Abstract
Background:
As Crohn’s disease (CD) is associated with a high risk of thromboembolic events (TE), including patients with subclinical inflammation, we aim to evaluate the correlation between the impact of endoscopic activity (EA) in the coagulation profiling of CD patients while in clinical remission.
Methods:
From 164 consecutive CD patients included in clinical remission [Crohn’s disease activity index (CDAI) < 150], 75 were in the EA group [Simplified Endoscopic Score for CD (SES-CD) ⩾ 7], 89 were in the endoscopic remission (ER) group (SES-CD ⩽ 2), and 50 were included as healthy controls in the study. Blood samples were analyzed for tissue factor (TF), factor VIII (FVIII), thrombomodulin (TM), ADAMTS-13, von Willebrand factor (VWF), and endogenous thrombin potential (ETP), as well as collecting data regarding risk factors for TE and CD profile.
Results:
Mean plasma TF activity showed significantly higher levels in the EA group when compared with the ER and control groups (127 pM versus 103 pM versus 84 pM; p = 0.001), although the VWF:Ag (160% versus 168% versus 110%; p = 0.001), VWF/ADAMTS-13 (191 versus 219 versus 138; p = 0.003), FVIII (150% versus 144% versus 90%; p = 0.001) and TM (5.13 ng/ml versus 4.91 ng/mL versus 3.81 ng/ml; p < 0.001) were only increased in CD regardless of EA status when compared with controls. Lastly, ETP with and without TM remained the same in all three groups.
Conclusions:
CD patients in clinical remission with EA present endothelial lesion inducing TF exposure and subsequent coagulation cascade activation. Recommended thromboprophylaxis for EA outpatient subgroups will require additional investigation in order to be validated.
Introduction
Crohn’s disease (CD) is a chronic inflammatory disorder associated with a two- to three-fold higher risk of thromboembolic events (TE) compared with subjects without CD. 1 Exposure is associated mainly with hospitalized patients displaying clinical activity of the disease, although other studies showed that outpatients also present an increased risk of TE for up to 2 months after discharge.2,3 A prevalence of 5.7% thrombosis in CD significantly linked to increased mortality has been previously observed [1.45% versus 10.71%, respectively with an odds ratio (OR) of 8.0]. 4 Nonetheless, the importance of subclinical inflammation in CD on the coagulation process remains unknown. Despite advances in the treatment of inflammatory bowel diseases (IBD) with new drugs, approximately 50% of patients in clinical remission still maintain endoscopic activity (EA). 5
Inflammatory cytokines such as IL-6, IL-17, IL-1β and TNF-α have been shown to up-regulate tissue factor (TF) expression. 6 TF is present mainly in subendothelial layers and is not in contact with circulating coagulation factors under physiological conditions. In the case of vascular breach, TF is released and converted into an activated coagulation factor through the formation of a complex with factor VII known as the initiation phase of the cell-based model of hemostasis.7,8 In the second, or amplification, phase, procoagulant factors are activated on the platelet surface. Both phases lead to formation of thrombin, a key enzyme in the coagulation cascade. Finally, the propagation phase leads to more thrombin generation, resulting in fibrinogen cleavage and fibrin polymerization.
The von Willebrand factor (VWF) is also involved in the regulation of hemostasis and thrombus formation. VWF is a multimeric protein secreted in plasma through a basal mechanism and released in larger quantities upon activation of the endothelium, stimulating platelet adhesion and aggregation.9,10 Under physiological conditions, it is cleaved by a disintegrin and metalloproteinase with thrombospondin motifs 13 (ADAMTS-13) in order to control thrombus formation.11,12 This enzyme, or the VWF/ADAMTS-13 ratio, 10 might be altered during an inflammatory process, facilitating VWF aggregation and thrombosis. Factor VIII (FVIII), carried in plasma by VWF, functions as a procoagulant protein. Increasing numbers of independent case–control studies have clearly demonstrated that high FVIII levels constitute a prevalent, dose-dependent risk factor for venous thromboembolism (VTE) and recurrent venous thrombosis. 13
Anticoagulation pathways are essential for restricting uncontrolled coagulation caused by excessive thrombus formation. While tissue factor pathway inhibitor (TFPI) and antithrombin (AT) operate continuously in inhibiting the initial hemostasis complex TF/FVIIa prior to FXa production in hemostasis, the induced anticoagulant pathways are triggered when thrombin escapes from a non-neutralized injury site. Induced pathways comprise endothelial protein C receptor (EPCR) along with thrombomodulin (TM), a thrombin receptor located in endothelial cells, that integrates crucial biological processes and biochemical pathways related to coagulation, innate immunity, inflammation, and cell proliferation. 14 As endothelial cells become activated by inflammatory conditions such as CD, TM expression decreases, soluble fragments are released via proteolysis, and TM-dependent protective mechanisms on the endothelium are reduced or lost. 14 Thrombin generation results from the reaction between procoagulant and anticoagulant factors, thus assessing this balance in vivo is an important tool in coagulation profiling. 15 The aim of this study was to evaluate the effect of EA in the coagulation profiling of CD patients in clinical remission to better understand the relevance of mucosal inflammation linked to the development of TE in CD.
