Abstract
Background:
Anemia is a common extraintestinal manifestation of inflammatory bowel disease (IBD). However, data on the influence of anti-tumor necrosis factor-alpha (anti-TNF-α) agents and iron supplementation on anemia in patients with IBD are sparse. We assessed the effect of iron supplementation in patients with IBD initially treated with an anti-TNF-α agent.
Methods:
Data from 79 IBD patients who started anti-TNF-α treatment at a tertiary hospital were analyzed. The patients were divided into the anti-TNF-α (n = 52) and anti-TNF-α with iron supplementation (n = 27) groups. Effects on laboratory parameters, the prevalence of anemia, and disease activity were evaluated at baseline (year 0) and 1 year later.
Results:
The hemoglobin (Hb) level significantly increased between years 0 and 1 in both groups [12.0 ± 1.8–13.3 ± 2.0 g/dL in the anti-TNF-α group (p < 0.001) and 9.8 ± 2.4–11.7 ± 2.3 g/dL in the anti-TNF-α and iron supplementation group (p = 0.004)]. In a subgroup analysis of severely anemic patients with IBD, iron supplementation increased the magnitude of the improvement in Hb level (8.5 ± 1.5–11.4 ± 2.1 g/dL; p = 0.001) compared with the anti-TNF-α group (9.3 ± 0.8–11.4 ± 2.7 g/dL; p = 0.081). Disease activity was significantly improved in both groups at year 1 compared with year 0. Persistent anemia was significantly correlated with severe anemia at baseline (p = 0.017).
Conclusion:
In anemic patients with IBD, anti-TNF-α agents led to clinically meaningful improvements in anemia independent of iron supplementation. Also, iron supplementation could be helpful in severely anemic patients with IBD.
Introduction
Inflammatory bowel disease (IBD) is a chronic inflammatory condition, the major forms of which are ulcerative colitis (UC) and Crohn’s disease (CD). The major signs and symptoms of IBD are in the gut; however, extraintestinal manifestations are frequent. 1 Anemia is a common extraintestinal manifestation of IBD. 2 Anemia in patients with IBD significantly increases healthcare costs and negatively affects quality of life.3,4 The prevalence of anemia in patients with IBD is approximately 20–30%, and it is more common in CD than in UC.5–7 Anemia in patients with IBD may stem from a variety of causes. The most frequent are iron deficiency caused by bleeding and absorption disorder, iron homeostasis disorder caused by inflammation, and vitamin B12 and folic acid deficiency caused by bone marrow suppression and absorption disorder.7,8 Among these, iron-deficiency anemia and chronic inflammation are the most important causes; indeed, iron-deficiency anemia is reported in up to 90% of anemic patients with IBD. 9 According to the European Crohn’s and Colitis Organization (ECCO) guidelines, iron supplementation is recommended for all IBD patients with iron-deficiency anemia. 10
Tumor necrosis factor-alpha (TNF-α) inhibitors (infliximab and adalimumab) are the first-line biologic treatments for IBD patients, and mucosal healing is key in the treatment of chronic inflammatory diseases.11,12 The inflammatory activity of IBD is a risk factor for anemia.13,14 Because IBD-related anemia results from a combination of iron deficiency and chronic inflammation, treatment of anemia in patients with IBD aims to resolve mucosal inflammation and provide supplemental iron. Although anti-TNF-α treatment reportedly increases hemoglobin (Hb) levels in patients with ankylosing spondylitis and rheumatoid arthritis,15,16 data on the effects of anti-TNF-α agents on anemia or Hb levels in patients with IBD are sparse. To our knowledge, three studies have evaluated the effects of anti-TNF-α and iron supplementation on anemia in patients with IBD.17–19 All of these studies involved Western populations. Therefore, we evaluated the efficacy of iron supplementation in Asian patients with IBD treated with anti-TNF-α agents.
