Abstract
Introduction:
The use of proton-pump inhibitors (PPI) may be associated with an increased risk of gastric cancer (GC).
Objective:
To review and meta-analyse available literature investigating the association between PPI use and GC risk.
Methods:
Two independent reviewers systematically searched Ovid MEDLINE, EMBASE, and Cochrane Library (inception to July 2020) for case-control and cohort studies assessing the association between PPI use and GC according to a predefined protocol in PROSPERO (CRD42018102536). Reviewers independently assessed study quality, extracted data, and meta-analysed available and newly calculated odds ratios (ORs) using a random-effects model, and stratified for GC site (cardia versus non-cardia) and PPI duration (<1 year, 1–3 years, >3 years).
Results:
We screened 2,396 records and included five retrospective cohort and eight case-control studies comprising 1,662,881 individuals in our meta-analysis. In random-effect models, we found an increased GC risk in PPI users [OR: 1.94, 95% confidence interval (95% CI): 1.47–2.56] with high statistical heterogeneity (I2 = 82%) and overall moderate risk of bias. Stratified analyses indicated a significant risk increase in non-cardia (OR: 2.20, 95% CI: 1.44–3.36, I2 = 77%) with a similar non-significant trend in cardia regions (OR: 1.77, 95% CI: 0.72–4.36, I2 = 66%). There was no GC increase with longer durations of PPI exposure (<1 year: OR: 2.29, 95% CI: 2.13–2.47, I2 = 0%; 1–3 years: OR: 1.46, 95% CI: 0.53–4.01, I2 = 35%; >3 years: OR: 2.08, 95% CI: 0.56–7.77, I2 = 61%).
Conclusion:
We found a twofold increased GC risk among PPI users, but this association does not confirm causation and studies are highly heterogeneous. PPI should only be prescribed when strictly indicated.
Introduction
Gastric cancer (GC) is one of the most frequent cancers worldwide with more than 1 million new patients per year, constituting 5.7% of all cancer cases worldwide. 1 Mortality of GC remains high with >780,000 deaths annually, corresponding to 8.2% of all cancer-related deaths. 1 Common GC risk factors are manifold and include environmental factors such as Helicobacter pylori (HP) infections, dietary habits, smoking and obesity, precursor lesions such as atrophic gastritis and intestinal metaplasia, and host-related factors.2–5 Nevertheless, there is a decreasing trend in lifetime GC incidence possibly related to a lower burden of HP infection. 6
Proton-pump inhibitors (PPI) are one of the most frequently prescribed drugs worldwide 7 and considered relatively safe. However, previous meta-analyses8–13 of mostly large retrospective cohort and case-control studies found a significant association between PPI use and an up to 2.5-fold increased GC risk across very heterogeneous study populations and PPI indications. Despite all potential limitations of retrospective studies, there is an urgent need for further investigation. Due to new evidence from Sweden, 14 Taiwan, 15 the United States, 16 and the United Kingdom 17 since the last meta-analyses, we performed an updated systematic literature review and meta-analysis.
Materials and methods
Data sources and search strategy
We conducted this systematic literature review according to a predefined protocol in PROSPERO, an international prospective register of systematic reviews (CRD42018102536) and adhered to the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA statement). 18
We developed search strategies for EMBASE, Ovid MedLine, and the Cochrane Library (Supplemental Appendix Figure 1), searched all databases from inception until 18 July 2020, and screened bibliographies of included records for additional publications.
Study selection, quality assessment, and data extraction
We screened titles and abstracts according to predefined eligibility criteria. We included cohort and case-control studies in any language assessing the association between PPI use and GC risk comprising individuals with a considerable PPI use and a control group with no or only rare PPI use. Moreover, we extracted burden of PPI exposure by any measure of duration, frequency, and dosage.
