Abstract
Background:
Patients with initially unresectable colorectal liver metastases (CRLM) could achieve survival benefit from successful conversion therapy. Recently, Tumor Burden Score (TBS) was proposed as a valuable index to predict outcome following resection of CRLM. The study is aimed to investigate the association of TBS with conversion outcome.
Methods:
A total of 234 patients who underwent first-line treatment in our center were enrolled as training cohort. The validation cohort was developed from 89 patients in our previous study. Cut-off value of TBS was calculated to stratify patients into two groups. Significance test and logistic regression model were used to examine the prediction value of TBS for conversion outcome after first-line systemic therapy. Kaplan–Meier method and Cox proportional hazard model were applied to assess the prognostic value of TBS.
Results:
TBS showed good discriminatory power [area under curve (AUC) 0.726, p < 0.001] with cut-off value defined as 14.3 in training cohort, which was validated in the validation cohort. Increasing TBS was related to adverse chemotherapy response and conversion outcome. Low TBS group had three times higher conversion rate than that in high TBS group (57.3% versus 19.0%, p < 0.001). Multivariate analysis indicated that high TBS [odds ratio (OR) = 3.676, 95% confidence interval (CI) 1.671–8.429, p = 0.002], first-line treatment response as stable disease (SD) or progressive disease (PD) (OR = 9.247; 95% CI 4.736–18.846, p < 0.001), and absence of targeted therapy (OR = 2.453, 95% CI 1.139–5.455, p = 0.024) were three independent risk factors for failure conversion outcome. High TBS was significantly associated with conversion outcome whatever chemotherapy response, addition of targeted therapy, and Kirsten rat sarcoma viral oncogene homolog (KRAS) status. High TBS was also associated with worse overall survival.
Conclusion:
TBS should be applied in clinical practice to predict conversion outcome in patients with initially unresectable CRLM.
Highlights
Patients with higher Tumor Burden Score (TBS) tend to have worse objective response and conversion outcome.
TBS predicts conversion outcome for patients with initially unresectable CRLM.
TBS is a prognostic marker of overall survival for patients with initially unresectable CRLM.
Introduction
Colorectal cancer (CRC) has become one of the leading malignant diseases with high incidence and mortality rates in the world, 1 and liver is the most common site for metastases, accounting for more than half of patients with metastatic CRC. 2 Hepatic resection is recommended as the most important curative measure for colorectal liver metastases (CRLM). 3 However, nearly 80% of patients with CRLM were considered unresectable at the time of diagnosis.4,5 After systemic chemotherapy, 22.5–51.9% of these patients were qualified for liver local curative therapy to achieve the status of no evidence of disease (NED).6,7 As a result, 5-year survival rate could be raised from 9% to 32% compared with systemic therapy alone.5,8 Therefore, conversion treatment followed by liver local curative therapy is of great benefit for patients with initially unresectable CRLM and thus should be carefully considered. Although various studies had been conducted to find out the optimal conversion approach,9,10 distribution of conversion rate in different patient cohorts and factors associated with conversion rate still remained unclear. Hence, exploring novel parameters to identify various conversion risk subgroups to guide personalized treatment is urgently needed.
Recently, Kazunari Sasaki et al. first reported that the ‘Tumor Burden Score’ (TBS), which was developed by tumor size and number of liver metastases, showed superior prognostic discriminatory power for CRLM patients compared with Fong score.11,12 While the original model was established based on tumor size and number obtained from pathological specimen after resection, imaging TBS using radiographic data also helped to predict survival outcomes preoperatively and had similar performance as pathological TBS. 13 As such, imaging TBS could be utilized to determine the optimal strategy for patients before initial treatment. However, few studies have investigated the clinical significance of baseline TBS for patients with initially unresectable CRLM.
Therefore, this study was designed to investigate the baseline TBS in patients with initially unresectable CRLM receiving first-line systemic treatment. Accordingly, we aimed to (1) describe the distribution characteristics of baseline TBS in unresectable CRLM and (2) investigate the association of TBS with conversion outcome in order to identify TBS as a valuable parameter to stratify patients into different conversion risk groups.
