221090834 for Corticosteroid-free efficacy and safety outcomes in patients receiving tofacitinib in the OCTAVE Sustain maintenance study by Stephan R. Vavricka, Thomas Greuter, Benjamin L. Cohen, Walter Reinisch, Flavio Steinwurz, Marc Fellmann, Xiang Guo, Nervin Lawendy, Jerome Paulissen and Laurent Peyrin-Biroulet in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors would like to thank the patients, investigators, and study teams involved in the OCTAVE trials. These studies were sponsored by Pfizer. Medical writing support, under the guidance of the authors, was provided by Sarah Mancini, PhD, CMC Connect, McCann Health Medical Communications, and was funded by Pfizer Inc, New York, NY, USA in accordance with Good Publication Practice (GPP3) guidelines (Ann Intern Med 2015; 163: 461–464). Editorial support, including submission of this manuscript, was provided by Antoaneta Rashkova, CMC Connect, McCann Health Medical Communications, and was funded by Pfizer Inc, New York, NY, USA. All authors authorized the submission of this manuscript via a third party and approved all declarations of funding, contributions, and conflicting interests. J.P. is an employee of Syneos Health, which was a paid contractor to Pfizer in connection with the development of this manuscript and related statistical analysis. Some of the data in this manuscript were originally presented at the 16th Congress of European Crohn’s and Colitis Organisation, 8–10 July, 2021 and 2a SEBRADII–Semana Brasileira de Doenças Inflamatórias Intestinais, 17–21 August 2021.
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.R.V. has received consulting fees and unrestricted research grants from Abbott, Falk Pharma GmbH, Ferring Pharmaceuticals, Janssen, MSD, Pfizer Inc, Sanofi-Aventis, Takeda, Tillotts, UCB, and Vifor. T.G. has a consultancy contract with Sanofi-Aventis; has received a travel grant from Falk Pharma GmbH and Vifor; and has an unrestricted research grant from Novartis. B.L.C. has served as an advisory board member for and received consulting fees from AbbVie, Celgene-Bristol-Myers Squibb, Pfizer Inc, Sublimity Therapeutics, and TARGET RWE; fees from the CME companies, Cornerstones, and Vindico; and has received speaker fees from AbbVie. W.R. has received research grants from Abbott, AbbVie, AESCA, Centocor, Dr. Falk Pharma, Immundiagnostik, and MSD; lecture fees from Abbott, AbbVie, AESCA, Aptalis, Celltrion, Centocor, Danone, Dr. Falk Pharma, Elan, Ferring Pharmaceuticals, Immundiagnostik, Mitsubishi Tanabe Pharma, MSD, Otsuka, PDL, Pharmacosmos, Schering-Plough, Shire, Takeda, Therakos, Vifor, and Yakult; and consulting fees from Abbott, AbbVie, AESCA, Amgen, AM Pharma, Astellas, AstraZeneca, Avaxia Biologics, Bioclinica, Biogen Idec, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Cellerix, Celltrion, Centocor, ChemoCentryx, Covance, Danone, Dr. Falk Pharma, Elan, Ferring Pharmaceuticals, Galapagos, Genentech, Gilead Sciences, Grünenthal, ICON, Index Pharma, Inova, Janssen, Johnson and Johnson, Kyowa Hakko Kirin Pharma, Lipid Therapeutics, MedImmune, Millennium, Mitsubishi Tanabe Pharma, MSD, Nestlé, Novartis, Ocera, Otsuka, PDL, Pfizer Inc, Pharmacosmos, Procter & Gamble, Prometheus Laboratories, Robarts Clinical Trials, Schering-Plough, Second Genome, SetPoint Medical, Takeda, Therakos, TiGenix, UCB, Vifor, Zyngenia, and 4SC. F.S. has served as an advisory board member for Pfizer Inc and has received consulting and speaker fees from AbbVie, Eurofarma, Ferring Pharmaceuticals, Janssen, Sandoz, Takeda, and UCB. M.F., X.G., and N.L. are employees and shareholders of Pfizer Inc. J.P. is an employee of Syneos Health, which was a paid contractor to Pfizer in connection with the development of this manuscript and related statistical analysis. L.P.-B. has received honoraria from AbbVie, Allergan, Alma, Amgen Biogen, Arena, Boehringer Ingelheim, Celgene, Celltrion, Enterome, Ferring Pharmaceuticals, Genentech, Gilead Sciences, Hikma, Index Pharmaceuticals, Janssen, MSD, Nestlé, Pfizer Inc, Pharmacosmos, Roche, Samsung Bioepis, Sandoz, Sterna, Takeda, and Tillotts; grants from AbbVie, MSD, and Takeda; and is a stockholder of CTMA.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The clinical trials described in this article were sponsored by Pfizer. Funding for medical writing support was provided by Pfizer Inc.
Data sharing statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