Materials and methods
The study protocol was approved by the Ethics Committee of the School of Medicine of the University of São Paulo Hospital das Clínicas (CAAE: 43502715.2.0000.0068).
This cross-sectional study focuses on clinical remission CD patients as the main subject of analysis considering the lack of studies regarding thromboprophylaxis in these patients. Sample gathering took place at the Hospital das Clínicas between May 2015 and February 2017.
All adult patients and control subjects gave their written informed consent to participate in the study, which was approved by the Hospital das Clínicas Scientific Committee. The study was conducted in accordance with the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines of good clinical practice, the Declaration of Helsinki, and applicable standard operating procedures.
Patients
The inclusion criteria were defined as CD patients in clinical remission with Crohn’s Disease Activity Index (CDAI) <150 who had been recommended colonoscopy examination, predominantly when biomarkers such as C-reactive protein (CRP) and fecal calprotectin (FC) indicated a high curve or need for surveillance. 16 The CD diagnosis criteria were based on classic histological, endoscopic, and radiological criteria, and patients were also classified by the Montreal classification of inflammatory bowel disease, the Inflammatory Bowel Disease Questionnaire (IBDQ), 17 and, in postoperative patients, by Rutgeerts classification. 18 Additional data was collected for smoking status, contraceptive use, previous and familial thrombosis associated TE conditions, and are further presented in Table 1.
Thrombosis associated conditions.

DVT, deep vein thrombosis; EA, endoscopic activity; ER, endoscopic remission.
Healthy subjects
A total of 50 matched healthy controls by age and sex (19 men and 31 women, mean age 42 years, range 22–67 years) were selected at the Hospital das Clínicas Endoscopic Center after answering a thrombophilia questionnaire according to the CD exclusion criteria described in the section Exclusion criteria for enrolled patients. Essentially healthy subjects were free from present and past thrombotic events, and took no oral anticoagulants or other medications (including oral contraceptives) known to interfere with blood coagulation.
Endoscopic evaluation
The scoring method chosen for EA evaluation was the Simplified Endoscopic Score for CD (SES-CD). 17 In order to avoid potential bias in the interpretation of coagulation parameters, patients with SES-CD between 3 and 6 were interpreted as a grey area group, so that only endoscopic remission (ER, SES-CD ⩽ 2) and EA (SES-CD, ⩾ 7) were included, and patients with SES-CD between 3 and 6 were excluded (Figure 1). All colonoscopies were performed by two experienced endoscopists in the IBD field, and, whenever discordant results in SES-CD arose, common sense analysis of the results calculation prevailed between scores.

Flow chart of screened, included, and excluded patients.
Exclusion criteria for enrolled patients
From 261 consecutive CD patients, only 236 had their colonoscopies performed due to withdrawals and no shows. Patients were first screened at the IBD Clinic by well being and number of stools per day, and had their CDAI calculated separately on the day of their colonoscopy, leading to an additional exclusion of 19 patients with CDAI >150.
Magnetic resonance imaging (MRI) was utilized to exclude any signs of activity in the proximal small bowel. To prevent potential influence bias from small bowel lesions in coagulation parameters, patients with proximal lesions visualized in MRI, not accessible by colonoscopy, were not included.
Other exclusion criteria were use of drugs interfering with coagulation (as salicylic acid, coumarins and heparins) and hepatic disorders, pregnancy, breastfeeding, active cancer, recent surgery, and changes in treatment during the last 3 months were additional excludants. From this standpoint, 72 were excluded due to presenting various other transversal conditions: 45 patients scored SES-CD between 3 and 6, 19 patients scored CDAI ⩾ 150, 2 blood samples were hemolyzed, 3 did not have a blood sample collected on the colonoscopy day, 2 were diagnosed with adenocarcinoma, and 1 withdrew informed consent. Such patients are represented in the flowchart (Figure 1).
