Abstract
Background:
Polymerase chain reaction (PCR) is a sensitive test for diagnosing Clostridioides difficile infection (CDI) and could remain positive following resolution of CDI. The kinetics of PCR positivity following antibiotics for CDI is unknown. We studied this and whether it predicted CDI recurrence.
Methods:
Adults with CDI from October 2009 to May 2017 were included. Serial stool samples within 60 days of treatment were collected. Recurrent CDI was defined as diarrhea after interim symptom resolution with positive stool PCR within 56 or 90 days of treatment completion. Contingency table analysis was used to assess the risk of recurrence.
Results:
Fifty patients were included [median age: 51 (range = 20–86) years, 66% women]. Treatment given was metronidazole, 50% (25); vancomycin, 44% (22); both, 4% (2); and fidaxomicin, 2% (1). Median duration of treatment for all 50 patients was 14 (range = 8–60) days. The median duration of treatment in patients who got prolonged therapy (>14 days) (n = 10) was 47 (range = 18–60) days. Median time to negative PCR was 9 (95% CI, 7–14) days from treatment initiation, which did not differ by antibiotics given (p = 0.5). A positive PCR during or after treatment was associated with a higher risk of recurrence at 56 days (p = 0.02) and at 90 days (p = 0.009).
Conclusion:
The median time to negative PCR in CDI was 9 days from treatment initiation. The PCR positivity during or after treatment may be useful for recurrence prediction; larger studies are needed to validate these results.
Introduction
Clostridioides difficile infection (CDI) is the commonest health care–associated infection, with increasing incidence of both primary and multiple recurrent CDI.1–3 Several stool-based diagnostic tests are available, including enzyme immunoassay (EIA) for glutamate dehydrogenase (GDH), EIA for toxin, cell cytotoxicity assay, and culture and molecular tests such as polymerase chain reaction (PCR) for toxin genes. The PCR has higher sensitivity (87–92%) than other tests, but cannot distinguish between infection and colonization.4,5 Currently, nearly half of the health care facilities in the United States use molecular tests for C difficile diagnosis.6,7
A positive PCR with typical clinical symptoms of CDI in the absence of alternative diagnoses is considered diagnostic. Interpretation of a persistent or repeatedly positive PCR during or after treatment of CDI is challenging; it could represent clinically insignificant delayed clearance of the bacteria, recolonization, or recurrence. The kinetics of a positive PCR test, particularly the duration over which it remains positive during or after treatment, and its utility in predicting CDI recurrence are largely unknown. One study using toxigenic culture found 21% and 36% of patients treated with vancomycin and metronidazole, respectively, had a positive stool test at the end of therapy. 8 In another study of CDI patients with initial positive toxin EIA, 3 of 14 and 14 of 26 patients remained toxin positive when retested within 14 and 30 days of treatment, respectively. 6 Patients received up to 14 days of metronidazole and vancomycin, although the results were not subcategorized based on treatment. In another study, the predictive ability of repeat EIA and PCR within 7 days of treatment completion was reported. 9 Of the 129 patients initially positive by EIA and with available repeat tests, 49 (38%) remained positive. In those diagnosed by PCR, 19 of 35 patients (54%) remained positive within 7 days. Details on treatment and clinical symptoms were not reported in this study. None of these studies assessed whether persistent or repeatedly positive tests predicted recurrence.
We conducted a prospective study to evaluate the kinetics of PCR test positivity during and after antibiotic therapy for CDI and to study the association between PCR positivity and recurrent CDI.
Methods
Patients aged 18–99 years seen at our institution with diarrhea and a positive C difficile PCR from October 2009 to May 2017 were eligible for the study. Diarrhea was documented prospectively as >3 watery bowel movements in 24 h. Patients were contacted for inclusion, and written consent was obtained. Patients were asked to submit five sequential stool samples (at day 2–4, day 6–8, day 12–16, day 19–23, and day 52–60 after initiation of treatment) and had weekly follow-up phone calls. Samples submitted outside this time frame and additional clinically indicated samples up to 90 days following treatment initiation were also included in the analysis. Positive samples without symptoms were considered colonization, and treatment was not offered. Positive samples with symptoms were considered diagnostic for recurrence and were not used as predictors of recurrence. Details regarding treatment and recurrences were obtained from the patient and supplemented by information from the medical records.
