Abstract
Patients with cholangiocarcinoma have poor survival since the majority of patients are diagnosed at a stage precluding surgical resection, due to locally irresectable tumors and/or metastases. Optimization of diagnostic strategies, with a principal role for tissue diagnosis, is essential to detect cancers at an earlier stage amenable to curative treatment. Current barriers for a tissue diagnosis include both insufficient tissue sampling and a difficult cyto- or histopathological assessment. During endoscopic retrograde cholangiopancreatography, optimal brush sampling includes obtaining more than one brush within an individual patient to increase its diagnostic value. Currently, no significant increase of the diagnostic accuracy for the new cytology brush devices aiming to enhance the cellularity of brushings versus standard biliary brush devices has been demonstrated. Peroral cholangioscopy with bile duct biopsies appears to be a valuable tool in the diagnostic work-up of indeterminate biliary strictures, and may overcome current technical difficulties of fluoroscopic-guided biopsies. Over the past years, molecular techniques to detect chromosomal instability, mutations and methylation profiling of tumors have revolutionized, and implementation of these techniques on biliary tissue during diagnostic work-up of biliary strictures may be awaited in the near future. Fluorescence in situ hybridization has already been implemented in routine diagnostic evaluation of biliary strictures in several centers. Next-generation sequencing is promising for standard diagnostic care in biliary strictures, and recent studies have shown adequate detection of prevalent genomic alterations in KRAS, TP53, CDKN2A, SMAD4, PIK3CA, and GNAS on biliary brush material. Detection of DNA methylation of tumor suppressor genes and microRNAs may evolve over the coming years to a valuable diagnostic tool for cholangiocarcinoma. This review summarizes optimal strategies for biliary tissue sampling during endoscopic retrograde cholangiopancreatography and focuses on the evolving molecular techniques on biliary tissue to improve the differentiation of benign and malignant biliary strictures.
Introduction
Biliary strictures are an important clinical condition that may lead to severe symptoms and complications, including jaundice, cholangitis, liver abscesses, and secondary biliary cirrhosis. Transpapillary biliary drainage with stent placement or balloon dilation through endoscopic retrograde cholangiopancreatography (ERCP) is a well-established treatment for these patients and allows for symptom relief of jaundice, pruritus, and pain, and improvement of liver functions.1–4
A biliary stricture may be caused by either benign or malignant etiology. Malignant strictures account for approximately 70% of cases, and most often concern pancreatic cancer, followed by cholangiocarcinoma (CCA), and at a lesser frequency other malignancies (such as gallbladder carcinoma, ampullary carcinoma, metastatic lesions). 5 The determination of the etiology of a stricture often requires the combination of several diagnostic modalities, including clinical evaluation, laboratory findings, and endoscopic and radiological imaging.6,7 In addition, tissue sampling for cyto- or histopathological assessment is of paramount importance. 8 The level of serum tumor markers, in particular CA-19.9 and CEA, is often evaluated in clinical practice during evaluation of biliary strictures; however, it should be noted that elevated levels of these markers are compatible with both benign and malignant causes of biliary obstruction. Therefore, before ordering tumor markers, consideration of the consequence of elevated levels is warranted, and interpretation of the test results requires combination with other diagnostic modalities, including cross-sectional imaging. 9
Although the majority of biliary strictures prove malignant after diagnostic work-up or follow-up, a number of benign conditions, such as primary sclerosing cholangitis (PSC), chronic pancreatitis, and IgG4-related sclerosing cholangitis, frequently mimic malignancy, and differentiation can be challenging.10–14
Despite the combination of the above-mentioned diagnostics, current strategies for the assessment of biliary strictures have a very low diagnostic accuracy. As a result, biliary strictures remain indeterminate in up to 20% of cases. 15 Since consequences are devastating when a malignant cause of a biliary stricture is missed or diagnosed at a later stage precluding surgery, improved diagnostic methods with adequate sensitivity are required. In addition to a high sensitivity, these diagnostic methods should also demonstrate a high specificity to avoid unnecessary large surgical procedures (such as pancreaticoduodenectomy). The importance of a high specificity is underlined by the observation that in approximately 15–25% of patients undergoing surgical resection for biliary strictures with a high suspicion of malignancy, pathological examination of the resected specimens reveals benign disease.16,17
A tissue diagnosis remains key for an adequate differentiation of the etiology of biliary strictures. Current barriers for a tissue diagnosis include both insufficient tissue sampling and a difficult cyto- or histopathological assessment mostly in an inflammatory background. Therefore, optimization of diagnostic strategies requires both improved tissue sampling during ERCP in combination with development and implementation of novel molecular diagnostic tools on biliary tissue.
In this review, we summarize current knowledge regarding optimal biliary tissue sampling of biliary strictures during ERCP, and elaborate on promising molecular techniques to differentiate between benign and malignant strictures. It should be noted that these diagnostic strategies will be included in a complex diagnostic algorithm in clinical practice.