Finally, out of 164 patients (67 men and 97 women, mean age 45 years, range 18–83 years), 75 in the EA group (SES-CD ⩾ 7) and 89 in the ER group (SES-CD ⩽ 2) were enrolled in the study.
Laboratory studies
Coagulation parameters
All CD group samples were collected prior to colonoscopy preparation. Blood samples were centrifuged at 3000 g for 20 min at room temperature. Platelet-poor plasma (PPP) were stored at −80°C until analysis for thrombin generation analysis with and without TM, TF activity, VWF antigen (VWF:Ag), ristocetin cofactor (VWF:RCo), ADAMTS-13, FVIII, and TM.
The thrombin generation test was performed according to Hemker et al., 18 using TF 5pM. Samples were evaluated in three distinct wells filled with plasma and reagent (80 µl PPP, 20 µl PPP-Reagent 5pM) and in three other additional wells filled with plasma, reagent, and TM at a final concentration of 6 nM. To calibrate the reaction of each sample, 80 µl of plasma and 20 µl of calibrator were added, in triplicate. After 15 min of incubation at 37°C, 20 μl of fluorogenic substrate diluted in buffer containing albumin and calcium chloride were automatically dispensed into each well. The reaction was monitored using Fluoroskan Ascent (Thermo Labsystems, Helsinki, Finland) with a 390 nm excitation filter and 460 nM emission filter. The addition of TM inhibited thrombin generation and reflected disorders of the activated protein C system.
TF activity was measured by a commercial Chromogenic Activity Assay Kit (Abcam, Cambridge, UK), which evaluates the ability of the TF/FVIIa complex to activate FX in FXa, as measured by the amount of FXa produced by a chromogenic reaction, reading an absorbance range of 405 nm. The change in absorbance was directly proportional to the concentration of TF. Reagent preparation (70 μl of mix) was followed by the addition of 10 μl of TF plasma samples added in each well and incubated at 37°C for 30 min in duplicate. A total of 20 μl of substrate was added to FXa in each well. Absorbances were read in the range of 405 nm every 5 min for 25 min. The variation in optical density (OD) of each sample in relation to the standard curve was divided by the minutes of observation (ΔOD/min). A graph was constructed with the TF concentration in pM and the OD405nm in a four-point curve.
Both the VWF:Ag and the VWF:RCo (in the presence of ristocetin, which promotes platelet aggregation) were determined by means of an automated turbidimetric reading system (HemosIL, Instrumentation Laboratory, Bedford, MA, USA) whose latex particles were sensitized with specific antibodies against VWF and platelet glycoprotein GPIIa/IIIb, respectively. The decrease in transmitted light after agglutination particles is directly proportional to VWF:Ag and VWF:RCo concentrations.
FVIII activity was determined by the functional coagulometric method (HemosIL) based on the activated partial thromboplastin time (aPTT). The same volume of diluted test plasma was added to FVIII-deficient plasma. Prolonged coagulation correction time was proportional to the FVIII concentration (expressed as % of activity).
ADAMTS-13 activity was measured by the chromogenic ELISA method (Technoclone). The ELISA plate, coated with anti-GST antibody (VWF73 ligand), upon incubation with the substrate, retained the VWF73 bound to the plate. The patient’s serum with ADAMTS-13 added to the plate cleaved the VWF73, emitting visible light read in the OD range of 450 nm and interpolated with a standard 5-point curve.
Serum TM (CD 141) was measured by a commercial ELISA kit (Abcam) following the manufacturer’s specifications. Briefly, the coated plate with anti-TM specific monoclonal antibody received the appropriate amount of samples, standards, and controls. The reaction intensity was determined through color generation, measured in the OD range of 450 nm and interpolated with a standard five-point curve.
Thrombophilia markers
Plasma protein C, S, antithrombin (AT) levels, protein C resistance, prothrombin and Leiden mutation were analyzed by standard laboratory methods. Thrombophilia tests were not performed in the control group since existing literature does not consider the role of hereditary thrombophilia markers in the TE etiology of CD.
Fibrinolysis markers
Plasma fibrinogen and D-dimer were analyzed by standard laboratory methods; however, these assays were also not performed in the control group.
Inflammatory markers
CRP was measured by a standard laboratory method and results were considered increased when ⩾0.5 mg/dl. Blood cell count, hematocrit, and blood platelets were also taken for analysis. FC (Bühlmann fCAL® ELISA; Bühlmann Diagnostics, Amherst, NH, USA) were collected on the day prior to colonoscopy, and results were considered increased when ⩾200 µg/g. Again, these tests were not performed in the control group.