Methods
Study population
A total of 234 patients who were diagnosed with initially unresectable CRLM and underwent first-line treatment from December 2012 to January 2020 at Sun Yat-sen University Cancer Center were enrolled in the training cohort. All patients met the following inclusion criteria: (1) histologically confirmed colorectal adenocarcinoma, (2) all metastatic lesion limited to liver, (3) no previous liver resection or interventional therapy, and (4) explicit conversion outcome. To evaluate the efficacy of TBS for prediction of conversion outcome and prognosis, the validation cohort included patients who received first-line treatment from May 2009 to June 2013 at our center in our previous study. 7 The patients in the validation cohort also met the inclusion criteria as the training cohort and there is no overlap of patients between the two cohorts. Informed consent for the use of the clinical data was obtained from the patients before first-line treatment. All procedures were performed based on the Declaration of Helsinki. The study was approved by the Institutional Research Ethics Committee of Sun Yat-sen University Cancer Center (approval number: B2020-309-01).
Definition of parameter and cut-off value
Clinical T stage and N stage of primary CRC were determined according to the seventh edition of the UICC-TNM staging system for CRC. The characteristics of liver metastases, including number, maximum diameter, distribution, and invasion of blood vessels, were assessed based on enhanced abdominal nuclear magnetic resonance imaging (MRI) or computed tomography (CT). TBS was calculated according to the following mathematical equation: (TBS) 2 = (maximum tumor diameter in cm) 2 + (number of tumor) 2 . 11 The carcinoembryonic antigen (CEA) and cancer antigen (CA) 19-9 (CA19-9) levels were measured before systemic therapy, and the cut-off value was 10 ng/ml and 35 U/ml, respectively, according to our previous study. 14
Treatments outcomes
The treatment strategy and operability of the liver metastases of each patient were determined based on the final agreement of the multidisciplinary team (MDT), including staffs from Department of Colorectal Surgery, Hepatobiliary Surgery, Medical Oncology, Medical Imaging, and Invasive Technology. Tumor response or progression after first-line treatment was determined according to the Response Evaluation Criteria in Solid Tumors 1.1. 15 Conversion success is defined as liver metastases deemed to be resectable after first-line systemic treatment and patients achieving NED status contributed by local treatment including surgery and radiofrequency ablation (RFA), while conversion failure is defined as liver metastases remaining unresectable after first-line systemic treatment and patients failing to receive curative local treatment. Overall survival (OS) was defined as the interval from the date of first-line systemic treatment to the date of death from any cause or to the last follow-up. The final follow-up visit occurred in April 2021 for training cohort and September 2021 for validation cohort.
Statistical analysis
Categorical variables were presented as whole numbers and percentages, and continuous variables as medians and interquartile ranges. Comparison between variables were assessed with chi-square test or Fisher’s exact test when appropriate. The receiver operating curve (ROC) analysis was performed to examine the discriminative power of TBS in conversion outcome and then to determine the cut-off value, the applicability of which was then assessed in the validation cohort. The logistic regression model was applied to identify the risk factors associated with conversion outcome. Parameters with a value of p < 0.05 in univariate analysis were included in the multivariate analysis. Odds ratios (ORs) and 95% confidence intervals (CIs) were subsequently calculated. Kaplan–Meier method was used to estimate OS, and differences between groups were assessed with log-rank test. The Cox proportional hazards model was applied to identify the risk factors associated with OS. Parameters with a value of p < 0.05 in univariate analysis were included in the multivariate analysis. Results were reported as hazard ratios (HRs) and 95% CIs. All analyses were conducted using SPSS 20.0 software (IBM, Chicago, IL, USA), GraphPad Prism 7 software (GraphPad Software Inc, San Diego, CA, USA), and R software packages. The reporting of this study conforms to the ‘Guidelines for reporting case series of tumours of the colon and rectum. Techniques in Coloproctology’. 16
