Abstract
Background:
Methotrexate monotherapy is recommended as a maintenance therapy for Crohn’s disease (CD). However, long-term follow-up data are scarce.
Objectives:
We aimed to examine the effectiveness and tolerability of methotrexate monotherapy in 94 CD patients from three inflammatory bowel disease Clinics in Korea.
Design:
This was a multicenter retrospective observational study.
Methods:
Patients with active CD treated with methotrexate monotherapy were included. Clinical characteristics, laboratory indicators, endoscopy indices were evaluated at baseline, 6, 12, and 24 months. Independent factors associated with long-term clinical and endoscopic outcomes were determined.
Results:
Methotrexate was administered orally (70.2%) or parenterally (29.8%). The mean methotrexate induction dose was 15.3 ± 0.4 mg/week, and the mean duration of therapy was 26.2 months. Of 76 patients who were treated for >6 months, the clinical remission rates were 76.3%, 74.6%, and 80.0% at 6, 12, and 24 months, respectively, by per-protocol analysis. The mean CRP levels were 7.5 ± 1.3, 5.3 ± 1.2, 3.8 ± 0.7, and 2.6 ± 0.5 mg/L at 0, 6, 12, and 24 months, respectively. Of 31 patients who underwent follow-up endoscopy after 27.5 months, the endoscopic remission rate was 38.7%. Baseline hemoglobin level <10 g/dL was a significant independent factor negatively associated with clinical remission at 6 [odds ratio (OR): 0.023, 95% confidence interval (CI): 0.003–0.206, p = 0.001] and 12 (OR: 0.079, 95% CI: 0.009–0.699, p = 0.023) months. Parenteral administration was a significant independent factor positively associated with clinical remission (OR: 11.231, 95% CI: 1.027–122.811, p = 0.047) and endoscopic remission (hazard ratio: 4.711, 95% CI: 1.398–15.874, p = 0.012) at 12 months.
Conclusions:
Methotrexate monotherapy was effective and tolerable as a maintenance therapy in CD patients.
Keywords
Introduction
Conventional immunomodulators, including thiopurines and methotrexate (MTX), are usually recommended for corticosteroid-refractory or corticosteroid-dependent inflammatory bowel disease (IBD) patients. 1 Immunomodulators act by decreasing the inflammatory response, maintaining clinical remission, and reducing the need for corticosteroids by preventing recurrence in IBD patients. 2 In the recent era of biologics, immunomodulators prevent antidrug antibody development when used in combination with biologic agents. 3 The recent Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE)-II statement emphasized the treat-to-target approach in IBD treatment with tight control of inflammation. 4 However, achieving this target remains challenging owing to limitations in the number and effectiveness of IBD drugs. In an era where various biologic agents and small molecules are being developed, it is necessary to reconsider the position of immunomodulators to maximize their therapeutic effects. 5
MTX, a folate antagonist, has been used as a first-line immunomodulator or an alternative agent to thiopurine therapy failure. However, in a recent randomized controlled trial (RCT), parenteral MTX monotherapy failed to prove superiority over placebo in achieving clinical remission at week 48 in ulcerative colitis (UC) patients.5,6 MTX monotherapy has been included in recent clinical guidelines for the treatment and remission maintenance of active Crohn’s disease (CD).7,8 Feagan et al. first demonstrated the effectiveness of intramuscular MTX at a dose of 25 mg/week for 12 months in an RCT including thiopurine-naïve patients with CD. 9 Although some studies have used MTX monotherapy in CD patients, its effectiveness, optimal administration route, optimal dose, tolerability, and mucosal healing rate in a real-world clinical setting remain uncertain. 10
Herein, we analyzed the long-term clinical and endoscopic outcomes and the occurrence of adverse events after MTX monotherapy in CD patients, including those with a history of thiopurine use. In addition, we identified independent factors associated with long-term clinical and endoscopic outcomes.