Optimizing tissue sampling techniques during ERCP
Cytology
Brush cytology is the most commonly used tissue sampling technique during ERCP. Since the majority of CCAs develop distal (40%) or in the perihilar (50%) region, most strictures are amenable to this technique. 18 After transpapillary biliary cannulation, a brush catheter is inserted over a guidewire into the biliary tree, and is advanced to the stricture. After advancing the brush from the catheter, the brush is moved back and forth along the stricture, resulting in attachment of epithelial cells to the brush. Thereafter, the brush is retracted in the catheter and withdrawn from the endoscope. The brush is cut off the catheter and can be smeared onto glass slides or can be stored in CytoLyt fixative solution. 19
Due to low diagnostic yield of brush cytology (which is described in detail in the following paragraph on ‘Cytology’), multiple studies have investigated modified techniques and new cytology brush designs to increase its diagnostic yield. These techniques aim at enhancement of exfoliation of epithelial cells and subsequently making material more representative by increasing cellularity. Repeat brushing (>1 brush per patient) increases cancer detection, 20 whereas obtaining brush cytology after stricture dilation by a balloon or passage dilating catheter does not result in higher cancer detection rates.20,21 The newest brushes are larger and/or have different types of bristles oriented at an angle greater than the conventional 45 degree angle. However, these more expensive Infinity® brushes (Infinity sampling device, US Endoscopy, Mentor, OH, USA) and the Cytolong® brush (Cook Endoscopy, Winston-Salem, NC, USA) demonstrated an equal diagnostic performance as compared with the conventional brush, and have therefore not widely replaced conventional brushes in clinical practice.22–24 The use of other devices to disrupt the biliary epithelium, such as a scraping brush and stent retriever have been studied in the past years, but study results were inconclusive due to small series. 25–27
In several patients it is a challenge to obtain cyto- or histological specimens with brushing or biopsies due to technical difficulties. Intraductal bile aspiration cytology is an easy and established method and a suitable alternative diagnostic strategy to investigate the morphology of biliary epithelial cells when other methods, in particular brush cytology, fail. 28 Biliary aspiration can be performed during either ERCP, via an endoscopic nasobiliary catheter, or a percutaneous transhepatic biliary catheter. 29 In general, 10–50 ml bile is aspirated and centrifuged to separate the cells for preparation of a smear, whereafter the slide is stained by standard Papanicolaou technique for cellular examination. These cells are well preserved and can be observed singly, in monolayer, or folded sheets. 30 The most important drawbacks of this technique is the low abundance of epithelial cells in bile and the lack of knowledge about the ideal bile volume and the ideal sampling site. 31
Finally, another technique that has been described in literature to obtain cytological specimens is by fine needle aspiration (FNA) of the biliary lesion during ERCP. This procedure is performed with a biliary catheter with a retractable needle that can be placed into the target lesion under fluoroscopic guidance. 32 The needle is moved back and forth into the lesion, after which the aspirations are processed into smears or are stored in CytoLyt. 33 This technique is not commonly applied in routine practice, since its additional value to brush cytology has not been proven.
Histology
Brushes are often combined with fluoroscopy-guided biliary biopsies to increase diagnostic accuracy for malignant biliary strictures. Obtaining ductal biopsies can, however, be technically challenging. Under fluoroscopic guidance, the biopsy forceps are advanced to the level of the stricture, and opened and closed to grasp a specimen from the stricture. It usually provides a sample of bile duct lining epithelium sometimes together with underlying stroma. In patients with an indeterminate biliary stricture it has been shown to improve diagnostic performance as compared with brush cytology alone. 34 Endoscopic retrograde transpapillary forceps biopsy demonstrates sensitivities ranging from 40% to 60% and specificities from 97% to 100%.35–40 Because of a limited sampling area for biliary biopsies, obtaining multiple biopsies or complementary biliary brushing is recommended. When biopsy is combined with brush cytology, sensitivity increases from 47% to 86%, while specificity remains close to 100%. 41 Furthermore, the combination of bile aspiration cytology, brush cytology, and forceps biopsy results in a sensitivity of 85% and a specificity of 100%. 42
Over recent years, many new tools have been developed to optimize the success rate of adequate tissue sampling in the bile duct. One of those methods is the wire-guided biopsy forceps, which can be advanced over a wire through the common bile duct. This technique may overcome the difficulties encountered by freehand insertion of the forceps across the major papilla. In the direct method, the forceps are directly inserted into the bile duct alongside the guidewire. Both direct and wire-grasping methods for obtaining transpapillary forceps biopsies have been explored, resulting in a significantly higher success rate of obtaining adequate specimens using the wire-grasping method (100% versus 50%). 43
Cholangioscopy targeted biopsies
Peroral cholangioscopy (POC) allows targeted biopsies by direct endoscopic visualization of the biliary stricture, resulting in a superior diagnostic performance as compared with ERCP-guided cytology brushing and forceps biopsies.44,45 In POC, a slim endoscope is introduced through a working channel of a duodenoscope. The cholangioscope is then introduced into the bile duct, either freehand or over a guidewire. This technique enables direct intraductal visualization of strictures, targeted biopsy sampling of suspected areas and in case of stones, intraductal lithotripsy. POC is considered to be a well-tolerated technique with relatively few complications. The nature and frequency of complications are comparable with those reported for conventional ERCP, including pancreatitis, perforation and bleeding, with a possible exception for the occurrence of cholangitis. 46 Retrospective studies have found higher rates of cholangitis among patients undergoing POC as compared with conventional ERCP, although prospective data are lacking. 47 In clinical practice, the use of antibiotic prophylaxis could be considered before POC to prevent cholangitis. The success rate with peroral cholangioscopy to achieve a visual diagnosis and a histological diagnosis is reported to be high (up to 90% and 80%, respectively).48–51 The addition of peroral cholangioscopy to biliary brushes or forceps biopsies resulted in a sensitivity of 100% and a specificity of 87%. 52 In up to 15% of cases the cholangioscope is not able to pass through the stricture, with PSC as an important risk factor for failed cannulation.44,53 The disposable through the scope Spyglass® single-operator cholangioscopy is a popular tool to obtain visual assessment and (fluoroscopic-guided) targeted biopsies of biliary lesions with a reported sensitivity as well as specificity of 83% for visual diagnosis, and a sensitivity and specificity of 66–86% and 94–100% for representative biopsies.54–59 Further improvement of sensitivity is reached when increasing the number of biopsies for histological assessment. 60 However, a critical look at the studies is needed. Many studies have included a small series of samples, a high risk of selection bias exists, and histological assessment is not blinded for clinical characteristics. Furthermore, in the above-mentioned studies, only representative samples were included, meaning that the real sensitivity is lower than the reported sensitivity. Nevertheless, peroral cholangioscopy with bile duct biopsies appears to be a valuable tool in the diagnostic work-up of indeterminate biliary strictures. Although at present the use of cholangioscopy with biopsies is often limited to expert centers, more widespread use can be awaited during coming years.