Results
Patients characteristics and systemic therapy
The demographic and clinicopathological characteristics and treatment approaches of patients in training cohort are summarized in Table 1. The median age was 55 years (IQR 47–62 years), with 168 (71.8%) males and 68 (28.2%) females. Of 194 patients with available Kirsten rat sarcoma viral oncogene homolog (KRAS) status, 69 (35.6%) had KRAS mutational type tumors; 150 (64.1%) patients received oxaliplatin-based chemotherapy regimen, 21 (9.0%) patients received irinotecan-based, and 40 (17.1) patients received FOLFOXIRI regimen. Most patients were given targeted drugs (n = 170, 72.7%), with 101 (59.4%) of them receiving Cetuximab and 69 (40.6%) receiving Bevacizumab. After first-line systemic therapy, 112 (47.8%) patients presented tumor partial response (PR), 64 (27.4%) patients were found to have stable disease (SD), and 58 (24.8%) patients were confirmed to develop into progressive disease (PD). Finally, 102 (56.4%) patients were successfully converted to NED. Among the 102 patients, 53 (52.0%) underwent both tumor resection and RFA, while 49 (48.0%) received tumor curative resection alone. Clinicopathological characteristics of validation cohort are summarized in Table S1 in the Supplemental material.
Baseline characteristics of patients in training cohort.

CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CRLM, colorectal liver metastases; FOLFOXIRI, folinic acid, 5-fluorouracil, oxaliplatin and irinotecan; FUDR HAI, 5-fluoro-2′-deoxyuridine hepatic artery infusion; IQR, interquartile range; KRAS, Kirsten rat sarcoma viral oncogene homolog; PD, progressive disease; PR, partial response; RECIST, Response Evaluation Criteria in Solid Tumors; SD, stable disease.
Data of 194 patients were available.
Distribution of TBS in initially unresectable CRLM
Distribution of TBS is demonstrated, with conversion rate and objective response rate (ORR) in each group of training cohort depicted in Figure 1(a) and (b), respectively. The range of TBS was 3.3–100.7, with conversion rate and ORR to be 69.2–10.5% and 76.9–13.2%, respectively. As TBS increased, both conversion rate and ORR decreased. In particular, there was a sharp drop on both curves when TBS was greater than 17.0. In addition, the two curves shared similar shape and trend by visual interpretation, suggesting that conversion rate and ORR were positively correlated. To better stratify patients, ROC curve analysis showed that the area under curve (AUC) for conversion outcome based on baseline TBS was 0.726 (95% CI 0.662–0.789, p < 0.001) (Figure 2). The optimal TBS cut-off value was 14.3 at the highest Youden index of 0.358. We then examined the applicability of 14.3 to distinguish patients with conversion success and patients with conversion failure in validation cohort. The result showed the discriminatory performance of this cut-off value for conversion outcome as AUC = 0.676 (95% CI 0.535–0.818, p = 0.028), sensitivity = 75.0%, and specificity = 60.3% (Figure S1 in the Supplemental material).

Distribution of TBS among total patients in training cohort. (a) Conversion rate among patients with different TBS intervals. (b) ORR among patients with different TBS intervals.

Receiver-operating curve analysis examining the discriminative ability of TBS to predict conversion outcome in training cohort.
Factors associated with TBS
In order to investigate clinicopathological parameters related to TBS in initially unresectable CRLM, patients were stratified into two groups according to the cut-off value in training cohort. As a result, 150 (64.1%) patients were divided into low TBS group (TBS ⩽ 14.3), and the remaining 84 (35.9%) patients into high TBS group (TBS > 14.3). Baseline clinicopathological characteristics were included in chi-square test or Fisher’s exact test and the results are shown in Table 2. Low TBS group had three times higher conversion rate than that in high TBS group (57.3% versus 19.0%, p < 0.001). In addition, compared with patients with low TBS, patients with higher TBS tended to have bilobar metastases (96.4% versus 68.7%, p < 0.001), higher level of CEA (94.0% versus 82.0%, p = 0.010) and CA19-9 (78.6% versus 64.0%, p = 0.021), chemotherapy response of SD or PD (70.2% versus 42.0%, p < 0.001), invasion of hepatic vein (81.0% versus 50.7%, p < 0.001), and portal vein (63.1% versus 29.3%, p < 0.001). However, no significant association was found between TBS and systemic treatment including both chemotherapy regimen and targeted therapy.
