Abstract
Background:
Little is known about the efficacy of proton-pump inhibitor (PPI) therapy in the management of esophageal ulcers after endoscopic submucosal dissection (ESD). Therefore, the objective of this study was to investigate the efficacy of PPI in ulcer healing following ESD for superficial esophageal neoplasms, using a propensity score analytic approach.
Methods:
This retrospective cohort study was conducted at a single referral center. Between April 2005 and August 2015, 199 consecutive patients with superficial esophageal cancer and esophageal dysplasia underwent ESD. For patients with PPI administration, intravenous PPI therapy was commenced immediately after ESD, and oral PPI was administered daily from post-operative day 3, until ulcer healing was identified. We compared the remnant-ulcer rate at 4 weeks after esophageal ESD between the PPI administration and non-PPI groups, using propensity scores and the inverse probability of treatment weighting (IPTW) method.
Results:
After exclusions, a total of 88 patients were analyzed. The remnant-ulcer rate at 4 weeks after ESD was 25.5% (12/47) and 14.6% (6/41) in the PPI administration and non-PPI groups (p = 0.21). After adjusting for background factors using IPTW, the risk of a remnant ulcer in the PPI administration group was not decreased significantly compared with that in the non-PPI group [odds ratio (OR) = 2.42, 95% confidence interval (CI): 0.73–7.97, p = 0.15]. Furthermore, PPI therapy did not decrease significantly the remnant-ulcer rate on logistic regression analysis after adjusting for the propensity score (OR = 2.40, 95% CI: 0.69–8.32, p = 0.15).
Conclusion:
PPI administration does not promote ulcer healing after ESD for superficial esophageal squamous cell carcinoma.
Keywords
Introduction
Esophageal cancer is the sixth most common cause of cancer-related mortality worldwide. 1 Patients with early-stage cancers have a good prognosis; however, advanced-stage esophageal cancers are rarely curable. In cases of superficial esophageal cancer, lymphatic metastasis is extremely rare for intramucosal lesions with an invasion depth reaching the epithelium and lamina propria mucosa. Endoscopic submucosal dissection (ESD) has developed widespread popularity as a minimally invasive treatment for superficial esophageal neoplasms. However, artificial ulcers are created after esophageal ESD, which can (infrequently) cause several complications, including postoperative bleeding, perforation, and symptoms of heartburn or precordial pain. 2
Numerous studies have reported that a proton-pump inhibitor (PPI) is useful in the prevention of hemorrhage from post-ESD ulcers in cases of early gastric cancer, as well as in the radical cure of ulcers.3,4 PPI administration is a standard treatment for gastroesophageal reflux disease (GERD) and endoscopy-negative reflux disease,5,6 and is, thus, frequently used in the postoperative management of superficial esophageal neoplasms. The aim of PPI administration after ESD is to promote healing of post-ESD ulcers and prevent delayed bleeding or perforation. To date, one randomized controlled trial indicated that PPI therapy has no apparent effects on ulcer healing. 7 Consequently, a guideline for endoscopic resection in esophageal cancer (which systematically determined the effectiveness of PPI following esophageal ESD) weakly recommend that PPI should not be administered for esophageal ESD, except in patients with GERD symptoms or reflux esophagitis. 8 However, further research is required because this recommendation is based on only one study. Therefore, the objective of this study was to evaluate the efficacy of PPI in terms of ulcer healing following ESD for superficial esophageal squamous cell carcinoma, using a propensity score analytic approach.
Methods
Patients
A retrospective cohort study was conducted at a single referral hospital in Japan. Between April 2005 and August 2015, 199 consecutive patients with superficial esophageal cancer and esophageal dysplasia underwent ESD in our department. The exclusion criteria were as follows: ESD for Barrett’s adenocarcinoma or multiple lesions; intraprocedural adverse events, such as perforation; lost to follow-up endoscopy; and PPI or H2-receptor antagonist administration prior to the procedure. Informed consent was obtained in the form of an opt-out system on the website to guarantee the opportunity for refusal for the provision of medical information to the study. The study protocol was approved by the institutional review board of Osaka Medical College (No. 2020-026), and the study was performed in accordance with the Declaration of Helsinki.