Confocal laser endomicroscopy
A novel technique for the distinction of benign and malignant biliary strictures is probe-based confocal laser endomicroscopy (pCLE), which provides in vivo evaluation of gastrointestinal mucosal histology. 61 A laser illuminates the tissue with a wavelength of 488 nm. 62 The light is locally absorbed by fluorophores and the reflected fluorescence is detected by the probe, resulting in multiple two-dimensional images, which can reconstruct a true microscopic three-dimensional structure in real time. 63 This technique has shown an excellent sensitivity of 89–98% to detect malignant strictures, but the diagnostic value is hampered by a disappointing interobserver agreement and a low specificity of 67%, mostly due to false positive results in cases with a benign inflammatory stricture.64–66 The Miami and Paris classifications have been developed to improve diagnostic accuracy of the confocal laser endomicroscopy for benign strictures caused by inflammation.67,68 Up to writing, only a small number of studies in a low number of expertise centers have investigated the diagnostic performance of pCLE. Therefore, prior to implementation of this technique in clinical practice, prospective studies need to be awaited to establish the additional diagnostic value of pCLE to existing diagnostic strategies.
Optimizing tissue analysis and new molecular techniques
Cytology
Several processing options exist for biliary brush samples to optimize the assessment of cytology, including smears on glasses, generally stained with May Grünwald–Giemsa (MGG) stain, and fixation in CytoLyt. After fixation in CytoLyt, cellular material can be further processed into sections by the use of ThinPrep® or Cellient™, which are suitable for cytological assessment (Supplementary Figure 1).69–71
Brush cytology is scored by morphological examination and can be classified into the following categories according to the Papanicolaou classification: nondiagnostic, negative for malignancy, atypical, neoplastic (benign or other), suspicious for malignancy, or malignant (Figure 1).72,73 Specimens scored as nondiagnostic cannot be interpreted due to technical or sample issues, including absence of epithelial cells. 74 In malignant smears, cells have among others, enlarged nuclei and an irregular nuclear membrane.

Examples of routine cytology of biliary brush samples. (a) Normal biliary epithelial cells. (b) Atypia; reactive changes with variation in nuclear size, often observed in PSC. (c) Adenocarcinoma, showing variation in nuclear architecture and size. Papanicolaou staining. Original magnification, ×25.
The diagnostic accuracy of brush cytology has been investigated in several studies. Table 1 provides an overview of the accuracy to detect CCA of biliary brush cytology in available studies, including reports on PSC patients.8,26,35,36,40,73,75–128 Specificity has been shown to be close to 100% in all these studies. However, sensitivity rates of brush cytology are rather disappointing, reported within a considerable range from 15% to 70%.104,107,129,130 Main reasons for the high rate of false-negative results are sampling errors and low tissue yield due to technical challenges, including difficult anatomic sites and poor visualization. 106 Moreover, the pathological assessment is difficult as illustrated by reports on a considerable interobserver disagreement.102,131 Especially in PSC the distinction between benign and malignant cytology on brush is notoriously difficult due to altered cellular morphology caused by inflammation. Predictors of a positive yield of brush cytology include advanced age, mass size >1 cm, stricture dilation before sampling, stricture length of >1 cm, and increased serum levels of CA-19.9, alanine aminotransferase (ALT), and total bilirubin.114,132
