Abstract
Background:
Systemic therapy can cause loss of skeletal muscle mass in colorectal cancer (CRC) patients in the neoadjuvant and palliative settings. However, it is unknown how the body composition is changed by chemotherapy rendering unresectable CRC to resectable disease or how it affects the prognosis. This study aimed at elucidating the effects of systemic therapy on skeletal muscles and survival in stage IV CRC patients who underwent conversion therapy.
Methods:
We reviewed 98 stage IV CRC patients who received systemic therapy in our hospital. According to the treatment setting, patients were divided into the conversion, neoadjuvant chemotherapy (NAC), and palliation groups. The cross-sectional area of skeletal muscles at the third lumbar level and changes in the skeletal muscle index (SMI), defined as the area divided by height squared, during systemic therapy were compared among patient groups. The effects of these parameters on prognosis were analyzed in the conversion group.
Results:
The mean SMI increased by 9.4% during systemic therapy in the conversion group (n = 38), whereas it decreased by 5.9% in the NAC group (n = 18) and 3.7% in the palliation group (n = 42, p < 0.0001). Moreover, patients with increased SMI during systemic therapy had a better overall survival (OS) than those whose SMI decreased in the conversion group (p = 0.025). The increase in SMI was an independent predictor of favorable OS on multivariate analysis (hazard ratio 0.25).
Conclusions:
Stage IV CRC patients who underwent conversion to resection often had an increased SMI. On the other hand, a decrease in the SMI during systemic therapy was a negative prognostic factor in such patients.
Introduction
Sarcopenia is a syndrome characterized by a gradual and general loss of skeletal muscle mass and strength, and increased functional limitations.1,2 In addition to sarcopenia primarily caused by aging, secondary sarcopenia is being gradually recognized as a key component of other health conditions including cancer progression. 1 Sarcopenia, observed in 15–70% of cancer patients, is associated with compromised physical function, impaired quality of life, and reduced tolerance to treatments.3–6 Moreover, close relationships were found between sarcopenia and survival from different solid tumors.3,5,6 On the other hand, systemic therapy can generally induce sarcopenia by direct and indirect depletion of skeletal muscle mass. 7
Regarding colorectal cancer (CRC), a cross-sectional study reported that sarcopenia has a negative impact on survival following surgery for colorectal liver metastases. 8 In addition, several longitudinal studies also found that a reduction in skeletal muscle mass during systemic therapy is related to early disease progression or poor survival in metastatic CRC patients.9–13 In the non-metastatic setting, a low skeletal muscle index (SMI) or psoas muscle index (PMI) at lumbar levels before surgery led to a poor survival in CRC patients.14,15 A population-based study revealed that longitudinal decreases in muscle mass and density during the first 1–2 years after diagnosis are related to overall and cancer-specific mortalities in stage I–III CRC. 16 In another study, a decrease in PMI during adjuvant chemotherapy was associated with a poor prognosis in patients with stage III colon cancer. 17 Moreover, sarcopenia was a negative prognostic factor in patients with lower rectal cancer who received neoadjuvant or postoperative chemoradiation therapy.18,19 However, little is known about the relationships among body composition, survival, and systemic therapy leading to conversion to curative resection in patients with initially unresectable stage IV CRC.
In the current study, we investigated the effects of systemic therapy on skeletal muscles and survival outcomes in initially unresectable stage IV CRC patients who underwent conversion therapy in comparison with stage IV CRC patients who received systemic therapy in other settings.
Patients and methods
This is a retrospective observational study using prospectively collected data in combination with computed tomography (CT) images retrieved later. The study was approved by the ethical board of the University of Tokyo (3252–10). Written informed consent was waived because of the retrospective design.
Patients
From our database collected at the Department of Surgical Oncology, the University of Tokyo Hospital, we retrieved data of consecutive patients who underwent curative-intent surgery for primary CRC with distant organ metastases after systemic therapy between January 2011 and June 2020. We also collected data of unresectable stage IV CRC patients who received first-line systemic therapy without surgical therapy at our department during the same period in a consecutive manner for comparison.
