Abstract
Background:
Tofacitinib is a small molecule that inhibits Janus kinase and has been reported to be effective in Western patients with ulcerative colitis (UC). However, the real-life data on tofacitinib in Asian UC patients are limited.
Objective:
To investigate the real-life effectiveness and safety of tofacitinib induction and maintenance treatment in Korean patients with UC.
Design:
This was a retrospective study on patients with UC who received tofacitinib treatment at 12 hospitals in Korea between January 2018 and November 2020.
Methods:
Clinical remission at week 52, defined as a partial Mayo score of ⩽2 with a combined rectal bleeding subscore and stool frequency subscore of ⩽1, was used as the primary outcome. Adverse events (AEs), including herpes zoster and deep vein thrombosis, were also evaluated.
Results:
A total of 148 patients with UC were started on tofacitinib. Clinical remission rates of 60.6%, 54.9%, and 52.8% were reported at weeks 16, 24, and 52, respectively. Clinical response rates of 71.8%, 67.6%, and 59.9% were reported at weeks 16, 24, and 52, respectively. Endoscopic remission rates at weeks 16 and 52 were 52.4% and 30.8% based on the Mayo endoscopic subscore and 20.7% and 15.2% based on the UC endoscopic index of severity (UCEIS), respectively. A higher UCEIS at baseline was negatively associated with clinical response [adjusted odds ratio (aOR): 0.774, p = 0.029] and corticosteroid-free clinical response (aOR: 0.782, p = 0.035) at week 52. AEs occurred in 19 patients (12.8%) and serious AEs in 12 patients (8.1%). Herpes zoster occurred in four patients (2.7%). One patient (0.7%) suffered from deep vein thrombosis.
Conclusions:
Tofacitinib was an effective induction and maintenance treatment with an acceptable safety profile in Korean patients with UC.
Plain language summary
Ulcerative colitis (UC) is an idiopathic, chronic inflammatory disorder of the colonic mucosa that usually presents with bloody diarrhea and abdominal pain. Tofacitinib is a small molecule that inhibits Janus kinase and has been reported to be effective in Western patients with UC. However, real-life data on the effectiveness of tofacitinib in Asian patients with UC are limited. To investigate the real-life effectiveness and safety of tofacitinib treatment in Korean patients with UC, we retrospectively analyzed the data of 148 patients with UC who received tofacitinib treatment at 12 hospitals in Korea between January 2018 and November 2020. Clinical remission (i.e. complete improvement of symptoms) was achieved in 60.6% and 52.8% of patients at weeks 16 and 52, respectively. Endoscopic remission was achieved in 52.4% and 30.8% of patients at weeks 16 and 52, respectively. A higher baseline score of the UC endoscopic index of severity, which is one of the endoscopic indices that evaluate the severity of inflammation of the colon, was negatively associated with clinical response (i.e. partial improvement of symptoms). Adverse events (AEs) including herpes zoster and deep vein thrombosis occurred in 19 patients (12.8%) and serious AEs occurred in 12 patients (8.1%). Our real-life study shows that tofacitinib is a clinically effective treatment for Korean patients with UC, and the incidence of AEs was also similar to those observed in other real-world studies.
Introduction
Ulcerative colitis (UC) is an idiopathic, chronic inflammatory disorder of the colon and is usually presented by bloody diarrhea and abdominal pain. 1 Several agents such as aminosalicylates, thiopurines, and biologics such as tumor necrosis factor (TNF) inhibitors and anti-integrin agents have been suggested for the treatment of UC. Introduction of TNF inhibitors such as infliximab, adalimumab, and golimumab has revolutionized the treatment of UC, resulting in decreased hospitalizations and colectomies.2–7 However, approximately 10–30% of patients with UC still show primary non-response to the TNF inhibitor treatment, while 23–46% exhibit secondary loss of response. 8 In Korean cohort studies, TNF inhibitors were effective in only 44.8–48.9% of UC patients even after 5 years of treatment.9,10 Therefore, new agents with different modes of action from those of TNF inhibitors and anti-integrin agents have been developed.