The primary outcome of our study was the risk of incident GC at any location. Whenever possible, we extracted adjusted odds ratio (OR) and respective 95% confidence interval (CI), as adjusted OR was the most frequent effect size in our included studies. In case only other point estimates, such as HR, standardized incidence ratios (SIRs), and RR were available, we extracted all necessary raw data to calculate the unadjusted OR. In case of missing or incomplete information, we contacted authors for additional information on the exposure and outcome definitions, distribution of exposure, GC cases, and controls. Only one potentially eligible study could not be considered in our meta-analysis after full-text review due to missing raw data to calculate OR and no response after contacting the corresponding author. 19
We excluded studies on other malignant gastric tumours (i.e. mucosa-associated lymphatic tissue lymphoma, gastrointestinal stromal, and neuroendocrine tumours) or gastric manifestations from other malignancies such as hematologic neoplasms, as well as those with exclusively paediatric populations (<18 years). Finally, cross-sectional studies, case reports, and pooled estimates from meta-analyses were excluded; however, suitable individual studies from meta-analyses were considered.
Two independent reviewers (D.S., D.G. or B.M.) screened titles and abstracts. Consensus for emerging discrepancies between reviewers was achieved after discussion with a senior author (B.M.). The inter-observer agreement regarding study inclusion was good for the screening of abstracts and titles (kappa = 0.87) and excellent for full-text articles (kappa = 1). A reviewer (D.S.) extracted data on a standardized form followed by verification by the other reviewers (N.K., B.M.). Two independent reviewers (D.S., B.M.) assessed study quality using a tool for assessing risk of bias in non-randomized studies of interventions (ROBINS-I). 20 Different bias domains (confounding, selection of study participants, classification of interventions, deviation from intended interventions, missing data, outcome measurements, selection of reported results) were analysed for every individual study. The risk of bias for every domain and the whole study was estimated according to predefined questions and classified as ‘low’, ‘moderate’, ‘serious’, and ‘critical’ according to published instructions in the Cochrane Handbook for Systematic Reviews of Interventions version 6.2. 21
Ethical considerations
This meta-analysis included only anonymised patient data upon informed written consent from published cohort and case-control studies approved by their respective institutional review boards complying with the 1975 Declaration of Helsinki.
Statistical approach
All calculations were performed using R version 4.0.4 and the package ‘meta’ version 4.19-0. 22 For our meta-analysis, we extracted adjusted OR and respective 95% CI and calculated new unadjusted OR in case of different point estimates (see above). Consequently, we calculated pooled OR by random-effect models using inverse variance weights and the Hartung–Knapp–Sidik–Jonkman (HKSJ) method.23–26 As an additional approach, we calculated pooled OR from provided raw binary outcome data by random-effect models using the Mantel–Haenszel and HKSJ methods. Heterogeneity between studies was assessed by means of I2 statistics. We chose a random-effects approach due to an expected high heterogeneity and disproportional weight of a single study with a very high number of participants 27 in an experimental fixed effects model.
In addition, we performed stratified analysis for study design (case-control versus retrospective cohort studies), GC site (cardia/proximal GC versus non-cardia/distal GC), and duration of PPI intake (<1 year, 1–3 years, >3 years).
Moreover, we calculated the pooled prevalence of GC among PPI users and non-users in five retrospective cohort studies.17,28–31
Finally, we assessed the risk of publication bias using Egger’s test. 32
Results
General characteristics of included studies
We identified 2,396 records during initial screening. After exclusion of 397 duplicates, 1,966 articles based on titles and abstracts, and 19 articles after full-text review, 14 studies remained for the qualitative synthesis (Supplemental Appendix Figure 2). Baseline characteristics of the individual studies are presented in Table 1. One large Danish case-control study 19 did not provide sufficient information to be included in our meta-analysis; however, it was considered in the qualitative synthesis.
Main characteristics of the included case-control and cohort studies.

EGD, esophago-duodeno-gastroscopy; GC, gastric cancer; GORD, gastro-oesophageal reflux disease; HP, Helicobacter pylori; HR, hazard ratio; H2RA, histamine 2 receptor antagonist; IRR, incidence rate ratio; n, number; N/A, not available or not applicable; OR, odds ratio; PPI, proton-pump inhibitors; ROBINS-I, risk of bias in non-randomized studies of interventions; SIR, standard incidence ratio.
