Abstract
Objective
To investigate BRCA1/2 gene mutations and their relationship with clinicopathological features in patients with breast cancer in Zhoushan Islands.
Methods
High-throughput whole-exome gene sequencing was used to detect BRCA1/2 mutations in 776 breast cancer patients in Zhoushan Islands.
Results
The BRCA1/2 mutation rate of breast cancer patients in Zhoushan Islands was 4.38% (34/776). BRCA1 mutations were significantly correlated with age, molecular type, and family history of breast and ovarian cancers. BRCA2 mutations were most commonly found in invasive lobular carcinoma. Moreover, the BRCA2 mutation rate of cancers with molecular type luminal B (receptor protein–tyrosine kinase [HER2]-negative) was also relatively high.
Conclusion
The rate of BRCA1/2 mutations in breast cancer patients from Zhoushan Islands is approximately 4.38%, and BRCA1 mutation is related to age, molecular type, and family history of breast and ovarian cancers.
Introduction
In China, breast cancer is the most common cancer in women. 1 BRCA1/2 gene mutations can be detected in more than 25% of hereditary breast cancer cases, and account for 5% to 10% of all breast cancers. 2 At most, pathogenic mutations of BRCA1 and BRCA2 raise the risk of ovarian cancer by 54% and 19%, and of breast cancer by 78% and 56%, respectively. 3 These genes are also related to many other malignancies4–6 such as fallopian tube cancer, 7 endometrial cancer, 8 melanoma, 9 and colorectal cancer. 10 Genetic testing for pathogenic BRCA1 and BRCA2 variants is strongly recommended for women with a personal or family history of breast, ovarian, fallopian tube, or peritoneal cancer. Moreover, prophylactic bilateral salpingo-oophorectomy is recommended for women with pathogenic BRCA1/2 pathogenic variants; the procedure reduces the risk of ovarian cancer by more than 80%. 11
BRCA1/2 expression status not only guides the prevention and screening methods for breast cancer, but also determines treatment and prognosis. Notably, BRCA mutation rates are inconsistent across different ethnic populations. Studies have indicated lower BRCA mutation rates in Asians than in Caucasians in Western countries.12–15 It is thus critical to investigate BRCA variations in different ethnic populations to improve cancer risk management in these populations. We therefore investigated BRCA1/2 mutations in breast cancer patients in Zhoushan Islands, Zhejiang Province, China, to clarify the genetic variation and clinicopathological characteristics of these patients. We aimed to provide the necessary data to enhance cancer risk assessment, prevention, treatments, and follow-up strategies for this Chinese population.
Materials and methods
Study participants and data collection
We enrolled breast cancer patients with complete data in Zhoushan Islands from 2001 to 2018. In this retrospective study, blood samples were collected from each participant for BRCA1/2 mutation screening after informed consent was signed.
Patients’ clinical records were used for the molecular classification of breast cancer, which was performed by referring to the guidelines and standards for the diagnosis and treatment of breast cancer of the China Anti-Cancer Association Committee of Breast Cancer Society. The molecular classification of breast cancer included luminal A (LA), luminal B (LB), HER2-positive, and triple-negative breast cancer (TNBC) types. LA types were estrogen receptor (ER)- and progesterone receptor (PR)-positive, receptor protein–tyrosine kinase (HER2)-negative, and had proliferation marker protein Ki-67 ≤ 20%. LB types were further divided into LB HER2-negative (ER- and/or PR-positive, HER2-negative, and Ki-67 ≥ 20%) and LB HER2-positive (ER- and/or PR-positive, HER2-positive) types. HER2-positive types were ER- and PR-negative but HER2-positive. TNBC types were ER-, PR-, and HER2-negative.
