Abstract
Background:
Iron deficiency anemia (IDA) is a common complication of inflammatory bowel disease (IBD) and can result in reduced quality of life and increased healthcare costs. IDA is treated with iron supplementation, commonly with intravenous iron formulations, such as ferric carboxymaltose (FCM), and iron sucrose (IS).
Methods:
This study assessed the cost-effectiveness of FCM compared with IS, in terms of additional cost per additional responder in patients with IDA subsequent to IBD in the Spanish setting. An economic model was developed to assess the additional cost per additional responder, defined as normalization or an increase of ⩾2 g/dl in hemoglobin levels, for FCM versus IS from a Spanish healthcare payer perspective. Efficacy inputs were taken from a randomized controlled trial comparing the two interventions (FERGIcor). Costs of treatment were calculated in 2021 Euros (EUR) using a microcosting approach and included the costs of intravenous iron, healthcare professional time, and consumables. Cost-effectiveness was assessed over one cycle of treatment, with a series of sensitivity analyses performed to test the robustness of the results.
Results:
FCM was more effective than IS, with 84% of patients achieving a response compared with 76%. When expressed as number needed to treat, 13 patients would need to switch treatment from IS to FCM in order to achieve one additional responder. Costs of treatment were EUR 323 with FCM compared with EUR 470 with IS, a cost saving of EUR 147 with FCM. Cost savings with FCM were driven by the reduced number of infusions required, resulting in a reduced requirement for healthcare professional time and use of consumables compared with the IS arm.
Conclusion:
The present analysis suggests that FCM is less costly and more effective than IS for the treatment of IDA subsequent to IBD in Spain and therefore was considered dominant.
Introduction
Approximately one in 1000 people in developed countries have inflammatory bowel disease (IBD), an umbrella term used to describe disorders that involve chronic inflammation of the digestive tract, the most common of which are Crohn’s disease (CD) and ulcerative colitis (UC). 1 A worldwide study estimated that the prevalence of IBD in Spain was 48.7 cases per 100,000 population in 2017, an increase of 12.3% since 1990. 2 The EpidemIBD study aims to provide a detailed nationwide analysis of the incidence and outcomes of IBD in Spain. 3 The full results of the EpidemIBD study are yet to report, but early analyses of the cohort found an annual incidence of iron deficiency anemia (IDA) of 14.3–16.0 cases per 100,000 population per year.4,5
IBD is characterized by chronic relapsing inflammation of the gastrointestinal tract, which can result in abdominal pain, diarrhea, rectal bleeding, abdominal abscesses, and fistulae. In addition, the condition is associated with extraintestinal complications that can involve a wide range of organ systems, including the musculoskeletal, dermatologic, hepatopancreatobiliary, ocular, renal, and pulmonary systems. 6 The most common extraintestinal complication of IBD is anemia, which can considerably impair functional status and health-related quality of life, as well as increasing hospitalization rates and healthcare costs. 7 A multicenter analysis of 1871 people with newly diagnosed IBD found that 49% of people with CD and 39% with UC experienced at least one instance of anemia in the first year after diagnosis. 8 Anemia in IBD is usually a result of a combination of IDA and anemia of inflammation (AI). Iron deficiency occurs in 36–90% of patients with anemia with IBD.9,10 Iron deficiency in IBD can result from a range of factors, including reduced dietary iron intake due to poor appetite, chronic intestinal bleeding, mucosal inflammation, and impairment of iron absorption (due to inflammation and the impact of drugs used to treat IBD). 11
Iron supplementation is recommended by the European Crohn’s and Colitis Organization (ECCO) in all patients with IBD-associated IDA. 12 The aim of treatment is to rebuild depleted iron stores and increase hemoglobin (Hb) levels to within normal ranges, or by ⩾2 g/dl within 4 weeks of treatment, depending on the severity of anemia. IDA can be treated with either oral or intravenous (IV) iron preparations.11,12 IV iron is recommended as a first-line treatment option in patients meeting any of the following criteria: clinically active IBD, previous intolerance to oral iron, hemoglobin below 10 g/dl, or requirement for erythropoiesis-stimulating agents. 12 A similar recommendation was made in the 2013 update of the Spanish Consensus Statement on alternatives to allogenic blood transfusion, with IV iron recommended for the treatment of IBD-associated IDA. 13 IV iron is safe, effective, and well tolerated for the treatment of IBD-associated IDA. 12 Oral iron may not be suitable for patients with IBD as unabsorbed iron can cause mucosal damage, generate reactive oxygen species causing oxidative stress in the intestinal tract, and alter the gut microbiota.14–17 However, new formulations containing ferric iron seem to overcome some of these drawbacks and may be suitable for patients with mild-to-moderate IDA and inactive IBD. 18