According to the purpose of systemic therapy and consequence as described below, patients were divided into three groups: (a) the ‘conversion group’ that included patients who underwent curative-intent surgery after chemotherapy for initially unresectable disease; (b) the ‘NAC group’ that included patients who received neoadjuvant systemic therapy (NAC) for resectable disease followed by surgery; and (c) the ‘palliation group’ that included patients whose disease remained unresectable even after systemic therapy. We excluded patients who underwent major surgery during systemic therapy, those with tumors invading to the abdominal wall, those who did not undergo CT scans at the lumbar levels before or after systemic therapy, those who received a short duration of systemic therapy for less than 8 weeks until post-treatment CT evaluation as explained below, and those who demonstrated a clinical complete response.
Systemic therapy for stage IV colorectal cancer and evaluation of tumor response
FOLFOX [oxaliplatin at 85 mg/m2 and folinic acid at 200 mg/m2, followed by 5-fluorouracil (5-FU) as a 400 mg/m2 intravenous bolus and a 2400 mg/m2 infusion over 46 h] was administered every 2 weeks. 20 CAPOX (oxaliplatin at 130 mg/m2 over 2 h on day 1 plus oral capecitabine at 1000 mg/m2 twice daily on days 1–14) was repeated every 3 weeks. 21 SOX (oxaliplatin at 130 mg/m2 over 2 h on day 1 plus oral S-1 at 40 mg/m2 twice daily on days 1–14) was repeated every 3 weeks. 22 FOLFIRI (150 mg/m2 of irinotecan, and 200 mg/m2 of folinic acid, followed by 5-FU as a 400 mg/m2 intravenous bolus and a 2400 mg/m2 infusion over 46 h) was administered every 2 weeks. 23 FOLFOXIRI (irinotecan at 150 mg/m2, followed by oxaliplatin at 85 mg/m2 and 200 mg/m2 of folinic acid over 2 h, and then and a 3000 mg/m2 infusion of 5-FU over 46 h) was administered every 2 weeks. 24 IRIS (irinotecan at 125 mg/m2 over 90 min on day 1 and day 15 plus oral S-1 at 40–60 mg twice daily on days 1–14) was repeated every 4 weeks. 25 Targeted biologics, such as anti-vascular endothelial growth factor (VEGF) antibodies (bevacizumab) and anti-epidermal growth factor receptor (EGFR) antibodies (panitumumab and cetuximab) were usually added to one of the above cytotoxic drug combinations based on the RAS status of tumors.26,27 Dose reductions and the cessation of chemotherapy were at the doctors’ discretion, and based on the patients’ condition and preference.
In order to evaluate tumor responses, CT scans were performed before systemic therapy, and every 2–3 months thereafter during the treatment. When increases in serum tumor markers were noted in an accelerated manner, or symptoms suggestive of tumor progression newly developed, CT scans and other imaging modalities such as magnetic resonance imaging and positron emission tomography, were additionally performed.
Resectability in stage IV CRC was assessed periodically in joint conferences or cancer board meetings by multidisciplinary teams comprising gastrointestinal, hepatobiliary, and pulmonary surgeons and radiologists at our hospital. The indications for chemotherapy and surgical resection were discussed based on resectability which depends on the possibility of complete removal of metastasized lesions while preserving sufficient function of the target organ and without sacrificing the adjacent essential organs. Conversion was defined as macroscopically complete removal (R0/R1 resection) for primary and all metastasized lesions that were initially unresectable. NAC was typically administered to CRC patients who had initially resectable metastases for a total of 2–4 months.
The best response was graded according to the Response Evaluation Criteria In Solid Tumors (RECIST) criteria version 1.1. 28
Follow-up after surgery
In patients who underwent radical surgery, postoperative adjuvant chemotherapy was usually recommended, but practically performed based on the doctor’s considerations and patient’s preference. 5-FU (+ folinate) monotherapy or in combination with oxaliplatin (FOLFOX or CAPOX) was administered for 6 months. 27 Patients were followed by physical examination, serum tumor marker measurement every 3 months, CT scans every 6 months, and an annual colonoscopy. When patients exhibited symptoms and/or had abnormal laboratory test results that suggested a recurrent disease, imaging studies were additionally performed. Local and systemic treatments for recurrence were determined based on the tumor’s size, location and rate of growth, preceding therapies, and patient’s preference and condition.
Relapse-free survival (RFS) was measured from the date of the complete removal of cancer until recurrence and censored at death or last follow-up. Overall survival (OS) was defined as the time between the initiation of treatment and death due to any cause. Surviving patients were censored at the last time point of direct or indirect contact.