Only numbers for both GC and oesophageal adenocarcinoma provided; no separate calculation for GC cases possible.
Not available in original publication. Calculated from Table 2: Addition of unexposed and PPI (2 + prescr.) groups both for person-years and GC cases.
Numbers available for PPI users, but not for control group in original publication.
Multivariate analyses without trimming.
No separate information on PPI and H2RA users available.
Our final meta-analysis summarizes eight case-control15–17,27,33–35,37 and five retrospective cohort studies17,28–31 with an overall population of 1,662,881 individuals from Europe (Denmark, Sweden, United Kingdom), Canada, United States, and Asia (Hongkong, Japan, Taiwan).
Three studies17,19,28 assessed GC risk in relation to the number of PPI prescriptions, three studies according to the number of defined daily doses (DDDs)16,17,34; five studies reported the duration of PPI use,14,16,17,29,31,33 among those one study with additional information on PPI frequency. 31 The five remaining studies did not provide detailed analyses either on PPI frequency, dosage, or duration.15,27,30,34,37
In two studies,14,28,29 there was no information on the proportion of cases and controls in unexposed individuals. Thus, we re-defined the PPI-unexposed group as individuals on histamine 2 receptor blocker (H2RA). In the Swedish cohort, updated PPI exposure and GC data 14 were combined with the H2RA population in the original paper 29 for the main analysis and the stratified analyses by PPI duration. Eight studies14,15,19,28,29,31,33,35,34 defined GC by localization according to International Classification of Disease (ICD) codes, three studies29,33,37 provided information on GC histology; however, one of those without ICD codes. 37 Three studies did not specify the diagnostic criteria for GC.27,30,36
Results from the quality assessment
The methodological quality of studies using the ROBINS-I tool 20 varied with an overall low risk of bias in 1 study,14,29 moderate risk in 11 studies15–17,27,28,31,33–35,37 and severe risk in 1 study 30 (Supplemental Appendix Table 1). Egger’s test 32 showed a significant risk of publication bias (intercept: 3.98, 95% CI: 2.46–5.50, p < 0.01).
Results from quantitative analyses
Association between GC and PPI use
Our meta-analysis of all 13 studies 15 –17,27–31,33–35,37 indicated an increased GC risk in PPI users (OR: 1.94, 95% CI: 1.47–2.56, I2 = 82%, p < 0.01) in random-effect models (Figure 1). The pooled GC prevalence of five retrospective cohort studies17,28– 31 was 0.6% (95% CI: 0.1–4%) among PPI users and 0.2% (95% CI: 0.04–1%) among non-users, both with high statistical heterogeneity (I2 = 99%, p < 0.01, Supplemental Appendix Figure 4). On a study level, Niikura et al. 30 report a GC prevalence rate of 11% among PPI users, which is 22-fold higher than in another Asian collective from Hong Kong 31 (0.5%). Our secondary analysis using newly calculated unadjusted OR from raw binary outcome data from all 13 studies 15 –17,27–31,33–35,37 yielded similar results (OR: 2.05, 95% CI: 1.54–2.64, I2 = 91%, p < 0.01). Stratified analyses according to study design showed a higher GC risk estimate in the five included retrospective cohort studies17,28– 31 (OR: 2.76, 95% CI: 1.56–4.88, I2 = 63%, p = 0.03) than in the eight included case-control studies 15 –17,27,33–35,37 (OR: 1.59, 95% CI: 1.23–2.05, I2 = 81%, p < 0.01, Supplemental Appendix Figure 3).

Association between PPI use and GC risk.
Association between GC and PPI use according to GC site
Seven out of 13 studies contributed to a stratified meta-analysis according to GC site.17,27,29,33–35,37 There was a significant risk increase in non-cardia GC (OR: 2.20, 95% CI: 1.44–3.36, I2 = 77%) with a non-significant trend towards an increased risk in the cardia (OR: 1.77, 95% CI: 0.72–4.36, I2 = 66%, Figure 2).