Review of literature. Sensitivity and specificity of brush cytology and FISH analysis in sporadic and PSC-associated CCA.
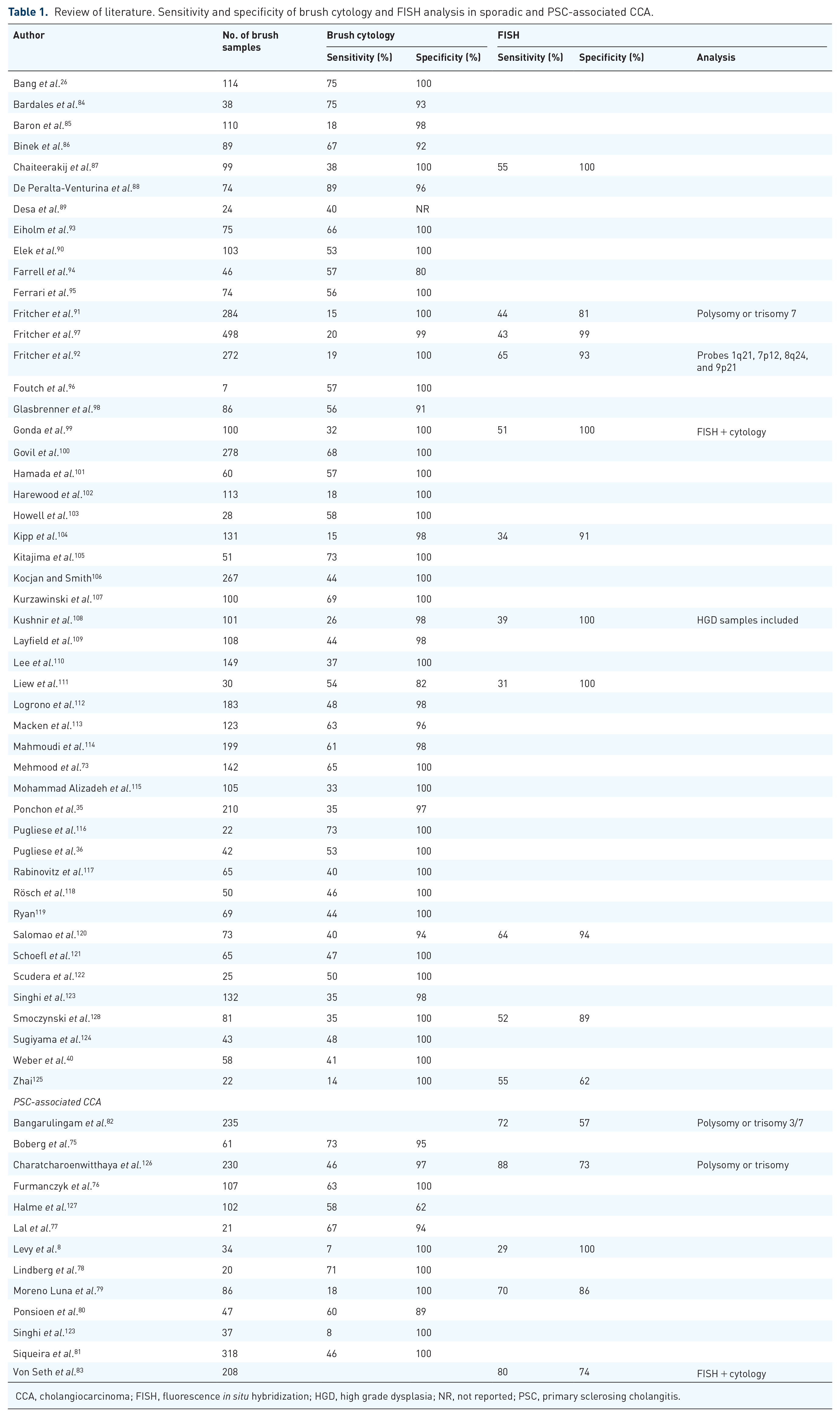
CCA, cholangiocarcinoma; FISH, fluorescence in situ hybridization; HGD, high grade dysplasia; NR, not reported; PSC, primary sclerosing cholangitis.
Similar to biliary brush cytology, bile aspiration cytology specimens from neoplastic biliary strictures demonstrate identical cellular alterations as compared with specimens from inflammatory biliary strictures, including hyperplasia or regenerative or degenerative alterations. In order to confirm neoplasia, increased nuclear–cytoplasmic ratio, variation in shape, and clumping of chromatin are essential features in cytological material. 30 Further research is required to explore the diagnostic accuracy of cytological examination from bile aspiration, since mainly small series were published with conflicting results and large ranges of sensitivity. Although promising results of bile aspiration were reported with a significantly higher sensitivity as compared with routine brushes (89% versus 37%), no difference in sensitivity between brushing and bile aspiration were observed when only adequate samples were examined and bile aspiration cytology needed to be repeated several times to obtain an acceptable sensitivity.29,133 Moreover, other studies did not confirm these findings and reported markedly lower sensitivities of 41–55%.30,134,135
Immunohistochemistry
Immunohistochemical staining is a widely available method in order to investigate specific tumorigenesis-related protein expression patterns in brush and biopsy samples. In CCA, patterns of multiple antibodies have been evaluated in several studies, including insulin-like growth factor II mRNA-binding protein-3 (IMP3), tumor-associated protein p53, Mac-2 binding protein (Mac-2BP), interferon α-inducible protein 27 (IFI27), urokinase-type plasminogen activator (uPA), and S100P. Sensitivity and specificity of immunohistochemical staining of these proteins are reported in Table 2.101,136–149
Review of literature. Sensitivity and specificity of immunohistochemistry in CCA.
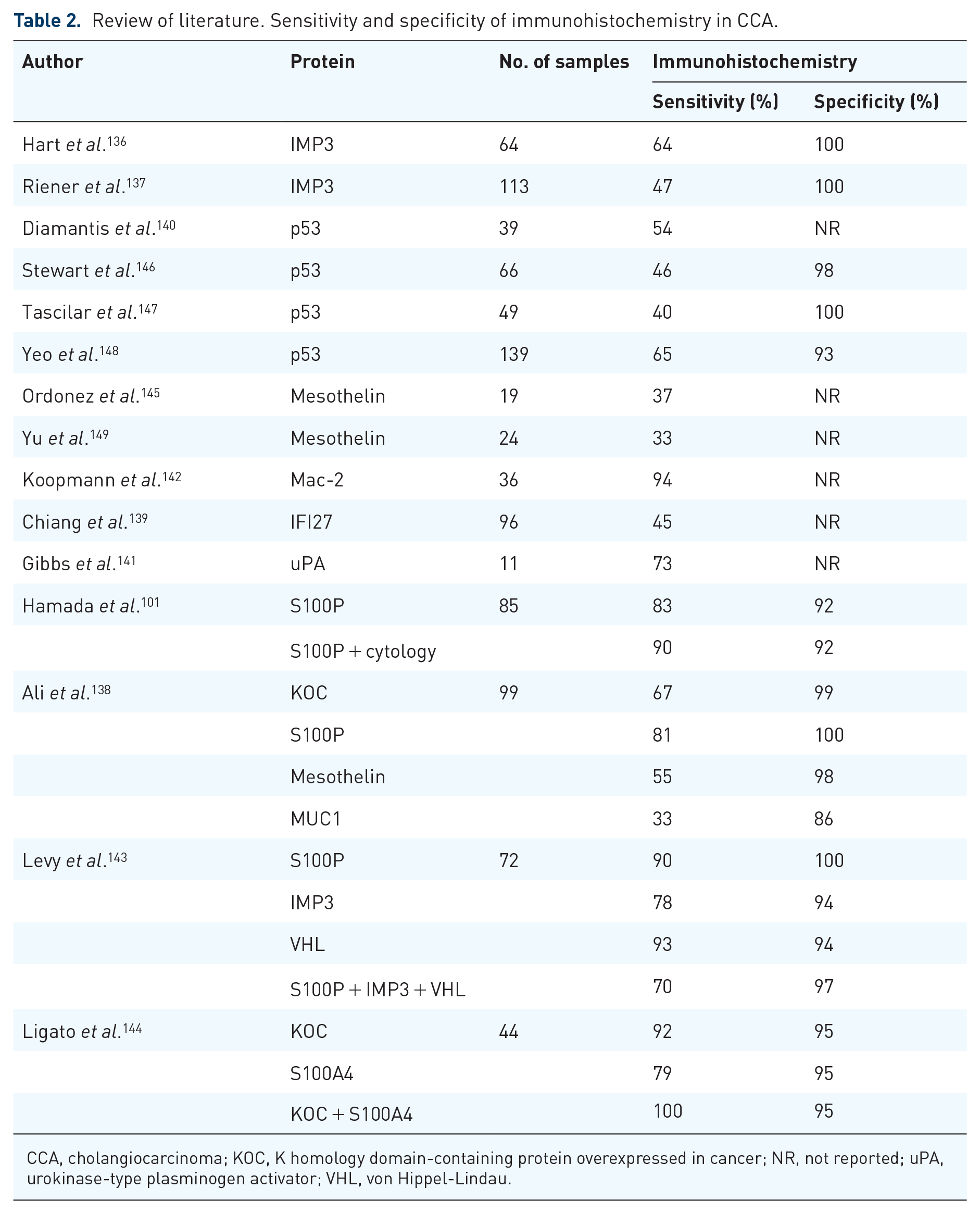
CCA, cholangiocarcinoma; KOC, K homology domain-containing protein overexpressed in cancer; NR, not reported; uPA, urokinase-type plasminogen activator; VHL, von Hippel-Lindau.
IMP3 is an oncofetal RNA-binding protein expressed in embryologic epithelial cells, myocytes and placental tissue during embryogenesis. 137 In several, mainly gastrointestinal malignancies, protein expression can be significantly upregulated. 136 It has been demonstrated that 58–78% of bile duct carcinomas show intermediate to strong cytoplasmic staining of IMP3.137,143 IMP3 is also expressed in biliary tissue with low or high-grade dysplasia. 137 Sensitivity and specificity of IMP3 immunohistochemistry for the diagnosis of CCA are 64% and 100%, respectively. When staining is combined with routine cytology, sensitivity increases to 72% with a specificity of 100%. 136 Positive IMP3 immunohistochemistry is often associated with concurrent p53 overexpression and enhanced tumor proliferation, indicated by a significantly higher ki-67 staining in tumor tissue with a strong IMP3 expression, and is associated with reduced survival rates.
