Abstract
Background:
Proton-pump inhibitors (PPI) are among the most prescribed drugs worldwide, and a large body of evidence raises concerns about their inappropriate use. Previous estimates of inappropriate use varied due to different definitions and study populations.
Aims:
We aimed to measure the population-based incidence and time trends of PPI and potentially inappropriate PPI prescriptions (PIPPI) with a novel method, continuously assessing excessive cumulative doses based on clinical practice guidelines. We also assessed association of patient characteristics with PPI prescriptions and PIPPI.
Methods:
This was an observational study based on a large insurance claims database of persons aged >18 years with continuous claims records of ⩾12 months. The observation period was January 2012 to December 2017. We assessed the incidence and time trends of PPI prescriptions and PIPPI based on doses prescribed, defining ⩾11.5 g of pantoprazole dose equivalents during any consecutive 365 days (average daily dose >31 mg) as inappropriate.
Results:
Among 1,726,491 eligible persons, the annual incidence of PPI prescriptions increased from 19.7% (2012) to 23.0% (2017), (p = <0.001), and the incidence of PIPPI increased from 4.8% (2013) to 6.4% (2017), (p = <0.001). Age, male gender, drugs with bleeding risk and multimorbidity were independent determinants of PIPPI (p = <0.001 for all).
Conclusions:
This study provides evidence that one of the most prescribed drug groups is commonly prescribed inappropriately in the general population and that this trend is increasing. Multimorbidity and drugs with bleeding risks were strong determinants of PIPPI. Addressing PPI prescriptions exceeding guideline recommendations could reduce polypharmacy and improve patient safety.
Introduction
Proton-pump inhibitors (PPI) are highly effective antacid drugs 1 and are among the most commonly prescribed drugs worldwide. Pantoprazole, one of the most popular examples, is the single most prescribed generic drug and generates the highest overall generic drug cost in Switzerland. 2 As the use of PPI has been steadily increasing, concerns about their inappropriate use have been growing. Both potential side effects3–5 and potentially unnecessary expenditures6–8 are of increasing interest. Many initiatives and professional societies advise against the prolonged use of PPI.9,10
From a patient- and health-economics perspective, excessive average long-term doses seem most relevant, as higher cumulative doses of PPI might produce more side effects 3 and cause more unnecessary costs.
Yet inappropriate use of PPI remains widespread and is still increasing, as multiple studies in various settings show.11–14 Many previous estimates of potentially inappropriate PPI prescriptions (PIPPI) are highly variable, but do not define inappropriateness based on continuous and cumulative doses prescribed on the patient level.11,13 Accordingly, considerable uncertainty remains about the valid extent of PIPPI within the population.
Aims
We aimed to measure the population-based incidence and time trends of PPI prescriptions and PIPPI by means of a novel method, which continuously assesses excessive cumulative doses based on clinical practice guidelines. We also assessed association of patient characteristics with PIPPI in comparison to PPI prescriptions.
Methods
Study design, data source and population
In this retrospective analysis, we used claims data of a major Swiss health insurance company (‘Helsana group’) covering approximately 14% of the Swiss population. 15 All persons registered in Switzerland are obliged by law to contract basic health insurance. Individuals who cannot afford to pay the monthly premiums receive subsidies or full insurance coverage by their social services. Equal access to the full range of medical treatments including drugs is thereby granted to everyone, regardless of insurance status. Lower monthly premiums are available when choosing different deductible schemes. Deductibles range from 300 to 2500 Swiss Francs (CHF) yearly. In addition, optional alternative health plans (managed care, telemedicine gatekeeping and others) enable lower monthly premiums.
Our sample includes persons aged 18 or older whose records were available for at least 12 months from 1 January 2012 through 31 December 2017. Prescriptions of implausibly high cumulative doses were excluded.