The study was approved by the Ethical Review Committee of Zhoushan Hospital (No. 2018177), and informed consent was obtained from all participants or their families. We have de-identified all patient details. The reporting of this study conforms to Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 16
BRCA1/2 gene variant detection and analysis
Blood samples were collected and genomic DNA was extracted using the Blood Genomic DNA Extraction Kit (Shanghai Lifefeng Biotech Co., Ltd., Shanghai, China). Shrimp alkaline phosphatase enzyme was used to eliminate excess primers and deoxynucleotide triphosphates in amplification reactions. To repair the ends of the digestion reaction, bases were added. Sequencing adaptors (Illumina, San Diego, CA, USA) were ligated to the end-repaired fragments with T4 DNA ligase (Agilent, Santa Clara, CA, USA), and the fragments were then purified with magnetic beads and 80% ethanol. The HiSeq X Ten high-throughput sequencing platform (Illumina) was adopted for library sequencing, with an average sequencing depth of over 1000×. The sequencing results were aligned to the reference sequences of BRCA1 (NM_0073000) and BRCA2 (NM_000059) for mutation detection using Burrows–Wheeler Aligner. 17 Meanwhile, the Genome Analysis Tool Kit 18 was used to recalibrate mutation sites with ANNOVAR. 19 The interpretation of all variants referred to the American College of Medical Genetics genetic interpretation principles. 20
Statistical analysis
Data were analyzed statistically using IBM SPSS Statistics for Windows, version 22.0 (IBM Corp., Armonk, NY, USA). Counting data are represented as frequencies and rates. The paired chi-square test was used to analyze paired classification data, and Fisher’s exact method was used when the theoretical frequency in the chi-square test was less than 1. Significance was determined as p < 0.05.
Results
Study population
In our study of 776 breast cancer patients, the oldest patient was 80 years old, and the average age was 50.16 ± 9.31 years. Sixty-eight patients were diagnosed at stage 0 (ductal carcinoma in situ [DCIS]), and 350, 260, 90, and 9 cases were diagnosed at stages I, II, III, and IV, respectively. Histopathological results revealed that 624 patients had invasive ductal carcinoma, 29 had invasive lobular carcinoma (ILC), 68 had DCIS, 24 had mucinous carcinoma, and 31 had other pathological types. Furthermore, 6.19% of the total population had a family history of breast or ovarian cancer (Table 1).
Clinicopathological characteristics of enrolled cases.
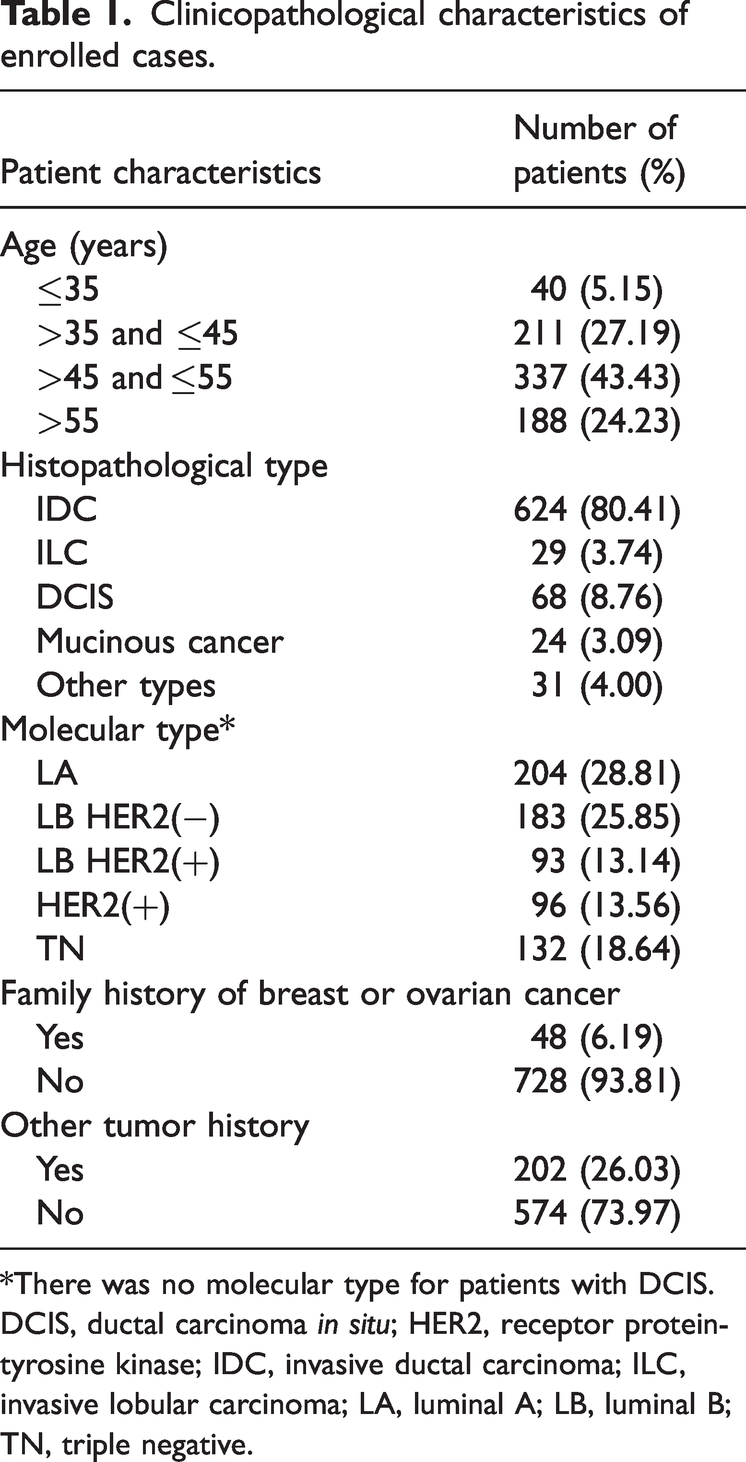
*There was no molecular type for patients with DCIS.
DCIS, ductal carcinoma in situ; HER2, receptor protein-tyrosine kinase; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma; LA, luminal A; LB, luminal B; TN, triple negative.
Correlations between BRCA1/2 gene mutations and clinicopathological characteristics