Sarcopenia measurement
The cross-sectional area of skeletal muscles at the third lumbar vertebra level on CT imaging was visualized within a range of −29 to 150 Hounsfield unit using the Ziostation2 system (Ziosoft, Tokyo, Japan), and calculated using Image J software (National Institutes of Health [NIH], Bethesda, MD, USA). The SMI was calculated by normalizing the cross-sectional areas of skeletal muscles for height squared (cm2/m2). 29 Considering ethnicity, sarcopenia was defined using sex-specific cutoffs for the SMI, <42 cm2/m2 for men and <38 cm2/m2 for women, according to the Japanese Society of Hepatology guidelines. 30
The SMI was analyzed before and after systemic therapy for individual patients. For calculating the post-treatment SMI, we used CT images at the first evaluation at least after 8 weeks of systemic therapy. The change in SMI, that is, the relative ratio of post-treatment SMI to pre-treatment SMI was also calculated.
Other data collection
The demographic and clinicopathological parameters such as sex, age, Eastern Cooperative Oncology Group Performance Status (ECOG PS), Charlson comorbidity index, 31 serum albumin, carcinoembryonic antigen (CEA; upper limit of normal 5 ng/ml) and carbohydrate antigen (CA) 19-9 (upper limit of normal 37 U/ml) levels before chemotherapy, the intra-tumoral status of microsatellite instability (MSI), RAS (KRAS and NRAS codon 12, 13, 59, 61, 117, and 146), and BRAF (codon 600), if available, primary tumor location, metastasized organs, the duration of chemotherapy before surgery or until the best response, and total number of palliative chemotherapy lines were investigated.
Statistical analyses
Continuous variables were compared by the Kruskal–Wallis or Wilcoxon rank sum test. The Steel–Dwass test was further used for multiple comparisons. Categorical variables were compared by Yates-corrected chi-squared or Fisher’s exact test where appropriate. The pre-treatment and post-treatment SMI was compared using the paired t test or Wilcoxon signed-rank test. Survival curves were analyzed using the Kaplan–Meier method and compared by the log-rank test. To identify prognostic factors, univariate analysis using a Cox proportional hazards model was employed, where variables were dichotomized. Then, only the variables with p < 0.10 were subjected to the multivariate analysis. A p value < 0.05 was considered significant. All statistical analysis data were obtained using JMP pro 15.1.0 (SAS Institute Inc., Cary, NC, USA).
Results
Patient characteristics
One hundred and thirty-two patients underwent systemic therapy for stage IV CRC during the study period. After excluding 34 patients who were unsuitable for the analyses, we compared 38 patients in the conversion group, 18 in the NAC group, and 42 in the palliation group (Figure 1).

Study flow for patient selection.
The patient background according to the treatment setting is shown in Table 1. The subjects included 58 men and 40 women, and the median age was 64 years old. There were more male patients in the palliation group than in the other groups (p = 0.009). RAS wild-type tumors were more frequent in the conversion group (70%) than the NAC and palliation groups (41% and 38%, p = 0.020). The conversion group was also characterized by frequent liver metastases (97%), whereas the palliation group had frequent metastases to the lung (43%) and distant lymph nodes (55%). All metastasized lesions in the NAC group were confined to a single organ. Regarding first-line therapy, the majority of backbone regimens (83–100%) were oxaliplatin-based regardless of treatment group. All patients in the conversion group received targeted antibodies, whereas they were administered to 71–78% of patients in the NAC and palliation groups (p = 0.005). The duration of systemic therapy until the evaluation of the post-treatment SMI was the shortest in the NAC group due to its therapeutic purpose. The tumor response rate to chemotherapy reached 88–90% in the conversion and NAC groups in contrast to that in the palliation group of only 50% (p = 0.002). No significant difference was observed in other parameters among the groups.
Patient characteristics.

Excluding unknown cases.
Evaluated by Response Evaluation Criteria in Solid Tumors, ver 1.1.
Comparison between the conversion and NAC groups.
Excluding patients without recurrence and unknown cases.
CA 19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; ECOG PS, Eastern Cooperative Oncology Group Performance Status; MSI, microsatellite instability; N/A, not available; NAC, neoadjuvant chemotherapy; PD, progressive disease; PR, partial response; SD, stable disease.