Ferric carboxymaltose (FCM) (Ferinject®) and iron sucrose (IS) (Venofer®), both manufactured by Vifor Pharma Group, and low-molecular-weight iron dextran (LMWID) (Cosmofer®), manufactured by Pharmacosmos, are the currently available IV iron formulations in Spain, with FCM recommended as the most effective intervention in clinical guidelines. 13 In Spain, IV iron is usually administered in an outpatient setting, most commonly either as a general hospital day case or as an IBD-specific unit day case. 19 While severe adverse events are uncommon, administration in a hospital setting with ready access to resuscitation equipment is recommended. 12 FCM and IS have differing dosing schedules, as described in the respective product labels. FCM can be administered in doses of up to 1000 mg iron in a single infusion, while IS dosage is restricted to 200 mg iron per infusion.20,21 The open-label, 12-week, head-to-head FERGIcor randomized controlled trial compared FCM with IS in patients with IDA and mild-to-moderate or quiescent IBD, and assessed the number of treatment responders at 12 weeks. 22 Response was defined as an Hb increase of ⩾2 g/dl in the primary endpoint, and, in line with ECCO guidelines, as normalization of Hb levels or an increase of ⩾2 g/dl in Hb levels in a secondary endpoint. For both endpoints, FCM was associated with a significantly greater percentage of patients achieving a response than IS.
In the current economic climate where there is increased pressure on public health services, it is imperative for healthcare policy makers to make decisions based not only on the efficacy and safety of interventions, but also based on their value for money, with the aim of maximizing the health of the population with finite monetary resources. Moreover, the SARS-CoV-2 pandemic has raised the need to reduce hospital visits to those that are strictly necessary and boosted the implementation of telemedicine and strategies that aim to reduce the time spent by patients in hospital settings. Cost-utility analyses are useful for such decisions, where costs and quality-adjusted life expectancy are estimated for the intervention and the comparator. An incremental cost-effectiveness ratio (ICER) is then calculated, describing the additional cost per each additional quality-adjusted life year (QALY) gained, using the following formula:
The ICER calculated can then be compared with a predefined willingness-to-pay threshold to assess whether the intervention can be considered good value for money. However, for this to be robust, evidence on the impact of treatments on quality of life is required. Where there is a paucity of data in this regard, cost-effectiveness analysis using ‘natural units’ can be considered as an alternative to cost-utility analysis. For example, cost-effectiveness analyses could estimate ICERs in terms of ‘additional cost per additional case detected’ when assessing screening programs or ‘additional cost per additional live birth’ when assessing fertility treatments.23,24 In treatment of IBD-associated IDA, cost-effectiveness analyses can assess the additional cost per additional responder. The ICER formula can be adapted as follows:
This method provides a framework for estimating the comparative cost-effectiveness of IDA treatments in IBD patients, thereby providing valuable information to healthcare decision makers. To date, no cost-effectiveness analyses of IV iron therapy in patients with IBD-associated IDA in Spain have been conducted. This study aimed to estimate the cost-effectiveness of FCM compared with IS in terms of additional cost per additional responder from a Spanish hospital payer perspective.
Methods
Modeling approach
A cost-effectiveness model was developed in Microsoft Excel to assess treatment costs and the proportion of patients with IBD-associated IDA achieving a response over one cycle of treatment (defined as a one-off treatment protocol to correct a patient’s current IDA) with FCM and IS from a Spanish hospital payer perspective. Response was defined in accordance with ECCO guidelines as normalization of Hb or an increase of ⩾2 g/dl in Hb levels. 12 The dichotomous variable of response versus no response was used as the measure of efficacy because the use of the continuous variable of mean increase in Hb levels may be influenced by baseline Hb levels.
The model analysis used a probabilistic approach, with characteristics sampled for each patient based on defined distributions to generate a cohort of patients with IBD-associated IDA. This resulted in a range of patients with different characteristics and therefore differing iron requirements (see ‘Cost inputs’ section). Whether a patient achieved a response (normalization of Hb or an increase of ⩾2 g/dl in Hb levels) was also sampled, reflecting that there is uncertainty around whether a patient responded to treatment or not (see ‘Efficacy inputs’ section). The model simulated characteristics for 1000 patients, as this produced stable results. No projections beyond a 1-year time horizon were made, and as such, discounting of outcomes was not required.