Materials and methods
Study design and patients
We retrospectively reviewed the medical records of patients with IBD who started anti-TNF-α (infliximab or adalimumab) treatment from January 2000 to December 2014 in a tertiary medical center in South Korea. The observation period was 1 year. This study was approved by the Institutional Review Board of Gil Medical Center (GCIRB2018-054) and informed consent was not needed because of the retrospective nature of the study and the fact that the analysis used anonymous clinical data. Clinical symptoms as well as radiological, histopathological, radiological, and international criteria were assessed to diagnose UC and CD.20,21 The epidemiological and clinical data obtained included patient age, sex, endoscopic and radiologic findings, medications, and disease location according to the Montreal classification system. 22
Only patients naïve to biologic agents and with information on laboratory results at initiation of anti-TNF-α treatment were included. Also, all included IBD patients had received anti-TNF-α treatment for at least 1 year (continuously during the study period) and had available laboratory and follow-up data at years 0 and 1 after starting anti-TNF-α treatment. Patients treated with iron supplementation or blood transfusions in the 3 months prior to screening or who had a history of erythropoietin treatment were excluded. The Hb level, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin level, mean corpuscular hemoglobin concentration, red blood cell (RBC) count, white blood cell count, platelet count, C-reactive protein (CRP) level, and erythrocyte sedimentation rate (ESR) were measured using routine laboratory tests. Disease activity was scored at years 0 and 1 by calculating the Mayo Score (Ulcerative Colitis Disease Activity Index; UCDAI) or Harvey–Bradshaw Simple Index (HBI).23,24
Iron supplementation and definition of anemia
We divided IBD patients into the anti-TNF-α and anti-TNF-α with iron supplementation groups. Iron supplementation was defined as taking oral iron [Feroba-You SR (Bukwang Pharmacy, Seoul, South Korea) or Ferrumpola (JW Pharmaceutical Corporation, Seoul, South Korea)] for more than 6 months during the study period. We diagnosed anemia according to the age- and sex-specific Hb thresholds set by the World Health Organization (WHO). 25 The WHO defines anemia as an Hb level of <13.0 g/dL in males and <12.0 g/dL in females. The prevalence of anemia was estimated as the ratio of the number of anemic patients to the total number of patients enrolled. Patients with an Hb level of <10.0 g/dL were defined as severely anemic, irrespective of age and sex.6,13 The Hb level and prevalence of anemia in years 0 and 1 were evaluated in both groups.
Statistical analysis
Data were collected and statistical analysis was performed using SPSS 20.0 (IBM Corp., Armonk, NY, USA). Categorical variables are expressed as percentages and frequencies, and continuous variables as the means ± standard deviations (SDs) (or medians with interquartile ranges). Pre- and post-treatment comparisons were performed using the paired t-test (or the Wilcoxon signed-rank test for medians). A multivariate logistic regression analysis was performed to identify factors associated with persistent anemia after 1 year of therapy. A p-value < 0.05 was considered indicative of significance.
Results
A total of 79 IBD patients treated with anti-TNF-α agents (37 with UC and 42 with CD) were included in this study. Table 1 lists their baseline demographic and clinical characteristics. The anti-TNF-α agents administered were infliximab (64 patients) and adalimumab (15 patients). The prevalence of anemia was 75.9% (60/79) and that of severe anemia was 34.2% (27/79). The mean disease duration of the patients with IBD was 52.1 months. Of all patients, 27 (34.2%) received an anti-TNF-α agent and iron supplementation simultaneously, and 52 (65.8%) received only an anti-TNF-α agent.
Baseline characteristics of inflammatory bowel disease patients treated with anti-tumor necrosis factor-alpha (TNF-α) agents.

The laboratory parameters of the patients were evaluated at years 0 and 1 (Table 2). At year 1, the mean Hb level in both groups increased significantly compared with the baseline [12.0 ± 1.8–13.3 ± 2.0 g/dL in the anti-TNF-α group (p < 0.001) and 9.8 ± 2.4–11.7 ± 2.3 g/dL in the anti-TNF-α and iron supplementation group (p = 0.004)]. The hematocrit and RBC count also increased significantly compared with the baseline in both groups. The mean levels of CRP and ESR decreased at years 0 and 1 in both groups. Table 3 shows the change in hemoglobin and disease activity in the UC and CD groups.
Hematologic and disease activity parameters pre- and post-treatment in inflammatory bowel disease patients at years 0 and 1.

CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; Hb, hemoglobin; Hct, hematocrit; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; PLT, platelet; RBC, red blood cell; RDW, red cell distribution width; WBC, white blood cell.
Hematological and disease activity parameters pre- and post-treatment in patients with ulcerative colitis and Crohn’s disease at years 0 and 1.

CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; Hb, hemoglobin; Hct, hematocrit; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; PLT, platelet; RBC, red blood cell; RDW, red cell distribution width; WBC, white blood cell.
We performed a subgroup analysis of patients with IBD and severe anemia (Table 4). The mean Hb level of patients with IBD treated with only anti-TNF-α agents was not significantly different between years 0 and 1 (9.3 ± 0.8–11.4 ± 2.7 g/dL; p = 0.081). However, the change in Hb level was greater in the anti-TNF-α agent and iron supplementation group (8.5 ± 1.5–11.4 ± 2.1 g/dL; p = 0.001). The hematocrit and RBC count also increased significantly only in the anti-TNF-α agent and iron supplementation group.
Major hematologic and disease activity parameters pre- and post-treatment in inflammatory bowel disease patients with severe anemia at years 0 and 1.

CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; Hb, hemoglobin; Hct, hematocrit; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; PLT, platelet; RBC, red blood cell; RDW, red cell distribution width; WBC, white blood cell.
The prevalence of anemia among the IBD patients was 75.9% at baseline and 41.8% at year 1 (Figure 1). The prevalence of anemia in the anti-TNF-α group was 67.3% at year 0 and 34.6% at year 1, and that in the anti-TNF-α and iron supplementation group was 92.6% at baseline and 55.6% at year 1. The median UCDAI and HBI scores were significantly lower in both groups at year 1 compared with year 0 (Table 5). The logistic regression analysis revealed that persistent anemia after 1 year of therapy was significantly associated with severe anemia at baseline [odds ratio (OR): 3.41; 95% confidence interval: 1.25–9.28; p = 0.017] (Table 6).
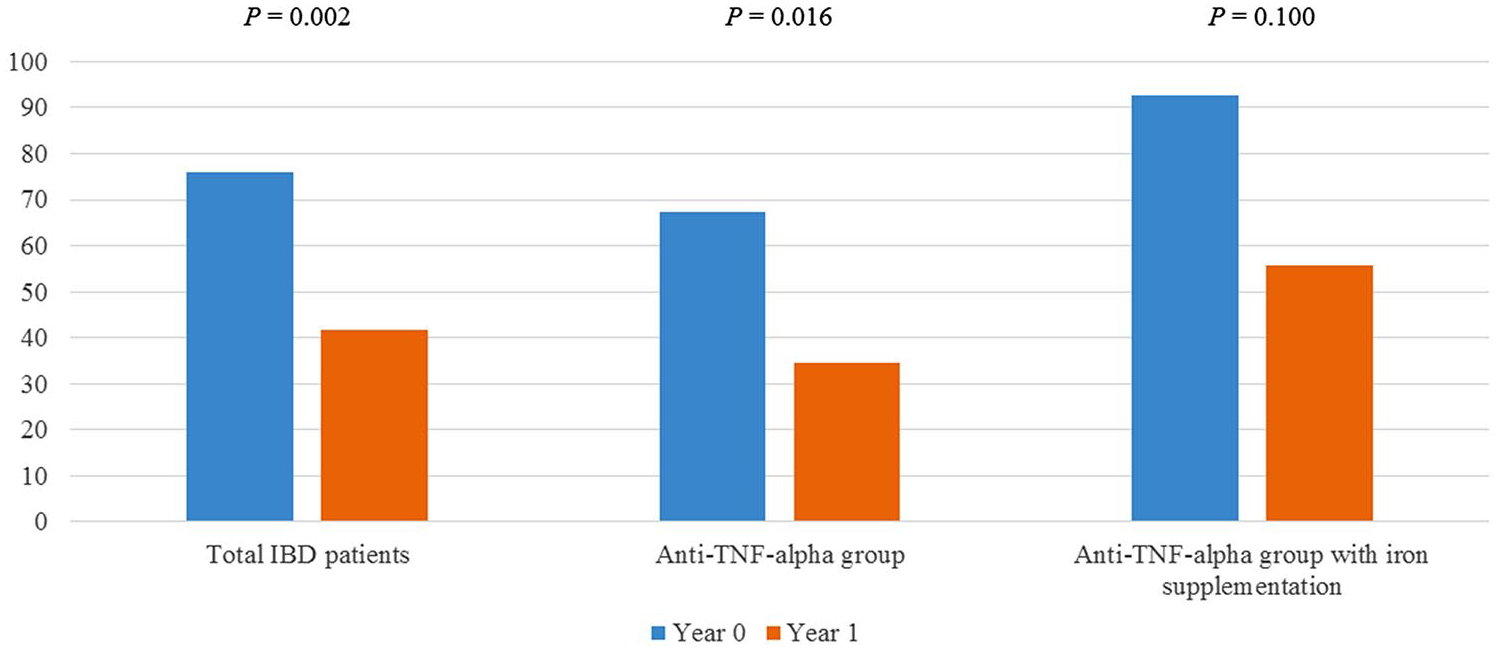
The prevalence of anemia in patients with inflammatory bowel disease before and after treatment.