Considerations
Thrombophilia, fibrinolysis and inflammatory marker assays were not performed in the control group since they were not the focus of this study, but rather measured in order to establish homogeneity between the EA and ER groups.
Statistical analysis
The sample size for originating statistical difference between groups was calculated in 75 patients per group (considering an alpha error of 0.05 and power of 0.9) using Saibeni et al. as a reference study considering thrombin generation a related test to clinical activity in CD and ulcerative colitis patients (no studies in literature focused on coagulation and EA in CD were found). 18 Data distribution was analyzed by Shapiro test and histogram analysis, and categorical data were summarized by percentage and comparison groups assessed with Fisher’s exact test. Quantitative data differences with normal distribution were assessed by Student’s t test and the Wilcoxon rank sum test for not normally distributed data. Comparisons between the three groups (EA, ER, and controls) were performed by the nonparametric Kruskal Wallis test with Bonferroni/Dunn test. The level of significance was set at p = 0.05. Associations among continuous variables were tested with Pearson’s correlation. RedCap (Research Electronic Data Capture) was the database of choice. All statistical tests were performed using R Statistics. Patients with missing data regarding the main variables were excluded from the study.
Results
Baseline demographic data
Females predominated in the study (59.2%), averaging 44 years of age (35–57), with age at clinical onset 29 years (22–41) and disease duration 11 years (7–17). Total IBDQ was 134 (114–147) and body mass index (BMI) was 24 (22–27). Disease location was as follows: ileocolonic in 66 (34%), terminal ileum disease in 60 (36.5%), and colonic in 38 patients (23%). Disease behavior was inflammatory in 57 (34.7%), stricturing in 48 (29.2), fistulizing in 59 (36%), and 33 (20%) had perianal involvement.
Groups were similar regarding age, gender, BMI, age at diagnosis, disease duration, disease location, and IBDQ (Table 2), and also similar in terms of treatment except for the increased use of biologic drugs (54% versus 46%; p = 0.041). The EA group showed more stricturing disease (43% versus 18%; p = 0.001), higher Rutgeerts scores (91% were i0 in the ER group, while 53% were i2, 3% i3, and 37% i4 in the EA group), higher CRP levels above 5 mg/l (59% versus 40%; p = 0.001), higher platelets levels (276.000 versus 242.000, p = 0.012) and higher FC levels (58% versus 41%) with a median FC higher in the EA group (819 versus 277, p = 0.004) (Table 2).