Materials and methods
Patient population and MTX therapy
From June 2007 to December 2022, all consecutive CD patients who received MTX monotherapy at three tertiary referral hospitals, including the IBD Clinic of Severance Hospital, Yonsei University College of Medicine (Seoul, Korea), Gangnam Severance Hospital (Seoul, Korea), and Seoul National University Bundang Hospital (Seongnam, Korea), were enrolled in this multicenter retrospective cohort study. A total of 94 patients who were treated with either oral or parenteral MTX were included. The inclusion criteria were as follows: (i) age ⩾18 years; (ii) definite diagnosis of CD according to the endoscopic, histological, or radiological criteria; and (iii) MTX monotherapy administration (concomitant use of 5-aminosalicylates or corticosteroids was allowed). The exclusion criteria were as follows: (i) patients treated with a combination of MTX and a biologic agent and (ii) those who were treated with MTX monotherapy for <4 months. Patients were identified using electronic medical records of all patients diagnosed with IBD at each institution. The reporting of this study conformed to the Strengthening the reporting of observational studies in epidemiology (STROBE) statement. 11
Data collection and outcome definitions
The following data were retrospectively collected from electronic medical records: age; sex; disease duration; smoking history; body mass index; Montreal location and behavior of CD; bowel surgery history; medical history; levels of C-reactive protein (CRP); hemoglobin; albumin; and the administration route, dose, indication, and reason for discontinuation of MTX therapy.
The primary endpoint was clinical remission at 6 months, and secondary endpoints were clinical remission at 12 and 24 months, CRP levels at 6, 12, and 24 months, endoscopic remission, and adverse events of MTX monotherapy. We used the Crohn’s Disease Activity Index (CDAI) score system for assessment of clinical disease activity. Clinical remission was defined as a CDAI score ⩽150, while endoscopic remission was defined as a Simple Endoscopic Score for Crohn’s Disease (SES-CD) of ⩽ 2.6,12 We defined transmural healing as a normal bowel wall thickness with the normalization of stratification and no hypervascularization using cross-sectional imaging studies. 13
Statistical analysis
The mean and standard deviation or median and range were calculated for all continuous variables, as appropriate. Multivariate logistic regression analyses were performed to identify independent factors affecting clinical remission with MTX monotherapy at 6 and 12 months, with adjustment for age, sex, disease duration, smoking history, body mass index, Montreal location and behavior of CD, bowel surgery history, medical history, levels of CRP, hemoglobin, and albumin, and administration route and dose of MTX. Cox proportional hazards analyses were performed to identify the independent factors affecting endoscopic remission with MTX monotherapy. Variables in univariate analysis with a p-value of <0.05 were included in the multivariate analysis. A p-value <0.05 was considered statistically significant. All statistical analyses were performed using Statistical Package for Social Sciences (SPSS version 23.0; SPSS Inc., Armonk, NY, USA).
Results
Baseline patient characteristics
Mean age at initiation of MTX was 31.3 ± 13.7 years, 62 patients (66.0%) were male, and the mean disease duration was 5.1 ± 6.8 years (Table 1). CD was predominantly located in the ileocolon (59 patients, 62.8%). The CD behavior was inflammatory in 55 patients (58.5%), stricturing in 23 (25.5%), and penetrating in 16 (17.0%). Perianal lesions were observed in 51 patients (54.3%), and 23 (24.5%) patients underwent bowel surgeries. Sixty-one (64.9%) patients had been treated with thiopurines and seven (7.4%) had been treated with biologic agents before enrollment.
Baseline characteristics (N = 94).

Variables are expressed as mean or n (%).
CDAI, Crohn’s Disease Activity Index; CRP, C-reactive protein; MTX, methotrexate; SES-CD, Simple Endoscopic Score for Crohn’s Disease.
MTX monotherapy characteristics
MTX was administered orally (70.2%) or parenterally (29.8%) (Table 1). The mean MTX dose was 15.3 ± 0.4 mg/week (median, 15.0 mg/week; range, 7.5–25.0 mg/week) during the induction period and 14.1 ± 3.6 mg/week (median, 15.0 mg/week; range, 7.5–25.0 mg/week) during the maintenance period (Table 1). The mean duration of MTX monotherapy was 26.2 ± 23.6 months (median, 17.1 months; range, 4.0–85.0 months) (Table 1). The main indication for MTX monotherapy was thiopurine intolerance (n = 48, 51.5%) (Table 2). Other indications for MTX monotherapy included patients who were not previously treated with immunomodulators and those who were steroid-refractory or dependent (34.0%), failure of thiopurines (7.4%), failure of a biologic agent and thiopurine combination therapy (6.4%), and failure of a biologic agent monotherapy (1.1%) (Table 2). Corticosteroids were continued in 15 patients (16.0%) during MTX monotherapy induction and was discontinued in 13 patients (13.8%).
Details of methotrexate drug use.