Association of TBS and clinical characteristics in training cohort.

CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CRLM, colorectal liver metastases; FUDR HAI, 5-fluoro-2′-deoxyuridine hepatic artery infusion; FOLFOXIRI, folinic acid, 5-fluorouracil, oxaliplatin and irinotecan; KRAS, Kirsten rat sarcoma viral oncogene homolog; PD, progressive disease; PR, partial response; RECIST, Response Evaluation Criteria in Solid Tumors; SD, stable disease; TBS, Tumor Burden Score.
Data of 194 patients were available.
Risk factors of failure conversion outcome
Because of collinearity, the number and size of liver metastases were not included in the univariate and multivariate logistic regression models in training cohort. As shown in Table 3, in univariate logistic regression analysis, bilobar metastases (OR = 2.895, 95% CI 1.527–5.635, p = 0.001), synchronous CRLM (OR = 4.674, 95% CI 1.595–17.019, p = 0.009), high level of baseline CEA (OR = 2.439, 95% CI 1.145–5.398, p = 0.023) and CA199 (OR = 2.380, 95% CI 1.356–4.224, p = 0.003), high TBS (OR = 5.711, 95% CI 3.094–11.050, p < 0.001), chemotherapy response (OR = 11.364, 95% CI 6.232–21.484, p < 0.001), absence of targeted drug (OR = 2.050, 95% CI 1.128–3.825, p = 0.021), invasion of hepatic vein (OR = 1.905, 95% CI 1.119–3.262, p = 0.018), and portal vein (OR = 3.136, 95% CI 1.812–5.537, p < 0.001) were remarkably related to failure conversion outcome. Multivariate analysis indicated that TBS > 14.3 (OR = 3.676, 95% CI 1.671–8.429, p = 0.002), first-line treatment response as SD or PD (OR = 9.247, 95% CI 4.736–18.846, p < 0.001), and absence of targeted therapy (OR = 2.453, 95% CI 1.139–5.455, p = 0.024) were three independent predictive factors for failure conversion outcome. According to the same stratification method, data of validation cohort demonstrated the significant association between TBS and conversion outcome (29.3% versus 8.3%, p = 0.013, Figure S2 in the Supplemental material).
Univariate and multivariate logistic regression analyses of baseline characteristics for conversion failure outcome in training cohort.

CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CI, confidence interval; CRLM, colorectal liver metastases; KRAS, Kirsten rat sarcoma viral oncogene homolog; OR, odds ratio; PD, progressive disease; PR, partial response; RECIST, Response Evaluation Criteria in Solid Tumors; Ref, reference; SD, stable disease; TBS, Tumor Burden Score.
Data of 194 patients were available.
Effect of TBS on conversion outcome stratified by first-line treatment response, addition of targeted therapy, and KRAS status
Results in training cohort revealed that whatever chemotherapy responses were, lower TBS was significantly related to higher conversion rates (PR: 78.2% versus 52.0%, p = 0.003, Figure 3(a); SD or PD: 28.6% versus 6.8%, p = 0.002, Figure 3(b)). In addition, whether targeted therapy was added to chemotherapy or not, low TBS and successful conversion outcome are significantly associated (addition of targeted therapy: 63.1% versus 23.1%, p < 0.001, Figure 3(c); chemotherapy alone: 47.2% versus 5.3%, p = 0.003, Figure 3(d)). Similarly, for patients with KRAS wild type, conversion rate was significantly higher in low TBS group than high TBS group (60.4% versus 21.6%, p < 0.001, Figure 3(e)). For patients with KRAS mutational type, conversion rate was also significantly higher in low TBS group than high TBS group (62.1% versus 11.1%, p < 0.001, Figure 3(f)).