Objectives
We defined our main objectives as follows:
Assessment of proton-pump inhibitor prescribing
The insurance claims cover all pharmacy and physician invoices of prescription drugs. PPI were identified by Anatomical Therapeutic Chemical (ATC) Classification codes A02BC. Incidence of PPI prescriptions was defined by any such prescription during any calendar year. All health care invoices submitted to Helsana for reimbursement during the study period were considered. Prescribed PPI doses of the various PPI products were converted into pantoprazole dose equivalents, thus referencing the most commonly prescribed PPI product in Switzerland. In an attempt to estimate PPI prescribing in the context of clinical practice in Switzerland, doses of all the PPI agents on the market were labelled as either maintenance or therapeutic dose and then translated into pantoprazole dose equivalents of 20 mg and 40 mg, respectively. Detailed definitions of the various pantoprazole dose equivalents are listed under Supplemental Material (1).
Definition and assessment of potentially inappropriate proton-pump inhibitor prescribing
Based on a literature review covering 34 clinical practice guidelines in 2017, we extracted PPI dosage and treatment regimens on 57 different PPI indications. 16 Thereof we derived a 365-day cumulative pantoprazole dose equivalent of 11.5 g as a cut-off for inappropriateness. This cut-off reflects an evidence-based maximal-dose scenario, based on three hypothetical sequential phases: (1) an 8-week double-dose therapeutic treatment phase (2 × 40 mg of pantoprazole dose equivalents, i.e. for severe erosive esophagitis), (2) a 4-week tapering to a therapeutic dose (1 × 40 mg), (3) a maintenance dose (1 × 20 mg) for the remainder of the year. The resulting cumulative yearly dose of 11.2 g of pantoprazole equivalents was rounded up to 11.5 g. PIPPI was then determined by screening every observed person day for a preceding 365-day cumulative dose exceeding 11.5 g. To clarify we give a practical example: if 100 tablets pantoprazole 40 mg were dispensed 1 December 2016, this corresponded to 4000 mg added to the balance sheet of one patient. If the same patient repeated this process twice (i.e. adding another 8000 mg and therefore 12,000 mg in total) before 30 November 2017, then this patient would be regarded as a dispenser of PIPPI for 2017 (on the day of the third dispensing, i.e. when 11.5 g are exceeded).
Patient characteristics
Patient characteristics included gender, age and the number of chronic conditions according to ATC codes and pharmaceutical cost groups (PCG). 17 Drugs which increase gastrointestinal bleeding risk and are therefore commonly associated with PPI co-prescriptions 18 were assessed by translating the prescription of all such drugs into defined daily doses per patient per day, using corresponding ATC codes. These drugs were grouped as follows: Anticoagulant therapy including vitamin K antagonists (ATC B01AA) and direct oral anticoagulants (ATC B01AF, B01AE), antiplatelet therapy including aspirin (ATC B01AC06) and other platelet inhibitors (ATC B01AC), systemic corticosteroids (ATC H02AB) and non-steroidal anti-inflammatory drugs (NSAIDs), including coxibs (ATC M01A, M01AH). The number of chronic conditions using PCGs was likewise applied on the basis of drug prescriptions, according to all concurrent ATC codes billed (including ATC code A02 for acid-related disorders) per calendar year per patient, and categorized as follows: 0 (reference group), 1–3 and ⩾4.
Statistics: analysis
We calculated the incidence of PPI prescriptions, being the first PPI prescription during a calendar year and the incidence of PIPPI, being patients crossing at least once the cumulative prescribed PPI cut-off dose of 11.5 g during any preceding 365 days, counted per calendar year. Both measures used the total patients enrolled within the calendar year as the denominator. In addition, the incidence of PIPPI among the incidental PPI prescription group was calculated. Percentages and mean values were used to describe categorical and nominal data, respectively. Descriptive statistics were used to provide characteristics of all patients with PPI prescriptions. These data were grouped into PPI prescriptions (i.e. non-PIPPI) and PIPPI groups. Chi-Squared tests for trend in proportions were used to analyse changes in proportions of PPI prescriptions and PIPPI incidence. For bivariate comparisons between the PPI prescriptions and PIPPI groups, Kruskal–Wallis, Fisher exact and Chi-Square tests were used accordingly. A two-sided p-value of 0.05 was considered statistically significant. To explore patient characteristics associated with PIPPI (dependent variable), we performed a multivariable logistic regression. Independent factors included in the regression model were age, gender, prescription of drugs increasing bleeding risk (yes/no) and number of chronic conditions. The goodness-of-fit of the logistic regression model was assessed by calculating the area under the curve (AUC).