Recurrent CDI (within 56 and 90 days of completion of previous treatment) was defined as diarrhea with a positive stool PCR after interim symptom resolution. Although 56 days is generally used as the cutoff for recurrence, the utility of this cutoff has been challenged.10–12 Hence, we report recurrence both through 56 days and through the end of follow-up (90 days). Systemic non-CDI antibiotic exposure during follow-up was also recorded and analyzed as a covariate. As this was a pilot study, a sample size of convenience was taken. The reporting of this study conforms to the STROBE statement (attached as a Supplementary file). 13 The study was approved by the Mayo Clinic Institutional Review Board (IRB ID: 09-003250).
Biospecimens
Stool samples were obtained as per standard procedure. All samples were tested using C difficile PCR assay [real-time PCR using LightCycler and Fluorescent Resonance Energy Transfer (Roche Molecular Systems, Inc., CA)]. This test was developed and its performance characteristics was determined by Mayo Clinic in a manner consistent with Clinical Laboratory Improvement Amendments (CLIA) requirements. The assay detects the regulatory gene (tcdC) which is a part of a pathogenicity locus (PaLoc) responsible for producing toxins A and B. 14 This pathogenicity locus is lacking in the nontoxigenic isolates. Using toxigenic culture as the gold standard, sensitivity and specificity of the assay are 86% and 97% respectively. 15 The tcdC gene is mutated in the ribotype 027 (RT027) strain; the assay, however, detects this mutated form as well.16,17 Follow-up research PCR results were not posted to the medical record and were not used for medical decision-making.
Outcomes
The primary outcome was time to first negative PCR from initiation of treatment. Secondary outcomes were time to first negative PCR categorized by treatment received (metronidazole versus vancomycin) and risk of recurrent CDI based on a positive PCR test during or after treatment completion. For risk of recurrence, the sample used in diagnosing the recurrent episode and any samples thereafter were excluded.
We also assessed asymptomatic C difficile PCR positivity (⩾1 positive PCR test without symptoms) during treatment, from day 1–7, day 8–14, day 15–21, and beyond day 21 after treatment completion. If a patient had multiple samples within one time frame, only the first sample in that time frame was included. Samples used in diagnosing recurrences and any samples thereafter were excluded.
Statistical analysis
Data analyses were performed using JMP version 14.0 (from SAS). Descriptive analyses were performed for demographics and other clinical variables. The time to resolution of symptoms in patients treated with metronidazole and vancomycin was compared using Wilcoxon rank sum test. Kaplan–Meier survival analysis was used to calculate time to first negative PCR after initiation of treatment. The time to first negative PCR in patients treated with metronidazole and vancomycin was compared by the log-rank test. Patients given fidaxomicin or combined metronidazole and vancomycin therapy were excluded from this analysis. A multivariable Cox proportional hazards regression model was used to evaluate the effect of age, sex, and treatment on time to first negative PCR. Chi-square and Fisher’s exact test were used to compare the risk of CDI recurrence in patients with a positive PCR versus those with negative PCRs (during treatment, after treatment completion, and either during or after treatment) and to compare asymptomatic C difficile positivity rates in patients treated with metronidazole and vancomycin. Parameters of diagnostic test performance were calculated for positive PCR during and after treatment in predicting recurrence. We assessed the effect of positive PCR on the risk of recurrence after controlling for systemic, non-CDI antibiotic exposure, the strongest risk factor for CDI, using multivariate logistic regression. For all analyses, p < 0.05 was considered statistically significant.
Results
Sixty patients were enrolled in the study, of which 10 dropped out; three patients withdrew consent and seven did not submit any samples. The 50 patients included in the final analysis had a median age of 51 (range = 20–86) years and 66% were women. Of these patients, 24% (12) had inflammatory bowel disease and 24% (12) had a prior history of CDI. Treatment for the current episode was metronidazole in 50% (25), vancomycin in 44% (22), both metronidazole and vancomycin in 4% (2), and fidaxomicin in 2% (1). Median duration of treatment was 14 (range = 8–60) days. Overall, 20% of patients (10) received a prolonged (>14 days) course of treatment. All patients completed their initial treatment regimen; none received second-line therapy due to clinical nonresponse or adverse events. The median time to resolution of symptoms was 12 (range = 0–68) days from the beginning of treatment, which did not differ by treatment given [metronidazole: 11 (range = 0–38) days, vancomycin: 15 (range = 0–68) days; p = 0.56)]. On removing outliers, the median time to resolution of symptoms was 12 (range = 0–32) days and was not different by treatment given [metronidazole: 10.5 (range = 0–32), vancomycin: 14 (range = 0–21) days; p = 0.59)].