Tofacitinib is an orally administered small molecule approved for the treatment of active UC. It inhibits intracellular kinases called Janus kinases, especially Janus kinases 1 and 3, which modulate gene transcription and ultimately alter the different aspects of cellular function and consequently regulate signaling for multiple immune-relevant inflammatory mediators, including type I interferon, interferon-γ, and various interleukins. 11 In the OCTAVE Induction trial, tofacitinib 10 mg twice daily could afford remission in 16.6–18.5% and endoscopic mucosal healing in 28.4–31.3% of patients with moderate-to-severe UC. 12 In the OCTAVE Sustain trial, 34.3–40.6% of induction responders showed remission at week 52 with a therapy involving tofacitinib 5 or 10 mg twice daily. 12 After these pivotal trials, several real-world studies from the West proved the effectiveness of tofacitinib in patients with UC.13–21 Real-world safety of tofacitinib for patients with UC has also been recently reported. 22
However, only limited data are available for Asian patients with UC. Post-hoc analysis of 121 patients from East Asia (Japan, Korea, and Taiwan) in the OCTAVE Induction and Sustain studies showed that the effectiveness and safety of tofacitinib were similar to those in Western patients. 23 However, real-world studies on the effectiveness and safety of tofacitinib in Asian patients have still been limited to a single-center-based design and no reports from Korea are yet available.24–28
Herein, we performed a multicenter study on the real-life therapeutic effectiveness and safety of tofacitinib induction and maintenance treatment in Korean patients with UC. Provisional data were presented as an E-Poster (P335) at the 16th Congress of the European Crohn’s and Colitis Organisation (Virtual) in 2021. 29
Methods
Study design and population
This was a retrospective observational multicenter cohort study conducted by the members of the IBD Research Group of the Korean Association for the Study of Intestinal Diseases. A total of 148 patients with UC from 12 hospitals in Korea, who were started on tofacitinib treatment between January 2018 and November 2020, were enrolled in this study. Patients (1) who were <18 years of age, (2) whose diagnosis of UC were uncertain, (3) who received tofacitinib for conditions other than UC (e.g. rheumatoid arthritis and psoriatic arthritis), or (4) who lacked clinical data were excluded from the study. Tofacitinib was started as 10 mg twice daily for at least 8 weeks, following which tofacitinib 5 or 10 mg was prescribed twice daily based on clinician’s discretion. We collected the demographic characteristics of the patients and various clinical aspects of UC such as age, sex, smoking history, prior use of biologics, extent of UC, UC activity indices (Mayo score or partial Mayo score), Mayo endoscopic subscore (MES), UC endoscopic index of severity (UCEIS), concomitant medications, and laboratory findings, including perinuclear anti-neutrophil cytoplasmic antibody, serum albumin, serum C-reactive protein (CRP), and fecal calprotectin (FC). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement 30 and the checklist is available as Supplemental Material.
Effectiveness outcomes and definitions
We assessed the clinical and endoscopic response at weeks 16, 24, and 52 after starting tofacitinib. The primary outcome was achievement of clinical remission at week 52. The secondary outcomes were the achievement of a clinical response, corticosteroid-free clinical remission, corticosteroid-free clinical response, endoscopic remission at each time point, and safety profiles. Effectiveness outcomes were assessed for all patients who received at least one dose of tofacitinib.