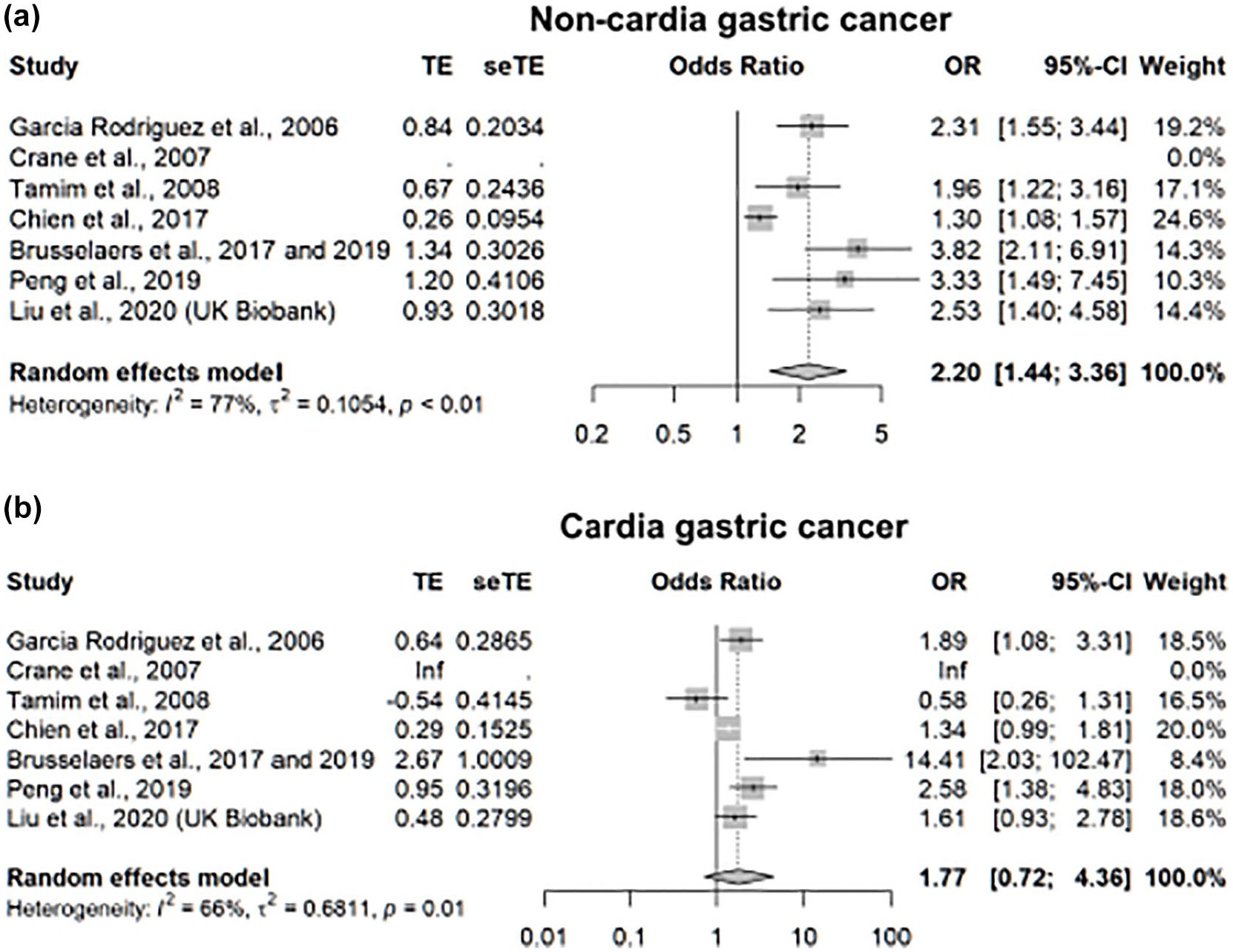
Association between PPI use and GC risk stratified by GC site.