The protein p53 is a product of the TP53 tumor suppressor gene, in which mutations occur in many cancers, including biliary carcinomas. 150 Staining biliary brush cytology and biopsies for p53 may be helpful to identify malignant strictures in samples showing indeterminate results on conventional brush cytology, since the majority of CCAs with TP53 mutations demonstrates overexpression of p53 protein.148,151 In 17–60% of CCAs, an increased p53 protein expression is observed, more often in poorly differentiated neoplasia as compared with well differentiated neoplasia.140,146,147,152
Mac-2BP is a cytoplasmic glycoprotein that interacts with galectin-3 on tumor cell surfaces and plays an important role in cell adhesion. 153 Approximately 94% of bile duct carcinomas show Mac-2BP expression, suggesting that this protein could be a diagnostic marker for biliary carcinomas. 142 Nevertheless, due to a lack of data, Mac-2BP immunohistochemistry is not recommended at present for differentiation between benign and malignant biliary strictures.
IFI27 is an oncogene involving the innate immune system and cell proliferation.139 High expression leads to cell proliferation and invasion. IFI27 knockdown in CCA has been shown to lead to cell cycle arrest in the S-phase, resulting in a lower cell proliferation rate. 154 In addition to diagnostic applications, this finding suggests that IFI27 might also be a potential treatment target.
Immunohistochemical analysis of uPA has been performed in a pilot study with 11 patients with known or suspected malignant strictures, demonstrating expression of this protein in 73% of the biliary cytology samples obtained during ERCP. 141 uPA plays a role in tumor invasion, growth, and cellular migration leading to the development of metastases.
S100P is a member of the S100 calcium-binding proteins and is involved in multiple cellular processes, including cell cycle progression and differentiation. An increased expression level is frequently observed in CCA, while no expression is seen in healthy bile duct tissue. 101 It has also been shown that S100P expression level increases during pancreatic carcinogenesis. 155 Moreover, cell proliferation, survival, motility, and invasion are stimulated by S100P expression in an in vitro culture of cancer cells. 156
Lastly, biomarker panels have been suggested in the detection of CCAs, including KOC, S100P, and mesothelin. 138 At least two of these biomarkers need to be positive to achieve a sensitivity of 100% and a specificity of 99%. K homology domain-containing protein overexpressed in cancer (KOC) is an oncofetal RNA-binding protein, frequently positively expressed in pancreatic carcinomas. 157 In addition, one-third of CCAs demonstrates mesothelin overexpression, a differentiation antigen with expression normally limited to mesothelial cells.145,149 Another suggested immunohistochemical biomarker panel includes S100P, von Hippel-Lindau gene product (pVHL), and IMP3. Negative staining of S100P and IMP3 combined with positive staining of pVHL can be found in a significant number (69%) of biliary adenocarcinomas. 143 The VHL protein has a tumor suppression function, meaning that carcinogenic development can occur in case of inactivation of both VHL alleles. 158
In summary, many immunohistochemical tumor markers have been investigated with varying success rates. However, full appreciation of these ancillary techniques is hindered by the low number of carcinomas evaluated in these studies. Due to this lack of data, none of the available immunohistochemical markers is recommended for routine use in clinical practice.