Efficacy inputs
Data on the efficacy of FCM and IS were taken from the FERGIcor randomized clinical trial, the results of which are described in Table 1. This study included 485 patients with IBD-associated IDA who received an iron infusion with FCM or IS. The study found that, in the full analysis set, 83.8% and 75.9% of patients were classified as responders after receiving treatment with FCM and IS, respectively. 22 In each iteration, whether a patient achieved a response was sampled based on standard deviations (SDs) of 2.4% for FCM and 2.9% for IS, derived assuming a binomial proportion confidence interval. Across all 1000 model iterations, the means and SDs of responders for FCM and IS were calculated, as was the difference between the treatment arms. This difference was used to calculate the number needed to treat, defined as the number of patients needed to be treated with FCM instead of IS to achieve one additional responder. Number needed to treat values are conventionally rounded up to the next integer value.
Outcomes of the FERGIcor trial.

Cost inputs
The cost of each IV iron treatment in patients with IBD-associated IDA was estimated by calculating the required iron dose based on the body weight and Hb levels of modeled patient. In each iteration, patient body weight and Hb inputs were sampled based on data from the FERGIcor trial. 22 The mean body weight was 68.4 kg (SD of 20.4 kg), and the mean Hb was 10.1 g/dl (SD of 2.2 g/dl). In the FCM arm, the iron dose was estimated using the simplified dosing table, taking into account patient body weight and baseline Hb (Table 2). In the IS arm, the iron dose was calculated using the Ganzoni formula, which takes into account patient body weight, Hb, target Hb, and iron stores, with target Hb and iron stores assumed to be 15 g/dl and 500 mg, respectively, in all patients. The formula is shown below:
Simplified dosing table.

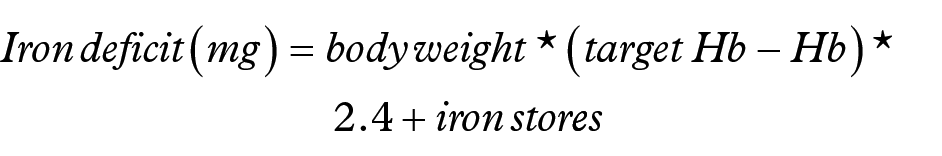
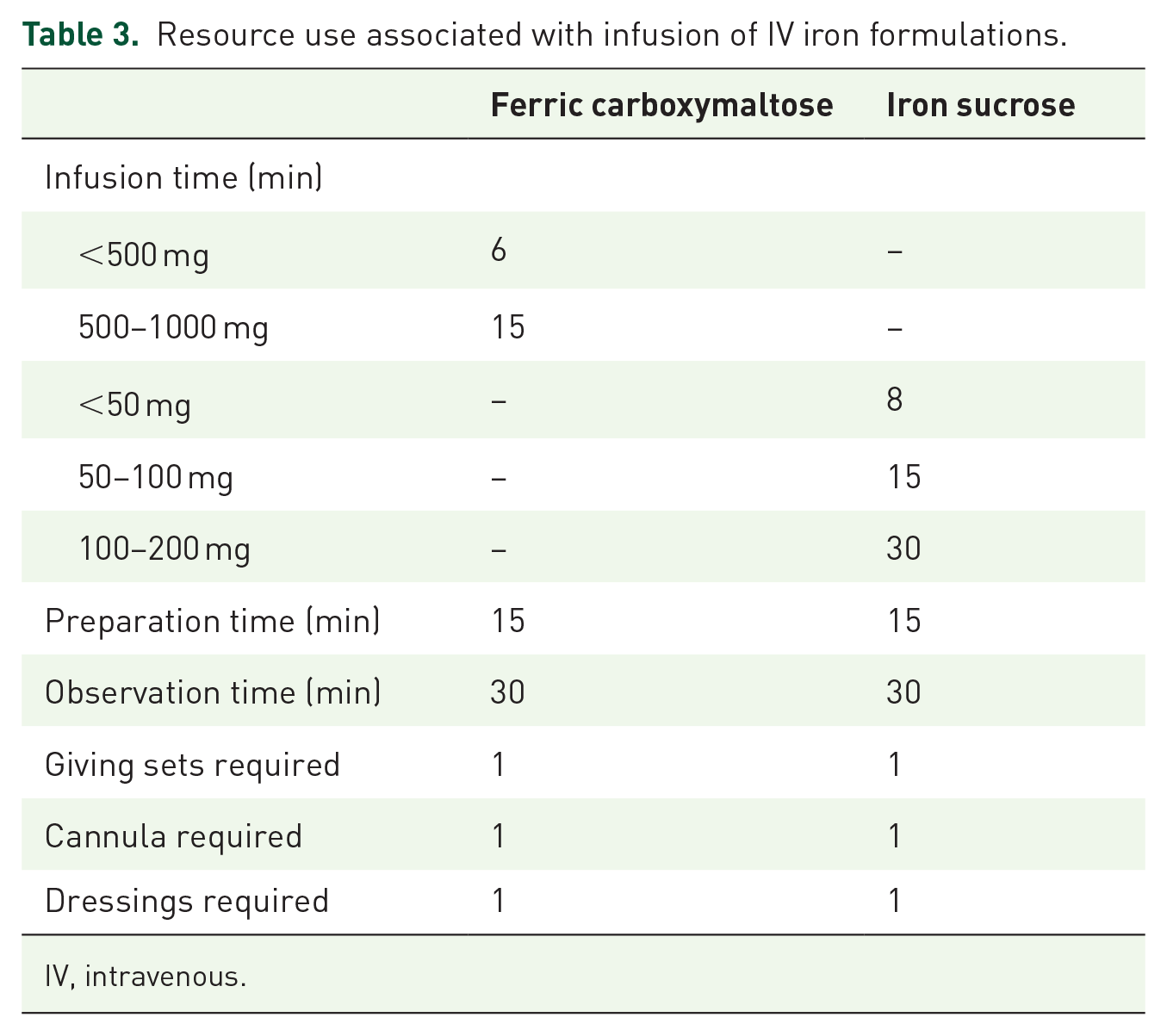
The use of differing approaches for calculating the required iron dose reflects both the product labels and the methods used in the FERGIcor trial.20–22 After the calculation of the iron dose, the number of infusions needed was estimated based on the dosing schedules described in the product labels for each intervention.20,21 A microcosting approach was used to calculate total costs, with the resource use associated with each iron infusion summed, capturing the iron formulation, healthcare professional time, and consumables (Table 3). A rebate of 7.5% was applied to the pharmacy cost of FCM, according to Royal Decree 8/2020. 25 This was not applied to the IS pharmacy cost, as generic formulations are available. All costs (Table 4) were estimated from a Spanish hospital payer perspective in 2021 Euros (EUR). It was assumed that wastage occurred when the contents of the IV iron vial were more than the required dose (i.e. the cost of the full vial was accrued). 25 The total cost of each course of iron treatment was summed as the cost of all required infusions.