Association between GC and PPI use according to PPI duration
Overall, 3 out of 13 studies14,29,31,33 could be meta-analysed for the duration of PPI intake. There was no trend for a duration-dependent effect of PPI use (<1 year: OR: 2.29, 95% CI: 2.13–2.47, I2 = 0%, p = 0.97; 1–3 years: OR: 1.46, 95% CI: 0.53–4.01, I2 = 35%, p = 0.21; >3 years: OR: 2.08, 95% CI: 0.56–7.77, I2 = 61%, p = 0.08; Figure 3).

Association between PPI use and GC risk stratified by duration of PPI use.
Results from qualitative analyses
GC risk according to location: non-cardia versus cardia
A meta-analysis regarding location-dependent GC risk was not possible, as most studies did not provide stratified information regarding GC location. Two additional studies could not contribute to the stratified meta-analysis according to GC site due to missing information.19,31 While a retrospective cohort study from Hong Kong 31 found a significant GC risk increase for PPI users in the non-cardia (HR: 2.56, 95% CI: 1.46–4.49), but not cardia region (HR: 1.24, 95% CI: 0.35–4.34), a large Danish case-control study 19 obtained a significant risk increase in both regions (non-cardia GC: HR: 4.68, 95% CI: 4.14–5.29; cardia GC: HR: 2.51, 95% CI: 2.26–2.79).
GC risk according to age and sex
Two studies provided analyses accounting for age and sex.15,29 Stratified meta-analyses according to age and sex were not feasible due to missing data in the Taiwanese study, 15 which precludes the calculation of an OR with a meta-analysis. In a large retrospective Swedish cohort, 29 the highest relative GC risk was found in PPI users < 40 years (SIR: 22.76, 95% CI: 15.94–31.52), whereas relative GC risk was lowest in those >70 years (SIR: 2.76, 95% CI: 2.61–2.92). The relative GC risk was significantly higher in male (SIR: 3.65, 95% CI: 3.45–3.85) than in female PPI users (SIR: 3.07, 95% CI: 2.87–3.28). In contrast, a Taiwanese case-control study 15 found no inter-sex difference in GC risk among PPI users (OR: 0.98, 95% CI: 0.77–1.25) and no GC risk increase with increasing life years (OR: 1.00, 95% CI: 0.99–1.01). In the analysis of two British databases, 17 there was no significantly increased GC risk in males and females after full adjustment in the United Kingdom Biobank (male: HR: 1.14, 95% CI: 0.70–1.87; female: HR: 1.73, 95% CI: 0.86–3.45), whereas the Primary Care Clinical Informatics Unit (PCCIU) database showed a significantly increased GC risk in women, but not men (male: OR: 1.26, 95% CI: 0.97–1.62; female: OR: 1.84, 95% CI: 1.38–2.47).
GC risk according to frequency and intensity of PPI use
No meta-analysis was feasible for frequency and intensity of PPI use due to very heterogeneous quantifications and stratifications of PPI intake ranging from number of prescriptions,17,19,28 DDD16,17,34 to insufficient or no definition.15,27,30,34,37 For instance, Liu et al. 17 used other subgroup cut-offs for daily dosages per year in the PCCI cohort (1–183, 184–365, 366–1095, ⩾1096) than Tamim et al. 34 (0–41, 42–119, 120–479, >480 daily dosages per year). Lee et al. 36 provided only mean daily dosages (0.75, 0.76–1.49, >1.5) without referring to a yearly dosage, so that a valid meta-analysis of these heterogeneous dosage units was not feasible. In contrast to the meta-analysis by Jiang et al., 10 we did not include Poulsen et al. 28 because the authors provided stratified analyses for the length of follow-up (with different time frames), but not for the duration of PPI intake.
Nevertheless, some individual studies deserve special attention. Cheung et al. 31 stratified for PPI frequency in context with duration. Only daily PPI intake was significantly associated with an increased GC risk during a prescription period of at least 1 year (HR: 5.04, 95% CI: 1.23–20.61), 2 years (HR: 6.65, 95% CI: 1.62–27.26), and 3 years (HR: 8.34, 95% CI: 2.02–34.41). On the contrary, weekly and less than daily PPI intake did not significantly alter GC risk.