To investigate a potential dose–response relationship between drugs increasing bleeding risk and PIPPI, we performed an additional analysis in which we included the absolute prescribed defined daily doses (DDDs) 19 of any of the specific drug categories (dependent variables) into a regression model. For this purpose, we defined the corresponding time period in the group of PPI prescriptions as the 365 days’ period following the first prescription of a PPI (as opposed to the retrospective 365 days in the PIPPI group). We present results of regression analysis as odds ratios with 95% confidence intervals (95% CI). Statistical analyses were conducted with “R” version 3.6.1 (2019-07-05).
Ethics
According to the Swiss national ethical and legal regulations, ethical approval from the local ethics committee was not needed for the study. As data were completely anonymized, no patient consent was necessary.
Results
The total amount of different persons accessible in our dataset throughout all years was 1,726,491. After applying inclusion criteria, this number amounted to 1,337,722. The number of included persons is reported on a yearly basis in Table 1. The mean age during the observation period was 51.2 years, 52.4% of persons were female.
Annual incidence of PPI and PIPPI prescribing.
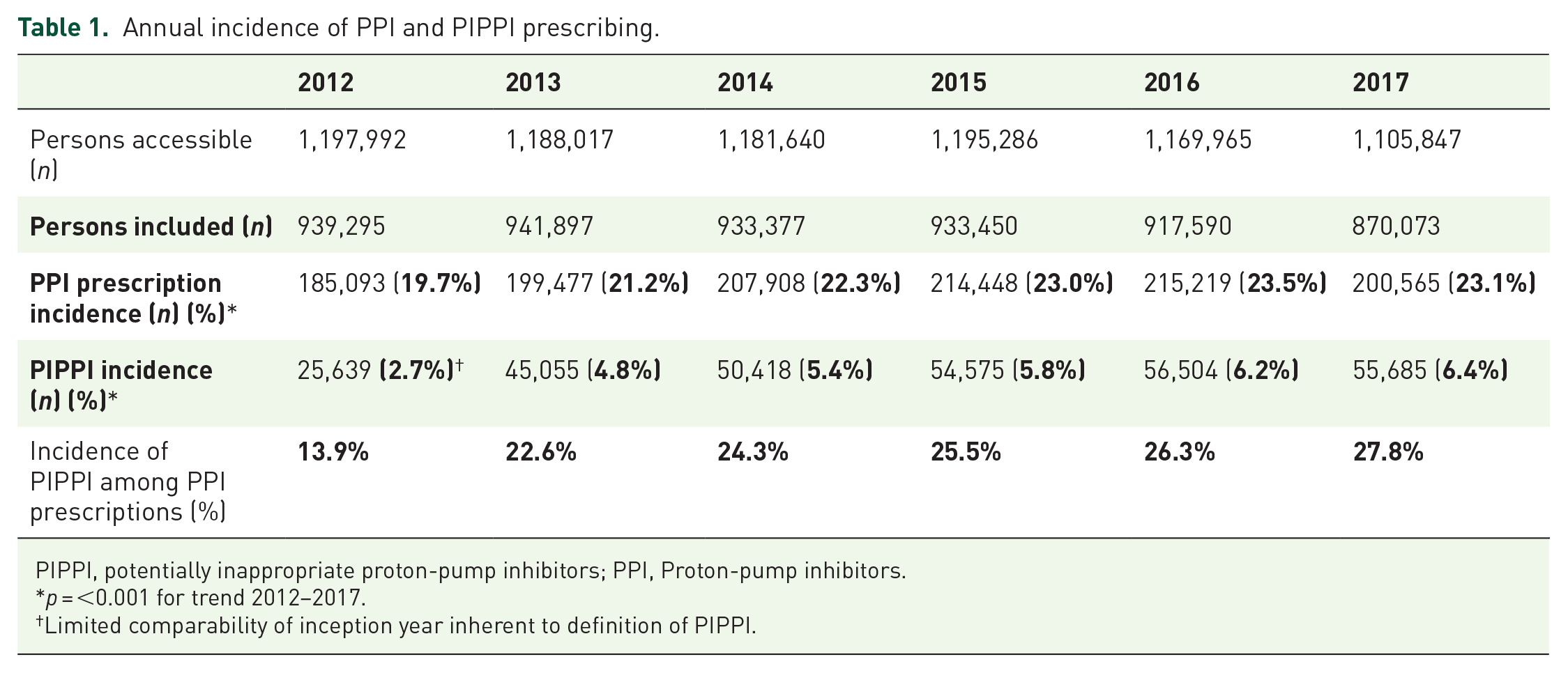
PIPPI, potentially inappropriate proton-pump inhibitors; PPI, Proton-pump inhibitors.
