Abstract
Background:
Hepatitis B e antigen (HBeAg)-positive chronic hepatitis B (CHB) in the immune-tolerant (IT) phase is significantly associated with high risk for hepatocellular carcinoma, suggesting requirement for antiviral therapy, particularly for those with histological liver injury. This study aimed to establish a non-invasive panel to assess significant liver fibrosis in IT chronic hepatitis B.
Patients and methods:
One hundred and thirteen IT-phase CHB patients were retrospectively recruited and divided into two histopathological groups according to their histological profiles: necroinflammatory score <4 (N <4)/fibrosis score ⩽1 (F0-1), and necroinflammatory score ⩾4 (N ⩾4)/fibrosis score ⩾2 (F2-4). Multivariate analysis was conducted to assess the predictive value of the non-invasive model for significant liver fibrosis.
Results:
IT-phase CHB patients with N <4/F0-1 had significantly higher HBsAg levels than those with N ⩾4/F2-4. The optimal HBsAg level of log 4.44 IU/mL for significant liver fibrosis (F ⩾2) gave an area under the curve (AUC) of 0.83, sensitivity of 81.1%, specificity of 81.6%, positive predictive value (PPV) of 68.2%, and negative predictive value (NPV) of 89.9%. An IT model with HBsAg and gamma glutamyl transpeptidase (GGT) in combination was established, and it had an AUC of 0.86, sensitivity of 86.5%, specificity of 81.6%, PPV of 69.6, NPV of 92.5, and accuracy of 83.2% to predict F ⩾2 in the IT-phase CHB patients. Notably, the IT model exhibited higher predictive value than the existing aspartate aminotransferase-to-platelet ratio index, Fibrosis-4 score, and GGT to platelet ratio.
Conclusion:
The established IT model combining HBsAg and GGT has good performance in predicting significant liver fibrosis in IT-phase CHB patients.
Introduction
Chronic hepatitis B virus (HBV) infection represents a public health problem across the world, with a particularly high prevalence in China. 1 HBV-infected individuals in immune tolerance (IT) are generally not recommended for anti-HBV treatment, owing to the evidence of a low risk of progressive liver injury in the IT patients. However, recent studies indicated that hepatocarcinogenesis may occur in IT-phase patients.2,3 On the basis of these new findings, the European HBV practice guidelines have recommended IT patients aged ⩾30 years to accept antiviral treatment regardless of the severity of hepatic histological damage. 4
To date, no antiviral therapy is recommended by international practice guidelines for anti-HBV treatment unless significant fibrosis (F ⩾2) is detected in IT-phase patients.5,6 A proportion of IT-phase patients had significant fibrosis,7,8 suggesting that antiviral treatment should be implemented. At present, liver biopsy is not recommended to assess liver injury as a routine procedure due to its invasiveness and complications. The diagnostic accuracy and reliability of non-invasive panels (aspartate aminotransferase-to-platelet ratio index (APRI) and FibroScan) are questionable 9 and, thus, there is a need for better non-invasive diagnostic models.
Indeed, the practice guidelines encourage research to improve knowledge pertaining to the natural history and indications for treatment in IT-phase patients. A recent study confirmed serum HBsAg can predict F ⩾2 in hepatitis B e antigen (HBeAg)(+) CHB patients. 10 In our previous study, we found that HBsAg levels can help differentiate patients with moderate liver injury from those patients in the IT phase, who were characterized by HBV DNA ⩾105 IU/mL, HBeAg positivity, and levels of alanine aminotransferase (ALT) <2 times the upper limit of normal (ULN). 7 In our multicenter, cross-sectional research, the purpose was to establish a HBsAg-based model for predicting F ⩾2 in IT patients, and compare its diagnostic value with APRI, 11 fibrosis index based on the four factors (FIB-4), 12 and gamma-glutamyl transpeptidase-to-platelet ratio (GPR). 13
Materials and methods
Study population
In this study, treatment-naïve Chinese IT patients were retrospectively recruited from January 2010 to January 2019 in First Affiliated Hospital of Fujian Medical University, Mengchao Hepatobiliary Hospital of Fujian Medical University, and First Hospital of Quanzhou Affiliated of Fujian Medical University. The criteria for eligibility were as follows:5,6 age of 18 years and older; HBsAg(+) for ⩾6 months; HBeAg(+); HBV DNA >1 million IU/mL; normal ALT (<40 IU/L); and scheduled liver biopsy. Patients with the following conditions were excluded from this research: (1) presence of other types of viral hepatitis, hepatocellular carcinoma, alcoholic liver disease, non-alcoholic fatty liver disease, decompensated cirrhosis, or autoimmune hepatitis; (2) concurrent infection with human immunodeficiency virus; (3) hereditary liver diseases; (4) drug-induced liver injury. The IT patients were stratified into two histopathological sets: necroinflammatory score <4 (N <4)/fibrosis score ⩽1 (F0-1); and necroinflammatory score ⩾4 (N ⩾4)/fibrosis score ⩾2 (F2-4).
