Abstract
Background:
There is limited data on the use of anti-TNF agents in patients with concomitant cirrhosis. The aim of this study is to assess the safety of anti-TNF agents in patients with compensated cirrhosis who used these medications for the treatment of an underlying rheumatologic condition or IBD.
Methods:
Multicenter, retrospective, matched, case-control study. A one to three case-control match was performed. Adults who received anti-TNF therapy were matched to three adults with cirrhosis who did not receive anti-TNF therapy. Patients were matched for etiology of cirrhosis, MELD-Na and age. Primary outcome was the development of hepatic decompensation. Secondary outcomes included development of infectious complications, hepatocellular carcinoma (HCC), extra-hepatic malignancy, and mortality.
Results:
Eighty patients with cirrhosis who received anti-TNF agents were matched with 240 controls. Median age was 57.2 years. Median MELD-Na for the anti-TNF cohort was seven and median MELD-Na for the controls was eight. The most common etiology of cirrhosis was NAFLD. Anti-TNF therapy did not increase risk of decompensation (HR: 0.91, 95% CI: 0.64–1.30, p = 0.61) nor influence the time to development of a decompensating event. Anti-TNF therapy did not increase the risk of hepatic mortality or need for liver transplantation (HR: 1.18, 95% CI: 0.55–2.53, p = 0.67). Anti-TNF therapy was not associated with an increased risk of serious infection (HR: 1.21, 95% CI: 0.68–2.17, p = 0.52), HCC (OR: 0.45, 95% CI: 0.13–1.57, p = 0.21), or extra-hepatic malignancy (OR: 0.82, 95% CI: 0.29–2.30, p = 0.71).
Conclusions:
Anti-TNF agents in patients with compensated cirrhosis does not influence the risk of decompensation, serious infections, transplant free survival, or malignancy.
Keywords
Introduction
Biological agents that target tumor necrosis factor α (TNF-α) were first described in the 1990s and have since revolutionized the treatment of multiple inflammatory conditions including rheumatoid arthritis (RA), psoriasis and inflammatory bowel disease (IBD).1–3 The safety profile of anti-TNF agents has been studied extensively and an increased risk of non-melanoma skin cancer,4,5 bacterial 6 and fungal 7 infections have been observed. Anti-TNF agents have also been associated with hepatotoxicity8,9 including the reactivation of viral hepatitis, drug induced liver injury (DILI), and de novo autoimmune hepatitis (AIH).
The clinical course of cirrhosis is characterized by decompensating events including the development of ascites, hepatic encephalopathy, and variceal bleeding. 10 The pathophysiology of decompensation is complex but involves a state of chronic inflammation mediated by pro-inflammatory cytokines and chemokines, 11 including TNF-α.12–15 As the development of a decompensating event is a critical point in the progression of cirrhosis with a decrease in median survival from 12 to 2 years, 16 it is important to evaluate the rate of decompensation in cirrhotic patients using these drugs. Furthermore, the risk of infection and malignancy in patients with cirrhosis who are using anti-TNF-α agents is unclear.
Early research demonstrated elevated TNF-α levels in patients with acute alcoholic hepatitis (AH)17,18 and this observation resulted in several studies that evaluated the potential therapeutic benefit of anti-TNF agents in this population. Although small observational studies observed a survival benefit in patients with moderate to severe AH who were treated with anti-TNF agents, 19 larger randomized controlled trials (RCT) failed to demonstrate a similar benefit.20,21 The outcomes from these RCTs were disappointing and both etanercept and infliximab showed increased mortality and rate of serious infections when compared to those who did not receive an anti-TNF agent. Perhaps due to this early experience, there is limited data on the use of these medications in patients with concomitant cirrhosis. As the indications for anti-TNF agents expands, understanding their potential effect on the clinical course of cirrhosis is imperative. The aim of this study is to assess the safety of anti-TNF agents in patients with compensated cirrhosis who used these medications for the treatment of an underlying rheumatologic condition or IBD.
Methods
This is a multicenter, retrospective, matched, case-control study evaluating the risk of hepatic decompensation, transplant free survival, malignancy and infectious complications in patients with compensated cirrhosis undergoing treatment with anti-TNFs. Patients evaluated at the Cleveland Clinic Florida (Weston, FL) and the Cleveland Clinic (Cleveland, OH) from 1 January 2000 to 30 June 2019 that met inclusion criteria were included in the study. The protocol was approved by the Cleveland Clinic Institutional Review Board (IRB #17-1387).