Disease activity in inflammatory bowel disease patients treated with anti-TNF-α agents at years 0 and 1.

CD, Crohn’s disease; HBI, Harvey–Bradshaw Simple Index; IQR, interquartile range; TNF-α, tumor necrosis factor alpha; UC, ulcerative colitis; UCDAI, Ulcerative Colitis Disease Activity Index.
Multivariate logistic regression analyses of factors associated with persistent anemia after 1 year of therapy.
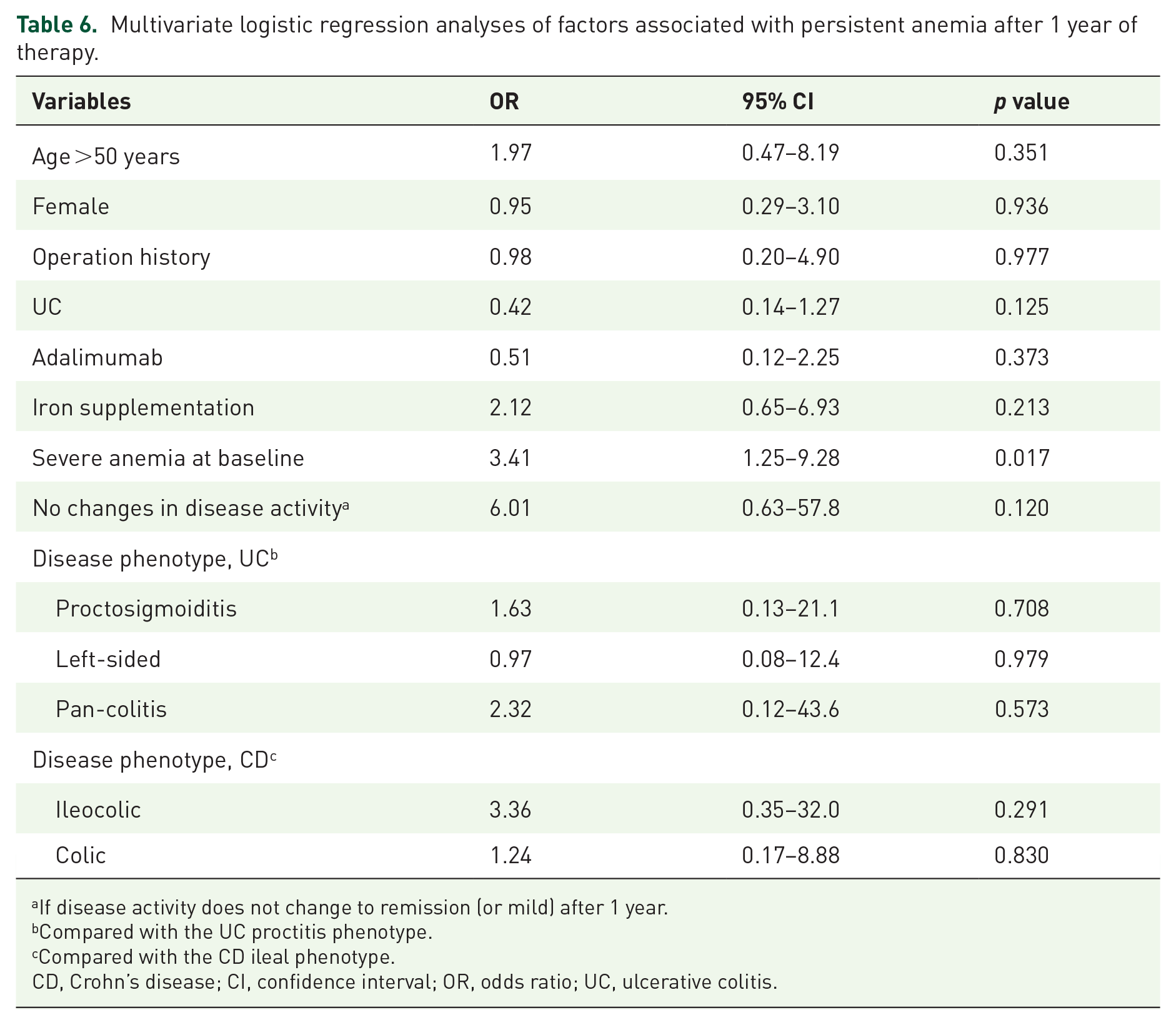
If disease activity does not change to remission (or mild) after 1 year.
Compared with the UC proctitis phenotype.
Compared with the CD ileal phenotype.
CD, Crohn’s disease; CI, confidence interval; OR, odds ratio; UC, ulcerative colitis.
Discussion
This is the first report on the efficacy of anti-TNF-α agents for treating anemia in biologic-naïve Asian patients with IBD. To date, only three studies have evaluated the effects of anti-TNF-α therapy and iron supplementation on anemia in patients with IBD,17–19 and none have yet been reported in Asia. Also, all patients enrolled in this study were receiving biologic therapy for the first time and had a confirmed diagnosis of IBD. This enabled us to link anti-TNF-α effects with outcomes of anemia treatment during the first year. Anti-TNF-α treatment increases the Hb level and decreases inflammatory activity. These effects were similar in the UC and CD groups. In addition, severely anemic patients with IBD who received an anti-TNF-α agent and iron supplementation exhibited a significantly greater increase in Hb level compared with those who received only an anti-TNF-α agent.
Research on the effects of anti-TNF-α agents and iron supplementation on Hb levels and anemia in patients with IBD is limited.17–19 Koutroubakis et al. evaluated the effect of anti-TNF-α treatment on Hb levels in 430 patients with IBD. 17 The median Hb level did not change between years 0 and 1, and the prevalence of anemia at year 1 was similar to that at baseline (36.6% versus 38.1%; p = 0.77). In addition, a hematopoietic response was observed in only 33.6% of the anemic patients with IBD, 94% of whom received iron supplementation. 17 This is inconsistent with our findings, but the difference may be explained as follows. First, the proportion of CD patients was 53% in this work, compared with 75.3% in the previous study. In addition, the rates of surgery related to IBD were 12.6% and 47.2%, respectively. This indicates that the patients in the previous study had a longer duration of IBD, and so their intestinal chronic inflammation was more severe and they experienced more complications. In fact, the disease duration in the previous study was approximately twofold that of our patients. IBD (and particularly CD) is a progressive disease; therefore, delayed administration of anti-TNF-α agents can exacerbate bowel mucosal inflammation and lead to irreversible structural damage. 