General data for 164 CD patients in clinical remission.
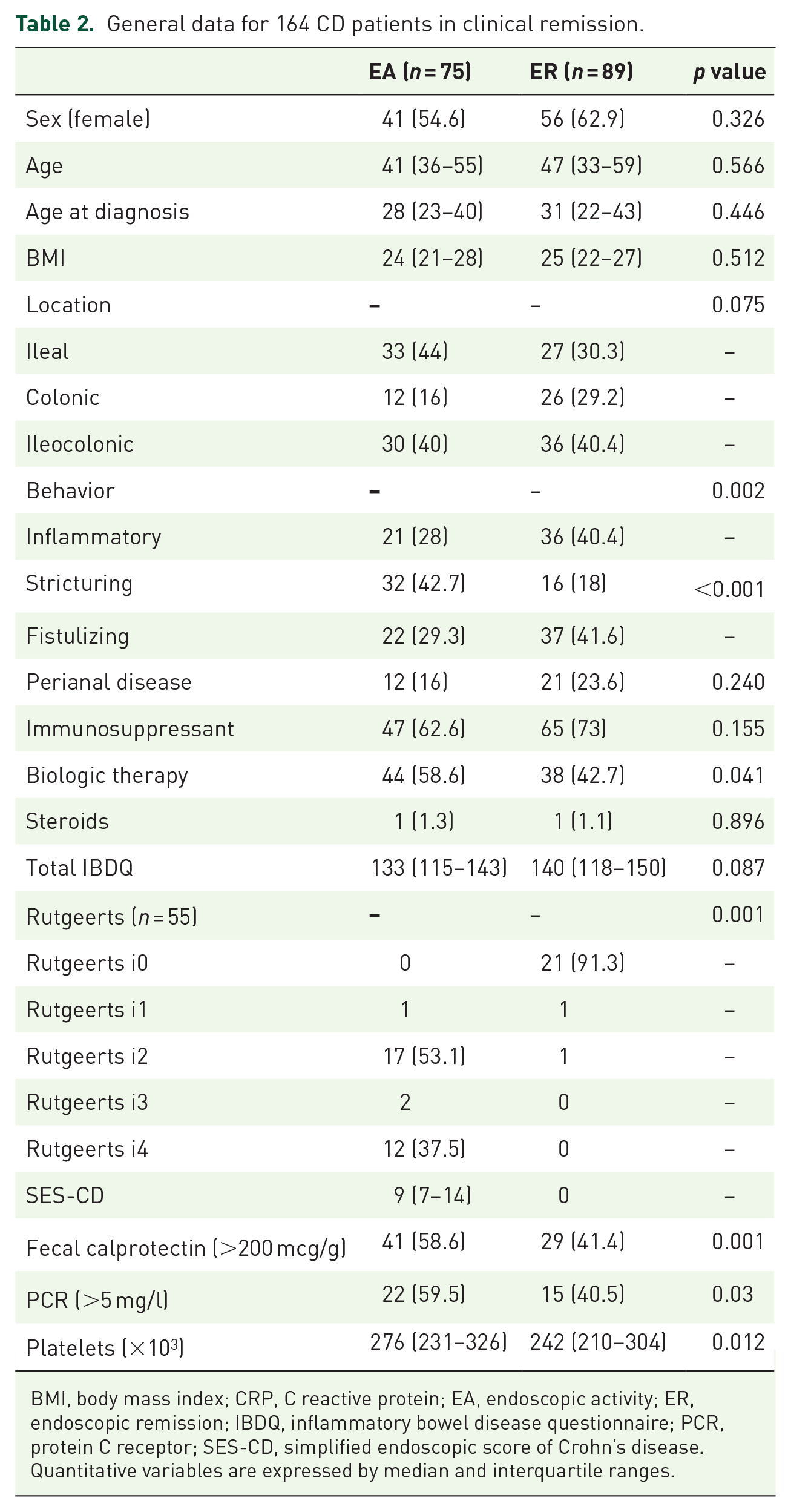
BMI, body mass index; CRP, C reactive protein; EA, endoscopic activity; ER, endoscopic remission; IBDQ, inflammatory bowel disease questionnaire; PCR, protein C receptor; SES-CD, simplified endoscopic score of Crohn’s disease.
Quantitative variables are expressed by median and interquartile ranges.
With reference to TE-associated conditions, groups were similar (Table 1) by: active smoking (11% versus 8%, p = 0.364), previous TE (1% versus 6%, p = 0.149), familial TE (4% versus 9%, p = 0.209), oral contraceptives (5% versus 4%, p = 0.663) and steroid use (1% in each group). In relation to hereditary thrombophilias, the only difference between the two groups was the prevalence of positive lupus anticoagulant (tested in 141 patients) shown higher in the EA group (8% versus 1%, p = 0.047). However, two of the five patients with positive lupus anticoagulant did not perform a second confirmatory test. No statistical difference was observed regarding antithrombin deficiency (n = 148; 12% versus 11%, p = 0.781), protein C deficiency (n = 148; 4% versus 2%, p = 0.272), protein S deficiency (n = 154; 4% versus 2%, p = 0.468), protein C resistance (n = 50; 10% versus 0.3%, p = 0.333), anti-cardiolipin IgM (n = 70, 11% versus 9%, p = 0.997), anti-cardiolipin IgG (n = 71, 11% versus 9%, p = 0.651), and prothrombin mutation (n = 46; 6% versus 7%, p = 0.831) between the EA and ER groups, as well as there being no difference regarding fibrinolysis markers (Table 1). Only one of the six patients tested for the factor V Leiden mutation had the mutation. Not all of the previously mentioned tests were performed on all patients due to lack of reagents in the central laboratory during the study period.
Coagulation parameters
Tissue factor
CD patients presented higher TF levels compared with controls, and levels were significantly higher in patients with EA (127 pM versus 103 pM versus 84 pM; p = 0.001) (Figure 2A). In addition, 82% in the activity group and 55% in the remission group had TF values above the 3rd quartile of the control group. The data show that even CD patients in endoscopic remission, while not in deep remission, maintained some degree of endothelial activation, resulting in increased TF levels, therefore triggering thrombosis. TF levels compared with FC showed a weak but positive correlation r = 0.26 (CI 0.03–0.46, p = 0.02). Among the other parameters, TF was the one with the best area under the curve in predicting EA, with a cutoff of 95.49, sensitivity of 0.80 and specificity of 0.47.