The study protocol was approved by the Ethic Committee of the First Affiliated Hospital of Fujian Medical University, an equivalent to the Institutional Review Board (IRB) in the US, and the need for written informed consent from each patient was waived due to its retrospective nature. [MTCA, ECFAH of FMU (2015)084-1.]’.
Examination of serological markers for hepatitis B
Serum HBsAg was tested by use of the Abbott ARCHITECT® assay (Abbott Laboratories, Chicago, USA) or the Elecsys® HBsAg II quant assay (Roche Diagnostics, Germany), as the results of ARCHITECT are strong correlated with that of Elecsys II (r = 0.96). 14 HBV DNA was examined by use of quantitative polymerase chain reaction assay (PG Company, Shenzhen, China).
Liver biopsy
Liver biopsy was performed in individuals who wanted to be initiatively examined for liver pathological damage or to receive medical treatment. Liver specimens were obtained using aspiration 16-gauge modified needles (TSK Laboratory, Tochigi, Japan). Qualified liver specimens (more than a length of 1.5 cm and six portal tracts) were obtained, fixed, embedded, and processed with hematoxylin-eosin, and Masson’s trichrome according to the standard protocols. Liver inflammation activity and fibrotic stage were independently assessed by at least two pathologists according to the Histological Activity Index numerical scoring system.
Liver stiffness measurement
Liver stiffness measurement (LSM) was tested using FibroScan. Only LSM with at least 10 successful acquisitions, success rate over 60%, and interquartile range ⩽30% were considered to be reliable. LSM was conducted within 1 week prior to liver biopsy.
Statistical analysis
A Student’s t test and chi-square test were applied for detection of differences in data. Correlations were tested by Spearman correlation coefficients. Univariate or multivariate analysis was applied to select independent parameters linked with F ⩾2. Following the TRIPOD guidelines, 15 we develop the predictive model for F ⩾2 in IT patients. In particular, bootstrap resampling (500 times) was employed in order to obtain the area under the curve (AUC) and the best cut-off value of HBsAg for F ⩾2. Statistical analyses were applied by use of R (http://www.R-project.org), Empower (R) (http://www.empowerstats.com, X&Y solutions, Inc., Boston, MA, USA), and SPSS V 23.0.
Results
Demographic and clinical characteristics
One hundred and thirteen IT patients were retrospectively enrolled. The laboratory and clinical parameters of patients are presented in Table 1. The prevalence of F ⩾2 was 32.7% (Table 1). HBsAg had a positive correlation with HBV DNA in IT-phase individuals (r = 0.49). Compared with IT individuals with N ⩾4/F2-4, those individuals with N <4/F0-1 showed higher levels of serum HBsAg and HBV DNA, and lower ALT and gamma glutamyl transpeptidase (GGT) levels (Table 2). Furthermore, IT patients <40 years of age (n = 60) exhibited F <2. We subsequently performed a subgroup analysis using the criteria of the age <40 years or the new ULN of ALT 35 IU/mL in healthy males and ALT 25 IU/mL in healthy females, 6 and found the prevalence of F ⩾2 was 19.4% (7/36).
The demographic and clinical characteristics.

Note. The IT patients were stratified into two histopathological sets: necroinflammatory score <4 (N <4)/fibrosis score ≤1 (F0-1); and necroinflammatory score ≥4 (N ≥4)/fibrosis score ≥2 (F2-4).