Chromosomal instability
Biliary tract tumors are genomically unstable, leading to chromosomal copy-number alterations. 159 Several techniques can be used to demonstrate DNA quantity and abnormalities, including digital image analysis, microsatellite instability, fluorescence in situ hybridization and next-generation sequencing (NGS).
Digital image analysis (DIA) quantifies DNA content, chromatin distribution, and nuclear morphology, by using a camera interfaced with a microscope. The determination of these characteristics may help to distinguish between benign and malignant biliary strictures. However, the accuracy of DIA is quite disappointing and comparable to routine brush cytology with a described sensitivity of 40% and specificity ranging between 77% and 92%.85,91
Analysis of microsatellite DNA is also widely used for detecting different types of cancer. Microsatellites are short highly polymorphic DNA fragments composed of nucleotide repeats, located on many positions within the genome. 160 In genomic unstable tumor cells, microsatellites may be gained or lost, implying allelic imbalance. In addition, in tumor cells, defective in DNA mismatch repair, microsatellites are prone to deletion and insertion mutations during DNA replication. This phenomenon is known as microsatellite instability (MSI) and can be the result of defects in mismatch repair genes, including mutL homolog 1 (MLH1), mutS homolog 2 (MSH2), and mutS homolog 6 (MSH6). 161 In addition, tumor clonality can be explored with microsatellite analysis. Microsatellite assays are able to detect identical genetic alterations in body fluids as found in the corresponding primary tumors, illustrating the clonality of the tumors. 162
Furthermore, fluorescence in situ hybridization (FISH) is a valuable strategy to add to the diagnostic work-up of biliary strictures. This technique utilizes fluorescently labelled complementary DNA probes targeting specific chromosomes or chromosomal loci of individual cells. 163 It allows detecting aneuploidy of chromosomes in biliary brushes or biopsies in order to distinguish between CCA and benign bile duct strictures (Figure 2). Several studies have investigated sensitivity and specificity of FISH on biliary brush samples in CCA and PSC-associated CCA. Table 1 provides an overview of 60 studies on both diagnoses. For CCA (non-PSC) reported sensitivity ranges from 31% to 65% and specificity is consistently reported close to 100%, and for PSC-associated CCA sensitivity ranges from 29% to 88% and specificity from 57% to 100%. All but one studies have included invasive carcinoma as study samples for FISH analysis; only Kushnir et al. 108 have included both invasive carcinoma and high-grade dysplasia. The range of reported sensitivities is considerable, which may be caused by various methods and endpoints used in the studies, including different probe sets and polysomy and/or trisomy as endpoints. Mostly, FISH leads to an enhanced accuracy of CCA diagnosis in addition to brush cytology assessment.87,111,120 Polysomy of chromosomes 3, 7, and 17, and loss of chromosome locus 9p21 are well-known genetic alterations in malignancies.99,164 Also in patients with PSC, polysomy is suspicious for the presence of biliary neoplasia, especially in combination with an elevated serum CA-19.9 level.165,166 For the detection of CCA an optimized set of four probes has been suggested, containing DNA probes directed to chromosomes 1q21, 7p12, 8q24, and 9p21. 92 CDKN2A is a 9p21 gene of which homozygous deletion and loss of heterozygosity is regularly found in CCA (5% and 20%, respectively). 167 In addition, FGFR genetic aberrations are also common in CCA, including FGFR2 fusions and amplification of FGFR19 and FGFR3.168,169 FGFR2 fusions are typically observed in small-duct intrahepatic CCA. 170 Amplification of ERBB2 and EGFR are less common in CCA, but both are also described in around 5–8% of patients.150,171 Amplification of these genes leads to overexpression of the corresponding proteins.
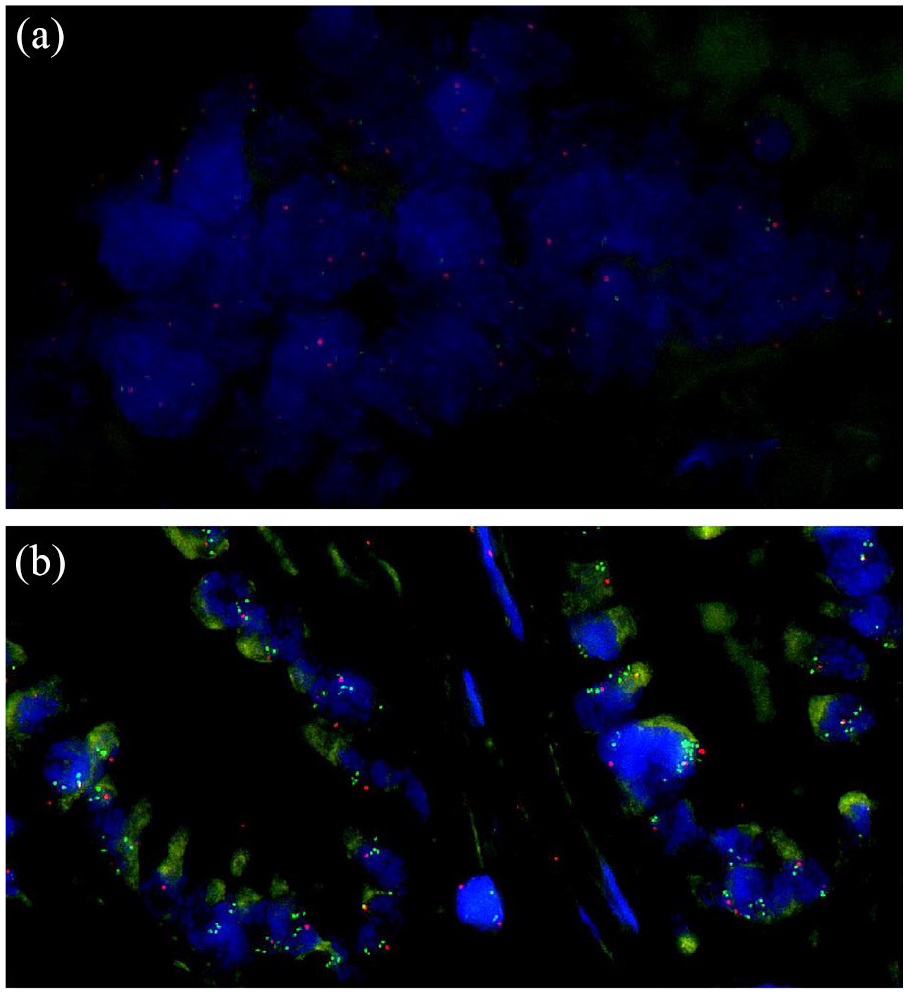
Examples of FISH negative (a) and positive (b) cases for MYC copy-number assessment. (a) Tumor cells with two signals for each of the two probes. (b) Tumor cells with MYC amplification.