Resource use associated with infusion of IV iron formulations.
IV, intravenous.
Pack contents and pack costs of IV iron formulations.

EUR, 2021 euros; IV, intravenous.
Costs extracted from https://botplusweb.portalfarma.com/.
Calculation of cost-effectiveness
The cost-effectiveness of FCM versus IS was expressed as the additional cost per additional responder. The mean cost of treatment with IS was subtracted from the mean cost of treatment with FCM, and the percentage of patients achieving a response with IS was subtracted from the percentage of patients achieving a response with FCM. The ICER was calculated by dividing the difference in costs by the difference in percentage of responders. In cost-effectiveness analyses, if an intervention shows greater clinical benefits and cost savings, it is considered ‘dominant’. In such cases, ICERs are not calculated as the decision-making process is clear.
Scenario analyses
A series of scenario analyses were performed to evaluate the impact of different methods of calculating iron requirements on cost-effectiveness results. In the base case analysis, the simplified dosing table was used in the FCM arm while the Ganzoni formula was used in the IS arm. In scenario analysis 1, the simplified dosing table was used in both arms, and the Ganzoni formula was applied in both arms in scenario analysis 2.
Sensitivity analyses
Sensitivity analyses were performed in addition to scenario analyses to assess the robustness of the model results and how varying model inputs affected calculated outcomes. To assess the uncertainty around the relative efficacy of the two medications, an analysis was performed using the odds ratio for achieving response with IS versus FCM reported in a network meta-analysis (NMA) to calculate the percentage of responders with IS. 26 This NMA captured four randomized controlled trials conducted in patients with IDA subsequent to IBD and found that the odds ratio for achieving a response with IS versus FCM was 0.70, with a 95% credible interval of 0.48–1.00.
Body weight and Hb are integral parameters when calculating iron doses using either the simplified dosing table or the Ganzoni formula. Therefore, sensitivity analyses were conducted with the mean body weight increased and decreased by 10 kg, to 78.4 kg and 58.4 kg, respectively, and with the mean Hb level at baseline increased and decreased by 1 g/dl, to 11.1 g/dl and 9.1 g/dl, respectively.