26 The impact of disease duration on the efficacy of anti-TNF-α agents is greatest for CD. Therefore, in the previous study, because the patients suffered from IBD for a longer time period and the percentage of patients with CD was high, it is likely that anti-TNF-α treatment was less efficacious than in our study. Second, the previous study enrolled a larger proportion of women compared with this work (29.1% versus 51.6%). Anemia is rarer in men than in women, so anemia in male patients with IBD is likely to be associated with IBD. However, the frequency of iron-deficiency anemia is higher in women than in men, irrespective of the presence of IBD. 27 In addition, it is likely that other factors, such as menstruation or hemolytic anemia, affect anemia in women. Hence, if the treatment only focused on IBD, the likelihood of achieving significant improvement in anemia will be lower in women than in men. Therefore, the effects of anti-TNF-α agents on anemia in the previous study were less marked than those reported here. By contrast, anti-TNF-α therapy was effective against anemia in patients with IBD in two other previous studies, similar to our findings.18,19
Generally, intravenous iron should be considered a first-line treatment in patients with clinically active IBD and previous intolerance to oral iron because orally administered iron has low bioavailability and may cause an absorption disorder related to increased hepcidin production. 10 In this study, oral iron was used instead of intravenous iron, which may explain the lack of a clear benefit in terms of Hb elevation in all patients with IBD. Interestingly, among severely anemic patients (34.2% of the total patient population), oral iron supplementation resulted in a significant increase in the Hb level. Unlike the ECCO guidelines (oral iron recommended for mildly anemic patients with IBD), our results showed that oral iron is beneficial in severely anemic patients. 10 Although oral iron has disadvantages compared with intravenous iron,9,28 the convenience of oral administration enables prolonged use. The effectiveness of oral iron supplementation in severely anemic patients with IBD needs to be confirmed in large clinical studies.