Clinical remission was defined as a partial Mayo score of ⩽2 points with a combined rectal bleeding subscore and stool frequency subscore of ⩽1 point. The clinical response was defined as the reduction in the partial Mayo score to ⩾3 points and at least 30% from baseline along with either a decrease in the rectal bleeding subscore of ⩾1 point or an absolute rectal bleeding subscore of ⩽1 point.2,15,31,32 The corticosteroid-free clinical remission and response were defined as the clinical remission and response without the need for concomitant systemic corticosteroids at the time of evaluation. Endoscopic remission was defined as a MES point of 0–1 or a UCEIS point of 0.33,34
When assessing clinical remission, clinical response, corticosteroid-free clinical remission, and corticosteroid-free clinical response, patients already in clinical remission at baseline were excluded. Likewise, when assessing endoscopic remission, patients who already achieved endoscopic remission (either by MES or UCEIS as mentioned above) at baseline were excluded. If the patients discontinued tofacitinib treatment before reaching each clinical evaluation time point or had missing clinical data, they were considered as not achieving clinical effectiveness outcomes and their follow-up data were censored. Patients who underwent colectomy during the study period or were lost to follow-up were also considered as not having achieved clinical effectiveness outcomes. In addition, if the patient had missing data, it was considered that they have not achieved clinical effectiveness outcomes since then. The clinical data were collected until week 52 after the initiation of tofacitinib.
Safety
The safety profile was assessed for all patients who received at least one dose of tofacitinib. Adverse events (AEs) included all events or symptoms such as colectomy, UC-related hospitalizations, infections, and deep vein thrombosis. The symptoms of the patients were checked at regular or unintended visits. Serous AEs were defined as life-threatening AEs, AEs resulting in persistent or significant disability, congenital anomaly, hospitalization, or death. 12 We also collected the data on patients’ lipid profiles and initiation of lipid-lowering medications. The safety profiles were also collected until week 52 after starting tofacitinib.
Statistical analysis
Categorical variables were presented as numbers and percentages, while continuous variables were presented as mean and standard deviation or median and interquartile range (IQR), depending on whether they were normally distributed or not. Univariable and multivariable logistic regression analyses were performed to identify factors associated with clinical remission, clinical response, corticosteroid-free clinical remission, corticosteroid-free clinical response, or endoscopic remission at weeks 16 and 52. Variables with p < 0.1 in the univariable analysis were included in the multivariable analysis and were analyzed using the stepwise regression method. A two-sided p < 0.05 was considered as statistically significant. The missing values were filled via multiple imputation using the Markov Chain Monte Carlo (MCMC) method. 35 A linear mixed model was used to evaluate longitudinal trends of disease activity and laboratory values at baseline and weeks 16, 24, and 52, including the participating center as a random effect. Disease activity indices and laboratory values at weeks 16, 24, and 52 were compared to those at baseline. CRP levels were log-transformed to approximate a normal distribution. Time was considered as a continuous covariate to investigate temporal trends in each group. All statistical analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA) and graphs were drawn using GraphPad Prism version 9 (GraphPad Software, La Jolla, CA, USA).
Results
Baseline characteristics
A total of 148 patients with UC were included in this study. The median age was 46 years (IQR, 33.3–56) and the median disease duration was 5 years (IQR, 2.1–9.3). Among these, 96 patients (64.9%) had previously been treated with biologic agents. Over half of the patients (81 patients, 54.7%) had extensive colitis. At baseline, the median Mayo score was 9 (IQR, 8–10) and the median UCEIS was 6 (IQR, 5–7). Concomitant oral 5-aminosalicylic acid (5-ASA), 5-ASA suppository, and azathioprine were prescribed to 119 (80.4%), 46 (31.1%), and 3 patients (2.0%), respectively. Concomitant systemic corticosteroids were administrated to 47 patients (31.8%). The median hemoglobin and serum CRP levels at baseline were 12.8 g/dL (IQR, 11.0–14.1) and 0.49 mg/dL (IQR, 0.20–1.12), respectively (Table 1).
Baseline characteristics.

All variables are presented as median (IQR) or n (%).
5-ASAs, 5-aminosalicylic acids; CRP, C-reactive protein; FC, fecal calprotectin; IQR, interquartile range; MES, Mayo endoscopic subscore; p-ANCA, perinuclear anti-neutrophil cytoplasmic antibody; TNF, tumor necrosis factor; UCEIS, ulcerative colitis endoscopic index of severity.