Selection of patients
Electronic medical records for the study period were queried using eResearch (Cleveland, OH). The inclusion criteria included patients older than 18 years of age, who were diagnosed with compensated cirrhosis and received an anti-TNF agent after the documented diagnosis of cirrhosis. The diagnosis of cirrhosis was determined using ICD-9 (571.0, 571.2, 571.4, 571.5, 571.6, 571.8, 571.9) and ICD-10 (K70.30, K74.3, K74.5, K74.60, K76.1, K74.69) codes. Records were subsequently queried for the following medications: adalimumab, certolizumab, etanercept, golimumab, and infliximab. Those patients who received an anti-TNF agent, irrespective of indication, were included. Controls were patients over the age of 18 years of age with a documented diagnosis of compensated cirrhosis who did not receive an anti-TNF agent. All medical records were evaluated to confirm the diagnosis of compensated cirrhosis after review of histology, radiography, and clinical data.
Matching procedure
A one to three case-control match was performed. All patients with cirrhosis who received an anti-TNF agent were matched to three patients with cirrhosis who did not receive an anti-TNF agent. Patients were matched for etiology of cirrhosis, severity of liver disease and age. The etiology of cirrhosis was classified as either due to alcohol, viral hepatitis, or non-alcoholic fatty disease (NAFLD). If a patient had an alternative etiology for their cirrhosis, they were labeled as “other”. Patients were matched on the severity of their underlying liver disease by utilizing the modified model for end stage liver disease score (MELD-Na). When possible, MELD-Na was matched exactly. However, if this was not possible, the MELD-Na was expanded to ±2 points of the patients who received anti-TNF therapy. Age at the time of diagnosis of cirrhosis was matched exactly if possible. If this was not possible, it was expanded to include an additional 5 years above or below the age of the patient who received anti-TNF therapy.
Study outcomes
The primary outcome was the development of hepatic decompensation defined as any of the following: ascites, variceal hemorrhage, and hepatic encephalopathy. The presence or absence of a decompensating event was determined by review of clinician progress notes, radiology records, and endoscopy reports.
Secondary outcomes included the development of infectious complications, hepatocellular carcinoma (HCC), extra-hepatic malignancy, and mortality. Infectious complications were defined as a serious infection, either bacterial, fungal, or viral, that required hospitalization and administration of antimicrobials. Infectious complications were only included if they occurred while the patient was receiving anti-TNF therapy. The presence of HCC was determined by review of histology and radiology reports that documented the presence of HCC utilizing the Liver Reporting and Data System (LI-RADS) scoring system. A LI-RADS score of 5 was used to diagnose HCC. Extra-hepatic malignancy was determined by review of the medical record including clinical documentation, histology, and radiology reports. For assessment of liver related mortality, we included the number of patients who either underwent liver transplantation (LT) or died due to a complication of their liver disease.
The duration of treatment was determined as the period of time that a patient received an anti-TNF agent after being diagnosed with cirrhosis. Patients were followed until discontinuation of anti-TNF agent, liver transplantation, or death.
Statistical analysis
Data were assessed for normality and appropriate descriptive statistics were calculated for discrete variables (frequencies and percentages) and continuous variables (means and standard deviations or median and interquartile range (IQR)). Characteristics between patients who received anti-TNF and those who did not were compared using Chi-square tests or Fisher’s exact tests and Mann–Whitney U tests. Comparisons between anti-TNF use and the outcomes of decompensation and mortality were performed using Kaplan–Meier survival estimates and compared using log-rank tests. Logistic regression and Cox proportional hazards models were used to evaluate associations between patient outcomes and anti-TNF use. Data were analyzed using SPSS version 25 (IBM Corp., Armonk, NY). All statistical tests were two-tailed and p < 0.05 was used to determine significance.
Results
We identified 80 patients with cirrhosis who received anti-TNF agents. Using a 1:3 case control match for age, MELD-Na and etiology of liver disease, 240 patients with cirrhosis who did not receive an anti-TNF agent were identified as controls. Demographic data is provided in Table 1. The median age for patients who received anti-TNF therapy was 57.2 years and the median age of the controls was 57.7 years. The median MELD-Na for the anti-TNF cohort was 7 and the median MELD-Na for the controls was 8. The most common etiology of cirrhosis was NAFLD. Patients who received anti-TNF therapy had a longer follow-up compared to those who did not receive anti-TNF medications (45.2 months versus 34.5 months, p < 0.01).