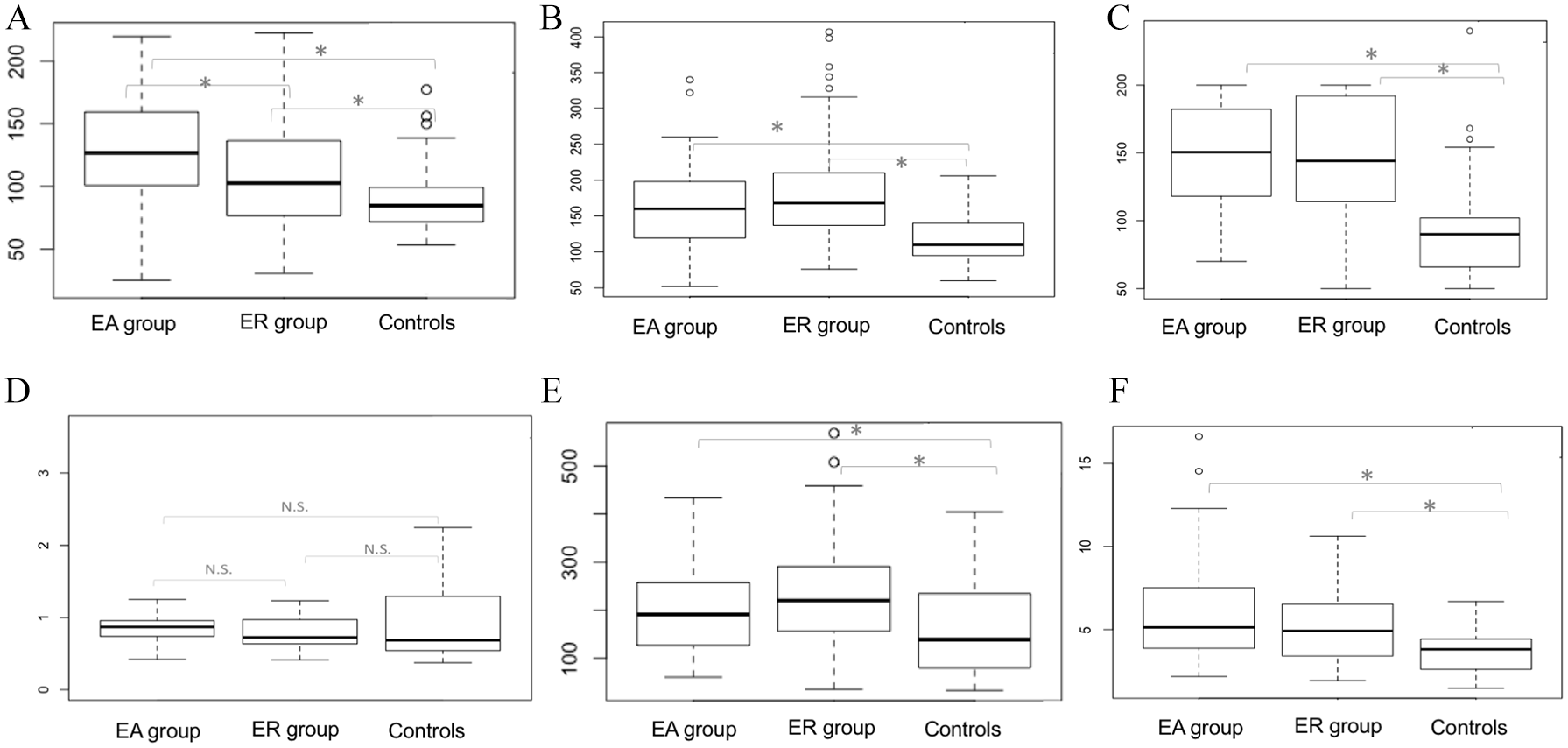
(A–F) Mean plasma tissue factor, VWF:Ag, FVIII, ADAMTS-13, VWF/ADAMTS-13, TM levels, between EA (n = 75), ER (n = 89) and control groups (n = 50) respectively.
VWF:Ag, VWF:RCo, FVIII and VWF/ADAMTS-13
CD patients also had higher VWF:Ag (160% versus 168% versus 110%; p = 0.001) (Figure 2B) and FVIII (150% versus 144% versus 90%; p = 0.001) (Figure 2B and C, respectively) when compared with controls regardless of disease activity, suggesting that the disease itself can be associated with endothelial dysfunction. ADAMTS-13 showed no difference between the EA, ER, and control groups (0.87% versus 0.72% versus 0.68%; p = 0.07) (Figure 2D). Nevertheless, the VWF/ADAMTS-13 ratio was higher in CD (191 versus 219 versus 138; p = 0.003) (Figure 2E), demonstrating a relative reduction of this enzyme in CD patients.