Clinical effectiveness of tofacitinib treatment
The median follow-up duration of the 148 patients was 52 weeks (IQR, 52–52). Among the 148 patients, a total of 115 patients (77.7%) maintained tofacitinib until week 52, while the remaining 33 patients (22.3%) stopped tofacitinib at a median of week 17 (IQR, 7.5–32, range, 2–48). After starting tofacitinib, 116 (78.4%), 41 (27.7%), and 2 patients (1.4%) maintained oral 5-ASA, 5-ASA suppository, and azathioprine until the last follow-up, respectively. Six patients (4.1%) were already in clinical remission at baseline and were excluded from the analysis of clinical outcomes. Those six patients received tofacitinib treatment because of active endoscopic lesions despite clinical remission. Of the remaining 142 patients, 86 (60.6%), 78 (54.9%), and 75 (52.8%) patients achieved clinical remission and 102 (71.8%), 96 (67.6%), and 85 (59.9%) patients achieved clinical response at 16, 24, and 52 weeks, respectively (Figure 1(a)). A total of 74 (52.1%) and 84 (59.2%) patients showed corticosteroid-free clinical remission and corticosteroid-free clinical response, respectively, at week 52 (Figure 1(b)). During tofacitinib treatment, the least-squares means of the partial Mayo score significantly decreased from week 16 and showed a decreasing trend thereafter (Figure 1(c)).

Clinical outcomes and changes in clinical scores during tofacitinib induction and maintenance treatment. (a) Clinical remission and clinical response rates. (b) Corticosteroid-free clinical remission and response rates. (c) Changes in the partial Mayo score, expressed as least-squares means ± standard error of the means.
Endoscopic effectiveness of tofacitinib treatment
Among the 148 patients, two did not have baseline MES data and three had already been in endoscopic remission by MES. In addition, three had missing baseline UCEIS data. At week 16, 75 (52.4%) and 30 (20.7%) patients achieved endoscopic remission by MES and UCEIS, respectively. At week 52, 44 (30.8%) and 22 (15.2%) patients achieved endoscopic remission through MES and UCEIS, respectively (Figure 2(a)). During tofacitinib treatment, the least-squares means of MES and UCEIS showed significantly decreasing trends after week 16 (Figure 2(b) and (c), respectively).
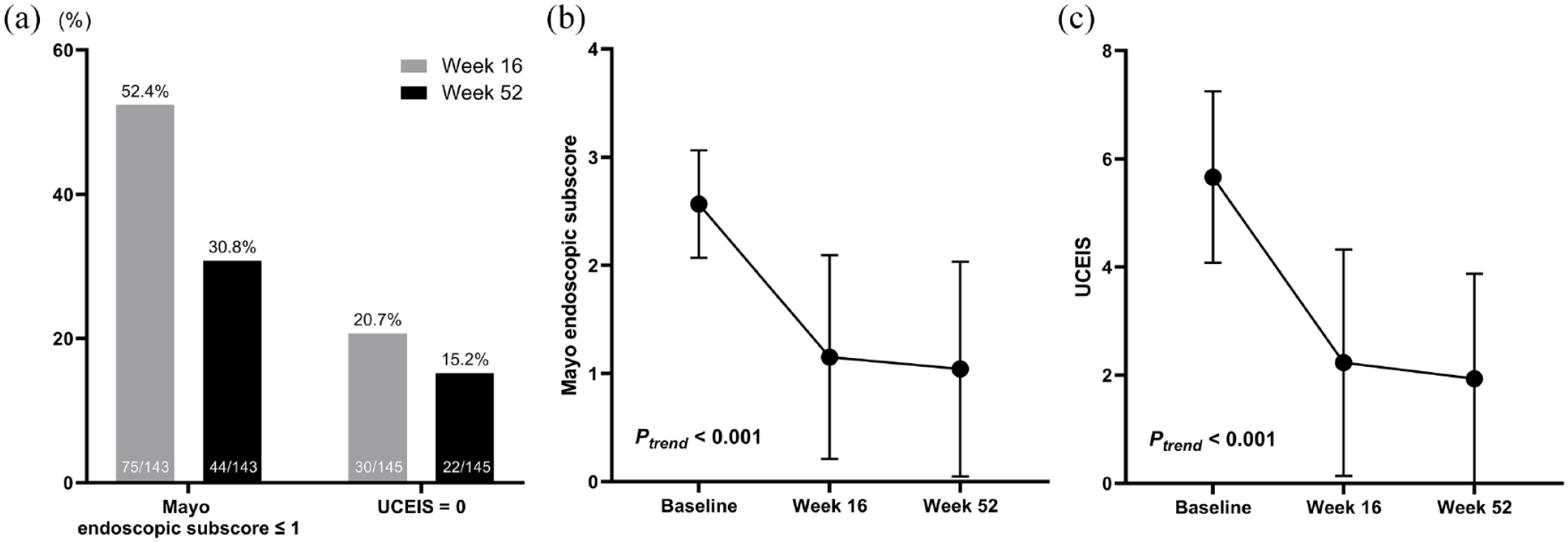
Endoscopic outcomes and changes in endoscopic scores during tofacitinib treatment. (a) Endoscopic remission. (b) Changes in MES and (c) UCEIS expressed as least-squares means ± standard error of the means.
