Abstract
Colorectal cancers (CRCs) remain one of the most common and challenging neoplasia in the Western world. The response rate of immunotherapeutic treatment approaches in a subset of advanced CRCs is remarkable and has sustainably changed treatment regimens. Unfortunately, currently available immunotherapeutics only displayed significant antitumoral activity – in terms of progression free survival (PFS) and objective response rate (ORR) – in microsatellite instability-high (MSI-H)/DNA mismatch repair deficient (dMMR) CRCs. Subsequently, these remarkable results had led to the US Food and Drug Administration’s approval of both immune checkpoint inhibitors (ICIs) pembrolizumab and nivolumab in the treatment of advanced MSI-H/dMMR CRCs. However, in microsatellite stable (MSS)/DNA mismatch repair proficient (pMMR) CRCs, ICIs have clearly failed to meet their expectations and are therefore not considered effective. As the vast majority of CRCs display a molecular MSS/pMMR profile, current treatment approaches endeavor to improve tumor immunogenicity that consecutively leads to increased proinflammatory cytokine levels as well as tumor infiltrating T-cells, which in turn may be targeted by various immunotherapeutic agents. Therefore, ongoing studies are investigating novel synergistic therapy modalities and approaches to overcome a “cold” to “hot” tumor conversion in MSS/pMMR CRCs. In this review, we summarize the efficacy and possible immune-related adverse events as well as novel therapeutic approaches of ICIs in the treatment of MSI-H/dMMR and MSS/pMMR CRCs.
Keywords
Introduction
Colorectal cancer (CRC) is the second-leading type of malignancy in the Western world, with an individual lifetime risk of about 6%. 1 Even though CRC is mainly diagnosed in older patients, a worrying increase in incidence and mortality has been observed among patients under the age of 50. 2 Oligometastatic CRCs confined to a single organ, usually the liver, are suitable for surgical and ablative interventions combined with systemic therapy, and go along with a 5-year and 10-year survival rate of 40% and 20%, respectively. 3 In contrast, unresectable metastatic CRCs (mCRCs) are incurable. Although there have been major advances in the understanding of the tumor biology as well as in treatment options over the last three decades, the overall 5-year survival in mCRC is still only around 20%. 4 Hence, more efficacious therapeutic strategies are urgently needed in the treatment of mCRC.
In the last years, significant breakthroughs have been accomplished in the understanding of CRC carcinogenesis and its interplay with the host immune system. This knowledge has been successfully translated into novel pharmacological interventions. 5 The approval of immune checkpoint inhibitors (ICIs) in melanoma was a complete game changer in cancer therapy. Their paramount success in the treatment of patients with metastatic melanoma has resulted in the introduction of these therapeutic agents in the treatment of many other types of cancer, for example, CRC. Patients with microsatellite instability-high (MSI-H)/DNA mismatch repair deficient (dMMR) mCRC demonstrated unprecedented objective response rates (ORR) and a substantial increase in overall survival after treatment with ICIs. 6 As a consequence, the US Food and Drug Administration (FDA) approved pembrolizumab (anti-PD-1-antibody) and nivolumab (anti-PD-1-antibody) as monotherapy or combined with ipilimumab (anti-CTLA-4-antibody) in the treatment of MSI-H/dMMR mCRC. Unfortunately, the vast majority of mCRCs (about 96%) are not MSI-H/dMMR but rather microsatellite stable (MSS)/DNA mismatch repair proficient (pMMR). Such patients, however, do not benefit from currently available immunotherapy modalities, which critically limits the impact of ICI for the treatment of CRC so far. 7 In line with this, studies in unselected and advanced mCRC patients did not report an improved clinical outcome of cancer immunotherapy interventions. 8 Different factors, such as low neoantigen burden, alterations in JAK/STAT pathways, local immunosuppression, among others, are currently believed to be responsible for the limited therapeutic impact of ICIs in these CRC tumors. 9 Moreover, biomarkers that predict potential responses in MSS/pMMR mCRCs and side effects of ICI treatment in CRC are currently not available. Therefore, this review article summarizes the current state of the art of ICIs in the treatment of mCRC, particularly focusing on their efficacy and potential immune related adverse events.