ESD procedure
ESD was performed with the patient under intravenous sedation using midazolam (Dormicum; Astellas Pharma, Tokyo, Japan) and pentazocine hydrochloride (Pentazin; Sankyo Pharmaceuticals, Tokyo, Japan). The following equipment were used: a conventional single-channel endoscope (GIF-Q260J; Olympus Medical Systems Corporation, Tokyo, Japan) with a transparent hood [Elastic Touch, slit and hole type, M (long); Top Company, Tokyo Japan]; an electrosurgical generator (ICC200 or VIO300D; ERBE Elektromedizin, Tübingen, Germany); a short needle-type endoknife, equipped with a water-jet function (Flushknife 1.5 mm, DK2618JB-15 or DK2618JN-15, Fujifilm Medical, Tokyo, Japan), and carbon dioxide for the inflation system. Firstly, circumferential marking dots were made 2–3 mm outside the margins of the lesion with an endoknife, after the lesions were identified by iodine staining. Secondly, hyaluronic acid solution (MucoUp; Boston Scientific, Tokyo, Japan) or physiological saline was injected into the submucosa to elevate the lesion. Third, the mucosa was incised outside the marking dots with a cutting current. Finally, submucosal dissection was followed by a cutting or coagulation current, based on the operator’s judgment. After removal of the lesion, the mucosal defect was visualized carefully for the presence of pulsating vessels, and coagulation was performed, as needed, using hemostatic forceps to prevent delayed bleeding.
Management of post-ESD ulcers
Patient management after ESD principally followed our hospital’s clinical pathway. All patients underwent endoscopy on post-operative day 2 to confirm hemostasis, and, if necessary, any blood vessels visible in the mucosal defect were cauterized using a soft coagulation current. Patients fasted until 2 days after ESD, and were allowed to eat from post-operative day 3, after the confirmation of no complications. From April 2005 to the end of 2012, all patients received PPI therapy; however, after 2012, PPI therapy was administered by the attending physician only as needed. For patients who received PPI administration, PPI therapy (omeprazole 40 mg/day, intravenously) commenced immediately after ESD, and oral rabeprazole (10 mg) was administered daily from post-operative day 3 until healing of the ESD ulcer was identified by endoscopy. Ulcer healing or remnant was evaluated by endoscopy at 4 weeks after ESD.
Primary endpoint
The primary endpoint was the remnant ulcer (not ulcer healing) rate after esophageal ESD. A remnant ulcer was defined as a persisting ulcer with a mucosal break, and ulcer healing was defined as epithelialization without a mucosal break.
Definition of clinical features
Atrophic gastritis was assessed endoscopically according to the Kimura-Takemoto classification system. 9 The degree of gastritis was classified into three groups as follows: none (no atrophy), mild (C1, C2, C3), and severe (O1, O2, O3).
ESD specimen size was calculated based on pathological measurement using the following formula; π × length × width/4 = area of resected specimen (cm2).
Mucoprotective agent use was defined as administration of the following drugs: rebamipide, sucralfate, ecabet sodium, polaprezinc, alginate, plaunotol, sofalcone, teprenone, irsogladine, misoprostol, or aluminium-magnesium. 10
Statistical analysis
Continuous variables are presented as the mean and standard deviation (SD) or median and range. A chi-squared or Fisher’s exact test was used for analyzing categorical variables. Group differences (PPI administration versus non-PPI) were evaluated using the unpaired t-test for continuous variables and a chi-squared or the Fisher’s exact test for categorical values.
The propensity score model for PPI administration was estimated using a logistic regression model that was adjusted for the patient characteristics listed in Table 1, based on our clinical experience and knowledge. Univariate analysis of each variable was performed to identify the risk of a remnant ulcer at 1 month after ESD with the odds ratio (OR) and 95% confidence interval (CI). PPI administration was analyzed additionally using the inverse probability of treatment weighting (IPTW) method, which was used to adjust for confounding factors. The covariate balance regarding each confounder before and after IPTW was assessed by the standardized difference: an absolute standardized difference (ASD) above 10% was considered to represent a meaningful imbalance. Moreover, the propensity score was included as a covariant in a multivariate logistic regression model to estimate the relationship between PPI administration and the risk of a remnant ulcer, preserving statistical power by reducing covariates into a single variable. Statistical analyses were performed using SPSS software version 22.0 for Windows (SPSS Inc., Tokyo, Japan) and the R statistical package (V.2.13.0; http://www.r-project.org). All statistical tests were two-sided, and p < 0.05 was considered significant.