Conversion rate in low and high TBS groups in training cohort. (a) Conversion rate among patients who had PR response to first-line systemic therapy stratified by TBS. (b) Conversion rate among patients who had SD or PD response to first-line systemic therapy stratified by TBS. (c) Conversion rate among patients who had targeted therapy stratified by TBS. (d) Conversion rate among patients who had chemotherapy alone stratified by TBS. (e) Conversion rate among patients who had KRAS wild type stratified by TBS. (f) Conversion rate among patients who had KRAS mutational type stratified by TBS.
Prognostic value of TBS and risk factors for OS
At a median follow-up of 43.5 months, 81 patients died of tumor. Patients with low TBS had significantly higher 3-year OS rate than patients with high TBS in training cohort (58.2% versus 43.2%, log-rank p = 0.003, Figure 4). The results of univariate and multivariate Cox analysis for OS in training cohort are shown in Table 4. Multivariate analysis revealed that poor tumor differentiation (HR 2.539, 95% CI 1.446–4.458, p = 0.001), clinical T4 stage (HR 1.920, 95% CI 1.104–3.339, p = 0.021), and high TBS (HR 2.202, 95% CI 1.302–3.722, p = 0.003) were the independent prognostic factors for OS. OS analysis in validation cohort revealed similar results that patients with low TBS had better 3-year OS rate than patients with high TBS (43.1% versus 20.0%, log-rank p = 0.047, Figure S3 in the Supplemental material).

Kaplan–Meier estimates of overall survival stratified by TBS in training cohort.
Univariate and multivariate Cox regression analyses of baseline characteristics for overall survival in training cohort.
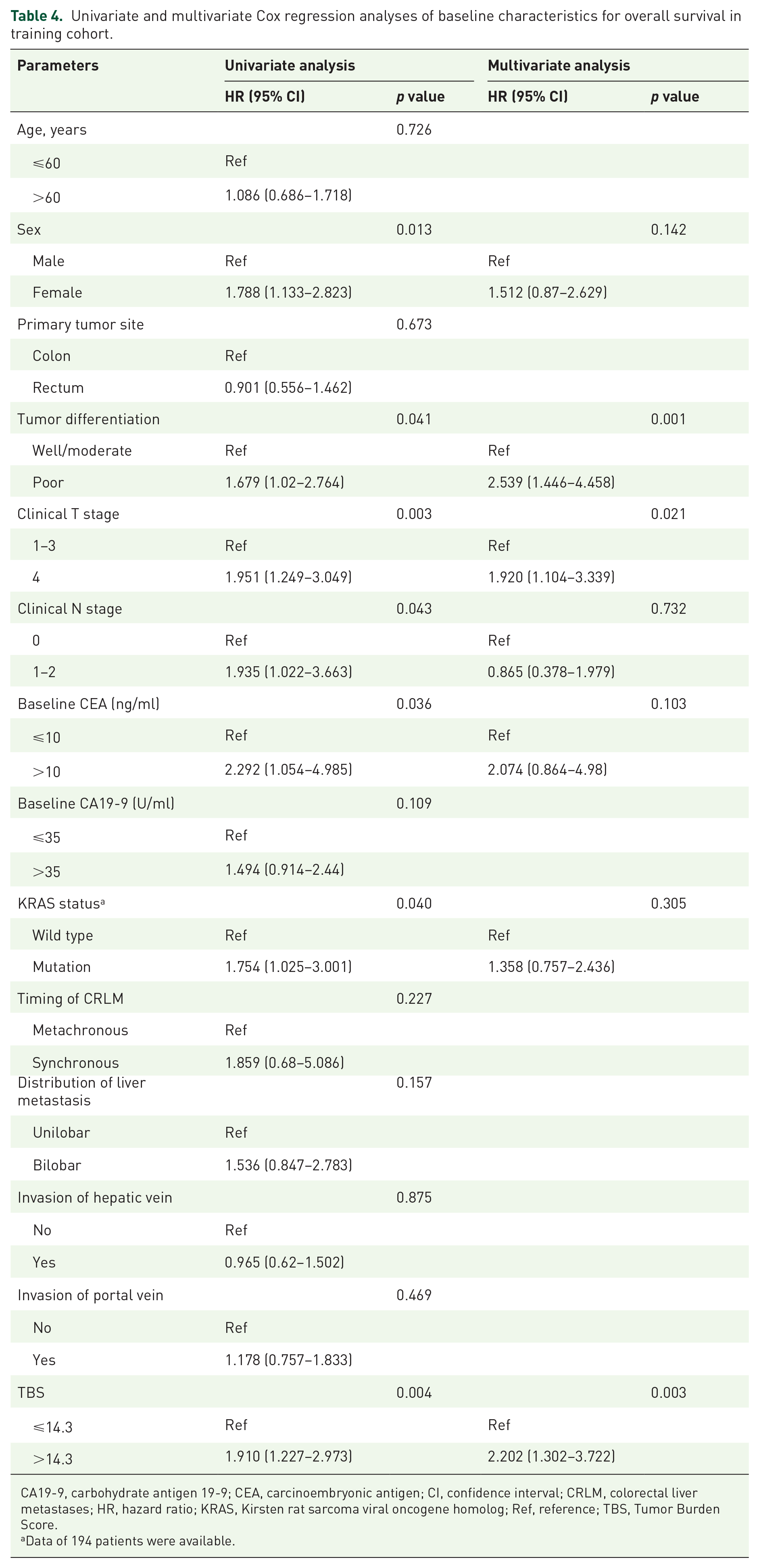
CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CI, confidence interval; CRLM, colorectal liver metastases; HR, hazard ratio; KRAS, Kirsten rat sarcoma viral oncogene homolog; Ref, reference; TBS, Tumor Burden Score.