Patient characteristics by treatment group.

IQR, interquartile range; TNF, tumor necrosis factor.
Primary outcome
There was no significant difference in the number of patients who experienced decompensation between patients with cirrhosis who received anti-TNF therapy versus those who did not receive anti-TNF drugs (51.2% versus 48.8%). Exposure to anti-TNF therapy did not increase the risk of decompensation (HR: 0.91, 95% CI: 0.64–1.30, p = 0.61) nor influence the time to development of a decompensating event (Figure 1). Anti-TNF therapy did not increase the risk for development of either ascites, hepatic encephalopathy, or variceal bleeding (Table 2).
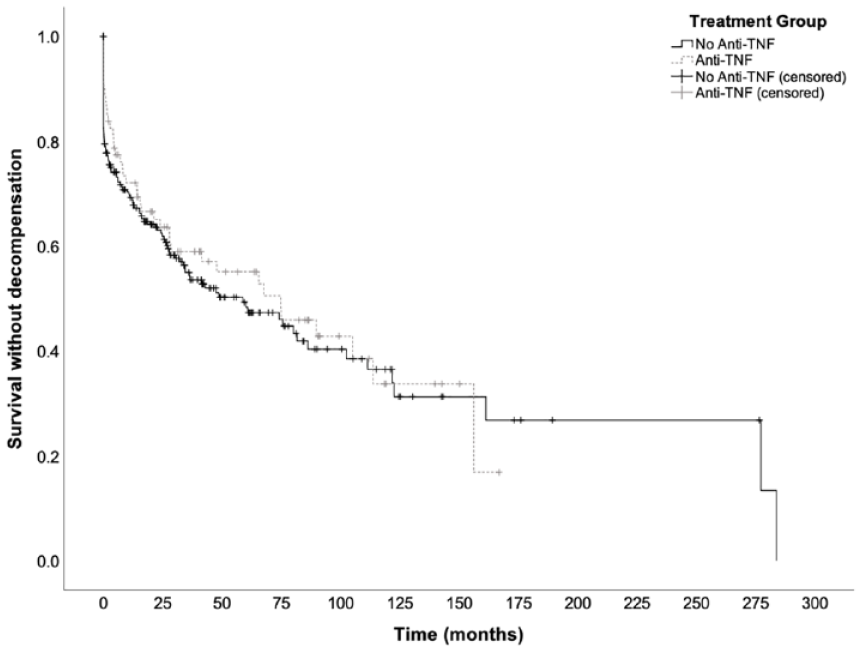
Kaplan–Meier curves for survival without decompensation. There was no significant difference in survival time without decompensation between patients who received anti-TNF and those who did not (p = 0.61).
Cox proportional hazards model of the association between anti-TNF use and patient outcomes.

CI, confidence interval; HR, hazard ratio; TNF, tumor necrosis factor.
Those patients that received anti-TNFs were evaluated for potential predictors of decompensation. Age, MELD-Na score nor the indication for anti-TNF therapy were associated with a significant risk for decompensation (Table 3). However, patients with cirrhosis due to alcohol use were more likely to decompensate compared to other etiologies of cirrhosis (HR: 2.83, 95% CI: 1.09–7.34, p = 0.03).
Cox proportional hazards analysis of decompensation among patients treated with anti-TNF (N = 80).

BMI, body mass index; CI, confidence interval; HR, hazard ratio; TNF, tumor necrosis factor.
Reference category.
Secondary outcomes
Transplant free survival was not affected by anti-TNF therapy. Ten (12.5%) patients who received anti-TNF therapy either died due to their liver disease or underwent LT compared to 22 (9.2%) patients who did not receive anti-TNF therapy (HR: 1.18, 95% CI: 0.55–2.53, p = 0.67). Figure 2 shows that there was no difference in time to mortality attributed to liver disease or LT (261 months versus 229.5 months, p = 0.08).