The rationale for ICI treatment in colorectal cancer
Mismatch repair (MMR) is an ancient and well-conserved system that detects, repairs and corrects base mismatches which emerge during DNA replication. Loss-of-function mutations in the MMR genes eventually lead to alterations in microsatellite lengths that are considered unstable as a result. A tumor is classified as MSI-H when more than 30% of microsatellites are unstable. 10 MSI-H/dMMR may develop sporadically as part of the driver mutation BRAFV600E sporadic 11 or in the context of hereditary non-polyposis colorectal cancer (or Lynch syndrome). 12 In clinical MSI/MMR diagnostics routine, polymerase chain reaction of at least five defined mononucleotide loci and immunohistochemistry are used to test for MSI-H/dMMR. Importantly, MSI-H/dMMR tumors represent a distinct CRC molecular subgroup, as they show a higher tumor mutational burden associated with higher neoantigen levels than MSS/pMMR tumors. Hence, dMMR CRCs are considered as being more immunogenic than pMMR CRCs.13,14 Moreover, it has been shown that MSI-H/dMMR tumors harbor a higher number of somatic mutations and, therefore, are believed to express an increased number of neoantigens that potentially activate the host’s immune system.
The tumor microenvironment (TME) is characterized by the dynamic interaction of cancer cells with their surroundings (e.g. extracellular matrix, vasculature, immune cells and cytokines). 15 But also nutrients, pH, oxygen levels and metabolites are recognized as part of the TME. 16 Moreover, TME is considered to be a driving force in terms of tumor heterogeneity and complexity. 17 One essential element of the TME is the immune system with its cellular components. Immune cells sense the environment for danger signals as well as for “foreign” (neo)antigens that may arise from genetic alterations within the tumor’s genome, such as single-nucleotide variants, deletions, insertions, among others.
According to recent reports, a high number of CD3+/CD45RO+ tumor-infiltrating lymphocytes is associated with improved outcome in CRC. Increased progression-free survival (PFS) as well as increased overall survival (OS) was observed in tumors that harbored a high fraction of infiltrating memory and CD8+ T-cells.18–20 These findings led to the development of the widely accepted and validated immunoscore. The immunoscore is calculated by assessing lymphocyte populations (CD3/CD45RO, CD3/CD8 and CD8/CD45RO positivity) in the invasive tumor margin as well as in the tumor core 21 and ranges from 0 (low density of cell types in both regions) to 4 (high density of cell types in both regions). It provides a validated prognostic parameter for OS, disease free survival and time to recurrence. 22 Currently, tumor staging by TNM classification is the gold standard. However, clinical outcome may vary within identical TNM tumor stages, as its prognostic accuracy is limited by the fact that it disregards the host immune response. 22
To characterize and standardize molecular subtypes of CRCs, the consensus molecular subtypes (CMS) classification is used and classifies CRCs into four distinct subtypes according to their gene expression pattern. 23 CMS1 is characterized by high mutational load, BRAF mutation or MSI status. About 13% of CRCs are defined as CMS1, which displays a beneficial prognosis. 24 CMS2, which accounts for 37% of CRCs, represents a group with increased EGFR expression, activated WNT- and MYC-pathways and high somatic alterations. 25 Thirteen percent of CRCs group into CMS3, which is defined by epithelial and metabolic dysregulation and KRAS mutation. Finally, 23% of CRCs are rated CMS4, which represents a group with stromal activation, TGF-β pathway and epithelial–mesenchymal transition and an unfavorable OS. The remaining 14% of CRCs are either mixed subtypes or unclassified patients. 26
Upregulation of the major histocompatibility complex (MHC) class I T cell-receptor as well as immune checkpoint proteins, such as programmed cell death 1 (PD-1), programmed cell death ligand 1 (PD-L1) and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), are used by tumor cells to stifle and therefore evade the immune system. Thus, they represent ideal therapeutic targets. The interaction of CTLA-4 with the surface proteins CD80 and CD86, which are expressed on antigen-presenting cells, dampens the activity of the effector immune cells. 27 PD-1 interacts with the two ligands PD-L1 and PD-L2 and promotes self-tolerance by suppressing inflammatory activity of T-cells. Therefore, PD-1 not only plays an eminent role in preventing autoimmune diseases but also hinders the immune system in killing cancer cells. 28 Overexpression of PD-1 in tumor cells located within an inflammatory microenvironment has been described. 29 PD-1/PD-L1 signaling pathway directly inhibits tumor cell apoptosis. Moreover, current data suggest that immunomodulatory drug-induced immune cell activation enables peritumoral lymphoid cells to successfully kill cancer cells upon PD-1/PD-L1 signaling pathway inhibition. 30 Furthermore, an upregulation of PD-L1 in the TME is associated with higher T-cell infiltration, which makes the tumor susceptible to ICI treatment. In addition, ICI treatment in these cases seems to be beneficial in regard of improved clinical outcome.