Thrombomodulin
Regardless of inflammatory activity, CD patients showed highly elevated levels of serum TM compared with controls (5.13 versus 4.91 versus 3.81 ng/ml, p < 0.001) (Figure 2F). Such increased levels represent endothelial cell activation supported by the sequential release via proteolytic activation into the blood, reducing the TM-dependent protective mechanisms linked to the endothelium. As inflammation progresses, the endothelium turns itself off, becoming incapable of performing its antagonistic neutralization role, thus increasing the risk of thrombosis.
Thrombin generation
No significant difference was observed between the CD groups and controls in endogenous thrombin potential (ETP) without TM (1674 nmol/l thrombin X min versus 1680 nmol/l thrombin X min versus 1654 nmol/l thrombin X min, p = 0.4) (Figure 3A) and with TM (609 nmol/l thrombin X min versus 539 nmol/l thrombin X min versus 650 nmol/l thrombin X min, p = 0.15) (Figure 3B). ETP ratio with and without TM (0.37 versus 0.31 versus 0.43, p = 0.052) (Figure 3C) showed no difference between the groups. Also, in relation to other parameters of thrombin generation, lag time showed no difference between the groups and the peak without TM was the only one showing a significant difference between the CD groups and controls (295 versus 298 versus 277 nmol/l; p = 0.001) (Figure 3D). Although this difference was not maintained when TM was added (127 versus 112 versus 130 nmol/l; p = 0.54), this parameter was considered as the maximum thrombin concentration at a given time, and has already been demonstrated as a possible hypercoagulability factor, suggesting increased high risk of recurrent VTE. 19 In general terms, the coagulation balance is stable in patients in clinical remission; however, as observed in the case of a patient with an important EA forming thrombosis during the study, it caused direct impact on thrombin generation as seen in the case in the following.

(A–D) ETP without TM, ETP with TM, ratio of ETP with and without TM and peak levels between EA (n = 75), ER (n = 89) and control group (n = 50), respectively.
All data collected from this study indicate that CD patients carry the inherent markers of endothelial dysfunction, which are characterized by an increase in VWF, VWF/ADAMTS-13 ratio, FVIII and serum TM, promoted by an increased exposure of TF in the damaged endothelial cells at inflammation sites. While not all anticoagulant mechanisms of thrombin have been explored in this study, TM definitely showed altered results, most likely as a compensatory mechanism.
Case report
During the course of the study, there was only one case of inferior vena cava thrombosis in a patient with intense endoscopic activity (SES-CD of 23). Upon analyzing the laboratory parameters, it was found that this patient had almost twice the mean of the control group TF (patient TF 157 pM versus mean TF in control group 84 pM). Regarding ETP, there was a reduction of only 19% in ETP after TM addition (Figure 4), while in healthy controls there was a reduction of over 45% in ETP (Figure 5).

Only 19% reduction in ETP after TM addition in a patient with intense EA that evolved with DVT. (ETP without TM: 1723 nmol/l thrombin X min; ETP with TM: 1264 nmol/l thrombin X min).

ETP without and with TM in healthy control; 47% reduction in ETP after TM was added to the test (ETP without TM: 1866 nmol/l thrombin X min; ETP with TM: 999 nmol/l thrombin X min).
Discussion
Venous thromboembolism remains a significant complication of IBD with associated morbidity and mortality. The role of clinically active inflammation has been well documented; however, less known is the coagulative state of IBD patients when IBD is quiescent. This work seeks to define the coagulation profile of CD patients who are in clinical remission undergoing endoscopy for nonsymptomatic indications, resulting in a significantly higher level of tissue factor activity in endoscopically active patients versus remission and versus controls, suggesting a prothrombotic state with EA. Furthermore, significant differences in other endothelial dysfunction markers were found when both EA and ER were compared with controls suggesting an imbalance of these factors in Crohn’s patients regardless of disease activity.
Increased TF levels alone would be irrelevant for consideration unless the activation of coagulation occurs as described in previous literature, where fibrinogen A and thrombin-antithrombin complexes are increased in IBD patients.20,21 Our results are in line with the current findings except for the use of endoscopic evaluations linking the increase in TF by the presence of bowel inflammation and ulcers. TF exposure reflects not only pro-coagulant markers such as FVW and FVIII, but also an increase in compensatory anticoagulant factors such as TM.