Change in SMI during systemic therapy
Sex-stratified SMI was compared before and after systemic therapy for each treatment group. The mean SMI before systemic therapy did not differ among the groups: 38.8, 41.7, and 39.9 for the conversion, NAC, and palliation groups, respectively, in men (p = 0.67), and 30.2, 34.0, and 28.0 for the conversion, NAC, and palliation groups, respectively, in women (p = 0.080). The frequency of sarcopenic patients at baseline was 79% in the conversion group, 78% in the NAC group, and 71% in the palliation group (p = 0.95). In general, the mean SMI increased after systemic therapy in the conversion group whereas it decreased in the NAC and palliation groups, as shown in Figure 2. The increase in the mean SMI in male patients in the conversion group and the decrease in the mean SMI in female patients in the NAC group were significant (p = 0.005 and p = 0.007, respectively).

Skeletal muscle index (SMI) before and after systemic therapy according to treatment group. (A) SMI data for men and (B) SMI data for women.
The comparative results of the relative change in SMI from baseline to post-treatment among the treatment groups are shown in Figure 3. After systemic therapy, the mean SMI increased by 9.4% in the conversion group, whereas it decreased by 5.9% in the NAC group and 3.7% in the palliation group (p < 0.0001). A treatment-induced decrease in the SMI was observed in 83% of patients in the NAC group and 60% in the palliation group. Conversely, 71% of patients (27 of 38) had an increased SMI after systemic therapy in the conversion group (p = 0.001, data not shown); among them, the SMI recovered to the normal range after systemic therapy in two patients who were initially diagnosed with sarcopenia. Representative CT images at the third lumbar level before and after systemic therapy in the three groups are shown in Figure 4.
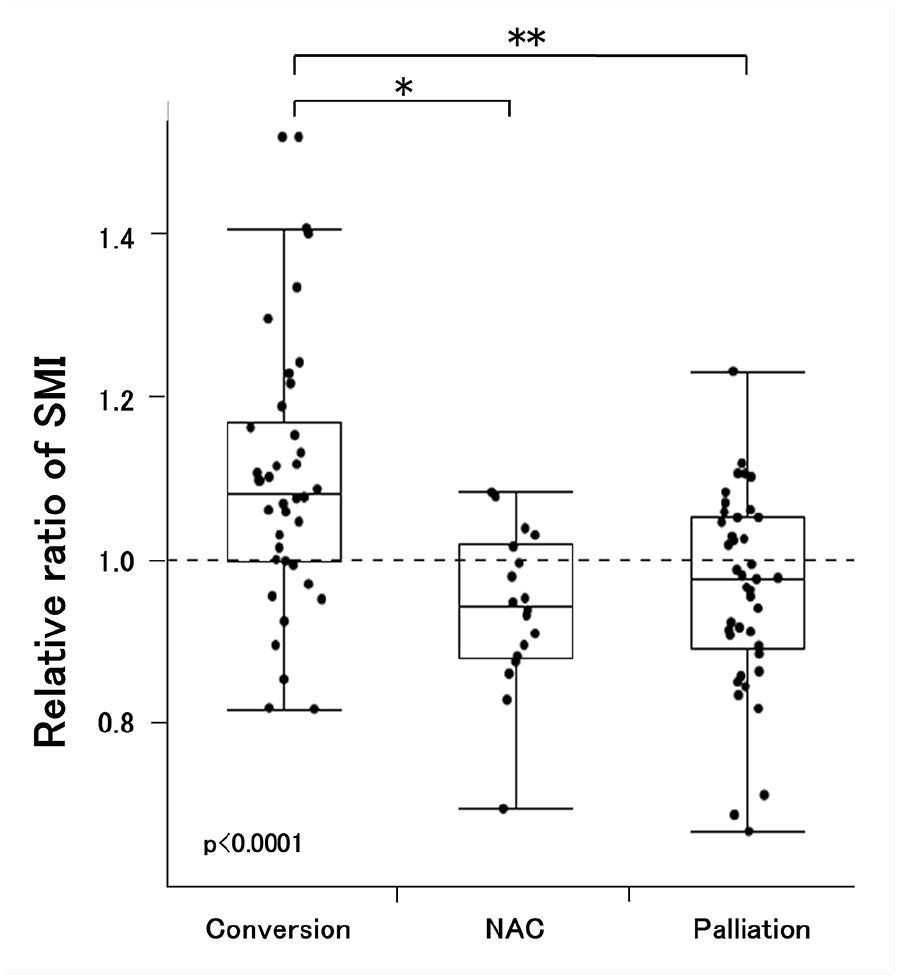
Box plots for the relative changes in the skeletal muscle index (SMI) after systemic therapy according to treatment group. Dots for individual data were overlaid. 1.0 is the baseline.