The retrospective cohort study by Poulsen et al. 28 found a trend towards a slightly increased GC risk with rising number of prescriptions; however, this effect was significant only in the subgroup with at least 15 prescriptions (incidence rate ratio: 2.1, 95% CI: 1.0–4.7). Furthermore, a Danish case-control study 19 found a gradually increasing GC risk with rising number of prescriptions (1–4 prescriptions: HR: 1.91, 95% CI: 1.64–2.22; ⩾15 prescriptions: HR: 3.56, 95% CI: 2.82–4.46) in the cardia but not in other regions. In the PCCIU database, Liu et al. 17 found the highest GC risk increase in PPI users with the lowest DDD and numbers of PPI prescriptions (1–183 DDD versus non-users: OR: 1.84, 95% CI: 1.43–2.38; 1–6 prescriptions: OR: 1.85, 95% CI: 1.44–2.37), whereas this effect mitigated to the highest DDD and numbers of PPI prescriptions (⩾1096 DDD versus non-users: OR: 1.30, 95% CI: 0.91–1.85; ⩾ 36 prescriptions versus non-users: OR: 0.97, 95% CI: 0.64–1.47). This trend was significant for DDD (p for trend = 0.02), but not for the number of prescriptions (p for trend = 0.07).
GC risk according to HP infection status
The literature summarized in our review is highly heterogeneous regarding the consideration of HP infection status; hence, no meta-analysis was possible. Brusselaers et al. 29 showed a substantially increased relative GC risk in PPI users with HP infection (SIR: 9.76, 95% CI: 8.87–10.71) compared with other PPI indications (SIR: 3.55, 95% CI: 3.27–3.86). Two studies30,31 specifically included individuals after HP eradication; therefore, a direct intra-study assessment of HP effects was impossible. However, there was no evidence of stronger PPI effects than in the other studies. Wennerstrom et al. 19 conducted a sensitivity analysis showing no difference in effect size upon censoring patients at the time of HP eradication. Poulsen et al. 28 incorporated HP infections as a confounder in their multivariate analysis, but the change in effect size could not be compared as unadjusted results were lacking.
Discussion
In our meta-analysis, we found a significant twofold increased overall risk of GC of PPI users compared with controls. Stratification according to GC site indicated a significantly increased non-cardia GC risk while there was a non-significant trend towards an increased cardia GC risk. Because the adenocarcinoma of the cardia and distal oesophagus are sometimes difficult to discriminate due to the overlapping pattern of local expansion, an accurate classification can be difficult also in epidemiologic studies.38,39 Due to different risk factors for adenocarcinoma at proximally or distally to oesophago-gastric junction, 40 an accurate identification of risk factors at this site is challenging. In our meta-analysis, the true effect of PPI to the carcinogenesis of cardiac GC may be weakened by the treatment of gastro-oesophageal reflux, which represents a common risk factor for distal oesophageal adenocarcinoma. However, there was no clear duration-dependent risk increase in PPI users with discrepant results in individual studies.