Patients who fulfilled the criteria of the age less than 40 years and the new upper limit of normal of ALT 35 IU/mL in healthy men and 25 IU/mL in healthy women according to the American Association for the Study of Liver Diseases 2018 guidelines for the treatment of chronic hepatitis B.
HBV, hepatitis B virus; ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma glutamyl transpeptidase; WBC, white blood cell count; PLT, platelet count; PT, prothrombin time.
Predictive value of serum HBsAg for significant fibrosis.
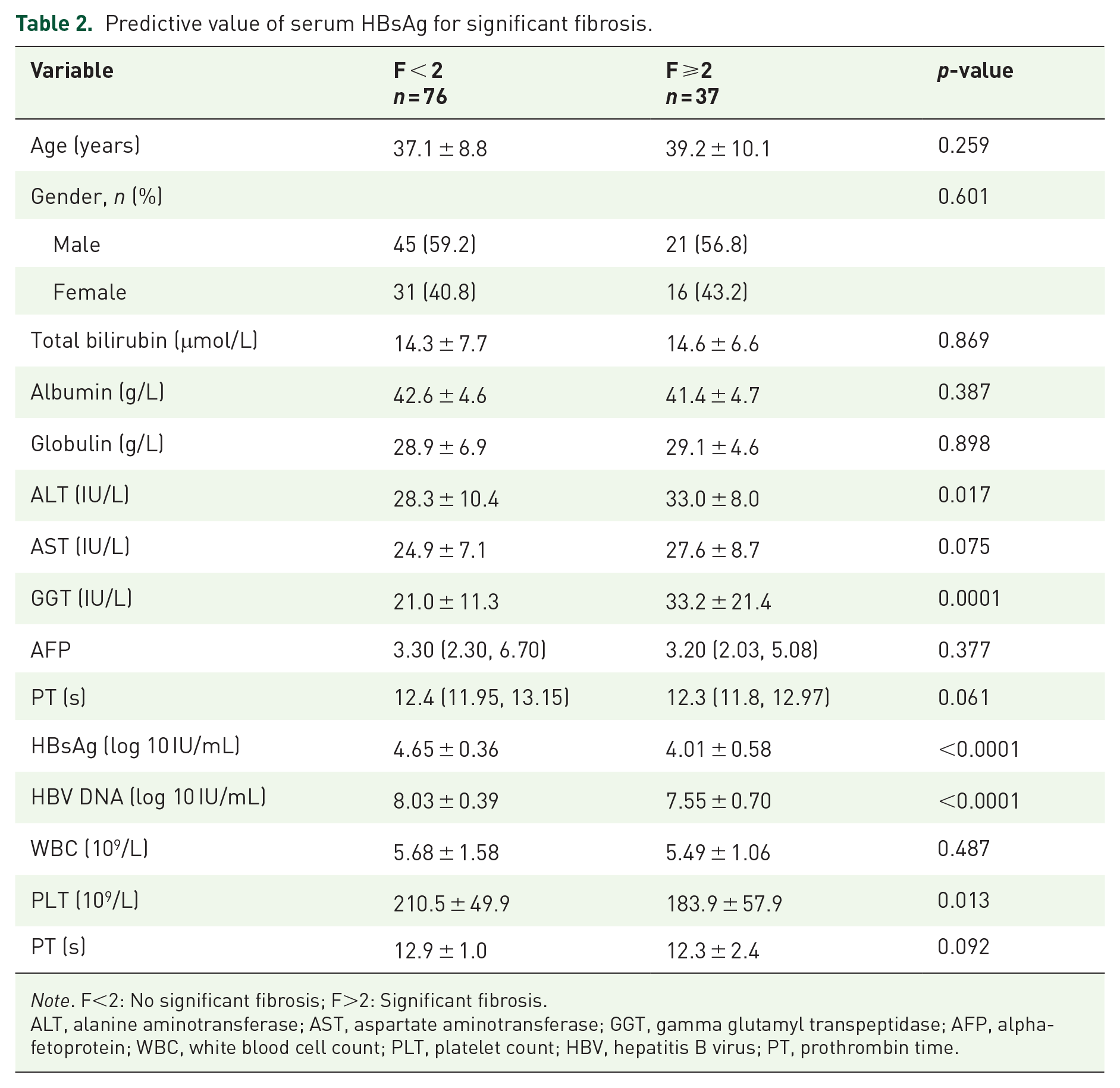
Note. F<2: No significant fibrosis; F>2: Significant fibrosis.ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma glutamyl transpeptidase; AFP, alpha-fetoprotein; WBC, white blood cell count; PLT, platelet count; HBV, hepatitis B virus; PT, prothrombin time.