Reporting
The reporting of this study conforms to the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) statement. 27 The completed CHEERS checklist can be found in the supplementary information.
Results
Base case analysis
In the base case analysis, FCM was found to be the more effective treatment option for patients with IBD-associated IDA, with 83.8% (SD 2.4%) of patients achieving a response compared with 75.9% (SD 2.9%) of patients achieving a response with IS, a treatment difference of 7.9%. When expressed as number needed to treat, 13 patients would need to switch treatment from IS to FCM in order to achieve one additional responder (Table 5).
Results of the base case modeling analysis.
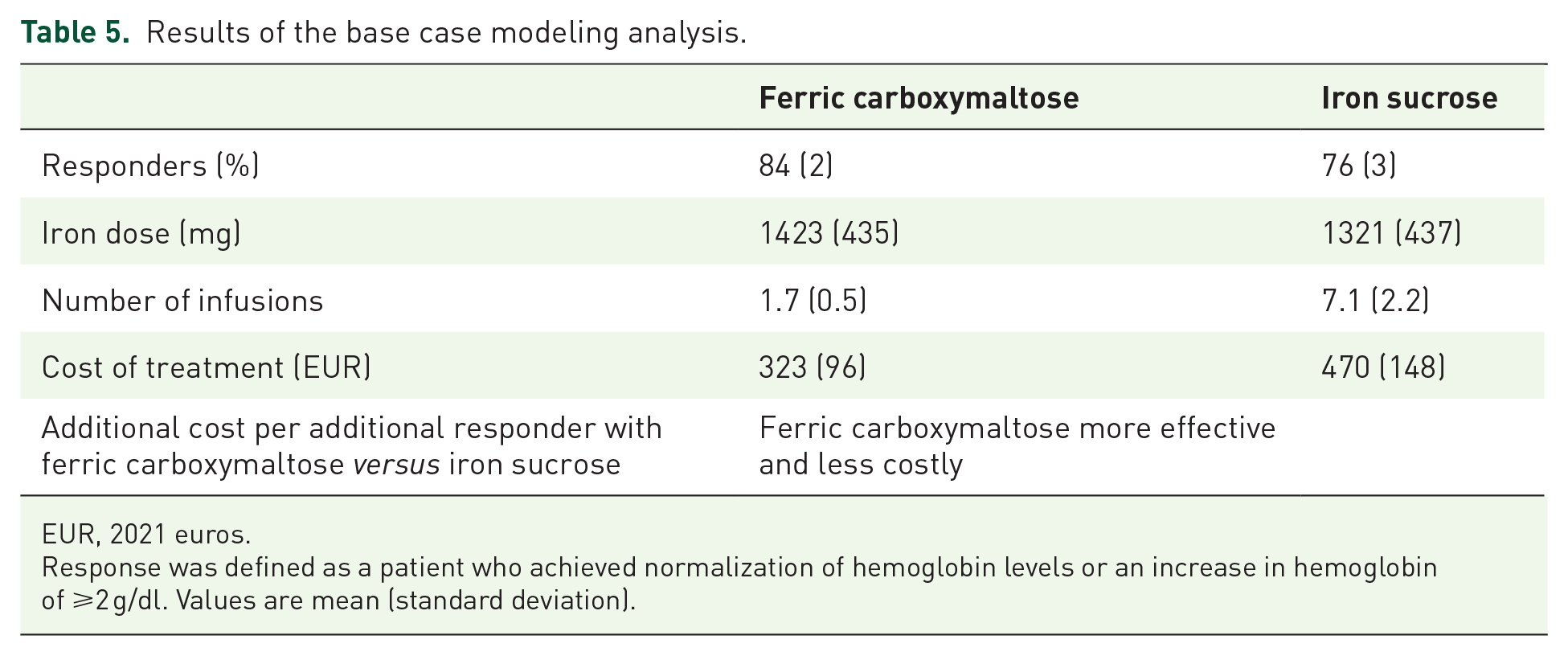
EUR, 2021 euros.
Response was defined as a patient who achieved normalization of hemoglobin levels or an increase in hemoglobin of ⩾2 g/dl. Values are mean (standard deviation).
A mean dose of 1423 mg (SD 435 mg) iron was required in the FCM arm, compared with a mean dose of 1321 mg (SD 437 mg) in the IS arm, resulting from the different methods used to calculate the iron requirement (Table 5). In order to deliver these doses, a mean of 1.7 (SD 0.5) infusions were required with FCM, compared with a mean of 7.1 (SD 2.2) infusions with IS. The mean cost of a course of treatment was EUR 323 (SD EUR 96) in the FCM group compared with EUR 470 (SD EUR 148) in the IS group, giving a cost saving of EUR 147 with FCM (Table 5). Cost savings with FCM were driven by the reduced number of infusions required, resulting in a reduced requirement for hospital-based healthcare professional time and use of consumables compared with the IS arm.