Examples of computed tomography (CT) images before and after systemic therapy. CT images at the third lumbar level were obtained from same patients in each group. (A–C) Before systemic therapy. (D–F) After systemic therapy (at the first evaluation). (A, D) Conversion group (37 years old, male). (B, E) NAC group (52 years old, female). (C, F) Palliation group (74 years old, male).
Impact of increased SMI on survival in patients undergoing conversion therapy
We next investigated how the change in SMI affected the prognosis of stage IV CRC patients who underwent conversion therapy. We compared the survival outcomes between patients with an increased SMI and those with a decreased SMI after systemic therapy in the conversion group. The median follow-up period was 44.3 months. As shown in Figure 5A, the change in SMI did not seem to affect RFS. On the other hand, patients with an increased SMI had a significantly better OS than those who lost SMI (p = 0.025 by the log-rank test, Figure 5B). The 3-year OS rate was 80% for the former and 47% for the latter.
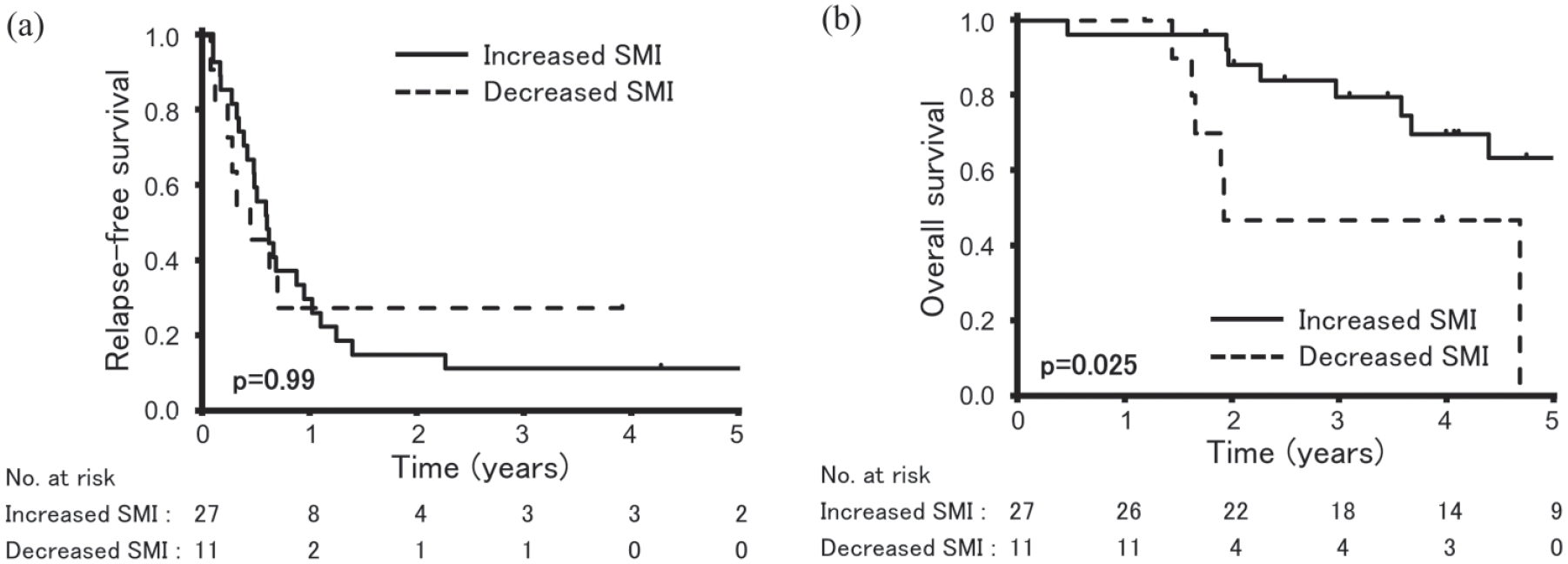
Survival curve analyses in the conversion group according to the relative changes in the skeletal muscle index (SMI) after systemic therapy. (A) Relapse-free survival and (B) overall survival.