Clinical features of the study population compared between PPI administration and non-PPI groups.

ASD, absolute standardized difference; BMI, body mass index; FIT, from the incisor teeth; PPI, proton-pump inhibitor; SD, standard deviation.
Results
A total of 88 patients treated by esophageal ESD from April 2005 to August 2015 were included in this study (Figure 1). Patients were excluded from analysis for the following reasons: 69 patients were lost to follow-up endoscopy; 27 patients had missing data; 13 patients had multiple lesions; 3 patients had Barrett’s adenocarcinoma; and 3 patients were under PPI or H2-receptor antagonist administration prior to the procedure (including overlapping patients). Results of the adjustment for background factors are shown for the PPI administration (n = 47) and non-PPI groups (n = 41) in Table 1. The covariate balance in the two groups after adjustment with IPTW was improved; the number of background factors with ASD above 10% was reduced from 17 to 10.
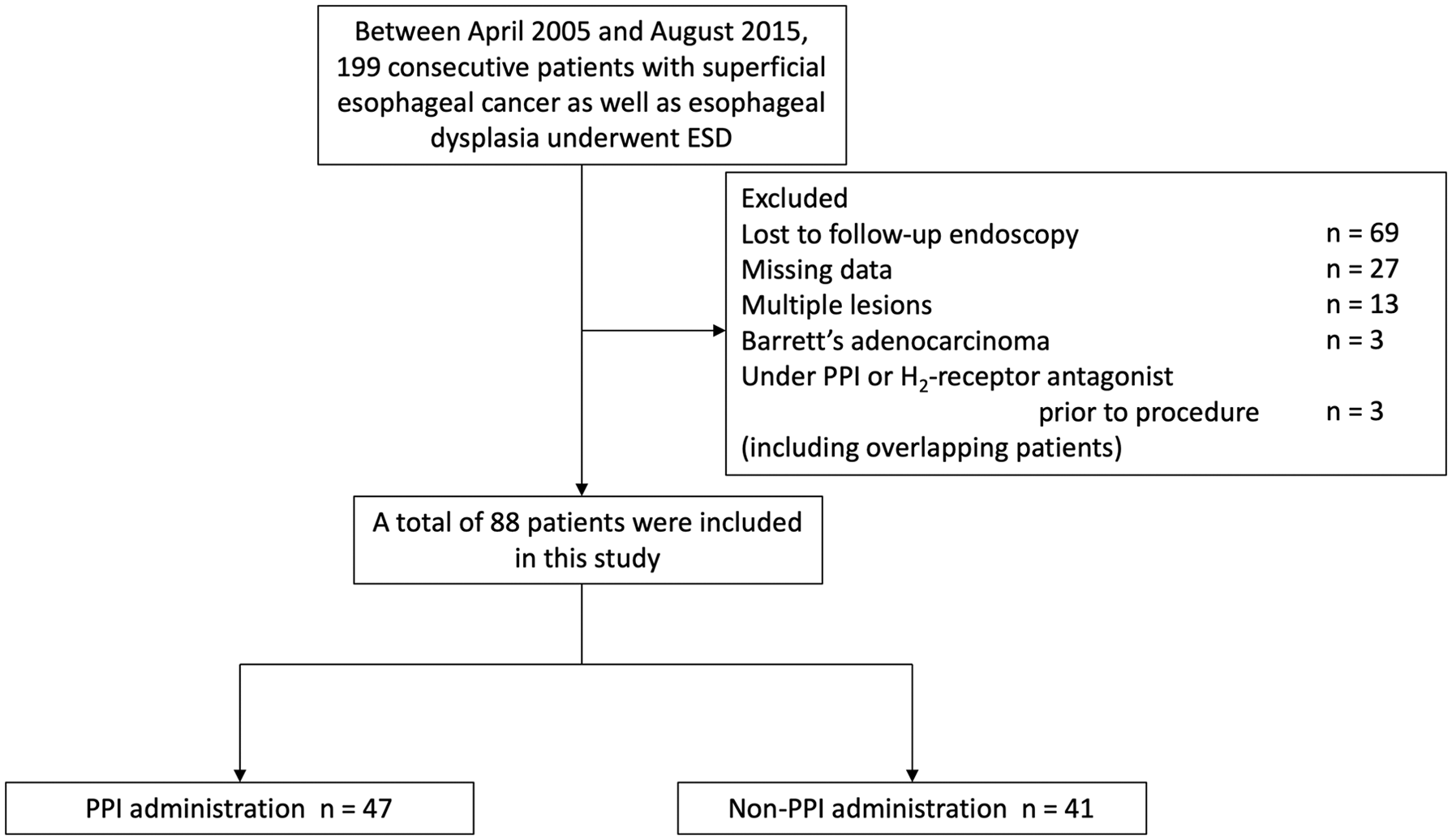
Patient selection flow chart.