When cost-effectiveness was assessed, the analysis suggested that FCM was more effective and less costly than IS. Therefore, FCM was considered dominant versus IS, and no calculation of an ICER was required.
Scenario analyses
In scenario analysis 1, the simplified dosing table was used to estimate the required iron dose in both treatment groups, with the cost of treatment increased in the IS arm to EUR 485, while there was no change in the cost of treatment in the FCM arm (EUR 323) compared to the base case results. When the Ganzoni formula was used to calculate the iron dose in both arms, the cost of treatment in the FCM arm fell to EUR 312 and there was no change in the cost of treatment in the IS arm (EUR 470). Efficacy results were identical to the base case in both analyses, with FCM remaining dominant versus IS irrespective of the methods used to calculate the iron requirement in each arm (Table 6).
Scenario analysis results.

EUR, 2021 euros.
Response was defined as a patient who achieved normalization of hemoglobin levels or an increase in hemoglobin of ⩾2 g/dl.
Sensitivity analyses
The conclusions of the analysis remained unchanged when alternative model inputs were applied in sensitivity analyses (Table 7). When an NMA was used to calculate the efficacy in the IS arm, the percentage of responders with IS increased to 78.4%, with the percentage of responders with FCM remaining unchanged at 83.8%. Costs were unchanged from the base case analysis, with FCM considered dominant versus IS.
Sensitivity analysis results.

EUR, 2021 euros; NMA, network meta-analysis.
Response was defined as a patient who achieved normalization of hemoglobin levels or an increase in hemoglobin of ⩾2 g/dl.
When body weight was increased by 10 kg, costs of treatment increased in both arms as a result of the greater iron requirements. Cost savings with FCM increased to EUR 155. The converse effect was observed when the mean body weight was decreased by 10 kg, with a decrease in costs in both arms, and FCM associated with cost savings of EUR 133. Increasing the mean baseline Hb by 1 g/dl resulted in a reduction in iron requirements and, therefore, costs in both arms (EUR 293 in the FCM arm and EUR 408 in the IS arm). Conversely, decreasing the baseline Hb resulted in higher costs in both therapy arms due to increased iron requirements. Across all of the sensitivity analyses, FCM remained dominant versus IS.
Discussion
The present analysis examined the cost-effectiveness of two IV iron formulations, FCM and IS, for the treatment of IBD-associated IDA in the Spanish setting, using country-specific costs. The results suggested that FCM is the more effective treatment option, being associated with a higher proportion of patients achieving a response, defined by the ECCO criteria as normalization of Hb levels or an increase of ⩾2 g/dl in Hb levels, in comparison with IS. Although FCM was associated with a higher cost per mg of iron than IS (EUR 0.19 versus EUR 0.16), FCM was associated with lower overall treatment costs as doses of up to 1000 mg of iron can be administered in a single infusion, compared with a limit of 200 mg per infusion with IS. This resulted in fewer infusions with FCM, and consequent cost savings as a result of a reduced requirement for nursing time and use of fewer consumables associated with administration of infusions.
Due to the regular episodes of inflammatory flares and mucosal bleeding in patients with IBD-associated IDA, the choice of a cost-effective treatment is vital as patients require repeated courses of iron treatment over a long period. As the present analysis assessed the proportion of responders and corresponding costs over one cycle of treatment for IDA, the impact of retreatment was not captured. A need for retreatment usually results from failure to respond to treatment or the recurrence of IDA due to occurrence of IBD-related flares. 28 A pooled analysis of three randomized controlled trials found that the recurrence of anemia in patients with IBD treated with IV iron occurred after a median of 10 months. 28 Therefore, the mean cost saving per patient per cycle of treatment of EUR 147 is likely to result in annual cost savings of EUR 176 per patient (assuming that the average patient requires 1.2 treatment cycles per year).
As well as generating cost savings, the reduced number of infusions with FCM compared with IS results in important benefits for patients. With FCM, the required iron dose can be delivered in approximately 1 week, whereas around 3 weeks are required to deliver the full dose of IS.20,21 In Spain, IV iron formulations are usually administered in a hospital setting as a day case, and therefore, the reduced number of infusions with FCM has a further benefit for patients that was not captured in the present analysis, with fewer days spent in hospital, reduced travel time to and from appointments, and reduced lost workplace productivity. The reduced number of infusions with FCM may be particularly pertinent during the SARS‑CoV‑2 pandemic, providing a method for the Spanish National Health System (Sistema Nacional de Salud) to achieve the stated policy goal of reducing the time patients spend in hospital settings.