Finally, chromosomal imbalance can also be detected by NGS. Several algorithms have been currently developed to identify copy-number variations based on targeted amplicon sequencing data.172–175 In addition, single-nucleotide polymorphism (SNP) analysis is another suitable NGS strategy to detect chromosomal imbalance. 176 During this analysis highly polymorphic SNP amplicons are included in the NGS panel, after which the status of each SNP can be interpreted as homozygous (noninformative), balanced heterozygous, or imbalanced heterozygous (loss or amplification of one allele).
Mutation profiling
Many studies have identified mutations in resection specimens of CCA, including in the genes TP53, KRAS, ARID1A, and IDH1.150,164,177–180 Mutations in TP53 are most common in biliary tumors with a prevalence of 26%. 150 Also KRAS mutations are frequently detected in CCA (17%). Some studies have even reported a prevalence of KRAS mutations in resection specimens and biliary brushes up to 30–40%.181–185 In addition to the previous described chromosomal imbalance of FGFR, also mutations have been identified in this gene family, more often in FGFR2 than in FGFR1, FGFR3, and FGFR4. 168 In contrast to an aggressive tumor progression in case of TP53 and KRAS mutations, FGFR mutations correlate with a significantly indolent disease course. 186 Furthermore, in addition to the previously described amplification of EGFR in a small number of CCA patients, somatic mutations of the EGFR gene have also been described in biliary tract cancer. 187 Patients with these mutations might benefit from EGFR inhibitors, such as gefitinib or erlotinib, which are used in EGFR mutant lung cancer.188–190 Mutations in GNAS and PIK3CA are less frequently observed alterations in CCA.183,191
Mutations differ between intrahepatic and extrahepatic CCA. TP53 and KRAS mutations are more common in extrahepatic than in intrahepatic CCA (45% versus 35% and 40% versus 24%, respectively). Furthermore, mutations in IDH1, ARID1A, BAP1, MCL1, NRAS, and PBRM1 are mainly detected in intrahepatic CCA, while ERBB2, SMAD4, FBXW7, BRAF, and CDKN2A alterations are mainly identified in extrahepatic CCA.192–196 A systematic review reported frequencies of IDH1 mutations in intrahepatic CCA ranging from 5% to 56%, while less than 1% of the extrahepatic CCA showed IDH1 mutations. 197 Mutations in PSC-associated CCA (PSC-CCA) are largely identical to mutations in sporadic (non-PSC-related) CCA, including alterations in TP53, KRAS, IDH1, and SMAD4. 198 In comparison to sporadic CCA, ERBB2 mutations and amplification are more often observed in PSC-CCA.198,199
NGS in both intraductal brushes and biopsies is promising for standard diagnostic care in biliary strictures. Different sequencing technologies are available, including Sanger, targeted sequencing, whole exome sequencing, and whole genome sequencing. 200 All methods have unique characteristics, leading to differences in coverage, read length, ideal DNA input, and limit of detection for mutations, and are therefore not all appropriate for mutation profiling in brush cytology. Singhi et al. 123 have identified mutations in a considerable number of biliary brushes and biopsies by performing targeted NGS with a large gene panel. With this gene panel a sensitivity of 73% and specificity of 100% was reached for biliary neoplasia. The most prevalent genomic alterations consisted of mutations in KRAS, TP53, CDKN2A, SMAD4, PIK3CA, and GNAS. Overall, when routine cytology was combined with targeted NGS with a large gene panel, sensitivity increased from 36% to 60–80% with a specificity of 99%.123,185 Kushnir et al. 108 demonstrated that the combination of brush cytology, FISH analysis, and mutation profiling of KRAS resulted in an increase of the diagnostic performance with a sensitivity of 66% and a specificity of 100%. When a large gene panel of 39 potentially interesting genes was added to brush cytology examination, sensitivity increased to 85%, with a maintained high specificity of 96%. 164 The promising data of mutation profiling need further confirmation.
Mutation profiling on liquid biopsies is a promising technique for the diagnosis of CCA, especially when tissue sampling fails, and to monitor tumor response to (targeted) therapy.201–204 Genetic profiles of cell-free (cf)DNA in blood samples have been shown to match the profiles in CCA.205–207 As an alternative, the mutation profiles may be investigated in bile samples. According to a small case series, cfDNA genetic profile analysis in bile has a higher diagnostic value as compared with blood samples for the detection of mutation profiles of CCA. 208 In addition, mutations may also be found in cfDNA from the supernatant fluids after biliary flushing during ERCP, similar to observations in samples obtained during bronchoscopy. 209 This technique requires further evaluation for biliary lesions. Another promising technique is the detection of circulating free microRNAs. MicroRNAs are small noncoding RNAs (21–23 nucleotides) that play an important role in cancer development by influencing many cellular processes, such as differentiation, proliferation, and apoptosis.210,211 Due to the remarkable stability of cfmicroRNAs in blood, these can be detected by various methods, including northern blotting, in situ hybridization, RT–PCR, microarray, and deep sequencing.212,213 Bernuzzi et al. 214 have identified several promising microRNAs as biomarkers for the diagnosis of PSC and CCA, including miR-200c for PSC and miR-194 for CCA. The analysis of microRNAs in serum might differentiate between PSC and CCA. According to a cohort study of 40 PSC, 40 CCA, 20 primary biliary cholangitis patients and 40 controls, the miR-483-5p and miR-222 were significantly more upregulated in CCA as compared with PSC. More data on the diagnostic value of microRNAs for the differentiation of benign and malignant biliary strictures are eagerly awaited.