Data of 194 patients were available.
Discussion
In this current study, we selected a newly established TBS and explored its value in predicting outcome of conversion therapy and some other associated factors and its role as a prognostic marker. Our final results revealed that baseline TBS had good discriminative ability to stratify patients into two groups. High TBS (>14.3) indicated the significantly lower possibility to achieve successful conversion outcome and also long-term survival than those with low TBS (⩽14.3). Moreover, multivariate logistic analysis and Cox analysis demonstrated that TBS was the independent factor of conversion outcome and OS.
According to previous researches investigating patients with initially resectable CRLM, the median size of largest CRLM was 2.5–3 cm, the median number of CRLM was 2, and the median TBS was 3.6,11–13 whereas our data revealed that they were 5.9 cm, 8, and 11.7, respectively. This suggested that TBS, which combines the size and number of liver metastases, reflected a much higher tumor burden in patients with initially unresectable CRLM than those with initially resectable CRLM. Therefore, a more accurate and effective TBS cut-off value is needed to distinguish patients with initially unresectable CRLM. We then conducted ROC analysis to stratify patients into two groups according to the effects of TBS on conversion rate. Based on the cut-off value which was defined as 14.3, conversion rate was 57.3% in low TBS group, whereas conversion rate was only 19.0% in high TBS group (Table 2). Importantly, this cut-off value also performed well in our validation cohort. Because of the strong correlation between TBS and conversion outcome, we consider TBS as 14.3 can be served as a powerful reference to define patients with initially unresectable CRLM as potentially resectable or non-resectable to be converted.