LMWID could not be included in the analysis due to the lack of direct (clinical trial) or indirect (NMA) evidence versus FCM and IS in patients with IDA subsequent to IBD. The dosing conditions of LMWID state that doses of up to 200 mg can be administered two or three times a week by intravenous drip infusion or intravenous injection or that the total dose can be infused over 4–6 h. 29 The need for repeated infusion or a long duration of infusion results in a significant requirement for healthcare professional time and therefore increased costs. Therefore, inclusion of LMWID as a comparator would be unlikely to change the conclusions of the analysis. Oral iron was also not included in the analysis. Oral iron is associated with relatively low pharmacy costs compared with IV iron, but may not be suitable for patients with IBD as unabsorbed iron can cause mucosal damage, generate reactive oxygen species causing oxidative stress in the intestinal tract, and alter the gut microbiota.14–17 Furthermore, in a randomized controlled trial comparing FCM with oral iron, FCM was associated with a faster response to treatment, and an NMA concluded that FCM was superior to oral iron based on response rates (defined as a patient who achieved normalization of hemoglobin levels or an increase in hemoglobin of ⩾2 g/dl).26,28 Newer oral iron formulations such as sucrosomial iron may reduce the gastrointestinal side effects associated with oral iron in the future, but this formulation has not yet been approved for use in Spain.30,31
The use of data from a randomized controlled trial represents a strength of the present cost-effectiveness analysis. The FERGIcor trial reported that FCM was associated with a significantly greater response rate than IS, and to test the impact of uncertainty around the key clinical outcome used to evaluate the effectiveness of the interventions, a sensitivity analysis was performed using the odds ratio for achieving a response with IS versus FCM reported in a previously published NMA to calculate efficacy in the IS arm. 26 The results were consistent with the base case findings, with FCM remaining more effective and less costly, suggesting that the conclusions of the present analysis are robust. However, the FERGIcor trial has a number of limitations. The trial enrolled patients with mild-to-moderate IBD or IBD in remission. Iron loss varies with IBD severity and therefore response to treatment may also vary. As such, there is some uncertainty around how outcomes would compare in patients with more severe disease. In addition, the study could now be considered old, as it was conducted between October 2008 and December 2009, and the follow-up period was limited to 12 weeks. However, non-anemic patients at the end of the FERGIcor study were invited to participate in the 8-month FERGImain study, in which FCM or placebo were administered if serum ferritin decreased to less than 100 µg/L. 32 Across the 8 months of follow-up, the probability of becoming anemic was significantly lower in the FCM arm compared with the placebo arm, demonstrating the efficacy of FCM over the long term. Moreover, the results of the FERGIcor study were supported by a further prospective, observational study performed in Spain, in which 42% of treatment courses resulted in a completed response (normalization of Hb or an increase of ⩾2 g/dl in Hb levels) after 2 weeks, increasing to 81% after 12 weeks. 33
The present analysis is the first to evaluate the cost-effectiveness of FCM versus IS specifically for the treatment of people with IDA subsequent to IBD in Spain, but a previously published cost-minimization analysis has assessed the relative costs of these interventions for treatment of IDA due to diverse etiologies. 34 In this analysis, chronic liver disease was the most common cause of IDA (49.5% of patients), followed by IBD (19.8%). The study found that FCM was associated with mean cost savings of EUR 67 per patient when all direct costs were included. The results of this previously published analysis concur with the results of the present analysis, in that FCM was associated with cost savings versus IS in both analyses. Cost savings with FCM were smaller in the cost-minimization analysis than in the present cost-effectiveness analysis, as patients received lower iron doses on average. Cost savings with FCM are greater in patients receiving higher doses of iron, as the number of infusions avoided with FCM compared with IS increases with the required dose.