Clinically approved ICIs for the treatment of mCRC
Anti-PD-1/PD-L1 antibodies
Both PD-1 and PD-L1 play an essential role in the inhibition of T-cell function. PD-1 is mainly expressed on T cells.31,32 Pembrolizumab and nivolumab are two (monoclonal) antibodies that lead to T-cell activation and proliferation by binding PD-1, thereby making interaction with its ligands impossible. Pembrolizumab was the first ICI approved by the FDA, in 2017. Based on pooled results from several phase I/II trials it was approved for the treatment of pre-treated MSI-H/dMMR metastatic cancers in a tumor-agnostic fashion. 8 More recently, pembrolizumab obtained first line FDA approval based on the pivotal Keynote-177 trial demonstrating superior PFS and ORR for pembrolizumab over combination chemotherapy in previously untreated MSI-H/dMMR mCRC. 33
CTLA-4 inhibitors
CTLA-4 is expressed on the surface of T-cells and T regulatory cells (Tregs) and serves as a negative regulator of T-cell activation. Ipilimumab binds to CTLA-4 and therefore inhibits the disruption of co-stimulatory signals required for T-cell activation. 34 Low-dose (1 mg/kg) ipilimumab in combination with nivolumab is FDA-approved for pre-treated MSI-H/dMMR mCRC based on data from the Checkmate-142 trial. This combination is being further developed for first line treatment for this subgroup of mCRC. 35
The mode of action of the discussed ICIs is demonstrated in Figure 1.
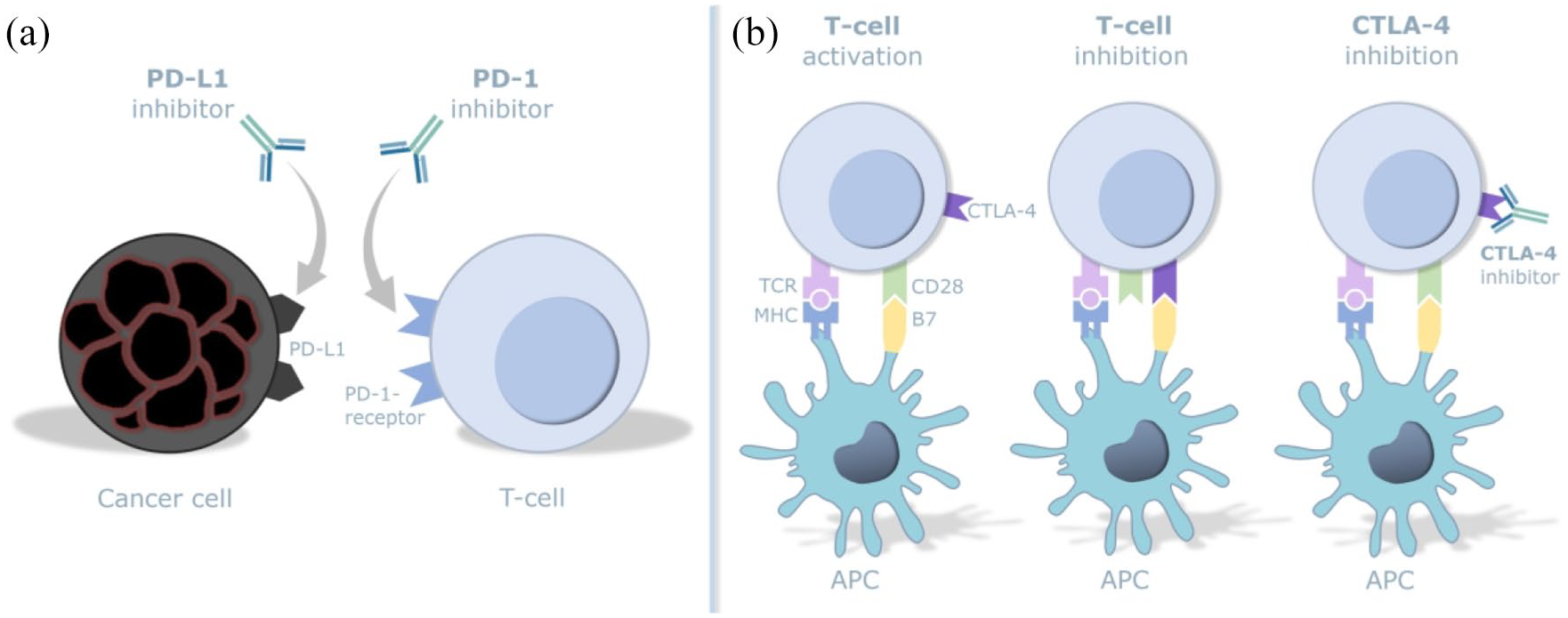
Effect mechanism of PD-1-, PD-L1- and CTLA-4-inhibitors. (a) Interaction of PD-1 on T-cells with PD-L1 on tumor cells is blocked by inhibitors preventing cancer evasion from the immune system. (b) CTLA-4, which mediates immunosuppression via the co-stimulatory receptor 28, leads to an activation of the immune system when inhibited.
The efficacy of ICIs in the treatment of CRC
Multiple molecular regulators controlling lymphocyte activation have been identified, and several of these have been studied as therapeutic targets for cancer treatment. As mentioned previously, the currently applied immunomodulatory therapeutics target critical checkpoint molecules that inhibit T-cell activation such as CTLA-4, PD-1 and PD-L1. Nevertheless, a clinical benefit has been demonstrated only in patients with MSI-H/dMMR mCRC previously treated with therapeutics that blocked inhibitory checkpoint molecules. 5
Role of immunotherapy in unselected CRC
Studies that investigated the effects of ICI treatment in unselected CRC patients displayed a poor clinical outcome. 8 For example, a phase I study that aimed to investigate the efficacy of nivolumab in a total of 19 patients with mCRC could not demonstrate a beneficial clinical response. 36 In addition, no significant clinical nor survival benefit was found in patients suffering from MSS/pMMR mCRC when treated with PD-1 monotherapy or dual checkpoint inhibition. 5 Although those study results were negative data, they have provided valuable information regarding potential indications of ICI treatment in CRC.
Since the vast majority of CRCs have an MSS/pMMR profile, novel synergistic therapeutic approaches (e.g. chemotherapy, radiotherapy, targeted monoclonal antibodies, among others), in combination with ICIs, might be required to turn those “cold” tumors “hot”.