Predictive value of LSM in IT-phase patients
LSM had relatively weak correlation with hepatic fibrosis in these IT individuals (r = 0.337, p = 0.048). Furthermore, the AUC of LSM to predict F ⩾2 was 0.70. As shown in Figure 1, the sensitivity was 57.1%, the specificity was 82.1%, the positive predictive value (PPV) was 44.4%, and the negative predictive value (NPV) was 88.5%.

Receiver operating characteristic curve analysis for evaluation of the ability of transient elastography (TE) to predict liver fibrosis stage among immune tolerance-phase chronic hepatitis B patients. The 95% confidence interval for the area under the curve (AUC) was estimated using bootstrap resampling (times = 500).AUC, area under the curve; APRI, aspartate aminotransferase-to-platelet ratio index; FIB-4, fibrosis index based on the four factors; GPR, gamma-glutamyl transpeptidase-to-platelet ratio; LSM, liver stiffness measurement.
Diagnostic value of HBsAg for F ⩾2 among IT patients
HBsAg had a moderate inverse correlation with liver fibrosis in IT-phase individuals (r = −0.56). Furthermore, at the value of log 4.44 IU/mL, the predictive characteristics of HBsAg for F ⩾2 were an AUC of 0.81, sensitivity of 81.1%, specificity of 81.6%, PPV of 68.2%, and NPV of 89.9%, respectively [Figure 2(a)].

Receiver operating characteristic (ROC) curve analysis for evaluation of the ability of the serum HBsAg level to predict liver fibrosis stages among immune tolerance-phase chronic hepatitis B patients. The 95% confidence interval for the area under the curve (AUC) was estimated using bootstrap resampling (times = 500). (a) ROC curves for the ability of the serum HBsAg level to identify significant liver fibrosis. (b) ROC curves for the ability of the serum HBsAg level to identify significant liver fibrosis in patients with the criteria of the age <40 years and new upper limit of normal values for alanine aminotransferase (ALT) of 35 IU/mL in healthy men and 25 IU/mL in healthy women according to the American Association for the Study of Liver Diseases 2018 hepatitis B guidelines. (c) ROC curves for the ability of the serum HBsAg level to identify significant liver fibrosis in patients with age <30 years and ALT <40 IU/mL according to the European Association for the Study of the Liver 2017 hepatitis B guidelines.
We found that the prevalence of F ⩾2 in IT individuals aged less than 40 years with ALT less than 35 IU/L in men and less than 25 IU/L in women was 19.4% (7/36); the AUC of HBsAg to diagnose F ⩾2 was 0.79 [Figure 2(b)]. In IT individuals aged less than 30 years with ALT less than 40 IU/mL, the AUC of HBsAg to diagnose F ⩾2 was 0.81 [Figure 2(c)].
Development of the IT model and its predictive performance in comparison with LSM, FIB-4, APRI, and GPR
We constructed the IT model: 9.39–2.60 × log HBsAg + 0.05 × GGT by applying multivariate regression analysis. The IT model with an AUC of 0.88 to predict F ⩾2 was validated using bootstrap resampling (times = 500) and had an AUC of 0.86 [Figure 3(a) and (b)]. The optimal cut-off value for diagnosing F ⩾2 in the IT model was −1.09. IT model was used to diagnose F ⩾2 in IT individuals aged <30 years and <40 years and showed an AUC of 0.88 and 0.81, sensitivity of 77.8% and 85.7%, specificity of 93.8% and 76.7%, PPV of 87.5% and 46.2%, NPV of 88.2% and 95.8%, and accuracy of 88.0% and 78.4%, respectively [Table 3 and Figure 3(c) and )(d)]. Notably, the IT model showed significantly better performance in the prediction of F ⩾2 in IT patients than the FIB-4 index, APRI, and GPR (p < 0.001, p = 0.002, and p = 0.0023, respectively; Figure 4) except transient elastography (TE) (p = 0.105).