Changes in laboratory values
During tofacitinib treatment, the least-squares mean levels of hemoglobin and serum albumin showed a significantly monotonous increase over time (ptrend < 0.001; Figure 3(a) and (b), respectively). For log-transformed serum CRP, the least-squares mean levels showed a significantly monotonous decrease during treatment (ptrend < 0.001; Figure 3(c)).
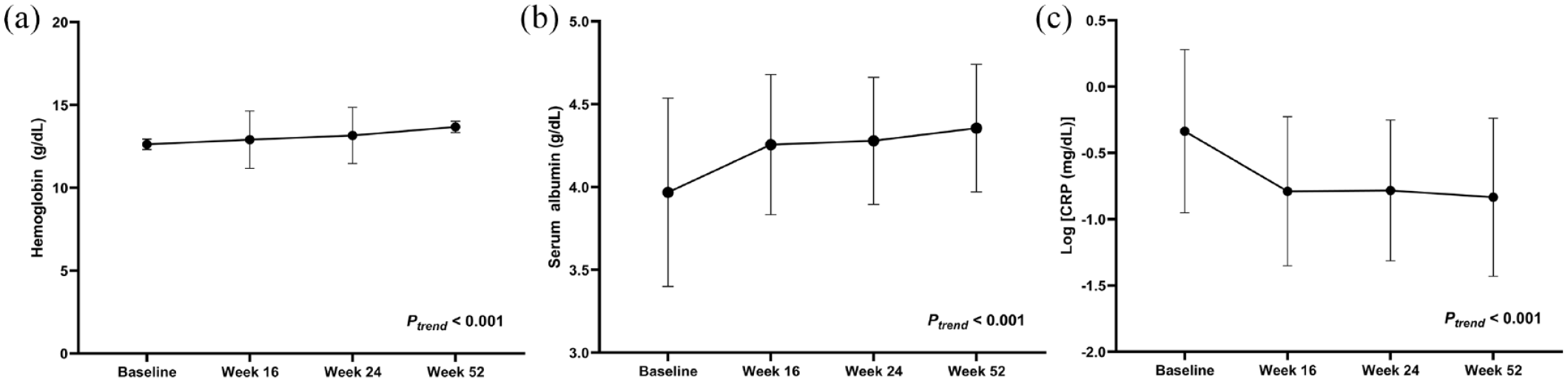
Changes in laboratory values during tofacitinib treatment. (a) Hemoglobin levels, (b) Serum albumin levels, and (c) Log-transformed serum CRP levels expressed as least-squares means ± standard error of the means.