p = <0.001 for trend 2012–2017.
Limited comparability of inception year inherent to definition of PIPPI.
Incidence of PPI prescriptions and PIPPI, relative proportions in the study population and the group with PPI prescriptions are reported in Table 1. The incidence of PPI prescriptions was 23.0% in 2017, which reflects a steady rise since 2012 (p = <0.001 for trend). The overall incidence of PIPPI was 6.4% in 2017, corresponding to 27.8% of incident PPI prescriptions. Again, the positive trend for proportions since 2012 was statistically significant (p = <0.001 for trend).
All incident PPI prescriptions and PIPPI cases and corresponding patient characteristics are listed in Table 2. Patients with PIPPI were significantly older and more often female (unadjusted analysis).
Patient characteristics and comparison between PPI and PIPPI prescribing groups.

NSAIDs, non-steroidal anti-inflammatory drugs; PIPPI, potentially inappropriate proton-pump inhibitors; PPI, proton-pump inhibitors; SD, standard deviation.
The majority of patients with PPI prescriptions or PIPPI were also prescribed drugs with increased bleeding risk at least once during the designated 365-day periods, 63.3% and 86.0%, respectively. The highest incidence for a specific category of drugs with increased bleeding risk was observed for NSAIDs, with 50.9% and 62.0% of PPI prescriptions and PIPPI patients exposed.
Overall, the majority of patients in both prescription groups had at least one chronic condition: 73.2% of patients in the PPI prescriptions as compared with 98% in the PIPPI group. In the PPI prescriptions group, 1–3 chronic conditions were most prevalent. In the PIPPI group, the proportion of patients suffering from more than three chronic conditions reached 64%.
Results of the multivariable logistic regression of PIPPI and patient characteristics are presented in Table 3 and indicate that the model explains most of the variation of PIPPI (AUC 81.3%). In contrast to the bivariate comparison, female gender was independently associated with lower odds of PIPPI. All categories of concurrent drugs with bleeding risk and the number of chronic conditions remained independently associated with higher odds of PIPPI.
Adjusted association between PIPPI prescribing and patient characteristics.

AUC, area under the curve; 95% CI, 95% confidence intervals; NSAIDs, non-steroidal anti-inflammatory drugs; OR, odds ratio; PIPPI, potentially inappropriate proton-pump inhibitor.
The additional analysis assessing the association of PIPPI with DDDs of all drugs with bleeding risk showed a significant dose–response relationship (Supplemental Material).
Discussion
In this large population-based study, one out of four adults was prescribed a PPI in 2017. Among these, one out of four was prescribed PIPPI based on cumulative doses not supported by clinical practice guidelines. We found increasing annual incidence trends for both PPI prescriptions and PIPPI during the observed 6-year period. Multimorbidity and drugs with bleeding risk were strong independent determinants of PIPPI.
In our study, the proportion of patients with at least one PPI prescription per year was 19.7% in 2012 and 23% in 2017. Corresponding rates from other health care systems varied between 7.4% and 29.8%.13,14,20–22 The definition of inappropriateness and differences between populations (i.e. inclusion criteria) and health care systems, including varying degrees of over-the-counter (OTC) dispensing, might explain some of this variance.