Role of immunotherapy in selected CRC (MSI-H/dMMR)
As described previously, approximately 4% of mCRCs are MSI-H/dMMR. To date, mainly anti-PD-1 and anti-CTLA-4 antibodies are applied in the treatment regimen of these selected CRC subtypes. One of the initial attempts to treat MSI-H/dMMR CRCs by immunotherapy was by blocking PD-1 with pembrolizumab. This approach led to remarkable results in mCRC therapy. The ORR in pembrolizumab treated MSI-H/dMMR tumors was 40% compared with 0% in MSS/pMMR mCRCs. Furthermore, there was a 20-week PFS of 78%. 37 To explain this rather large disparity between the two tumor molecular subtypes, whole-exome sequencing was performed on these tumors and revealed an increased rate of somatic mutations among MSI-H/dMMR tumors as a possible explanation. Additional reports have also shown positive effects of another PD-1 inhibitor, nivolumab, in the treatment of MSI-H/dMMR refractory mCRCs that had previously been treated with chemotherapeutic agents such as fluoropyrimidine, oxaliplatin or irinotecan. 38 Nivolumab treatment revealed an ORR of 31% and a 1-year PFS of 50%. 39 Later, Overman and colleagues also tested the combinatory treatment of nivolumab plus ipilimumab. This therapy consolidation showed positive and even more promising results than nivolumab monotherapy as the ORR increased up to 54.6%. In addition, the 1-year PFS was as high as 71%. Furthermore, the 1-year OS was 12% higher in the dual therapy than in the nivolumab-only treatment. 40 Despite the above-mentioned promising results, a meta-analysis reported that ipilimumab administration is associated with a greater incidence of adverse events (AEs) compared with pembrolizumab and nivolumab. 41 PD-L1 inhibitors such as durvalumab have also been considered in the treatment of dMMR tumors, including mCRCs. A recent phase II study (NCT02870920) raises hope that OS in advanced refractory CRCs may be prolonged by durvalumab in combination with tremelimumab (CTLA-4 inhibitor). 42 Nevertheless, more data are needed to draw proper conclusions about its safety and efficacy profile. 43 Most recently, Keynote-177, the first randomized trial specifically recruiting MSI-H/dMMR mCRCs, showed superior PFS for patients treated with first line pembrolizumab compared with standard-of-care chemotherapy, establishing first line immunotherapy as standard-of-care for MSI-H/dMMR mCRC. 33
Side effects of ICIs in the treatment of CRC
Chemotherapeutics such as antimetabolites and cytotoxic agents have been widely applied in the first line anticancer treatment of oligo- and metastatic CRC in the last decades. It has been well documented in numerous studies that serious side effects may occur upon administration of these broadly acting and rather unspecific agents. Due to their adverse effects these treatment regimens often lead to regrettable therapy delays and treatment refusals, as well as decreased patient compliance.