The strong correlation between TBS and conversion rate can be explained from two aspects. First, higher baseline TBS directly reflects more aggressive tumor burden and further remarkably increases the technical difficulty and the feasibility of curative liver resection. Although there is no consensus on the indications and contradictions of resection for CRLM, several latest guidelines suggested whether complete resection can be achieved and whether adequate liver volume can be remained as important factors for liver resection.17,18 For example, Shanghai international consensus on diagnosis and comprehensive treatment of CRLM 2019 suggested complete removal (R0) and preservation of adequate liver function (remnant liver volume ⩾ 30–40%) as one of the criteria for surgical treatment and vice versa. 18 Although the number and size of tumor have been excluded from the constantly evolving criteria, these two parameters are still closely related with the operability of liver metastases. Radical resection cannot be guaranteed in case of numerous lesions. Besides, large tumor tends to invade the major hepatic blood vessels, which is an important risk factor of inadequate liver volume. TBS, based on both the number and size of tumor, is a potential parameter to quantify the criteria for liver resection. In addition, our results revealed that high TBS is strongly correlated with bilobar metastases distribution as well as vascular invasion, further indicating the significant relation between high TBS and operability. Second, patients with increasing TBS reflected gradually declining ORR in our study, suggesting that increasing tumor burden was associated with poor chemotherapy sensitivity. As a result, patients with high TBS might receive less benefits from first-line systemic therapy, and thus were more likely to have failure conversion outcome compared with those with low TBS. Even if patients showed PR to down-sizing chemotherapy or gained considerable shrinkage of tumor, conversion rate was still significantly lower in high TBS group than in low TBS group (52.0% versus 78.2%, p = 0.003, Figure 3(a)). These implied that baseline tumor morphological characteristics exerted strong influences on treatment strategies and outcome.
While it is widely believed that response to chemotherapy is a powerful indicator for survival, 19 some studies have also demonstrated that response rate is significantly correlated with conversion rate20,21 and addition of targeted therapy, such as bevacizumab or cetuximab to chemotherapy, could further yield improvements in response rate and resectability.10,22–25 In the present study, the result of multivariate logistic regression revealed that poor chemotherapy response (SD or PD) and absence of targeted drug were two independent factors associated with failure conversion outcome. Our data also revealed that high TBS, as an another independent risk factor, was demonstrated to be significantly correlated with conversion outcome whatever chemotherapy response was, whether targeted therapy was implemented or whether KRAS mutated. The results indicated that TBS was more likely to be an inherent indicator reflecting conversion outcome to system treatment, no matter how effective the treatment regimen and KRAS status was. In addition to predict conversion outcome, TBS was also a prognostic predictor for OS. Several scoring systems have been established to stratify patients with initially resectable CRLM into different groups with different prognosis. For example, the original TBS model used 3 and 9 as two cut-off values to divide patients into three zones and showed good prognostic performance.11,13 Genetic and Morphological Evaluation (GAME) score is another scoring system invented for patients with initially resectable CRLM, which incorporated not only TBS but also some other clinicopathological characteristics. This model, also using the same cut-off values for TBS as the original model, demonstrated good discriminatory ability. 26 However, the cut-off values used in the above two models are not applicable for patients with initially unresectable CRLM because of their relatively much higher TBS, as we have discussed before. Hence, the larger cut-off value 14.3 was used to stratify patients, and the result demonstrated its excellent performance. Patients with TBS > 14.3 had significantly worse long-term OS compared with those with TBS ⩽ 14.3, which was also confirmed in the validation cohort. Also, TBS was independently correlated with OS together with tumor differentiation and clinical T stage.
Hence, TBS should be utilized to help determine the optimal individualized treatment strategies by considering the patients’ baseline characteristics, physical conditions, and the possible conversion outcome. For patients with low TBS and presenting good general conditions, comprehensive and intensive strategies could be assigned accordingly. Studies have reported that triplet regimen FOLFOXIRI, although at the expense of increased toxicity, confers superior efficacy in OS, recurrence-free survival (RFS), and ORR compared with standard doublet regimens FOLFOX and FOLFIRI.10,27–29 However, for patients with high TBS and poor underlying conditions, because of the low probability to achieve successful conversion, palliative and supporting therapy is more recommended. Taken together, we suggested MDT should carefully select the optimal treatment strategy for patients based on TBS and physical conditions.