In addition to diagnostic purposes, identifying mutations might also be important for the treatment of CCA. In several cancer types, suitable molecular targets have been identified for individualized targeted therapy, including EGFR and BRAF mutations in lung cancer and melanoma, respectively.215,216 The development of molecular targeted therapies in CCA is challenging due to the large variety of genetic aberrations.217–219 However, mutations in genes as ERBB2, EGFR, and PIK3CA are detected and agents with these genes as targets have been suggested for targeted therapy in CCA. 220 Bankov et al. 221 identified 79 mutations in 39 intraductal biopsies of 16 CCA patients with targeted NGS, resulting in potentially therapeutic targets in 6/16 patients: EPHA2, ERBB2, PIK3CA, and PTEN. Unfortunately, several clinical trials have been performed in CCA with inhibitors of various targets in oncogenic pathways, including MEK1/2, EGFR, ERBB2, and BRAF/VEGFR, all of which did not show a pronounced clinical benefit.222–225 Also in PSC-CCA, potentially actionable molecular targets were identified, including ERBB2, EGFR, MET, and MYC. 199 However, clinical data of targeted therapy in PSC-CCA is scarce.
Methylation
Aberrant DNA methylation contributes to carcinogenesis in several types of tumors by inactivating tumor suppressor genes. 226 Normally, DNA methylation occurs at CpG sites, which are DNA regions where a guanine nucleotide follows a cytosine nucleotide. Hypermethylation is mostly located in the promoter regions, while dense CpG islands are usually devoid. Promoter hypermethylation of the CpG islands leads to transcriptional repression. 227 In addition, global hypomethylation is also often found in neoplastic cells. 228 However, the precise role of the hypomethylation and the mechanism to silence tumor suppressor genes are unclear. The epigenetic change of aberrant DNA methylation induces silencing of tumor suppressor genes and appears to be an early event in carcinogenesis. 229 In many tumor types, including lung cancer, methylation plays a critical role in the initiation and progression of neoplasia.230,231 Tumor suppressor genes MLH1, p14ARF, CDKN2A, CDH1, NPTX2, TFPI-2, APC, TIMP3, RASSF1, and DAPK are commonly methylated in CCA.226,232,233 Approximately 80–85% of the biliary tract tumors shows methylation in at least one known tumor suppressor gene, while around 70% of CCA demonstrates methylation in at least three genes.226,234
In addition, inactivation of tumor suppressor microRNAs by DNA methylation is frequently found in CCA. 232 MicroRNAs are involved in several pathways and deregulation of these small molecules can lead to progressive carcinogenesis of different types of cancer. 235 For example, tumor suppressor miR-370 can be inactivated by DNA methylation in tumor cells of bile duct carcinomas. 236 MiR-370 is downregulated by a high expression of interleukin-6, a pro-inflammatory cytokine. Overexpression of interleukin-6 is often found in CCA and may contribute to tumor cell growth by miRNAs deregulation, including miR-370. 237
Detection of DNA methylation of tumor suppressor genes and microRNAs could be a valuable diagnostic tool for CCA. Besides, DNA methylation inhibitors might be promising for the treatment of these tumors. Further investigations of these techniques are required.
Conclusion
CCA is commonly diagnosed at an advanced stage and has a devastating prognosis. A low diagnostic yield of currently available diagnostic methods contributes to the late diagnosis of CCA. Genetic factors and a variety of signaling pathways are involved in the carcinogenesis and are potential diagnostic targets. Current diagnostic work-up consists mainly of routine cytology examination, and in some centers the standard is assisted by FISH analysis or p53 immunohistochemistry. Genetic alterations and aberrant DNA methylation in biliary brushes and/or biopsies are promising diagnostic and prognostic markers for CCA, which are already implemented in the diagnostic work-flow of some laboratories. Amplification of 1q21, 7p12, 8q24, ERBB2, EGFR, and FGFR genes are observed in biliary tumors, as well as CDKN2A loss and FGFR2 fusions. The majority of these potential markers could be identified by FISH or NGS and aid in a timely diagnosis of CCA. Prompted by the described considerations, in case of a high suspicion of malignant stricture, we would recommend obtaining both brush cytology and fluoroscopic-guided biopsies. NGS should be performed on this biliary material when results are inconclusive. A prominent position of molecular analyzes for the diagnosis of CCA may be awaited in the near future.
Supplemental Material
sj-jpg-1-tag-10.1177_17562848211002023 – Supplemental material for Optimal tissue sampling during ERCP and emerging molecular techniques for the differentiation of benign and malignant biliary strictures
Supplemental material, sj-jpg-1-tag-10.1177_17562848211002023 for Optimal tissue sampling during ERCP and emerging molecular techniques for the differentiation of benign and malignant biliary strictures by Eline J. C. A. Kamp, Winand N. M. Dinjens, Michail Doukas, Marco J. Bruno, Pieter Jan F. de Jonge, Maikel P. Peppelenbosch and Annemarie C. de Vries in Therapeutic Advances in Gastroenterology
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