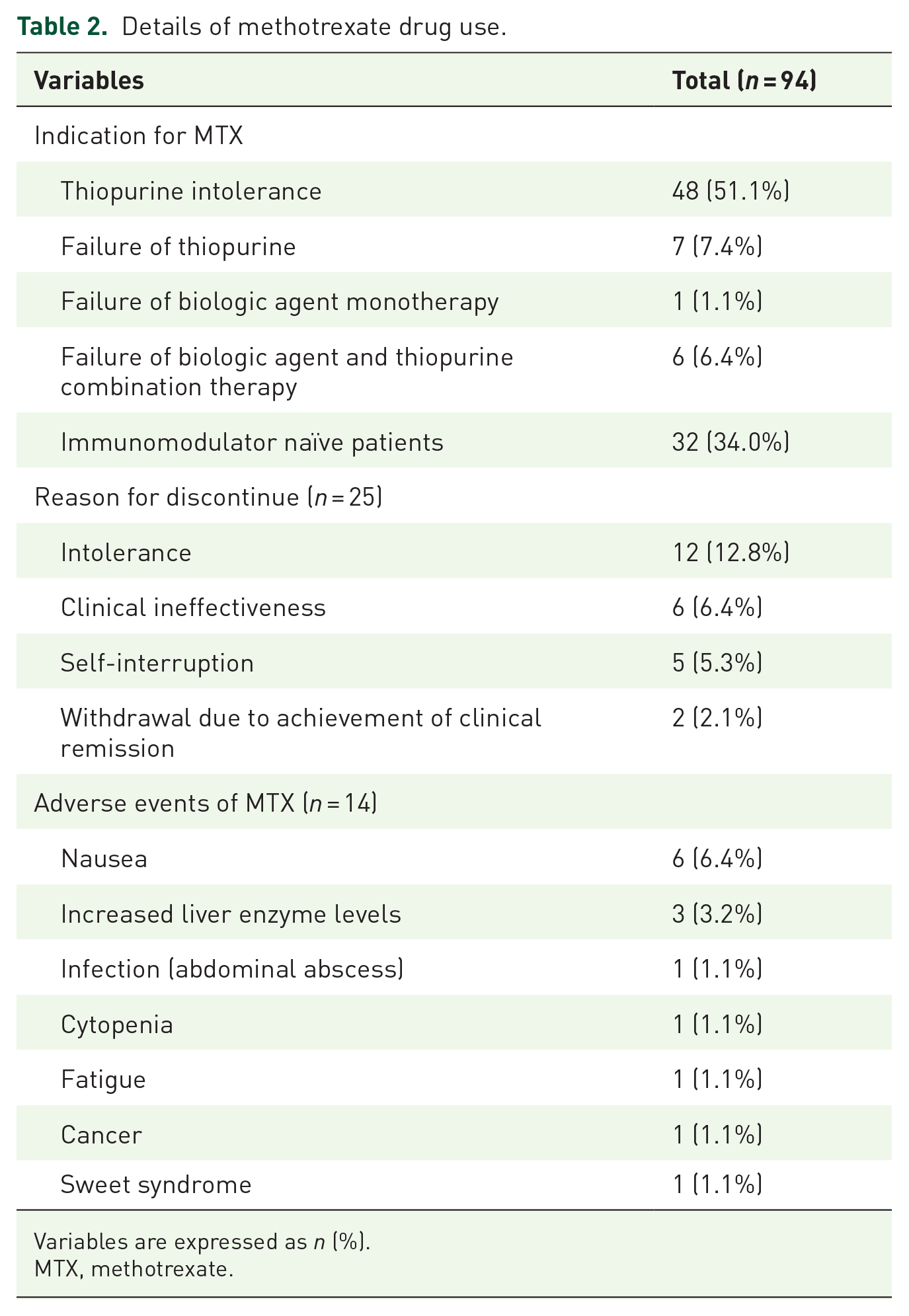
Variables are expressed as n (%).
MTX, methotrexate.
Adverse events
At least one adverse effect was reported in 14 (14.9%) patients. Nausea was reported in six patients (6.4%); of these, five required MTX discontinuation. Liver enzyme abnormalities were reported in three patients (3.2%); of these, two required MTX discontinuation. Other adverse events included intra-abdominal abscess (n = 1, 1.1%), cytopenia (n = 1, 1.1%), fatigue (n = 1, 1.1%), rectal cancer (n = 1, 1.1%), and Sweet syndrome (n = 1, 1.1%) (Table 2).