The annual incidence of PIPPI in our study rose from 4.8% in 2013 to 6.4% of all adults in 2017, corresponding to 27.8% of the population with an incidental PPI prescription. To our knowledge, there is no previous literature describing PIPPI with continuous, cumulative dose regimens on the patient level and based on clinical practice guidelines. Multiple studies have assessed the extent of PIPPI in various settings and with different definitions of long-term use. 12 Previous estimates have been highly variable and ranged from proportions of overuse from 11 to 84%. 23 PIPPI was often ascertained either based on prescriptions exceeding a certain amount of weeks,24,25 within specific age groups, 25 specific situations such as drugs with bleeding risk, 8 or as the absence of an appropriate clinical indication.14,26 A nationwide drug utilization study in France reported 32.4% of new PPI users in the year 2015 without other drugs or diagnoses to support an indication for PPI therapy, 14 yet only 4.1% of new PPI users received a continuous PPI therapy lasting longer than 6 months. Another study assessed DDDs per patient but reported averages per calendar year, thereby leaving uncertainty about the actual degree of PIPPI at the patient level. 11 Nevertheless, some previous studies using different definitions of PIPPI, presented findings aligned with our results: a nationwide population-based study in Iceland found that 21% of incident high-dose PPI prescriptions were still using high doses after 1 year. 13
Our data show a significant increase of PPI prescriptions and PIPPI over time. This phenomenon was described previously.13,20,27 Whether this is merely a consequence of an increased prevalence of polypharmacy in an aging society, 25 or pointing to challenges in the deprescribing process, with patients reporting increased symptoms and lower satisfaction after withdrawal of PPI, 28 remains unclear based on our data.
Higher age was associated with PIPPI compared with PPI prescriptions, whereas female gender was not. Yet evidence with regard to gender as determinant of PIPPI is conflicting.24,29
Drugs with increased bleeding risk and especially NSAIDs were more common in the PIPPI group (62%), as compared with the PPI prescriptions group (50.9%). Similar proportions were reported elsewhere: NSAID co-use reached 48% in one study assessing long-term PPI prescriptions, and a recent population-based estimate attributed NSAID use to 53.3% of patients newly prescribed a PPI.21,14 Our finding remained stable after regression analysis, showing that all drugs with bleeding risk were independently associated with PIPPI. The effect of NSAIDs decreased with short-term NSAID use, presumably due to a large number of healthy patients within the sample. While these data underline the substantial contribution of NSAIDs to PPI prescriptions, it remains unclear as to why exactly drugs with increased bleeding risk are associated to PIPPI (as opposed to PPI prescriptions). Interestingly, most guidelines commenting on prevention of NSAID-associated ulcers and bleedings (as compared with other guidelines recommending PPIs) leave room for interpretation by omitting recommendations on dose reduction in this specific context.30–33 As physicians tend to overestimate the need for preventive PPI prescriptions, 34 this might lead to prescriptions of higher doses more frequently. Also, it is known that previously asymptomatic patients can become symptomatic upon withdrawal of PPI drugs. 35 Patients with preventive PPI prescriptions might thus be at particular risk for the re-initiation of therapeutic doses, as such prescriptions are often stopped abruptly in clinical practice, together with drugs increasing bleeding risk.
Furthermore, we found chronic conditions in 98% of patients with PIPPI and in 73.1% of patients with PPI prescribing. In fact, an increasing number of chronic conditions was the strongest predictor of PIPPI compared with PPI prescriptions in our logistic regression analysis. This finding has been described previously.21,25,36
Strengths of this study are its large and representative sample including roughly 1.2 million persons, reflecting about 14% of the adult Swiss resident population. 15 Furthermore, our method of defining PIPPI overcomes some of the variability of past estimates. It does not depend on the documentation and identification of (often absent) disease- or drug-related reasons justifying PPI prescriptions. It also does not rely on methods based on treatment intervals or days covered, which might not capture the long-term recurrence of PPI prescriptions or the actual dosage prescribed. In addition, by using a continuous, ‘rolling’ approach, our analysis is not restricted to calendar year boundaries and thereby can assess daily average doses over longer periods. The chosen cut-off value for PIPPI (11.5 g) is based on a clinical worst-case scenario of maximally necessary and sensible PPI intake, which only is surpassed in very seldom clinical situations.