There were 774 benign variants, 27 likely benign variants, 44 variants of uncertain significance, 1 likely pathogenic variant, and 33 pathogenic variants in the 776 cases. Of the total patients, 4.38% had BRCA1/2 mutations. The mutation rate for each of BRCA1 and BRCA2 was 2.19%, and no patients tested positive for both mutations. Correlations between clinicopathological variables and BRCA mutation status were evaluated using the chi-squared test (Table 2). BRCA1 mutations were significantly correlated with age (p = 0.032), molecular type (p = 0.022), and family history of breast and ovarian cancers (p < 0.001). By contrast, no significant correlations were identified with BRCA2 mutations.
Correlations between BRCA1/2 mutations and clinicopathological characteristics.

*There was no molecular type for patients with DCIS. #Fisher’s exact test.
DCIS, ductal carcinoma in situ; HER2, receptor protein-tyrosine kinase; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma; LA, luminal A; LB, luminal B; TN, triple negative.
BRCA1 mutations did not occur in any patients with ILC, DCIS, or mucinous cancer; they only occurred in invasive ductal carcinoma (2.56%) and other less common pathological types (3.23%). By contrast, BRCA2 was mutated in all four cancer types, with the highest mutation rate in invasive lobular cancer (10.34%). The BRCA1 mutation varied significantly among different molecular types, with the highest mutation rate in TNBC and no identified mutations in HER2-type cancers. Furthermore, the LB HER2-negative type had the highest percentage of BRCA2 mutations (4.37%), whereas no mutations were identified in the LB HER2-positive type (Chart 1). Although the BRCA1 mutation rate was significantly higher in patients with a family history of breast or ovarian cancer than in those without a family history, there was no significant difference between these two groups in BRCA2 mutation status.

BRCA1/2 mutations and molecular types.
Discussion
According to statistical data from 2015, breast cancer incidence is the highest among female malignancies, 21 and is estimated to keep this top rank in 2022. 22 The survival rates for breast cancer types are lower in China than in the USA, 23 indicating that efforts are needed in China to deliver more effective cancer care, including targeted screening of high-risk populations.
Both BRCA1 and BRCA2 variants are associated with increased risk of breast cancers. 24 BRCA1/BRCA2 are cancer suppression genes that play crucial roles in activating DNA repair in response to cellular stress. Their tumor-suppressive effects have been attributed mainly to their cell-cycle checkpoint and DNA repair roles.25,26 BRCA testing is recommended in high-risk populations, including those with a personal or family history of BRCA mutation-positive ovarian or breast cancer, breast cancer diagnosed before 45 years of age, and TNBC. 27 The identification of BRCA mutation status can impact subsequent prevention and treatment decisions, as well as prognosis evaluation. Increasing evidence indicates that BRCA1 and BRCA2 mutation carriers have poorer outcomes than non-carriers.28–30 In China, however, many high-risk individuals do not undergo BRCA mutation testing because of difficulties with medical payments.