Another important point of discussion is the assessment of the CDAI, which has long been criticized as rather inaccurate index to confirm clinical remission in CD patients, due mainly to a slight unbalanced superiority of those parameters exhibiting subjectivity. EA has been proven to be more precise. A single previous study has evaluated coagulation beyond clinical activity by performing endoscopic studies in ulcerative colitis showing higher FVW levels, 22 being justified by a marker of both endothelial dysfunction and acute phase response to inflammation. On the opposite hand, low levels of ADAMTS-13 also found in this study can be justified either by an excess consumption of VWF multimers released by the activated endothelium, or by greater proteolytic cleavage by activated neutrophils in cases of inflammatory circulating cytokines such as TNF and IL-6.10,11,23 These outcomes are also reflected in higher platelet and FVIII levels shown in the EA group, thus inferring it might be a reaction to the inflammatory burden or related to endothelial disruption.22,24
With regards to anticoagulation pathways, TM soluble levels were studied as a compensatory mechanism when triggered by the increase of procoagulant factors. 25 The release of TM bound from the endothelium reduces its TM-dependent protective mechanisms, ultimately lowering the activation of the protein C pathway. 14 Lastly, looking at the coagulation balance through the thrombin generation potential, no differences were found in ETP between all three groups, although the highest peak of thrombin generation among patients with CD versus controls might be relevant for consideration, as also previously shown in the literature. 26 The peak has already been associated with a higher risk of deep vein thrombosis with an OR of 1.74. 19
It is worth mentioning that ETP in general is altered in CD patients with intense inflammatory potential as demonstrated in the clinical case previously mentioned. 18 This patient, despite being in clinical remission, has presented with severe EA and deep thrombosis without other TE risk factors, except for the inflammatory burden. In addition to having a possible resistance to protein C as demonstrated by high ETP after TM, they still had much higher levels than TF when compared with the average.
The previous observations point to the existence of a hypercoagulable state in CD. Such compensatory mechanisms of endogenous anticoagulation factors might produce a rebalancing of the coagulation and limit the capacity of compensations when other risk factors are present, thus predisposing patients to thrombosis. Further studies are needed to validate whether EA outpatient subgroups will be recommended thromboprophylaxis, especially for those being treated at public health services in developing countries, where there are often insufficient hospital beds to accommodate the intake of patients. Deserving further investigation is the analysis of TF as a potential thrombosis biomarker (as in patients with an increased risk of bleeding with prothrombin time and aPTT).
It is worth mentioning that, at the time of sample collection, the chosen methodology was not patient reported outcomes but rather CDAI, since data collection had started prior to this updated method became available in IBD.
Conclusion
This paper remains an unprecedented study in evaluating the role of EA in the hemostasis of CD patients while in clinical remission. The EA subgroup presents an active coagulation process due to endothelial lesions and TF exposure, being in line with the cell-based model of hemostasis. Compensatory mechanisms might promote coagulation rebalancing, predisposing patients to thrombosis when other risk factors are present.
In conclusion, the dialogue between the gastroenterologist and the hematologist becomes paramount in the attempt to unveil all the factors that influence the high risk of thrombosis in CD patients, making this empirical research a call for further deepening of research into issues related to coagulation, including the role of TF as one of the main markers of inflammation versus the coagulation interface.
Footnotes
Acknowledgements
Special thanks to all investigators, study staff, and participants who were involved in the study; Magaly Gemio Teixeira for her contribution with patients from Coloproctology Outpatient Center, University of Sao Paulo School of Medicine, Brazil; Joao Italo for his statistical analysis contribution; Laurent Peyrin-Biroulet and Jeremy Lagrange who critically revised the article contributing with relevant intellectual content; and editorial support provided by Priscila Leal Felipe.
Author contributions
ARA: contributed to the study design, implemented the study protocol, collected and interpreted the data, wrote the first draft of the manuscript, contributed to subsequent revisions, and contributed to its intellectual content; MFCA, AOMC, AMS, PV, ED, and FJC oversaw the implementation of the study protocol and contributed to its intellectual content; ARA and ASC contributed to the implementation of the study protocol and collected the data; TRFR, CLOA and IN contributed to the reading of the results. AZAL conceived and designed the study, oversaw the study implementation and collection of data, interpreted the data, contributed to the writing of the first draft of the manuscript and its subsequent revisions and contributed to its intellectual content. ARA and AZAL are the guarantors of this work and, as such, had full access to all the data in the study.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant #2015/06196-0, São Paulo Research Foundation (FAPESP).