Outcomes
First, we assessed the clinical remission rates according to the intention-to-treat analysis. The clinical remission was achieved in 58 of the 94 patients at 6 months (61.7%), 44 of the 94 patients at 12 months (46.8%), and 28 of the 94 patients (29.8%) at 24 months. Next, per-protocol analysis was performed. A total of 76 patients who were treated with MTX monotherapy and followed up for >6 months were statistically analyzed. The mean CDAI scores were 117.4 ± 80.3, 97.8 ± 97.8, 96.6 ± 80.3, and 84.7 ± 73.4 at 0, 6, 12, and 24 months, respectively. Clinical remission was achieved in 58 of the 76 patients at 6 months (76.3%), 44 of the 59 patients at 12 months (74.6%), and 28 of the 35 patients (80.0%) at 24 months (Figure 1(a)). The mean CRP levels were 7.5 ± 1.3, 5.3 ± 1.2, 3.8 ± 0.7, and 2.6 ± 0.5 mg/L at 0, 6, 12, and 24 months, respectively (Figure 1(b)).

(a) Per-protocol analysis of clinical remission rates at 6 months (76 patients), 12 months (59 patients), and 24 months (35 patients), and (b) C-reactive protein levels at 0 months (76 patients), 6 months (76 patients), 12 months (59 patients), and 24 months (35 patients) after methotrexate (MTX) combination therapy in patients with Crohn’s disease.
Logistic regression analysis was used to determine the effect of different variables on the maintenance of clinical remission with MTX monotherapy. Baseline hemoglobin level <10 g/dL was a significant independent factor negatively associated with clinical remission at 6 [odds ratio (OR): 0.023, 95% confidence interval (CI): 0.003–0.206; p = 0.001) (Table 3) and 12 (OR: 0.079, 95% CI: 0.009–0.699, p = 0.023) (Table 4) months. Parenteral administration was a significant independent factor positively associated with clinical remission at 12 months (OR: 11.231, 95% CI: 1.027–122.811, p = 0.047) (Table 4).
Logistic regression analysis for clinical remission at 6 months.

p Value for comparing clinical remission and non-remission in Crohn’s disease patients treated with MTX monotherapy.
BMI, body mass index; CRP, C-reactive protein; MTX, methotrexate; OR, odds ratio.
Logistic regression analysis for clinical remission at 12 months.

p Value for comparing clinical remission and non-remission in Crohn’s disease patients treated with MTX monotherapy.
BMI, body mass index; CRP, C-reactive protein; MTX, methotrexate; OR, odds ratio.
A total of 31 patients underwent a follow-up endoscopy after 27.5 months (median, 16.2 months; range, 6.0–85.3 months). Of these, 12 patients (38.7%) had achieved endoscopic remission (SES-CD ⩽2). The mean SES-CD scores were and 6.3 ± 7.9 and 6.8 ± 6.7 at baseline and follow-up, respectively. Cox proportional hazard analysis determined that parenteral administration was the only significant independent factor positively associated with endoscopic remission [hazard ratio (HR): 4.711, 95% CI: 1.398–15.874, p = 0.012] (Table 5, Figure 2). A total of 37 patients underwent a follow-up cross-sectional imaging study after 25.3 months (median, 20.1 months; range, 3.4–85.4 months). Of these, 12 patients (32.4%) achieved transmural healing.
Cox proportional hazards analysis for endoscopic remission.

p Value for comparing endoscopic remission and non-remission in Crohn’s disease patients treated with MTX monotherapy.
BMI, body mass index; CRP, C-reactive protein; HR, hazards ratio; MTX, methotrexate.

Cumulative endoscopic remission rates stratified according to the administration route of methotrexate in patients with Crohn’s disease (31 patients).
MTX monotherapy was discontinued in 25 patients primarily owing to intolerance (12.8%). Other reasons included clinical ineffectiveness (6.4%), self-interruption (5.3%), and withdrawal after achievement of clinical remission (2.1%) (Table 2).
Discussion
IBD is characterized by chronic relapsing inflammation of the gastrointestinal tract with an often unpredictable course, even while on the best currently available therapeutic options.14–16 Despite availability of several biologic agents and small molecules, many patients require a more effective therapeutic option for CD.17,18 Therefore, we sought to demonstrate the effectiveness of MTX monotherapy, which may be an optimal treatment strategy in CD patients.