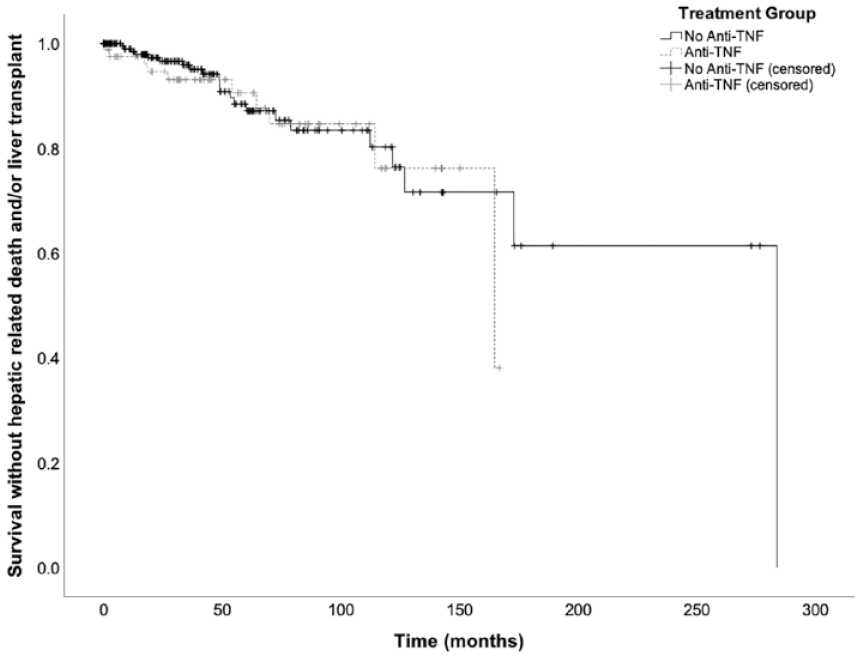
Kaplan–Meier curves for survival without hepatic mortality and/or liver transplant. There was no significant difference in survival time without hepatic mortality and/or liver transplant between patients who received anti-TNF and those who did not (p = 0.67).
Serious infections were seen in 21.3% of patients in the anti-TNF therapy group and in 15.4% of controls. The use of anti-TNFs was not associated with a significant increase in the risk for development of a serious infection (HR: 1.21, 95% CI: 0.68–2.17, p = 0.52). There was no difference in time to development of serious infection between the two groups (Figure 3). In addition, anti-TNF use was not associated with an increased risk of HCC (OR: 0.45, 95% CI: 0.13–1.57, p = 0.21) or development of an extra-hepatic malignancy (OR: 0.82, 95% CI: 0.29–2.30, p = 0.71).

Kaplan–Meier curves for survival serious infectious complication. There was no significant difference in survival time without infectious complication between patients who received anti-TNF and those who did not (p = 0.51).
Discussion
Despite the role that TNF-α plays in the inflammatory cascade that results in decompensation, we observed that patients with cirrhosis who received anti-TNF therapy had similar rates of decompensation when compared to matched controls who did not receive anti-TNF therapy. Contrary to what may have been expected, anti-TNF therapy did not significantly increase the risk of serious infection. In addition, anti-TNF therapy did not influence transplant free survival, nor was it a significant risk factor for hepatic or extra-hepatic malignancy.
The effect of anti-TNFs in patients with cirrhosis remains poorly understood. Our study attempts to expand our understanding of the potential role that anti-TNFs have in the progression of cirrhosis and also to better understand their safety profile in this unique patient population. A recent review article concluded that in patients with NAFLD and no fibrosis, anti-TNFs may have a beneficial effect. 22 However, the authors noted that there was insufficient data in patients with cirrhosis, largely due to the paucity of studies assessing anti-TNF use in patients with cirrhosis. Thus far a handful of case reports have described the use of anti-TNFs in patients with, for the most part, compensated cirrhosis. Two case reports described the experience of 4 patients with IBD and concomitant cirrhosis.23,24 One patient with decompensated cirrhosis and a MELD-Na of 23 at the time of anti-TNF initiation required LT, however in the other cases, all of whom had compensated cirrhosis, anti-TNF therapy was reportedly well tolerated. Furthermore, two patients with psoriatic arthritis and compensated cirrhosis were successfully treated with anti-TNF therapy and followed for an average of 7.5 months after biologic initiation.25,26
Several studies have studied the effect of anti-TNF agents on mortality. When compared to non-biological agents, anti-TNF therapy was not associated with increased mortality in patients with IBD, ankylosing spondylitis, RA, psoriasis, and psoriatic arthritis, 27 A retrospective cohort study of patients with RA observed that after adjusting for disease severity, anti-TNF administration was indeed associated with a significant reduction in mortality in women. 28 The apparent survival benefit of anti-TNFs in certain rheumatological diseases is believed to due to their disruptive effect on the inflammatory cascade. Aside for studies that observed increased mortality in patients with AH,20,21 the effect of anti-TNFs in patients with compensated cirrhosis has not been evaluated. In our study, anti-TNF treatment did not affect transplant free survival in patients with compensated cirrhosis.