Factors associated with clinical outcomes
In the multivariable analysis, a higher UCEIS at baseline was negatively associated with clinical remission [adjusted odds ratio (aOR): 0.733, p = 0.010], clinical response (aOR: 0.769, p = 0.044), and corticosteroid-free clinical remission (aOR: 0.742, p = 0.013) at week 16 (Supplemental Tables 1–3). No factors were independently associated with clinical remission or corticosteroid-free clinical remission at week 52. However, a higher UCEIS at baseline was negatively associated with clinical response (aOR: 0.774, p = 0.029] (Table 2) and corticosteroid-free clinical response (aOR 0.782, p = 0.035) (Table 3) at week 52.
Univariable and multivariable analyses of associated factors for clinical response at week 52.

CI, confidence interval; CRP, C-reactive protein; OR, odds ratio; MES, Mayo endoscopic subscore; p-ANCA, perinuclear anti-neutrophil cytoplasmic antibody; UCEIS, ulcerative colitis endoscopic index of severity.
Univariable and multivariable analyses of associated factors for corticosteroid-free clinical response at week 52.

CI, confidence interval; CRP, C-reactive protein; OR, odds ratio; MES, Mayo endoscopic subscore; p-ANCA, perinuclear anti-neutrophil cytoplasmic antibody; UCEIS, ulcerative colitis endoscopic index of severity.
Adverse events
Out of the 148 patients, 19 (12.8%) experienced more than one AE, except for an abnormal lipid profile. If we exclude the lipid-profile abnormality, the most common AE then was infections, which occurred in 15 patients (10.1%). Serious AEs occurred in 12 patients (8.1%), who were all admitted to hospital, mainly due to systemic infection (n = 8). Three patients were admitted due to UC flare, one of whom finally received colectomy 2 weeks after tofacitinib initiation. The remaining one patient was admitted due to neutropenia. No UC-related death was recorded. Herpes zoster occurred in 4 (2.7%) patients, 4–21 weeks after starting the tofacitinib therapy. Of these four patients, three had been prescribed tofacitinib 10 mg twice daily and the remaining patient was under tofacitinib 5 mg twice daily. Only a single dermatome was affected in all four patients, who were treated by topical or oral acyclovir. Only one patient suffered from post-herpetic neuralgia; there were no other complications related to herpes zoster and all patients could maintain their tofacitinib treatment. One patient (0.7%) developed deep vein thrombosis. A 48-year-old male patient complained of lower leg pain with swelling 30 weeks after starting the tofacitinib treatment. He was under tofacitinib 10 mg twice daily. Computed tomography showed a suspicious minimal thrombosis in his right popliteal vein. The tofacitinib treatment was stopped and compression stockings were applied, which improved his symptoms afterwards. However, no cases of pulmonary thromboembolism or other major adverse cardiac events were noted in our study population. Due to lipid abnormality, 19 patients were newly started on a lipid-lowering agent among the evaluable patients (a total of 88 patients: excluding 6 patients with dyslipidemia at baseline and 54 patients with insufficient data). Details of AEs including their incidence rates per 100 patient-years are summarized in Table 4.
AEs during tofacitinib treatment (N = 148).

Excluding 6 patients with dyslipidemia at baseline and 54 patients with insufficient data.
AEs, adverse events; UC, ulcerative colitis.
Discussion
In this retrospective observational multicenter cohort study, tofacitinib was administered as an effective induction treatment for Korean patients with active UC with clinical remission and response rates at week 16 of 60.6% and 71.8%, respectively. Moreover, its effectiveness was maintained for 1 year, which was presented as clinical remission and response rates at week 52 (52.8% and 59.9%, respectively). The endoscopic remission rate at week 52 was 30.8% by MES and 15.2% by UCEIS. The biochemical profiles showed an improving tendency over 1 year. A higher UCEIS at baseline was negatively associated with clinical response and corticosteroid-free clinical response at week 52. AEs including herpes zoster and deep vein thrombosis and serious AEs were in line with the previously reported safety profile.