Immune-related adverse events (irAEs) in response to ICI therapy on the other hand are different from the AEs induced by chemotherapeutic agents. ICIs are somewhat unique regarding their toxicity, organ involvement, severity and onset pattern compared with chemotherapeutic agents.44,45 Immune checkpoints are important in controlling the self-tolerance upon the induction of an immune response. 31 As anti-CTLA-4, anti-PD-1 and anti-PD-L1 monoclonal antibodies interfere with different stages of T-cell activation, distinct, albeit partially overlapping, toxicity profiles are seen among those drugs. 46 Nonetheless, the exact pathophysiological mechanisms of irAEs still remain unclear. 41 Severe irAEs are as high as 16% in anti-PD-1 and up to 27% in anti-CTLA-4 antibody treatment. Moreover, in combination, irAEs rise to as much as 55% of the patients. 47 Gastrointestinal, hepatic, pulmonary, skin and endocrine toxicity irAEs have been described and defined in multiple randomized controlled trials.41,48,49 Prior to an irAE diagnosis, secondary causes such as tumor progression or infections must be ruled out in cooperation with the respective specialists. Fortunately, most of the irAEs are well controllable by immunosuppressive therapeutics such as glucocorticoids, anti-TNF antibodies, mycophenolate mofetil or cyclophosphamide, among others, and by supportive therapy approaches. Nevertheless, life-threatening and fatal side effects are a serious concern in regard to safety of ICI treatments. 36 Most frequent irAEs are summarized in Table 1.
Summary of the most common irAEs in different organs and systems.

irAE, immune-related adverse event.
Gastrointestinal and hepatologic toxicity
Diarrhea, which is the most frequent gastrointestinal side effect, is seen in up to 33% of patients treated with ipilimumab and in up to 19% among patients that are under anti-PD-1 antibody monotherapy. 50 If nivolumab and ipilimumab are administered in a combinatorial treatment, the incidence increases up to 44%. 51 Colitis, another common irAE, is defined as (bloody) diarrhea with either abdominal complaints or the evidence of inflammatory stigmata in radiographic imaging. 52 Depending on the severity, treatment options vary from antidiarrheal drugs to systemic glucocorticoids and discontinuation of the ICI. 53 An exemplarily endoscopic picture of an ICI colitis is displayed in Figure 2. As described previously, other causes of diarrhea and colitis, such as infectious diseases, must be excluded. Intriguingly, diarrhea and colitis typically occur 6 weeks after the first ICI administration and are more frequent with anti-CTLA-4 antibody immunotherapy. 54 A consensus about the role of the computed tomography (CT) scan in the diagnosis of colitis has not been found yet and is the matter of current debates. If the diagnosis remains unclear (after a CT scan), the performance of a colonoscopy or sigmoidoscopy must be considered with high priority. 46 Vomiting, epigastric pain and nausea are clinical symptoms that suggest upper gastrointestinal drug-mediated toxicity. They are less common than lower gastrointestinal toxicity, in which diarrhea is the most common symptom. 55

Endoscopic finding of a severe immune checkpoint inhibitor colitis with large ulcers and mucosal erythema.