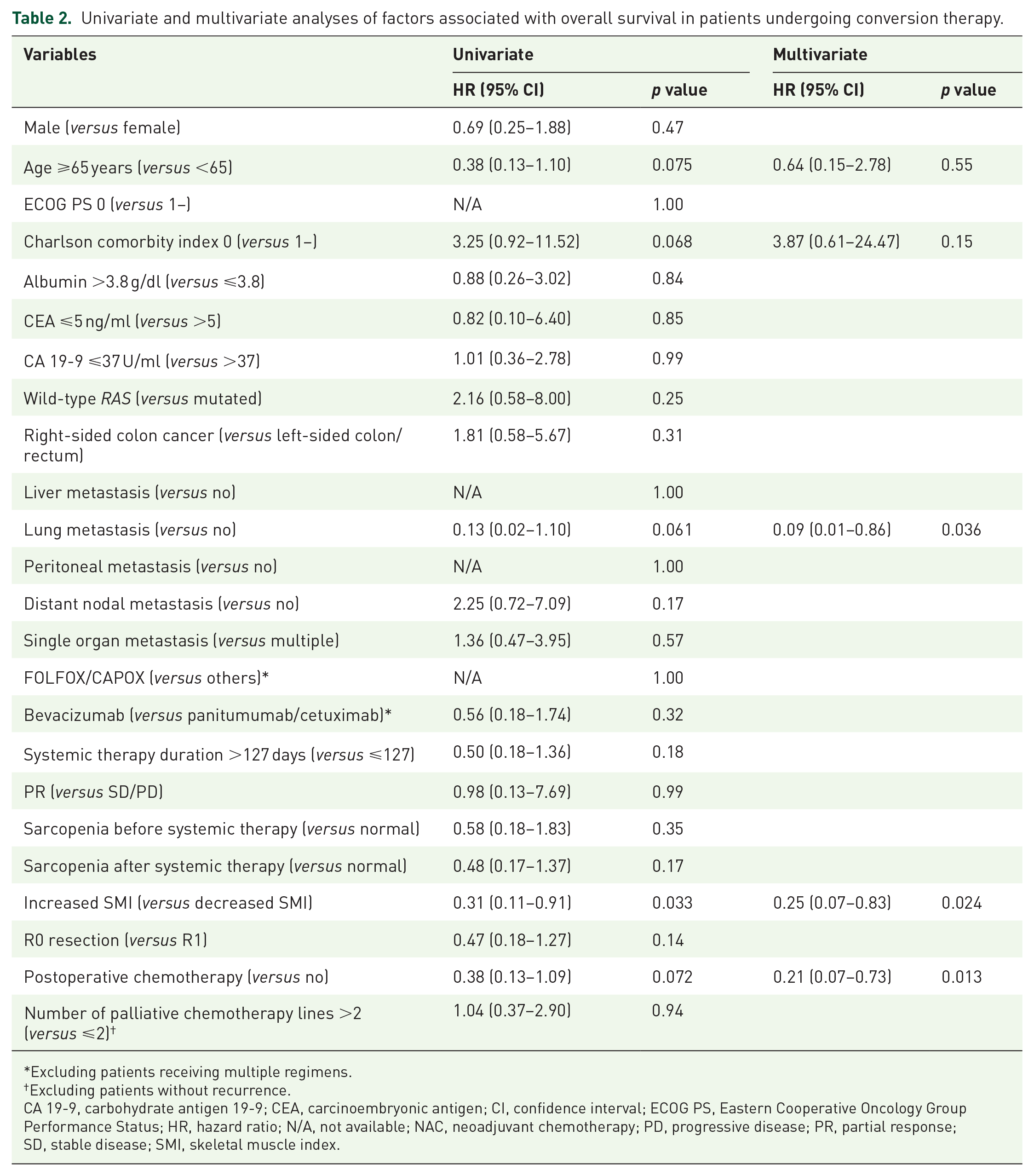
In response to the separated OS curves stratified by the change in SMI, we performed univariate and multivariate analyses of factors associated with OS in the conversion group. As shown in Table 2, an old age, Charlson comorbidity index ⩾1, lung metastasis, an increased SMI, and postoperative chemotherapy were significant variables associated with OS by univariate analysis, whereas other variables such as preoperative therapy-related factors and sarcopenia before and after systemic therapy were not. On multivariate analysis, lung metastasis, increased SMI, and postoperative chemotherapy were confirmed to be independent prognostic factors (hazard ratio 0.09, 0.25 and 0.21, respectively, Table 2).