This is the first cohort-based study that proved the real-life effectiveness of tofacitinib as an induction and maintenance treatment for Korean patients with UC, demonstrating clinical effectiveness of tofacitinib, observed in the pivotal OCTAVE Induction and Sustain Trial. 12 Previous studies from the West have reported clinical remission rates and clinical response rates at week 16 as 32–53% and 57–76% after tofacitinib induction, which were not significantly different from our results.14,18–20 A French study, which used the same definition for clinical outcomes as our study did, evaluated the use of tofacitinib as a maintenance treatment and reported corticosteroid-free clinical remission and clinical response rates at week 48 as 34.2% and 36.8%, respectively. 15 Differences in baseline characteristics such as disease severity and prior treatment might have contributed to different outcomes between studies. Ethnic factors could also have affected clinical outcomes, as suggested by a recent population-based study that showed a better long-term prognosis of Korean patients with UC, compared with Western patients. 36 However, long-term observation and further larger studies are required to prove the difference in the outcomes between Korean and Western patients with UC after tofacitinib therapy.
Motoya et al. performed a subgroup analysis of Asian patients in the OCTAVE Induction and Sustain trials and showed a similar efficacy compared to that of the global study population. 23 Real-life Japanese studies have reported week 8 clinical remission and response rates of 27.7–50.5% and 46.6–66.3%, respectively.24–27 They have also reported that 41–43.5% and 45–47.1% of patients showed remission and response at weeks 48–52, respectively.26,27 These results were also slightly numerically different from those of our study, probably because of the difference in the patients’ characteristics and the definitions of clinical outcomes. Overall, the clinical effectiveness outcomes of our study coincide with the data from recent meta-analyses, including Western and Eastern studies, which showed clinical remission and response rates of 47% and 64.2% at weeks 12–16 and 41.4% and 41.8% at month 12, respectively. 37
One of the strengths of our study is the report on endoscopic outcomes, because week 16 endoscopic evaluation is mandatory for evaluating the response to induction therapy with tofacitinib in Korea. The OCTAVE trial showed that mucosal healing, defined as MES ⩽ 1 in our study, was achieved in 28.4–31.3% of patients at week 8 and in 37.4–45.7% of patients at week 52. 12 The Asian subgroup analysis of OCTAVE showed similar results regarding endoscopic mucosal healing, that is, in 24.2% of patients at week 8 and 45.5–57.1% of patients at week 52. 23 Cremer et al. 14 conducted a real-world study and reported endoscopic remission (MES ⩽ 1) as 50% at week 16, which was similar with our data.
During tofacitinib treatment in patients with UC in our study, the laboratory values generally showed improving trends: an increase in hemoglobin and serum albumin and a decrease in serum CRP. Previous studies also showed similar trends for biochemical improvements after tofacitinib treatment.13,16–18,20,21,28 Previous studies have also shown that higher serum CRP and lower serum albumin levels are correlated with severe clinical and endoscopic disease activities and poor prognosis.38–41 In addition, improvement in the serum CRP and albumin within a short period after starting the biologic treatment has been reported to be related with better outcomes for UC patients.42–45 Hence, it can be suggested that biochemical improvements observed in our study could reflect clinical improvement during the tofacitinib treatment.
When performing the logistic regression analysis to determine the association between clinical outcomes and several variables, missing values cause the problem of excluding all data in the multivariable regression analysis. Unfortunately, there were some variables with missing values in our data albeit though the number of missing values in each category was very small (range, 1–10). To overcome this problem, we used the missing value imputation technique, which allows for uncertainty regarding the missing data by creating several different plausible imputed data sets (10 imputed data sets in our study) and appropriately combining the results obtained from each set. As the pattern of missing data was arbitrary, we used the MCMC method, which creates multiple imputations using simulations from a Bayesian prediction distribution for normal data.