Our meta-analysis includes global evidence from five retrospective cohorts/registries and eight case-control studies with an overall population of 1,662,881 individuals. Most studies14–17,19,27–29,31,33–35 used large populations from national and regional databases in Asia, Europe, and North America, which increases generalizability. In comparison with the latest meta-analyses10–13 including up to eight studies on the association between PPI use and GC risk, we could include five additional studies and more than 500,000 additional patients. This enabled more robust stratified analyses according to GC site and standardized time frames for PPI duration compared with previous meta-analyses. Two recent meta-analyses deserve special mention. Lin et al. 12 included only a fraction of our studies (eight instead of 13 studies in our meta-analysis) with a literature search in 2018. The same applies to the stratified analysis according to GC site where they meta-analysed five studies compared with seven studies in our updated meta-analysis. In addition, this meta-analysis has some methodological shortcomings: Stratified analyses according to PPI duration were not homogeneous in terms of PPI duration. While we used standardized cut-offs for PPI duration (<1 year, 1–3 years, ⩾3 years), Lin et al. mixed different PPI durations in various categories enabling for the integration of Poulsen et al. 28 and Lai et al. 15 in some but not all subgroups. However, this approach is potentially misleading due to the missing standardization of PPI durations. Furthermore, only the subgroups with a PPI use ⩾3 years and ⩾1 year were considered from the retrospective cohort study by Cheung et al. 31 so that one of the largest retrospective cohort studies was only partly considered in this stratified analysis. Finally, they used apparently inconsistent inclusion criteria for HP subgroups in their stratified meta-analysis for HP status with current and past HP infection: while the authors classified two entire cohorts with probable – but not confirmed – HP eradication as ‘past HP infection’ groups,30,31 only a fraction of the study population was analysed in the other studies,15,29 which markedly enhances the risk of misclassification and selective reporting which impacts the generalizability of the data.
The other recent meta-analysis by Song et al. 13 meta-analysed only a fraction of our included studies (5 versus 13 studies). In contrast to our meta-analysis on PPI exposure, the main exposure was acid-suppressive drugs and only one subgroup analysis of five studies for PPI use was available. Therefore, the authors were not able to present detailed stratified analysis on cardia and non-cardia GC and duration-dependent GC risk for PPI users. 13
Stratified analyses according to study design indicated an increased GC risk not only in case-control studies but also in cohort studies, which are typically less prone to selection and recall bias. Our meta-analysis thus reflects the growing body of evidence for an association of PPI usage with GC in observational studies in highly different settings.
Addressing the risk of reverse causation, 11 out of 13 studies incorporated a lag time for GC diagnosis removing PPI usage limited to the previous 631,34 or 12 months,14,15,17,19,28,29,33,35 2 years,16,17 3 years,17,27 or 4 and 5 years 17 before GC diagnosis. Furthermore, 10 out of 13 studies excluded patients with a history of any cancer19,28,29,33–35 or history of GC.15–17,31
Nevertheless, this work encountered several limitations due to very heterogeneous original studies concerning study design, quality, cases and controls definitions, PPI indications, quantification of PPI exposure, and effect sizes, which may also result in highly heterogeneous pooled OR and prevalence in the included studies. First, some original publications did not provide risk estimates or did only report incomplete information on the exposure and outcome distribution within the study population. We attempted to overcome this issue by calculating new unadjusted OR based on the published figures, and by contacting the corresponding authors for additional information. Nevertheless, unadjusted point estimates exhibit the risk of confounding and should be interpreted cautiously. However, in a sensitivity analysis comparing newly calculated OR with remaining studies, we found highly similar results. Second, in two studies,14,28,29 we could not retrieve information on the unexposed group so that we used H2RA users as such. In a sensitivity analysis excluding these studies, however, the results remained unaffected. Third, although all studies indicate an increased GC risk among PPI users compared with controls, its extent varies, reflected by the high statistical heterogeneity in our and previous meta-analyses. Leave-one-out analyses, however, revealed no disproportional influence of any single study, and Graphical Display of Study Heterogeneity plots 41 demonstrated no overt sub-clusters.
Fourth, we could only include retrospective cohort and case-control studies, which are subject to residual confounding. Fifth, immortal time bias 42 leading to a misclassification of PPI users and non-users remains an issue in 11 out of 13 studies; however, 2 studies accounting for this issue still found a significant association of PPI exposure and GC risk.14,15 Sixth, a publication bias cannot be excluded based on the Eggers test.