No postoperative adverse events (including delayed perforation and delayed bleeding) were identified. The remnant-ulcer rate at 4 weeks after ESD was 25.5% (12/47) and 14.6% (6/41) in the PPI administration and non-PPI groups, respectively. The univariate analyses for predictive variables of a remnant ulcer are shown in Table 2. No predictive variables were associated with a remnant ulcer. The univariate and multivariate analyses for the risk of a remnant ulcer in the PPI administration group are shown in Table 3. Consequently, PPI administration was not a significant predictive factor for a remnant ulcer, with an OR of 1.92 (95% CI: 0.64–5.80, p = 0.21) before IPTW and an OR of 2.42 (95% CI: 0.73–7.97, p = 0.15) after adjusting for the background factors using IPTW. After adjustments for the propensity score using logistic regression analysis, the risk of a remnant ulcer in the PPI administration group was not decreased compared with that in the non-PPI group (OR = 2.40, 95% CI: 0.69–8.32, p = 0.15).
The risk of a remnant ulcer in the univariate analysis.

BMI, body mass index; CI, confidence interval; FIT, from the incisor teeth; OR, odds ratio; PPI, proton-pump inhibitor; SD, standard deviation.
The risk of remnant ulcer in the proton-pump inhibitor administration group.
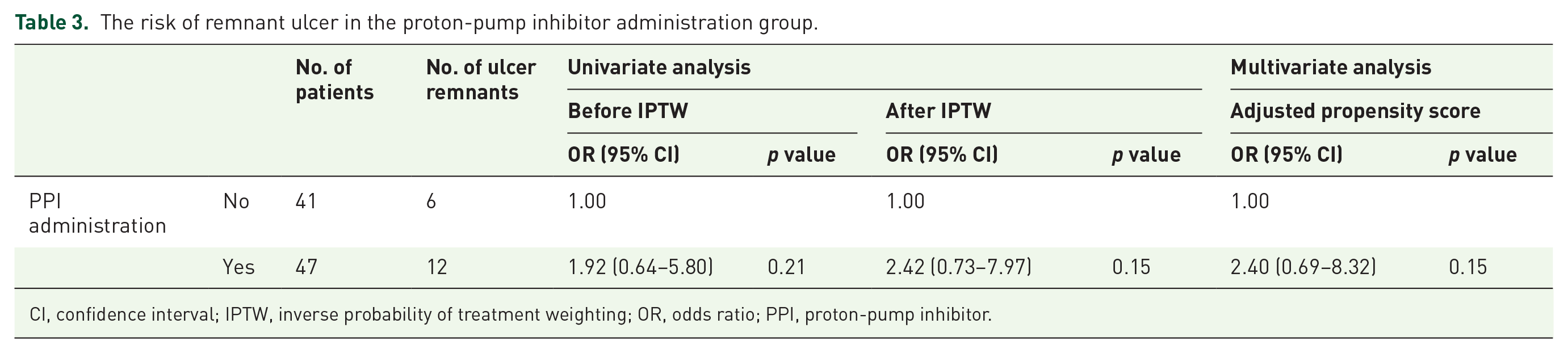
CI, confidence interval; IPTW, inverse probability of treatment weighting; OR, odds ratio; PPI, proton-pump inhibitor.