There are some limitations to this retrospective study. First, the number of included patients may be inadequate, and selection bias may exist. Hence, larger amounts of patients are required for external validation. Second, several biomarkers were identified as crucial prognostic factors for survival and recurrence, including KRAS, NRAS, and BRAF, while effects of some other genes, such as phosphatidylinositol 3 kinase (PI3 K) and tumor protein P53 (TP53), were still under debate.30–33 In this study, we only included KRAS into analysis and future studies should take more gene status into consideration to obtain further understanding of the influence of these biomarkers on TBS and conversion outcome.
Conclusion
TBS presents significant association with conversion outcome after first-line systemic therapy in patients with initially unresectable CRLM. By stratifying patients into low and high TBS groups, TBS should be applied in clinical practice as an important parameter in the determination of optimal individualized treatment strategy.
Supplemental Material
sj-docx-1-tag-10.1177_17562848211066206 – Supplemental material for Application of Tumor Burden Score for predicting conversion outcome in patients with initially unresectable colorectal liver metastases after first-line systemic therapy
Supplemental material, sj-docx-1-tag-10.1177_17562848211066206 for Application of Tumor Burden Score for predicting conversion outcome in patients with initially unresectable colorectal liver metastases after first-line systemic therapy by Jianhong Peng, Yujun Liu, Weihao Li, Yuzhu Lin, Hui Sun, Zhizhong Pan, Xiaojun Wu, Wenhua Fan and Junzhong Lin in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-2-tag-10.1177_17562848211066206 – Supplemental material for Application of Tumor Burden Score for predicting conversion outcome in patients with initially unresectable colorectal liver metastases after first-line systemic therapy
Supplemental material, sj-tif-2-tag-10.1177_17562848211066206 for Application of Tumor Burden Score for predicting conversion outcome in patients with initially unresectable colorectal liver metastases after first-line systemic therapy by Jianhong Peng, Yujun Liu, Weihao Li, Yuzhu Lin, Hui Sun, Zhizhong Pan, Xiaojun Wu, Wenhua Fan and Junzhong Lin in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-3-tag-10.1177_17562848211066206 – Supplemental material for Application of Tumor Burden Score for predicting conversion outcome in patients with initially unresectable colorectal liver metastases after first-line systemic therapy
Supplemental material, sj-tif-3-tag-10.1177_17562848211066206 for Application of Tumor Burden Score for predicting conversion outcome in patients with initially unresectable colorectal liver metastases after first-line systemic therapy by Jianhong Peng, Yujun Liu, Weihao Li, Yuzhu Lin, Hui Sun, Zhizhong Pan, Xiaojun Wu, Wenhua Fan and Junzhong Lin in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-4-tag-10.1177_17562848211066206 – Supplemental material for Application of Tumor Burden Score for predicting conversion outcome in patients with initially unresectable colorectal liver metastases after first-line systemic therapy
Supplemental material, sj-tif-4-tag-10.1177_17562848211066206 for Application of Tumor Burden Score for predicting conversion outcome in patients with initially unresectable colorectal liver metastases after first-line systemic therapy by Jianhong Peng, Yujun Liu, Weihao Li, Yuzhu Lin, Hui Sun, Zhizhong Pan, Xiaojun Wu, Wenhua Fan and Junzhong Lin in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We greatly appreciate the help from all of our colleagues in the Department of Colorectal Surgery at Sun Yat-sen University Cancer Center who were involved in administering the treatments in the current study.
Author contributions
JHP, YJL, and WHL performed data analysis and drafted the manuscript; JHP, HS, and YJL collected the clinical data and follow-up results; ZZP, XJW, WHF and JZL supervised the current study; JZL and WHF proofread the manuscript. All authors read and approved the final manuscript.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by grants from the National Natural Science Foundation of China (no. 82003051 and no. 81772595).
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request. The authenticity of this article has been validated by uploading the key raw data onto the Research Data Deposit public platform (![]() ), with the approval RDD number as RDDA2021410973.
), with the approval RDD number as RDDA2021410973.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