Hepatitis onset is most often seen between 8 and 14 weeks and usually presents with an asymptomatic elevation of alanine aminotransferase and aspartate aminotransferase. Among patients that are treated with anti-CTLA-4, or anti-PD-L1 or anti-PD-1 antibodies, the incidence is as high as 10% and 5%, respectively. 56 It is reported that hepatitis can occur in as much as 30% of the cases if a combined ICI therapy regimen is applied. The liver synthesis function should be controlled on a regular basis by assessing coagulation factors (INR, aPTT, Factor V) and albumin. Other causes of liver function alteration including extensive metastatic liver disease, alcohol abuse, other drug toxicity or infections, among others, should be considered.
Esophagitis and pancreatitis have been reported as irAEs but are rather rare.
Dermatological toxicity
Almost 50% of patients treated with ICIs develop and suffer from pruritus and skin rashes, making these the most common irAEs. 54 These symptoms usually occur within the first 3 weeks upon treatment initiation. 47 They are less frequent in anti-PD-1 and anti-PD-L1 (40%) than in anti-CTLA-4 antibody immunotherapy (50%). In combination therapy, up to 60% of patients suffer from pruritus and skin rashes. 57 Other cutaneous manifestations such as Stevens–Johnson-syndrome, toxic epidermal necrolysis, different forms of dermatitis and pemphigoid are less common.
Endocrinological toxicity
Although not that often, primary adrenal insufficiency after ICI treatment, which often presents as adrenal crisis, is a life-threatening and serious endocrine irAE and requires in-patient management. 58 Hypothyroidism (occurring in 6% of the cases) is often asymptomatic or only mildly symptomatic, appears a few weeks after treatment start and is highly associated with the development of antithyroid antibodies.47,59 A lifelong hormone substitution is often necessary. 57 Among some others, type 1 diabetes mellitus, hypophysitis and hyperthyroidism have also been reported as endocrine irAEs.
Cardial, pneumological, hematological, nephrological, rheumatological, ocular and neurological toxicity
Although the incidence of heart failure, myocarditis, pericarditis and arrhythmias is less than 1%, those irAEs are potentially fatal. Whenever these irAEs are suspected due to prominent elevated troponin levels and conduction system abnormalities in the electrocardiogram, patients need to be hospitalized immediately in order to preclude other causes and to initiate high dose glucocorticoids therapy.60,61 Pneumonitis occurs in about 5% of patients that are treated with ICIs. It is usually diagnosed or becomes symptomatic beyond 12 weeks of treatment initiation. 62 Typical symptoms are wheezing, coughing, dyspnea and exercise intolerance. As pneumonitis is an exclusion diagnosis, secondary causes need to be excluded. Common hematologic irAEs include (hemolytic) anemia, neutropenia, immune thrombocytopenic purpura and disseminated intravascular coagulopathy. The former occurs in 5–10% of ICI treated patients.57,61 Due to these possible adverse effects, a complete blood count should always be performed before and within ICI treatment periods.
Inflammatory oligo- and polyarthritis of small, medium and large joints may cause rapid deterioration of the affected joints about 5 months after ICI treatment. It is mostly seen concomitantly to other irAEs and is reported with a frequency of approximately 7% of patients receiving ICIs. 61 Among others, rhabdomyolysis, myositis and giant cell arteritis are other rheumatologic irAEs that may develop upon ICI treatment.
Ocular irAEs include uveitis, conjunctivitis and episcleritis, as well as keratitis. They can occur after 8 weeks after ICI administration. 61 The incidence is comparable to cardial irAEs and concerns less than 1% of patients under ICI treatment. Symptoms that can occur are blurry vision, sicca syndrome and photophobia. Patients should always be seen by an ophthalmologist.