Although a recent publication has estimated a willingness-to-pay threshold used in the Spanish healthcare payer system when assessing cost-utility analyses, there are no accepted willingness-to-pay thresholds for ‘cost per responder’ analyses. 35 This cost-effectiveness analysis is the first such study to compare FCM with IS in the Spanish setting, but cost per responder analyses are routinely used in the evaluation of IBD and other inflammatory diseases such as rheumatoid arthritis and psoriatic arthritis, and this can provide valuable context to the results of the present study. For example, a cost-effectiveness analysis comparing treatments for methotrexate-naïve psoriatic arthritis patients in the Spanish setting found that the incremental costs per responder (defined as a ⩾20% improvement in American College of Rheumatology component scores) versus placebo ranged from EUR 38 to EUR 17,377 for the three treatments evaluated over 16 weeks of treatment. 36 While the cost per responder that reflects good value for money can be debated, in the present analysis FCM was associated with increased efficacy and reduced costs, and, therefore, FCM is likely to be the optimal treatment option.
A limitation of the present cost-effectiveness analysis is that it did not capture the impact of adverse events. The most common adverse events reported in the FERGIcor trial were nasopharyngitis, worsening of UC, transient hyperferritinemia, and transient hypophosphatemia, none of which are likely to be clinically significant. 22 To further assess the adverse events associated with IV iron, data have been collected in two NMAs. Adverse events have been found to be low with all IV iron formulations, with infusion site reactions, transient increases in liver enzymes, headache, hyperferritinemia, and hypophosphatemia the most common. 26 A more recent NMA confirmed the low incidence of adverse events, with headache and transient hyperferritinemia the most common with both FCM and IS. 37 A few post-marketing reports have identified some patients with symptomatic hypophosphatemia requiring clinical intervention following administration of FCM, in those with risk factors for developing low serum phosphate levels.38,39 IBD patients may have absorption disorders, vitamin deficiency (such as vitamin D), be receiving co-medications, and experience recurrent bleeding episodes, all of which may prompt them to have lower phosphate baseline values, independent of intravenous iron treatment.40,41 In patients at risk for hypophosphatemia, repeated parenteral iron treatment, especially with FCM, needs to be adapted to reflect the underlying disease and its inherent risk for hypophosphatemia, other hypophosphatemia-inducing drugs, and the time necessary to restitute iron stores. Future economic evaluations should investigate how the costs and outcomes could be impacted by adverse events. In addition, the analysis only captured one cycle of treatment due to the limitations of the trial data, with extrapolation based on retreatment frequency required to estimate annual costs. It should also be noted that the conducted analysis was specific to Spain, and therefore, the results have limited applicability to other country settings. However, the conclusions are likely to hold across European countries due to similar patterns in medication and healthcare professional costs. 42
Conclusion
To the best of our knowledge, this economic evaluation is the first study to compare the cost-effectiveness of FCM and IS for patients with IBD-associated IDA in the Spanish setting. The results suggest that FCM is likely to be more effective, with a higher percentage of patients achieving a response, and less costly than IS, and therefore was considered dominant. Use of FCM to treat IBD-associated IDA is likely to be an appropriate use of scarce healthcare resources in Spain.
Footnotes
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: F.A.-A. has served as speaker, consultant, and advisory member for or has received research funding from MSD, Abbvie, Pfizer, Kern Pharma, Takeda, Janssen, Ferring, Faes Farma, Shire Pharmaceuticals, Tillotts Pharma, Chiesi, and Dr. Falk. F.B. has served as a speaker, a consultant, and advisory member for or has received research funding from MSD, Abbvie, Takeda, Janssen, Pfizer, Biogen, Amgen, Ferring, Faes Farma, Tillotts Pharma, Falk Pharma, Chiesi, Gebro Pharma, and Vifor Pharma. J.B.-B. has participated in educational activities, research projects, scientific meetings, or advisory boards sponsored by Abbvie, Janssen, Sandoz, Vifor Pharma, and Pfizer. E.D. has served as a speaker, or has received research or education funding or advisory fees from AbbVie, Adacyte Therapeutics, Biogen, Celltrion, Gilead, Janssen, Kern Pharma, MSD, Pfizer, Roche, Samsung, Takeda, Tillots, and Thermofisher. B.S. has received support for conference attendance, speaker fees, research support and consulting fees of Abvvie, Dr Falk, FAES Pharma, Ferring, Jannsen, MSD, Pfizer, Shire, and Takeda. J.M.H. has participated in educational activities, research projects, scientific meetings, or advisory boards sponsored by Merck Sharp Dohme (MSD), Ferring, Abbvie, Janssen, Biogen, Sandoz, Kern Pharma, Faes Farma, Vifor Pharma, and Takeda. A.R.d.A. is an employee of Vifor Pharma Group. W.J.V. and B.H. are employees of Ossian Health Economics and Communications, which received consulting fees from Vifor Pharma Group to support preparation of the analysis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by funding from Vifor Pharm Group.
Compliance with ethics guidelines
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.