This study used only oral iron supplementation for the following reasons. First, according to the guidelines, intravenous iron should be used as a first-line treatment in patients who are intolerant of oral iron supplementation.10,29 Because all patients enrolled in this study tolerated oral iron supplementation (used >6 months), there was no reason to perform additional intravenous iron supplementation. Second, although intravenous iron supplementation is safer than in the past, life-threatening reactions can occur, albeit rarely.30,31 Also, intravenous iron supplementation is not used initially unless there is a specific reason, such as the inability to take oral iron supplementation, a severe absorption disorder, severe blood loss, or the need to use erythropoiesis-stimulating agents. Patients in this study did not undergo parenteral iron supplementation because they did not fulfill any of these criteria.
Iron deficiency occurs in patients with IBD because of, for example, intestinal blood loss (a major reason), decreased dietary intake, iron malabsorption, and functional deficiencies. In particular, intestinal inflammation interferes with iron absorption, increases hepcidin production, and internalizes or decomposes iron.32,33 Because inflammation hampers the transport of iron from intestinal epithelial cells to the circulation, iron accumulates in macrophages and monocytes.34,35 Ultimately, a chronic inflammatory status prevents iron from entering the circulation. Therefore, the most important consideration in patients with IBD is controlling chronic inflammation. Indeed, iron supplementation in patients with uncontrolled inflammation is not beneficial.
It was confirmed that the risk of persistent anemia appearing after 1 year of treatment was high when initially accompanied by severe anemia. IBD disease status is likely to be severe in the presence of initial severe anemia, and the response to treatment with an anti-TNF-α agent is likely to be worse than that of patients with a milder disease status. Considering the importance of inflammation control in the improvement of anemia, it is highly likely that the treatment response to anti-TNF-α agents affects the recovery from anemia. In addition, the OR value tended to increase when disease activity did not improve. This may provide another basis for showing that IBD disease control plays an important role in the recovery of anemia. Also, if the initial hemoglobin level is too low, the anemia will not completely resolve, even after some recovery.
This study had several limitations. First, it was a retrospective single-center study; therefore, we were unable to identify the causes of anemia. Second, the latent effects of concomitant therapies were not evaluated. Third, the patients were assessed in a tertiary hospital; therefore, the data may exhibit a selection bias towards patients with severe IBD. Thus, the associations with anemia observed in this study may not reflect associations in the general IBD patient population. Finally, a small number of patients were enrolled in our study, which restricted the subgroup analysis. We conducted a normality test to reduce the statistical error that may occur due to the small number of patients. If normality was not satisfied, the statistical analysis was performed using a non-parametric method to secure some statistical power. In conclusion, anti-TNF-α therapy increases Hb levels and decreases the rate of anemia in IBD patients after 1 year. Iron supplementation also improves anemia in patients with IBD, particularly the Hb level in those with severe anemia. Therefore, iron supplementation may be useful in patients with IBD and severe anemia, but controlling the underlying inflammation using biologics is more important.
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (grant number: 2020R1I1A306686511)