Univariate and multivariate analyses of factors associated with overall survival in patients undergoing conversion therapy.
Excluding patients receiving multiple regimens.
Excluding patients without recurrence.
CA 19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group Performance Status; HR, hazard ratio; N/A, not available; NAC, neoadjuvant chemotherapy; PD, progressive disease; PR, partial response; SD, stable disease; SMI, skeletal muscle index.
Discussion
This is the first study to demonstrate that as many as 71% of patients who underwent conversion therapy for initially unresectable CRC gained skeletal muscle mass during systemic therapy, as evaluated by SMI. This change in body composition was in clear contrast to that in patients who received NAC or palliative systemic therapy.
Loss of muscle mass is multifactorial. It can be induced not only by systemic treatment but also be reflective of cancer progression accompanied by proinflammatory cytokine release and muscle protein catabolism.7,32 Accordingly, preceding studies demonstrated that the response to treatment and reduction in tumor burden are associated with less muscle loss in patients with lung or gastric cancer receiving systemic therapy.33,34 Of note, Stene et al. reported that nearly half of the patients had stable or increased muscle mass during palliative systemic therapy for stage IIIB–IV non-small cell lung cancer, which corresponded to the response to treatment. 33 Based on the patients in the conversion group in our study, a marked reduction in tumor burden may lead to the recovery of skeletal muscle mass. The types of targeted therapy and cytotoxic drugs can also affect the body composition. Anti-EGFR antibodies frequently cause clinically meaningful mucositis, and thereby reduce food intake.35,36 However, there was no imbalance between the use of anti-EGFR versus anti-VEGF antibodies among the treatment groups. Irinotecan is known for diarrhea as a dose-limiting adverse event, 37 but the drug was used only in limited numbers of patients in the current study. Therefore, their distinct changes in mean SMI did not seem to be affected by these factors.
In CRC with initially unresectable metastases, patients with lower tumor burden are considered to have more chance to undergo successful conversion therapy. In addition, patients who had the potential to improve SMI and thereby responded well to systemic therapy could receive curative-intent surgery, which is also a possible selection bias of the conversion group. For example, peritoneal metastases could considerably interfere with nutrient absorption, causing the decrease in SMI. In the palliation group, frequent peritoneal metastases (21%) might contribute to the decrease in SMI, and deprive patients of an opportunity to undergo conversion to resection.
Consistent with a previous study on resectable colorectal liver metastases, 38 NAC reduced skeletal muscle mass in many patients in the current study. We consider that unlike conversion therapy, NAC had a more profound effect on the decrease in SMI than the SMI-increasing effect resulting from tumor shrinkage. Of note, the magnitude of the decrease in SMI was slightly greater in the NAC group (5.9%) than in the palliation group (3.7%), although the duration of systemic therapy was shorter. Interestingly, Daly et al. reported similar results in patients with foregut cancer by comparing NAC and palliative systemic therapy. 39 They evaluated changes in many body composition metrics per 100 days, and found that patients receiving NAC had a greater loss in skeletal muscle mass (7.3%) than those who received palliative therapy (2.8%, p < 0.001). However, the reasons for their findings were not discussed probably because of the heterogeneity of cancer types. 39 Why NAC causes a marked loss of skeletal muscle in CRC and other malignancies needs to be clarified.
A growing number of studies have explored the relationships between body composition metrics and prognosis in different malignancies. In unresectable stage IV CRC, Miyamoto et al. reported that patients with skeletal muscle loss >5% after systemic therapy had a poorer prognosis than the other patients. 9 Blauwhoff-Buskermolen et al. performed a similar analysis, concluding that the loss of muscle mass during the 3 months of systemic therapy was associated with low survival. 10 In the CAIRO-3 trial investigating first-line bevacizumab and oxaliplatin-including regimens for metastatic CRC, a decrease in SMI during the treatment was related to early disease progression and reduced survival. 11 Even in later-line chemotherapy, skeletal muscle loss during regorafenib or trifluridine/tipiracil was associated with OS in two recent retrospective studies, both of which also showed that only patients who received regorafenib showed a significant reduction in skeletal muscle.12,13 Regarding resectable stage IV CRC, van Vledder et al. reported a correlation between preoperative sarcopenic status and poor RFS or OS after hepatectomy for colorectal liver metastasis. 8 Consistent with these studies, a decreased SMI was significantly associated with an unfavorable OS among CRC patients who underwent conversion therapy in our study. On the other hand, the change in SMI was not directly related to RFS. A majority of the recurrence after conversion occurred early after surgery; at the time of the first relapse, patients usually did not manifest symptoms or change in body composition. In contrast, OS is generally influenced by the tolerability of systemic and local therapies and physical/nutritional status. These could explain why RFS was independent of the change in SMI during preoperative therapy in the current study.