Nephrotoxicity includes different forms of nephritis (e.g. acute interstitial nephritis, “lupus-like” nephritis) and occurs in 2% of patients that are treated with anti-CTLA-4 antibody and in 5% of patients undergoing anti-PD-1 antibody immunotherapy. 61 Anti-PD-L1 agents have not yet been reported to cause nephrotoxicity. 47 Depending on the respective antibody, toxicity may arise after 2–10 months.
Although rare (<1%), neurological irAEs such as myasthenia gravis and Guillain–Barré syndrome demand immediate treatment to prevent persistent harm. Typical occurrence of these neurological irAEs is about 13 weeks after ICI administration. 50
Current attempts to achieve “cold-to-hot” tumor conversion
So called “cold” tumors are characterized by low immunogenicity that is defined as reduced tumor antigen burden, inadequate numbers of tumor infiltrating lymphocytes as well as an immunosuppressive TME. 63 The latter has been well discussed in numerous reviews.64,65 Moreover, “cold” tumors typically show poor response rates upon immune checkpoint blockade. On the contrary, high levels of proinflammatory cytokines and T-cell infiltration are characteristic for “hot” TMEs, which in general respond well to immunotherapy. Therefore, current efforts in the field lie on converting “cold” CRCs into “hot and inflamed” CRCs that can consecutively be tackled by immunotherapeutic drugs. To conquer a tumor conversion, various approaches in combination with immunotherapy are currently under clinical investigation.
Chemotherapy and immunotherapy
Tumor cell fragmentation is a result of chemotherapy-induced cell death. These fragments are phagocytosed by antigen presenting cells (APCs) followed by consecutive antigen presentation to T-cells, which in turn initiates and increases immune responses. 5 Many chemotherapeutic drugs target proliferating cells such as bone marrow cells. A side effect of various chemotherapeutics is their capability of suppressing predominantly immune inhibitory cells such as Tregs. This results in a relatively increased proliferation of homeostatic T-cells and an elevated immune response. 66 Intriguingly, the combination of oxaliplatin and cyclophosphamide with ICIs has been successfully tested. 67 Currently, the combination of ICIs with 5-fluorouracil, leucovorin and oxaliplatin (FOLFOX) is under investigation.
Radiotherapy and immunotherapy
The abscopal effect describes a phenomenon where radiation of solitary tumor tissue results in an immune activation also at other, non-irradiated, sites. This leads to an extended antitumoral effect of the radiotherapy. 5 In a non-randomized phase II study with a total of 22 MSS/pMMR mCRC patients that underwent radiotherapy of metastasis followed by pembrolizumab treatment, only one patient displayed a measurable regression of non-irradiated lesions. 5 Nevertheless, different studies are currently ongoing to investigate the role of the combination of radio- and immunotherapy in CRC treatment. Results from the ongoing phase II EORTC ILOC study (NCT03101475) are eagerly awaited.
Combination of immunotherapeutic agents (TLR agonists, TFG-β inhibitors, VEGF and EGFR)
Pathogen recognition receptors such as toll-like receptors (TLRs) are expressed on the surface of innate immune cells. Upon recognition of damage-associated molecular patterns or pathogen-associated molecular patterns an innate immune response is initiated. 67
In vivo studies are currently assessing the treatment efficacy of TLR agonists in combination with anti-PD-1 antibodies in colon cancers. There is increasing evidence that speaks in favor of this combination, as recent data of murine models of colon cancer revealed a prolonged survival and decreased tumor growth upon TLR agonist and anti-PD-1 antibody treatment. 68
It has been described that TGF-β exerts an essential role in promoting metastasis in CRCs by inhibiting the process of antigen presentation and by reducing the production of cytolytic factors in dendritic cells (DCs). 69 According to a recent report, TGF-β inhibition through antibodies leads to a cytotoxic T-cell response in MSS/pMMR CRCs. 69 Moreover, preclinical in vivo data of TGF-β inhibition in mice with colon cancers show a decreased tumor burden and an increased overall survival among these animals. 70 Clinical trials harnessing anti-PD-L1 and anti-TGF-βR2 immunoglobulins in the treatment of CRCs are currently ongoing.