Kinetics of PCR positivity
All patients had a positive PCR test for C difficile at the time of enrollment; 41 patients (82%) submitted three or more samples for the study; 14 patients (28%) submitted all five samples. The timeline of submission of stool samples and the results of PCR testing are depicted in Figure 1. Overall, 39 patients (78%) submitted at least one stool sample during treatment and 42 patients (84%) submitted at least one sample after treatment completion. Using Kaplan–Meier survival analysis, the percent of patients with a positive PCR test decreased from 100% at the start of treatment to 14.9% at day 28; 5.9% remained positive beyond day 60 (Figure 2). Overall, six patients (12%) remained persistently PCR positive throughout the study (four were treated initially with metronidazole and two with vancomycin).
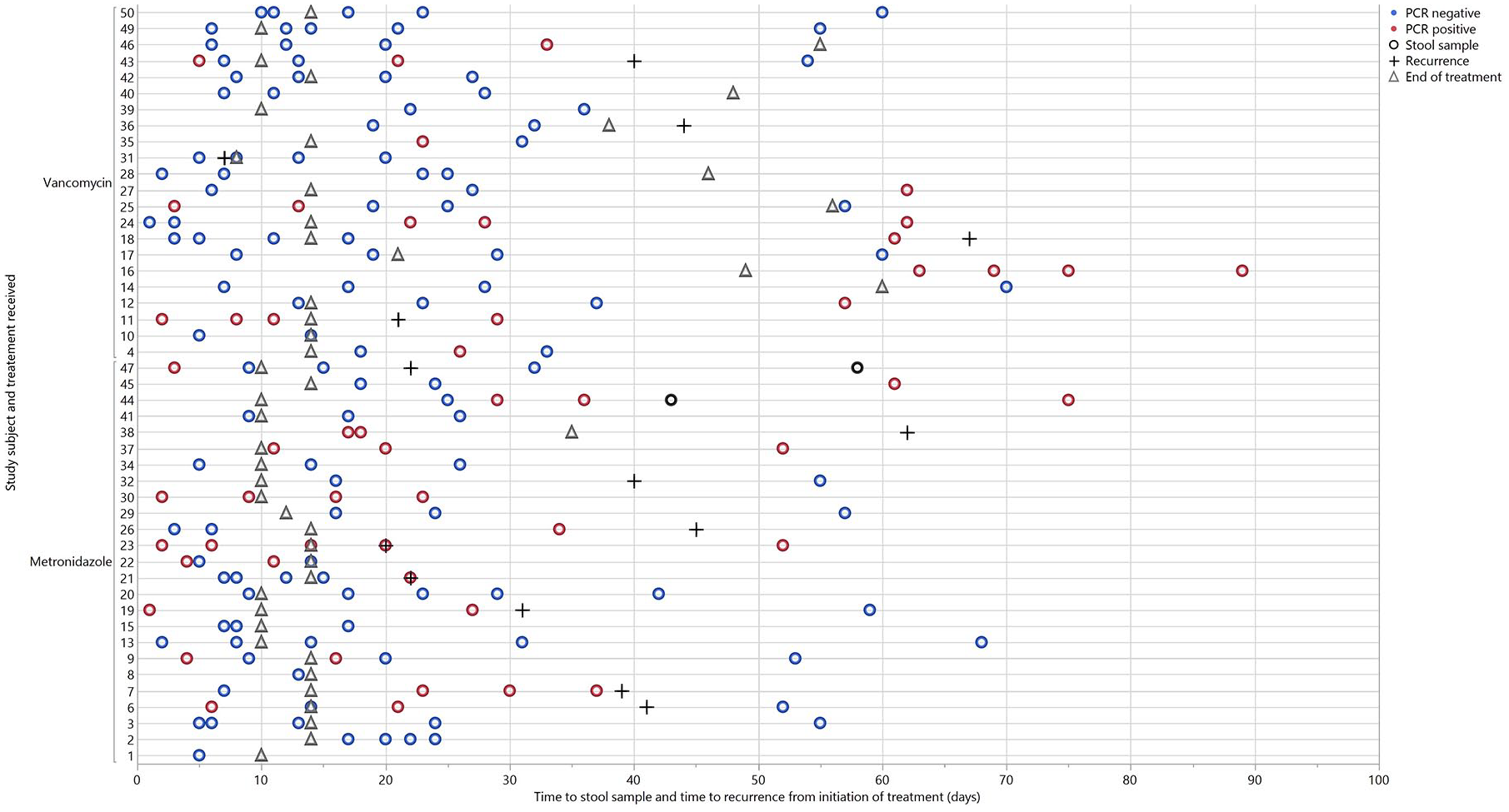
Timeline of PCR results and recurrence for each study subject. Each horizontal line represents a single subject. Day 0 indicates day of treatment initiation.