Validity of the IT model and its performance in comparison with APRI, FIB-4, GPR, and TE for predicting significant fibrosis.

AC, accuracy; AUC, area under the curve; IT, immune tolerance; NPV, negative predictive value; PPV, positive predictive value; Sen., sensitivity; Spec., specificity.

Receiver operating characteristic curve analysis for evaluation of the ability of the immune tolerance (IT) model to predict liver fibrosis stages among the IT-phase chronic hepatitis B (CHB) patients. The 95% confidence interval (CI) for the area under the curve (AUC) was estimated using bootstrap resampling (times = 500). (a) The specificity and sensitivity of the IT model for the prediction of significant liver fibrosis in IT-phase CHB patients. (b) Blue shaded areas showed the bootstrap estimated 95% CI with the AUC (times = 500). (c) The ability of the IT model to predict significant fibrosis among IT-phase CHB patients aged <30 years with ALT <40 IU/mL. (d) The ability of the IT model to predict significant fibrosis among IT-phase CHB patients aged <40 years with ALT of 35 IU/mL in healthy men and 25 IU/mL in healthy women.

Receiver operating characteristic curves for the ability of the immune tolerance (IT) model, compared with FibroScan, FIB-4, APRI, and GPR to predict significant fibrosis among IT-phase chronic hepatitis B patients.
Discussion
Identification of F ⩾2 on liver biopsy among IT-phase patients has potentially important clinical significance during the decision-making for an antiviral treatment. This retrospective study of IT-phase chronic hepatitis B (CHB) patients from multiple centers produced the following major novel findings: (1) the prevalence of moderate necroinflammation, or F ⩾2, was as high as 38.9% in IT-phase patients, who may need but miss immediate administration of antiviral therapy; (2) serum HBsAg was significantly associated with liver fibrosis; (3) the IT model provided significantly better prediction of F ⩾2 than the FIB-4 index, APRI, and GPR. Our results suggest that the IT model established in this study is capable of predicting F ⩾2 among IT-phase patients.
Serum ALT is an imperfect biomarker for reflecting hepatic damage. In our study, the IT patients with moderate liver injury had higher ALT levels than patients with minimal liver injury. It has been thought that IT-phase patients of a younger age (<40 years 6 or <30 years 4 ) have minimal or absent histological injury. However, in our study, 19.4% of patients aged <40 years with ALT <35 IU/L in healthy males and <25 IU/L in healthy females, and 36% of patients aged <30 years with ALT <40 IU/mL showed significant fibrosis. Thus, it is critical to determine F ⩾2 in IT individuals for appropriate clinical management.
Currently, liver biopsy is not recommended in IT-phase patients unless the patient is 40 years or older.5,6 Due to the invasiveness and complications, liver biopsy is generally not accepted for IT-phase patients. In our research, liver biopsy was applied in these 113 IT individuals for evaluation of liver injury, and the sample size was relatively large for this type of study.
HBsAg, a hallmark of HBV infection, is modulated by both virus and host immunity and negatively associated with host immune control of HBV. 16 Serum HBsAg levels in IT-phase patients reach the peak during the period of HBV infection. Our previous research indicated that HBsAg can distinguish patients with active liver injury from IT-phase patients. 7 Building upon our previous findings, we conducted this multicenter cross-sectional research by strictly limiting levels of HBV DNA and the ULN of ALT in IT-phase individuals. In this study, we found a strong reverse correlation between HBsAg and liver fibrotic stages in IT-phase individuals. The HBsAg AUC to predict F ⩾2 was 0.81. Our results, together with the current practice guidelines, can support selective performance of liver biopsy in individuals with HBsAg <4.44 log IU/mL, an indicator of F ⩾2. Notably, quantification of HBsAg to diagnose patients with potential hepatic damage for liver biopsy is expected to minimize the number of IT patients with hepatic damage who are mistakenly considered. From a clinical perspective, serum HBsAg, based on its simplicity, may become a potential predictive biomarker for liver fibrosis and may lead to a reduction in liver biopsy. Although the mechanisms underlying the inverse relationship between serum HBsAg and fibrosis stages remain unclear, there is a possibility that the host immune responses could be involved. First, the immune-mediated response to HBV infection may result in liver injury. Second, the retention of HBsAg within hepatocytes during immune-mediated liver damage may lead to a reduction in HBsAg.