The vascular endothelial growth factor (VEGF) expression is not only upregulated in tumor tissue (neoangiogenesis) due to increased nutrition and oxygen demand, but it also seems to play a role in T-cell inhibition and immunomodulation. A decreased number of Tregs, enhanced T-cell function, less tumor infiltration, as well as an augmented differentiation and maturation of DCs is observed upon VEGF inhibition, therefore, enhancing antitumoral immune responses. 71 Due to promising results in MSI-H/dMMR tumors, the combination of ICIs and VEGF inhibition is currently tested in unselected CRCs. 6 In 2017, the first phase Ib study with a total of 10 MSI-H/dMMR mCRC patients treated with atezolizumab and bevacizumab was published. An ongoing, randomized, open-label, two-arm, multicenter phase II study is currently investigating the effects of a bevacizumab/oxaliplatin/fluoropyrimidine combination regimen (mFOLFOX-6/mOXXEL) in addition with valproic acid on PFS in mCRC (REVOLUTION study protocol). 72 The phase Ib REGNIVO trial investigating the combination of regorafenib with nivolumab in patients with CRC or gastric cancer displayed an overall ORR of 40% in the entire population and a manageable safety profile. 73 Lenvatinib and pembrolizumab were investigated in the LEAP-005 study (NCT03797326, phase II), and showed promising antitumor activity and manageable toxicity across different pretreated tumors. According to the REGOMUNE trial (NCT03475953), a phase II study, the combination of regorafenib and avelumab in non-MSI-H mCRC displayed and increased PFS and OS compared with regorafenib as monotherapy.
Endothelial growth factor receptor (EGFR) antagonists not only improve T-cell-mediated toxicity but also decrease immunosuppressive cells and are therefore believed to augment immunity. 74 Neoadjuvant anti-EGFR antibody treatment revealed an increased PD-1 expression and CD8+ T-cell infiltration in metastases. 67 Current studies are investigating the combination of either anti-PD-1 or anti-CTLA-4 with anti-EGFR (panitumumab and cetuximab) in mCRCs.
DNA polymerase epsilon (POLE) and immunotherapy
Enzymes of the DNA polymerase family are responsible for DNA replication and proofreading. POLE mutations are found in as much as 1.5% of CRCs and are associated with an elevated tumor T-cell infiltration and hypermutations that improve their immunogenicity. 75 Promising response rates to immunotherapy were found in CRC. 76
Lymphocyte-activation gene 3 (LAG-3) and immunotherapy
The main ligand of the LAG-3 protein is MHC class II and it negatively regulates activation and homeostasis of T-cells. 77 Furthermore, it suppresses Treg function and is involved in the activation of DCs. 78 Therefore, the therapeutic effects of anti-LAG-3 monoclonal antibodies in combination with immunotherapy are currently being investigated not only in mCRCs. 79 Data of ongoing clinical trials are awaited.
Vaccine therapy
MSI-H leads to an increased antigen burden, which acts as a highly immunogenic stimulator of T-cell activation. Hence, it serves as an ideal target for therapeutic vaccines. 12 DCs ensnare antigens and activate CD4+ and CD8+ T-cells. Furthermore, DCs have an essential role in organizing naïve B cells, natural killer cells and T-cells through secreting cytokines. DC vaccination therapy comprises priming the patient’s DCs and exposing them to specific cancer antigens to activate cytotoxic T-cells. 67 Since CRCs express various tumor specific antigens such as CEA, Mucin 1 and Wilms Tumor 1, they represent an ideal option for DC therapy. 67 Despite the safety of DC-based immunotherapy, the tumor response rate upon this therapy approach is disappointing and hardly exceeds 15%. 80
Conclusions
In this review, we summarize the efficacy and possible immune related AEs of ICIs in the treatment of MSI-H/dMMR and MSS/pMMR CRCs. ICIs changed the game in the treatment of melanoma, non-small cell lung cancer, renal cell carcinoma and MSI-H/dMMR CRC completely, but clearly failed to meet expectations as monotherapy in MSS/pMMR CRC treatment. Therefore, novel and synergistic therapeutic approaches are urgently awaited, as the vast majority of CRCs have a MSS/pMMR profile. Hence, current studies focus on combing ICIs with different and (established) therapeutic approaches, in the hope of overcoming immune resistance and of making them safe and effective regardless of the MSI/MMR molecular tumor status. Due to the rapid growth of insights in CRC treatment, harnessing numerous (immuno)therapeutic modalities and their combinations, their role and importance is yet to be defined in this common neoplasia.