This study has also limitations. We were not able to estimate OTC or hospital dispensing. However, available OTC formulations are low dose and small package size only, and patients are unlikely to use OTC PPI on a long-term basis, as costs are high and health insurance coverage is plenary. Furthermore, the effective ingestion of PPI of course remains unclear, as our data documents drug reimbursement only. Again, it is unlikely that a relevant amount of patients would regularly dispense PPI on a long-term basis without actually ingesting them. Lastly, our sample does not contain clinical data. We therefore cannot exclude potentially justified long-term, high-dose PPI treatments as occurs in very rare conditions. However, clinical recommendations for long-term prescriptions of therapeutic dose PPI are limited: Barrett-esophagus 37 with a prevalence of <2% in the general adult population 38 and Zollinger–Ellison syndrome, which is exceedingly rare. 39 It seems thus unlikely, that such disease-specific therapeutic dose PPI prescriptions would alter the general finding of this study, which affects a substantial share of the overall population.
Many studies observed side effects of PPI prescriptions use without establishing causality, 40 but some trials did.41,42 In light of the widespread prescription of PPI, even the probability of rare or minor side effects should raise concern. Appropriate PPI prescriptions (and deprescribing) in the context of short-term preventive use (e.g. with NSAIDs) might be of special interest for further investigations. Co-occurrence is widespread and specific recommendations on dose reductions are lacking. Our data portray a period when dose lowering and deprescribing of PPI should have been commonplace. The Choosing Wisely campaign in the US adopted this target in 2012, 10 which was widely received in Switzerland. Its Swiss offshoot Smarter Medicine 43 absorbed the PPI lowering recommendation later, in 2015.
However, a systematic review reported PPI to be among the drugs most resistant to deprescribing. 44 A call for more deprescribing 45 might therefore not be enough. Some authors, as a consequence, believe that prescribing or paying restrictions (self-pay for high dose) should be put in place by authorities.21,45 We suggest that a standardized operationalization of PIPPI, as presented in this study, offers opportunities to measure PIPPI use more validly, to compare its occurrence across health service providers, settings, and care models, and possibly to incentivize a more cautious use of PPI drugs, while increasing transparency and awareness.
Conclusion
This study provides evidence that one of the most prescribed drug groups is commonly prescribed inappropriately in the general population and that this trend is increasing. Multimorbidity and drugs with bleeding risks were strong determinants of PIPPI. Addressing PPI prescriptions exceeding guideline recommendations could reduce polypharmacy and improve patient safety.
Supplemental Material
sj-docx-1-tag-10.1177_1756284821998928 – Supplemental material for Potentially inappropriate proton-pump inhibitor prescription in the general population: a claims-based retrospective time trend analysis
Supplemental material, sj-docx-1-tag-10.1177_1756284821998928 for Potentially inappropriate proton-pump inhibitor prescription in the general population: a claims-based retrospective time trend analysis by Leander Muheim, Andri Signorell, Stefan Markun, Corinne Chmiel, Stefan Neuner-Jehle, Eva Blozik, Pascale Ursprung, Thomas Rosemann and Oliver Senn in Therapeutic Advances in Gastroenterology
Footnotes
Author contributions
All authors have been involved in the study concept, design and interpretation of results. Andri Signorell collected and analysed the data, Leander Muheim drafted the article and all of the authors revised it critically. All authors approved the final version of the article, including the authorship list.
Conflict of interest statement
Helsana Group provided support in the form of salaries for authors AS and EB being employed by the Helsana Group. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript. AS and EB declare no conflict of interest. Helsana Group shall have no liability to any third party in respect of the contents of this article. All the other authors as well have no conflicts of interests or financial disclosures to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Swiss Academy of Medical Sciences with an unrestricted grant.
Guarantor of the article
Leander Muheim
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