In the present study, we screened unselected breast cancer patients in Zhoushan Islands for BRCA1/2 mutations for the first time. In this population, BRCA1 gene mutation was age-related. It was more likely to be mutated in younger breast cancer patients (≤45 years), and especially in those younger than 35 years, which is the recommended age for BRCA1/2 gene mutation detection according to the Chinese Society of Breast Surgery. 31 BRCA1/2 mutations (especially those of BRCA1) were more likely to occur in populations with a family history of breast or ovarian cancer. Furthermore, when considering histological cancer types, BRCA2 mutations appeared more common in invasive lobular carcinoma than in other pathological types in our study. This finding was distinct from the results of a Korean study, in which only 10% of BRCA2 mutations were associated with lobular cancer types.32,33 Additionally, DCIS is reportedly rare in BRCA1 variant carriers, but relatively common in BRCA2 mutation carriers. 34 However, only 1.47% of patient with DCIS had BRCA2 mutations in our study.
BRCA1/2 gene mutations are closely related to molecular type in breast cancer. In a study of 146 genetic counseling cohorts, 64 patients with early TNBC were detected as carrying BRCA1/2 gene mutations, with a mutation rate of 43.8%. 35 Two China-based studies with small samples36,37 reported gene mutation rates of 18.80% and 16.36% in patients with TNBC; however these percentages remain much higher than our finding of 7.58%. In the present study, BRCA2 mutation was most common in the LB HER2-negative molecular type, accounting for 4.37% of cases. Notably, the reported gene expression profiles of BRCA1 and BRCA2 are completely different, with BRCA1 mutants mostly ER-negative and BRCA2 mutants mostly ER-positive, 38 and BRCA2 mutation rates are reportedly higher in ER-positive patients than in ER-negative patients. This finding is similar to our results, although BRCA mutation rates showed only a very small difference between the ER-positive and -negative groups. However, the main limitation of the present study is that the sample size was relatively small.
Zhoushan Island is relatively isolated. The cross-sea bridge was only opened to traffic in 2010, and there is relatively little population movement. Furthermore, the islanders' diet is dominated by seafood and a high salt intake, 39 which may be associated with BRCA gene mutation. The BRCA1/2 gene mutation features of Zhoushan Island are thus likely different from Western populations, and even from those of other Asian countries. Compared with populations from other regions with geographical proximity to the sea, the BRCA1/2 mutations of patients with breast cancer in Zhoushan Islands were lower than those reported in Korea,33,40 Japan,34,41 and Sicily 42 , regardless of whether patients were unselected or had breast/ovarian cancer (Chart 2).

BRCA1/2 mutations in different geographical regions.
In the present study, BRCA1 gene mutations were closely associated with a younger age, TNBC, and a family history of breast or ovarian cancer, and BRCA2 variants were more common in ER-positive and ILC subtypes. The findings of these characteristics of BRCA mutation status will enrich Chinese data regarding breast cancer gene mutations, and may improve individualized cancer prevention and treatment. Further studies into BRCA variants among healthy populations should be performed to build comprehensive cancer risk management strategies for women with BRCA1/2 variants.
Footnotes
Acknowledgements
We appreciate the guidance and help provided by Professor Zhen Hu’s team at the Affiliated Cancer Hospital of Fudan University in Shanghai and Dr Jiaxin Li at Zhejiang University School of Medicine. We also thank the breast surgery nursing team of Zhoushan Hospital for their active participation.
Author contributions
Conceptualization: SB, XQ; formal analysis: XQ, NS, YL; funding acquisition: XQ; investigation: NS, YL, JS, JX, YZ; project administration: XQ; resources: SB, XQ, NS, YL, JS, JX, YZ; supervision: XQ; software: XQ; validation: XQ; writing – original draft: SB; writing – review & editing: XQ.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research was partly funded by the Science Foundation of Zhoushan Science and Technology Bureau (No. 2018C31108) to Xia Qiu.