Furthermore, we developed a non-invasive IT model with the combination of HBsAg and GGT to predict F ⩾2 in IT patients. Edwards and colleagues found that GGT was elevated in early cholestasis, and this or an increase of epidermal growth factor could explain the relationship between increased GGT and the severity of liver fibrosis. 17 Bootstrap resampling was used to validate the IT model based on the TRIPOD guidelines. 15 The AUC for the IT model was 0.86 for the prediction of F ⩾2. Considering that patients younger than 30 years or 40 years had lower rates of F ⩾2 and likely represented the typical IT phenotype with higher HBV DNA and HBsAg levels based on current HBV practice guidelines,4 –6 we used the IT model to predict F ⩾2 in IT patients aged <30 years and <40 years and found that the predictive values were similar (AUC of 0.88 and 0.81, respectively). Prior to the current research, the effectiveness of TE, FIB-4, APRI, and GPR for evaluating hepatic fibrosis in IT individuals has not been explored. In contrast to FIB-4, APRI, GPR, and TE, the IT model showed a much greater AUC. To date, TE, FIB-4, and APRI have been highly recommended by several international HBV practice guidelines for identifying candidates for liver biopsy.5,6,18 Recent studies indicated that APRI and FIB-4 have low predictive value for hepatic fibrosis in CHB individuals with persistently normal ALT levels (AUC of 0.518 and 0.597, respectively) and are not suitable for evaluating fibrosis in CHB patients on long-term antiviral treatment.9,19 In this study, to identify F ⩾2, the FIB-4, APRI, and GPR had AUCs of 0.62, 0.59, and 0.72, respectively.
This is the first research, to our knowledge, to evaluate the predictive ability of TE in IT individuals, suggesting a weak correlation of LSM with liver fibrosis. Furthermore, the performance of LSM for predicting F ⩾2 was below expectation, with an AUC of 0.70 at a cutoff of 7.7 kPa. Due to the retrospective nature of this research, we obtained the TE test data from only a small fraction of the study patients (35 patients). The AUCs for TE and the IT model were not significantly different among these 35 patients (AUCs of 0.70 versus 0.895, respectively); however, TE had a relatively low AUC. A relevant research with a majority of IT patients is necessary to assess the predictive performance of TE in the near future.
Despite its strengths, this research has some potential limitations. First, our non-invasive IT model was established and internally validated by retrospective analysis. Future external validation of our results is needed. Second, more IT-phase patients aged <40 years and with the newly proposed ULNs of ALT according to the American Association for the Study of Liver Diseases, or aged <30 years with ALT <40 IU/mL according to the European Association for the Study of the Liver, should be enrolled to investigate HBsAg as a useful clinical parameter for predicting liver fibrosis among IT-phase patients. Third, this non-invasive IT model was established by analyzing data on Chinese patients with CHB; further validation of this model in other ethnic populations should be conducted in the future.
In conclusion, our study demonstrated that approximately 30% of IT patients have moderate-to-severe hepatic damage. The serum level of HBsAg correlates negatively with the severity of hepatic damage. The IT model established by integrating HBsAg and GGT can help diagnose IT-phase patients in whom F ⩾2 has occurred and for whom antiviral therapy may need to be initiated.
Footnotes
Author contributions
Zeng DW and Liu YR conceived and designed the experiments; Zeng DW, Huang ZX, Lin MX and Liu YR performed the experiments; Kang NL, Lin X, Li YN and Zhu YY analyzed the data; Kang NL, Huang ZX and Lin MX contributed reagents/materials/analysis tools; Zeng DW, Huang ZX and Lin MX wrote the manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the Fujian Provincial Department of Science and Technology (2019Y0015 and 2019J01432), Quanzhou Science and Technology Bureau Planning Project, No. 2019N019S.