In this study, a higher UCEIS at baseline was negatively associated with clinical response and steroid-free clinical response at week 52. Similarly, Ishida et al. reported that a baseline UCEIS of ⩾5 had a significantly lower tofacitinib-continuation rate than those with a UCEIS of <5. 28 Interestingly, a higher MES was not associated with clinical response at week 52. It could be regarded that baseline UCEIS can reflect clinical outcomes and prognosis more accurately than MES after the tofacitinib therapy, as the former represents mucosal inflammation more precisely than MES does.46,47 In contrast, previous treatments with biologics did not affect the clinical outcomes. Similar results have also been reported by previous real-world studies.13,18,19,24 Together with the results from the OCTAVE Induction and Sustain trials, 12 these results reinforce the role of tofacitinib as a second-line agent for those exposed to biologics.48,49
Our study also reported tolerable AEs during tofacitinib treatment. The most common AE was infections: Cytomegalovirus (CMV) colitis in 4.1%, herpes zoster in 2.7%, and Clostridioides difficile (C. difficile) infection in 1.4% of patients. These results were similar to those of other real-world studies, which reported 3.4% of patients with CMV colitis, 0.9–10% of patients with herpes zoster, and 0.4–6.9% with C. difficile infection.13–16,18–22,24–27 Venous thromboembolism developed in one patient (0.7%), whereas pulmonary thromboembolism was not observed in our study. These results are similar to those of other real-world studies, which reported 0–1.3% of patients developing venous thromboembolism after tofacitinib therapy.14,16–22,24,27
Our study, the largest-scale Asian study so far showed the real-world effectiveness and safety of tofacitinib in Korean patients with UC. However, it has several limitations too. First, this was a retrospective study with some missing data for some patients. Applying strict methods of evaluation, if the patients had missing clinical data, it was considered that they did not achieve clinical effectiveness outcomes since then, and hence, they were marked as ‘no responders’. If we had prospectively collected the patient’s clinical data with less missing data, we might have shown more accurate and even better clinical outcomes. To minimize the possibility of bias from the retrospective nature of our study, all authors shared the definitions of variables and outcomes and collected the data based on those definitions. Second, because this was a multicenter study, there could be a discrepancy in managing and evaluating the patients. However, because the guidelines for UC management are widely applied50,51 and strict reimbursement criteria are enforced in Korea for small molecule treatments for UC, the discrepancy between how doctors manage UC patients could be minimized. Third, because FC levels were collected for a few patients only, we could not incorporate the change in the FC levels after tofacitinib treatment.
Conclusions
Tofacitinib was a clinically effective real-world induction and maintenance treatment for Korean patients with UC. The clinical outcomes were similar to those of previously reported real-world studies. In addition, the endoscopic activity scores and laboratory results improved during the treatment. The incidence of AEs was also similar to that observed in other real-world studies, demonstrating the absence of any new safety signal associated with tofacitinib treatment.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231154103 – Supplemental material for Real-life effectiveness and safety of tofacitinib treatment in patients with ulcerative colitis: a KASID multicenter cohort study
Supplemental material, sj-docx-1-tag-10.1177_17562848231154103 for Real-life effectiveness and safety of tofacitinib treatment in patients with ulcerative colitis: a KASID multicenter cohort study by Seung Hwan Shin, Kyunghwan Oh, Sung Noh Hong, Jungbok Lee, Shin Ju Oh, Eun Soo Kim, Soo-Young Na, Sang-Bum Kang, Seong-Joon Koh, Ki Bae Bang, Sung-Ae Jung, Sung Hoon Jung, Kyeong Ok Kim, Sang Hyoung Park, Suk-Kyun Yang, Chang Hwan Choi and Byong Duk Ye in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848231154103 – Supplemental material for Real-life effectiveness and safety of tofacitinib treatment in patients with ulcerative colitis: a KASID multicenter cohort study
Supplemental material, sj-docx-2-tag-10.1177_17562848231154103 for Real-life effectiveness and safety of tofacitinib treatment in patients with ulcerative colitis: a KASID multicenter cohort study by Seung Hwan Shin, Kyunghwan Oh, Sung Noh Hong, Jungbok Lee, Shin Ju Oh, Eun Soo Kim, Soo-Young Na, Sang-Bum Kang, Seong-Joon Koh, Ki Bae Bang, Sung-Ae Jung, Sung Hoon Jung, Kyeong Ok Kim, Sang Hyoung Park, Suk-Kyun Yang, Chang Hwan Choi and Byong Duk Ye in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