Using the large-scale data from the IBD BioResource (United Kingdom), Wang et al. 19 reported that 1042 biologic-naïve IBD patients were treated with MTX monotherapy (791 CD, 251 UC, and 15 unclassified IBD); MTX monotherapy was discontinued in 17.4% owing to intolerance. 19 After a median of 2 years of MTX monotherapy, the effectiveness, which was defined as no escalation of medication or surgery, is reportedly 35.5% in UC and 26.7% in CD. 19 Older age, shorter disease duration, and ileocolonic type of CD are positively associated with the effectiveness of MTX monotherapy. 19 Although the study had the strength of a large sample size, the optimal route and dose of MTX or past thiopurine medication history were not analyzed. In addition, it was limited by the fact that the clinical outcome evaluations relied on patients’ subjective perceptions rather than objective indicators. Hong et al. 20 reported that the clinical response to MTX monotherapy was 65.9% in 85 Korean CD patients. Furthermore, they demonstrated that parenteral administration was the only factor associated with response to MTX monotherapy. 20 Although clinical outcomes at 6 months after MTX monotherapy in CD patients were analyzed in the study by Hong et al., the long-term clinical outcomes or endoscopic remission were not evaluated. Mosonero et al. reported the short-term clinical response and remission rate after MTX monotherapy in CD patients after failure of treatment with anti-tumor necrosis factor alpha to be 60% and 30%, respectively. Additionally, they reported a long-term clinical effectiveness of 82% and 74% at 12 and 24 months, respectively. 21 Furthermore, they determined that short-term clinical remission at 12–16 weeks is significantly associated with long-term MTX maintenance. 21 Reports on the mucosal healing rate of MTX monotherapy in CD patients are scarce. Some small sample-sized studies have reported mucosal healing rates of 39.0–47.4% with MTX monotherapy22–24; others have reported mucosal healing rated of only 9.5–11.0%.19,25
We analyzed the effectiveness of MTX monotherapy during the mean 26.2 months in CD patients; the clinical remission rates were 76.3%, 74.6%, and 80.0% at 6, 12, and 24 months, respectively. Additionally, we demonstrated an endoscopic remission rate of 38.7%. Adverse events were reported in 14 patients (14.9%), and MTX monotherapy was discontinued in 12 patients (12.8%).
To our knowledge, this is the first study to evaluate the independent factors associated with endoscopic remission after MTX monotherapy in CD patients. Parenteral administration of MTX was positively associated with clinical and endoscopic remission. The relative bioavailability of oral MTX to subcutaneous MTX is 0.73–0.86; furthermore, the pharmacokinetic profiles are significantly varied.26,27 In particular, since MTX is absorbed in the small bowel, the bioavailability of oral drugs in CD patients with severe small bowel inflammation varies greatly and is difficult to predict. Our results suggest that the parenteral route could contribute to achieving deep mucosal healing, which is the recommended target in the recent STRIDE-II strategy. 4 There was no significant difference in the clinical and endoscopic outcomes of MTX monotherapy between thiopurine-naïve and thiopurine-treated patients in our study. Therefore, MTX monotherapy could be considered as a first-line immunomodulator or an alternative for patients who responded poorly to thiopurine therapy. The reasons for this are that the mechanism of thiopurine resistance is different from that of MTX. Also, thiopurines are not immunogenic. Moreover, a considerable number of patients who were unable to tolerate thiopurine-associated side effects were included in the study.
Gastrointestinal complications such as nausea, vomiting, and diarrhea are the most common and well-known early side effects of MTX. Although nausea is common, it can be prevented by the concomitant use of folic acid or antiemetics. Hepatotoxicity due to the accumulation of metabolites, cytopenia, and teratogenicity are late side effects of MTX. Our study demonstrated that the incidence of the side effects of MTX was lower than preconceived. In patients of East Asian origin, the frequency of thiopurine-induced bone marrow suppression is high; however, that of MTX-induced cytopenia is low. Therefore, MTX treatment is beneficial in patients at a higher risk of cytopenia, such as those with NUDT15 mutations.
Our study had several limitations. First, although the sample size was larger than that in other studies, it was still small, and the study population was heterogeneous because of its retrospective design. In particular, the number of patients who underwent follow-up endoscopy was small. Nevertheless, the details of MTX treatment, laboratory findings, and endoscopic data were analyzed. Additionally, the effect of MTX administration route, MTX dose, and previous immunomodulator use on the clinical and endoscopic outcomes following MTX monotherapy in CD patients were evaluated. Second, endoscopic response was evaluated only in some patients at inconsistent time points.
Conclusion
In conclusion, our study showed that MTX monotherapy can be used in adult IBD patients, with an acceptable effectiveness and tolerability profile. Additionally, we found that the parenteral administration of MTX was superior to oral administration in terms of clinical and endoscopic remission.