Discussion
In the present study, PPI administration was not associated with accelerated ulcer healing after esophageal ESD, even when using the IPTW method and propensity scores in the multivariate analysis. The ulcer healing rate at 4 weeks after esophageal ESD in our cohort was 79.5%, which corresponds approximately with that in previous studies.11,12
A number of studies have reported that PPI therapy is effective in curing ulcers in cases of post-ESD ulcers in the stomach.4,13–16 However, PPI therapy did not promote ulcer healing after esophageal ESD in the present study. Kakushima et al. conducted a randomized controlled trial of 229 patients with superficial esophageal squamous cell carcinoma, and reported that PPI treatment had no apparent effects on the ulcer healing rate (84% without PPI versus 85% with PPI) or GERD symptom-appearance rate (25% without PPI versus 30% with PPI). 7 The ulcer healing rate was slightly smaller in the current study than in this previous study, which might be explained by differences in timing of ulcer healing assessment (4 weeks after ESD in the current study versus 5 weeks in the previous study).
Furthermore, the present study did not find significant differences in the ulcer-healing rate based on the presence of esophageal hiatal hernia or reflux esophagitis, which contribute to gastric acid reflux. These findings are novel, as such cases were excluded in the study by Kakushima et al. 7 The lack of a significant difference may be due to the fact that, in the present study, the mean distance from the incisor teeth (FIT) was approximately 30 cm. Acid reflux into the esophagus can occur in healthy individuals, as well as in those with GERD, but is limited to just above the lower esophageal sphincter.17,18 On the assumption that PPI therapy could promote healing, PPI generally tends to be used for post-ESD ulcers in the esophagus. However, our results suggest the possibility that, in Japan, where cancer in the thoracic esophagus is common, PPI administration for curing post-ESD ulcers may be unnecessary.
In the present study, we could not elucidate an effect of PPI administration on the prevention of perforation and delayed bleeding. The incidence of bleeding after esophageal endoscopic mucosal resection or ESD is generally low.2,19,20 Consistent with this, perforation and delayed bleeding did not occur in the present study. Thus, further larger-scale studies are required to investigate the association between PPI administration and severe adverse events.
The present study has some limitations. First, a selection bias should be considered because of the retrospective nature of the study. However, the use of propensity score matching may have reduced the selection bias, unless the true confounders related to the outcome were not included in the propensity model. Second, the statistical power of the present study might be low, as this was a single tertiary-center study with a small sample size. However, we used propensity scores and the IPTW method to avoid reducing the sample size. Third, patients with Barrett’s adenocarcinoma were excluded. Esophageal adenocarcinoma mainly develops around the esophagogastric junction, which is likely to be exposed to gastric reflux. Thus, in Barrett’s adenocarcinoma cases, PPI therapy might promote the ulcer-healing rate after esophageal ESD. Fourth, in the present study, rabeprazole 10 mg (standard dose for GERD) was administered orally until healing of the ESD ulcer. Therefore, the effect of the higher than standard dose on the ulcer healing rate after esophageal ESD remains unclarified. Besides, potassium-competitive acid blockers with the advantages of higher acid suppression over conventional PPIs might promote ulcer healing21,22 Fifth, the cutting current, especially ENDO CUT mode, was reported to attenuate the inflammation and fibrosis of ulcer after esophageal ESD compared with the coagulation current.23–25 Thus, the difference among electrosurgical unit modes might be a potential confounding factor. However, this factor could not be quantified, because different electrosurgical unit modes were used during the procedure, depending on the submucosal condition (e.g. likely to use coagulation current for submucosal layer rich in blood vessels).
In conclusion, PPI administration does not promote ulcer healing after ESD for superficial esophageal neoplasms. Unlike that for the stomach, PPI administration should not be considered for the prevention of adverse events and improvement in ulcer healing after esophageal ESD, in terms of its advantages and cost effectiveness.
Footnotes
Authors’ contributions
HK, TI, and TT: conception and design. HK, TT, YT, SN, SK, KO, and YK: data acquisition. TI, TT, AH, YK, and MS: analysis and interpretation of the data. HK and TI: drafting of the article. TT: critical revision of the article for important intellectual content. All authors: final approval of the article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest
The authors declare that there is no conflict of interest.