In addition to the increased SMI, the presence of lung metastases was a significant factor associated with better OS. In an analysis of >4000 CRC patients with metastases in a single organ using prospective randomized trials, patients with lung metastasis had better OS than those with metastases in the liver only. 40 In patients with colorectal liver metastasis, extrahepatic metastases used to be reported as a poor prognostic factor.41,42 However, recent studies demonstrated that patients who had resections of liver and lung metastases had similar or even better OS than those who underwent removal of isolated liver metastases.43–45 Because many patients received preoperative systemic therapy in the above studies,43,44 the findings may be affected by the selection bias that pulmonary metastasectomy was performed only when they responded well to the treatment. Not surprisingly, the conversion group comprising patients treated in a similar manner showed a consistent result with their works.
The implementation of postoperative chemotherapy also reduced the risk of overall mortality in conversion therapy for CRC according to multivariate analysis. We previously reported that postoperative chemotherapy was a favorable prognostic factor in a cohort of CRC patients who received preoperative radiation therapy and/or chemotherapy. 46 The importance of the continuation of systemic therapy was again demonstrated in the current study.
Due to its retrospective nature, this study has several other limitations. The study subjects comprised a small number of patients, particularly those who underwent NAC. There were many biases in the patient background such as sex, RAS status, metastasized organs, use of targeted antibodies, duration of treatment, and response to systemic therapy among the treatment groups. Data on MSI status and BRAF mutation were missing in many patients because the costs of these genetic testings were not covered by the Japanese health insurance system until 2018. FOLFOXIRI (plus bevacizumab) was chosen in only 5% of patients probably because the Pan-Asian adapted ESMO consensus guidelines first recommended the triplet regimen in Asia in 2018. 27 We excluded patients receiving systemic therapy for only a short period (<8 weeks) and those in whom a complete response was achieved without surgery, which may lead to selection bias. In addition, the slice levels and angles of lumbar CT scans were not precisely matched between pre and post-treatment evaluations in each patient or among patients. The dates of CT did not always coincide with those of the initiation and/or ending of systemic therapy. Sarcopenia is not evaluated only by quantification of muscle mass; other metrics by diagnostic imaging tools, such as overall skeletal muscle mass, non-imaging diagnostic parameters, including physical function, and physical performance assessed by dynamometers, including grip and quadriceps strengths, were not considered in the current study. Moreover, the cut-off points to define sarcopenia vary among studies, and our cut-off defined by the Japanese Society of Hepatology guidelines 30 for Japanese patients is uncommon; however, we mostly analyzed and discussed the change in SMI during systemic therapy in order to minimize the influences of sex difference and different criteria for sarcopenia on the interpretation of the results.
In conclusion, the current study was the first to demonstrate the frequent increase in SMI during first-line systemic therapy changing formerly unresectable stage IV CRC to resectable disease. As such an increase in SMI also conveys a survival benefit, it is important to make efforts to preserve muscle mass by meticulous approaches, such as nutritional support, muscle exercise programmes, and pharmacological intervention1,7 even during systemic therapy in patients with metastatic CRC.
Footnotes
Acknowledgements
The authors thank the Medical English Service (Kyoto, Japan) for English editing.
Conflict of interest
The authors declare that there is no conflict of interest.
Data availability
The datasets generated and/or analyzed during the current study are not publicly available because they are derived from the patient database of the hospital and hence are subject to confidentiality, but are available from the corresponding author on reasonable request.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants-in-Aid for Scientific Research [C: grant number; 18K07194, C: grant number; 19K09114, C: grant number; 19K09115, and Challenging Research (Exploratory): grant number: 20K21626] from the Japan Society for the Promotion of Science. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