Both cirrhosis and anti-TNF agents independently increase the risk of serious infection. A review of data from the National Hospital Discharge Survey showed that patients with cirrhosis are at a nearly 2.5 times increased risk of developing sepsis after adjusting for age, gender and race 29 and infections are associated with a four-fold increase in mortality. 30 TNF-α is a critical cytokine in the inflammatory cascade that follows a bacterial infection. A 2015 meta-analysis reported that patients with RA who received high-dose biological therapy were at an almost two times increased risk of developing a serious infection when compared to patients treated with traditional disease modifying agents. 31 Similarly, a meta-analysis studying patients with ulcerative colitis showed an increased rate of infection in patients treated with anti-TNF agents when compared to placebo. 32 Reports of infection-related mortality with the use of anti-TNF medications in AH led to concerns about the use of these medications in patients with cirrhosis. However, the population of patients with AH is unique and the risk of infection in compensated cirrhotics with the use of anti-TNF drugs has not been well defined. In our study, similar rates of serious infection were observed between the two groups. Although there was a difference in the length of follow-up between the two cohorts, we accounted for time in our analysis, and anti-TNF therapy did not affect the risk of infection or time to development of infection.
Cirrhosis is present in 80–90% of patients with HCC and the 5-year cumulative risk for development of HCC in patients with cirrhosis is estimated to be between 5% and 30%. 33 In addition to HCC, patients with cirrhosis are at a nearly 10 times increased risk for development of cholangiocarcinoma. 34 Extra-hepatic malignancies including esophageal, pancreatic, lung, and colon cancers are also more common in patients with cirrhosis. 35 The neoplastic potential of anti-TNF agents has been evaluated in several studies. Two large registry-based studies of patients with RA did not observe any increased risk of solid organ malignancy.36,37 However, the incidence of non-melanoma skin cancer is increased in patients treated with anti-TNF agents. Information, however, on the risk of HCC or extra-hepatic malignancy in cirrhotics using anti-TNF is limited. One case report described 2 patients with psoriasis and a long-standing history of compensated cirrhosis due to hepatitis C who developed HCC after an average of 39.5 months of etanercept therapy. 38 We observed that despite treatment with anti-TNF agents, there was no difference in the development of either hepatic or extra-hepatic malignancy in our cohort of patients with cirrhosis.
This study is the largest reported experience on the use of anti-TNF agents in patients with cirrhosis. We accounted for potential risk factors for decompensation by utilizing a matched, case-control study design. A recent cohort study showed that the clinical course and survival of patients with cirrhosis varied significantly depending on the etiology of the underlying liver disease. 39 The authors observed the highest risk of decompensation in patients with cirrhosis due to alcohol use and relatively lower rates of decompensation in patients with cirrhosis due to AIH. We therefore used age, etiology of liver disease, and MELD-Na as criteria for matching controls to patients who received anti-TNFs.
Our study had several limitations. This was a retrospective study and there was no specific treatment protocol for use of anti-TNFs so we could not account for specific medication differences or doses. In certain instances, a decompensating event or infectious complication may have occurred at another institution and therefore there is the possibility that events, for both cohorts, may have been under-reported. Although, to our knowledge, this is the largest study evaluating the risk of anti-TNFs in cirrhotic patients, the number of patients in our study was limited and the results only apply to patients with compensated cirrhosis.
TNF-α plays a key role in the pathophysiology of cirrhosis but the use of anti-TNF agents in patients with compensated cirrhosis does not influence their risk of decompensation, serious infections, transplant free survival, or malignancy. Clinicians, however, must be judicious with the use of anti-TNF agents and remain vigilant for complications in patients with cirrhosis who receive anti-TNF agents.
Footnotes
Author contributions
Guarantor of the article: Nikhil Kapila, MD. AG, JMR, GF, FS, MA, IH acquired, analysed and interpreted data and drafted the manuscript, MM collected data, performed the statistical analysis and drafted the manuscript. JM and JFC analysed and interpreted data, drafted and critically reviewed the manuscript, All authors approved the final version of the article, including the authorship list.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