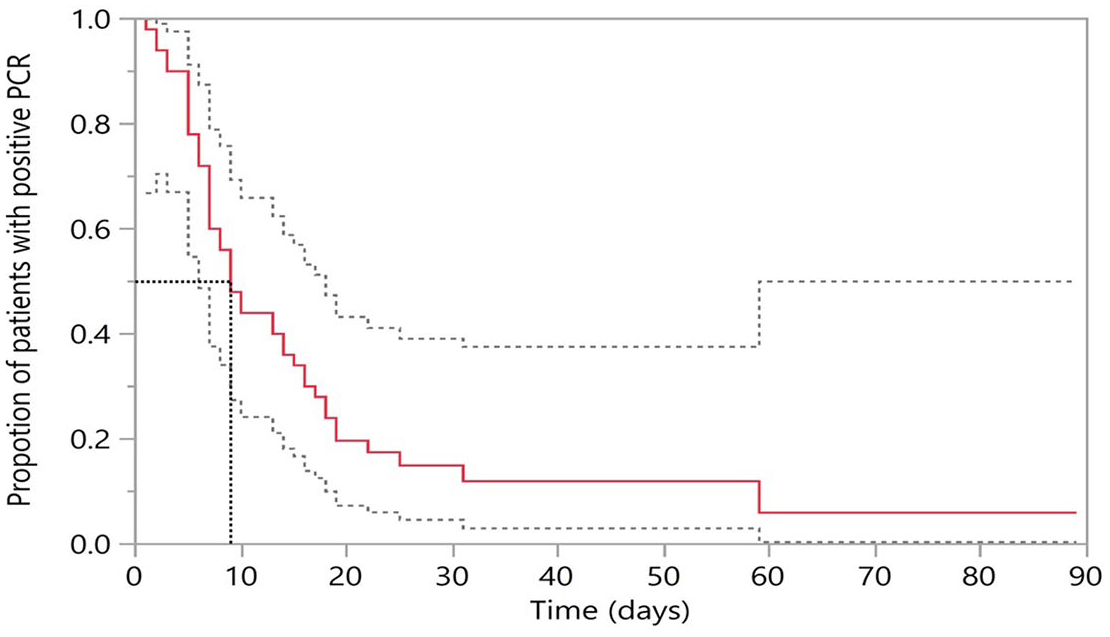
Kaplan–Meier curves showing time to first negative PCR from initiation of treatment. Day 0 indicated date of initiation of treatment; 95% confidence intervals are shown in dotted lines.
The median time to first negative PCR was 9 [95% confidence interval (CI), 7–14)] days. This was not different in patients treated with metronidazole [9 (95% CI, 7–17) days] or vancomycin [7.5 (95% CI, 6–18) days], log-rank test, p = 0.5 (Supplementary Figure 1).
On multivariate Cox proportional hazards analysis, age, sex, or treatment (vancomycin or metronidazole) did not affect time to first negative PCR (p = 0.67).
PCR test for predicting CDI recurrence
Recurrence of CDI occurred in 14 patients (28%) within 56 days and 15 patients (30%) within 90 days of treatment completion. In those with recurrence, the median time to recurrence was 26 (range = 0–81) days after completion of treatment.
When considering recurrence within 56 days, patients with ⩾1 positive PCR(s) during treatment had a higher risk than those with negative PCR(s) [odds ratio (OR): 3.9 (95% CI, 0.9–16.1), p = 0.054)], although this did not reach statistical significance. Asymptomatic patients with ⩾1 positive PCR(s) after treatment completion (asymptomatic PCR positives) did not have a higher risk of recurrence compared with those with only negative PCR(s) [OR: 2.8 (95% CI, 0.7–11.5), p = 0.15)]. Patients with ⩾1 positive PCR(s) either during or after treatment had a higher risk of recurrence [OR: 5.1 (95% CI, 1.2–21.6), p = 0.02)] compared with those with negative PCR(s). When adjusted for systemic non-CDI antibiotic exposure, patients with PCR positivity during treatment, and either during or after treatment had higher risk of recurrence (Table 1).