Seventh, missing data for important risk factors in the individual studies precluded adjustments for common GC risk factors, which may mitigate this association. Despite representing one of the most important confounders in the association between PPI and GC risk, 43 most studies did either not adjust for HP infection status17,33,34,37 or analysed study populations after documented HP therapy (however, without proven eradication).30,31 Therefore, there might be synergistic carcinogenic effect by HP infection and PPI through hypergastrinaemia44,45 or early dyspeptic symptoms related to gastritis, precancerous lesions such as intestinal metaplasia, dysplasia, and early GC might favour regular PPI use. This may also explain the markedly increased GC risk in individuals with PPI use <1 year without further relevant risk increase in subgroups with longer PPI use. However, considering the decreasing prevalence of HP in Western countries, confounding by HP is unlikely to be the main explanation of the association of PPI usage and GC risk. Eighth, most studies failed to control for other common GC risk factors,2–5 such as smoking15,19,27,29,30,34,35,37 and obesity,15,19,27–30,34,35,37, and no study adjusted for dietary salt and nitroso compound intake.3,5,14,17,27–30,33–37
Another inherent limitation of most included studies is the missing information on precancerous lesions such as intestinal dysplasia and metaplasia 5 in most included studies,15,17,19,27,28,31,33,34,37 which may both favour regular PPI use and the development of GC. This could lead to an overestimation of the effect due to residual confounding. Finally, no universally accepted definition of long-term PPI usage is available and definitions of long-term PPI use were missing in 10 included studies14,17,27–30,34–37 and highly heterogeneous in the remaining studies,3,33,35 so that we strictly avoided this term in our meta-analysis. However, our data did not show a duration-dependent risk increase in GC risk among PPI users.
In the randomized controlled COMPASS trial, Moayyedi et al. 46 found no statistically significant difference in the risk for gastrointestinal tumours between the pantoprazole and placebo group (HR: 1.04, 95% CI: 0.77–1.40); however, the study suffers from several methodological shortcomings and conflicts of interest in the authorship, as previously discussed. 47 For instance, it was underpowered for safety outcomes, comprised a highly selected study population, and did not provide information on drop-out rates as well as separate analyses on GC risk. In addition, two of three study arms included daily aspirin intake by study design, which in combination with regular PPI intake was associated with a considerably decreased GC risk increase compared with PPI alone in retrospective analyses.48,49
Even though our meta-analysis demonstrates a clear association between PPI use and GC risk, causality cannot be proven. Large randomized controlled trials in this field are ethically challenging and not feasible. However, this meta-analysis includes evidence from large registry studies with a retrospective cohort design, but with prospective data collection, therefore the highest level of evidence available by now. Yet, future research should focus on a comprehensive and accurate prospective documentation of GC risk factors, endoscopic/histopathological findings, co-medication, and co-treatments, which would enable application of sophisticated causal inference techniques 50 as used in other studies on risks of PPI. 51
Conclusion
Our meta-analysis found an almost twofold increased GC risk among PPI users compared with controls. Despite a clear association between PPI exposure and GC risk, causation cannot be proven. Moreover, there was substantial heterogeneity in study designs, populations, definitions of exposure and outcome, and residual confounding as well as a potential publication bias remain of concern. Indications for PPI treatment should be verified, PPI use avoided, and non-indicated PPI unprescribed whenever possible.
Supplemental Material
sj-docx-1-tag-10.1177_17562848211051463 – Supplemental material for Association between proton-pump inhibitors and the risk of gastric cancer: a systematic review with meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848211051463 for Association between proton-pump inhibitors and the risk of gastric cancer: a systematic review with meta-analysis by Daniel Segna, Nele Brusselaers, Damian Glaus, Niklas Krupka and Benjamin Misselwitz in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
Niklas Krupka and Benjamin Misselwitz contributed equally to this work and share last authorship.
Author contributions
D.S.,B.M., and N.B. designed the concept of the meta-analysis. D.S., D.G., and B.M. designed and performed the literature research and screened abstracts and manuscript and extracted the data. N.K. performed the statistical analysis. D.S., B.M., N.K., and N.B. wrote the paper. All authors approved the paper and contributed important intellectual content.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.M. reports grants (MSD) and personal fees (MSD, Vifor, Takeda), and D.S. reports travelling fees (AbbVie, Vifor, Gilead), all outside the submitted work. The other authors have nothing to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