Risk of CDI recurrence with PCR positivity after adjusting for systemic non-CDI antibiotic exposure.

CDI, Clostridioides difficile infection; PCR, polymerase chain reaction. Results which show signficant results (p < 0.05) have their p-values blodfaced.
Results from multivariate logistic regression model. Variables in the model were PCR positivity (yes versus no) and antibiotic exposure (yes versus no).
When considering recurrence within 90 days, patients with ⩾1 positive PCR(s) during treatment had a higher risk of recurrence than those with negative PCR(s) [OR: 5.3 (95% CI, 1.3–22.6), p = 0.02]. Patients with ⩾1 positive PCR(s) after treatment completion (asymptomatic PCR positives) tended toward higher risk of recurrence compared with those with only negative PCR(s) [OR: 3.5 (95% CI, 0.8–14.2), p = 0.08], although this was not statistically significant, possibly due to small sample size. Patients with ⩾1 positive PCR(s) either during or after treatment had a higher risk of recurrence [OR: 6.0 (95% CI, 1.4–25.2), p = 0.009)]. These results did not change when adjusted for systemic non-CDI antibiotic exposure (Table 1).
The PCR test during treatment had a high specificity [80.0% (95% CI, 60.9–91.1%)] and negative predictive value [76.9% (95% CI, 57.9–89.0%)] in predicting recurrence within 90 days, although sensitivity was modest [57.1% (95% CI, 32.6–78.6%)] (Supplementary Table 1). Positive PCR either during or after treatment had a high sensitivity [80.0% (95% CI, 54.8–93.0%)] and negative predictive value [87.5% (95% CI, 69.0–95.7%)] in predicting recurrence at 90 days (Supplementary Table 1), although with moderate specificity [60% (43.6–74.4%)].
Univariate analysis performed on patients with a history of inflammatory bowel disease (IBD) (n = 12) or previous history of CDI (n = 12) showed no difference in the PCR positivity during treatment, after treatment, or during or after treatment (all p > 0.05). Analysis of recurrence of CDI within 90 days of treatment completion in these groups also showed no significant difference (p = 0.30 for IBD versus non IBD; p = 1.0 for previous CDI versus no-previous CDI).
Asymptomatic C difficile PCR positivity during and after treatment
Overall, 13 of 39 patients (33.3%) had ⩾1 positive PCR(s) during treatment, more in patients treated with metronidazole [42.1% (8)] than vancomycin [22.2% (4)], although this difference was not statistically significant (p = 0.19).
After treatment, 19 of 42 asymptomatic patients (45.2%) had ⩾1 positive PCR(s). The proportion of asymptomatic PCR positives was similar in the metronidazole and vancomycin groups [45.5% (10) versus 47.1% (8) patients respectively, p = 0.92)]. During the first 7 days after treatment completion, asymptomatic PCR positivity was more common in the metronidazole than the vancomycin group, but the difference was not statistically significant, possibly due to small sample size; rates of asymptomatic PCR positivity were similar thereafter (Table 2).
Clostridioides difficile PCR positivity rates by therapy.

CI, confidence interval; PCR, polymerase chain reaction.
Data presented as % (n/number of patients with available information).
Day 1 refers to the first day after treatment completion.
Discussion
The PCR for C difficile infection is a highly sensitive test used for diagnosis both as a standalone test and as part of a multistep algorithm.18,19 In this prospective study looking at the kinetics of PCR positivity during antibiotic therapy, the median time to a negative PCR was 9 days from the initiation of treatment. This was not different in patients treated with metronidazole or vancomycin and was not affected by age or sex of the patient. Regarding the ability of PCR to predict recurrent CDI, patients with a positive PCR during or after treatment had a higher risk of recurrence than those with persistently negative testing, even after controlling for systemic non-CDI antibiotic exposure. These results were largely similar when using either a 56-day or 90-day cutoff for defining recurrence. We also restricted our analysis to only asymptomatic positive PCR tests after treatment (which was seen in about half the patients treated with either metronidazole or vancomycin). While asymptomatic PCR positivity post-treatment did not reach statistical significance in predicting CDI recurrence, this could have been a type 2 error related to sample size, as the point estimates suggested a threefold increased risk of recurrence. To date, several studies have attempted to predict recurrent CDI using clinical parameters and microbiome signatures, but none have proven accurate enough to be used routinely in clinical practice. It is possible that incorporating post-treatment PCR positivity into a predictive model that includes these other features could improve the operating characteristics to the point that these models would be clinically useful. Given the clinical implications of the potential utility of a positive post-treatment PCR in predicting recurrent CDI, and the suggestive results of our study, this question should be assessed in a larger study.
Few studies to date have reported data on serial stool testing in patients with CDI.6,8,9,20 In one study, 19 of 35 patients (54%) with repeat PCRs within 7 days of the initial test remained positive, which is comparable with our results. 9 In another study, serial stool samples in 76 CDI patients were analyzed using toxigenic culture. 8 At the end of treatment, C difficile was isolated more frequently in patients treated with metronidazole than vancomycin (36% versus 21%, respectively), although the difference was not statistically significant. Patients treated with vancomycin were more likely than those treated with metronidazole to have C difficile in their stool 10–15 days after treatment. We found similar results in the first week following treatment completion, when patients treated with metronidazole had a higher propensity to have C difficile in their stool; the difference in our study was also not statistically significant. In a clinical trial by Gerding et al., the isolation of toxigenic C difficile in the placebo group increased from 14% at the end of treatment to >50% at weeks 1–3 following treatment. 20 Compared with our study, C difficile isolation rates in this study were higher in the first week post-treatment, possibly due to the higher use of metronidazole, considering that this treatment may have higher stool C difficile isolation rates. 8 These results have important implications for infection control purposes, as patients with asymptomatic PCR positives may work as potential reservoirs of C difficile for disease transmission. 21
To our knowledge, our study is the first to report the kinetics of PCR positivity during and after treatment of CDI and its utility in predicting CDI recurrence. Strengths of the study are that follow-up was well structured and most patients submitted at least three stool samples. However, the study has several limitations. Sample size was modest, and the timing of stool submission was somewhat heterogeneous. We also did not have data on C difficile ribotype and toxin producing capability. This could have implications for prediction of recurrence, as patients who are recolonized with a nontoxigenic C difficile strain may be protected against future CDI.20,21 Another practical limitation of this study is the inability to distinguish diarrhea due to recurrence of CDI and due to other causes like postinfection IBD. As a consequence of prospective, long duration (9 years) inclusion of patients in our study, metronidazole (rather than vancomycin or fidaxomicin) was used for initial treatment of CDI in half of our patients. This may limit the generalizability of our study as the recent IDSA guidelines recommend vancomycin as the treatment of choice for initial CDI episode. 18
Conclusion
In this prospective study, patients with CDI became PCR negative after a median of 9 days from treatment initiation. The PCR positivity during or after treatment predicted recurrence. The results suggest that repeat PCR testing may have a role in recurrence prediction, although larger studies are needed before these results can be implemented in routine practice.
Supplemental Material
sj-docx-1-tag-10.1177_17562848211050443 – Supplemental material for Kinetics of polymerase chain reaction positivity in patients with Clostridioides difficile infection
Supplemental material, sj-docx-1-tag-10.1177_17562848211050443 for Kinetics of polymerase chain reaction positivity in patients with Clostridioides difficile infection by Srishti Saha, Devvrat Yadav, Ryan Pardi, Robin Patel, Sahil Khanna and Darrell Pardi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-2-tag-10.1177_17562848211050443 – Supplemental material for Kinetics of polymerase chain reaction positivity in patients with Clostridioides difficile infection
Supplemental material, sj-jpg-2-tag-10.1177_17562848211050443 for Kinetics of polymerase chain reaction positivity in patients with Clostridioides difficile infection by Srishti Saha, Devvrat Yadav, Ryan Pardi, Robin Patel, Sahil Khanna and Darrell Pardi in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors wish to thank Dr Matthew Mrazek for his assistance with study design.
Authors’ note
This study was selected for an oral presentation in the Presidential Plenary session at the American College of Gastroenterology 2019 Annual Meeting, San Antonio, Texas.
Author contributions
SS contributed to data collection and interpretation, analysis, drafting, and revision of manuscript; DY contributed to data interpretation, analysis, drafting, and revision of manuscript. RP contributed to drafting and revision of manuscript; RP contributed to drafting and revision of manuscript; SK contributed to concept, data interpretation, drafting, and revision of manuscript. DSP contributed to concept, drafting, and revision of manuscript.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor of the article
Darrell Pardi accepted full responsibility for the conduct of the study, had access to the data, and had control of decision to publish.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
