Abstract
The use of complementary and alternative medications (CAM), products, and therapies not considered to be part of conventional medicine is common among patients with inflammatory bowel disease (IBD). Patients often turn to these therapies as they are considered natural and safe, with significant benefit reported beyond disease control. There is emerging evidence that some of these therapies may have anti-inflammatory activity; however, robust evidence for their efficacy in modulating disease activity is currently lacking. Patients often avoid discussing the use of CAM with their physicians, which may lead to drug interactions and/or reduced adherence with conventional therapy. It is important for physicians to be aware of the commonly used CAM and current evidence behind these therapies in order to better counsel their patients about their use in the management of IBD. This narrative review provides an overview of the evidence of the more commonly used CAM in patients with IBD.
Keywords
Introduction
The inflammatory bowel diseases (IBD), which comprise Crohn’s disease and ulcerative colitis (UC), are common disorders that cause diarrhea, abdominal pain, and bleeding. 1 They are immune-mediated diseases, characterized by periods of activity, quiescence, and relapse. 2 Increased understanding of the pathogenic mechanisms of IBD has led to the development of new efficacious therapeutic agents in the form of biologic therapies. 3 These therapies, however, are associated with a number of potential risks, including infection, local reactions, and malignancy. 4 Furthermore, durability of therapies is a concern, with a significant proportion of patients losing response over time.5,6
Patients often turn to complementary and alternative medications (CAM), which are medications or practices not considered as conventional medicine, to control symptoms as well as manage their chronic disease. 7 There are a number of reasons patients may turn to CAM, the most common being a belief that management of their IBD may be cured or improved with diet and lifestyle changes, the purported anti-inflammatory properties of CAM, anxiety about potential side effects of conventional therapy and the perceived ineffectiveness of conventional therapy. 8 Recent studies suggest that half of all IBD patients use some form of CAM to help manage their disease.9–13 Given the increasing use among patients with IBD, it is important for physicians managing these patients to be aware of the current evidence in order to better counsel patients about the role of CAM in the management of IBD.
CAM are derived from a number of sources and include herbal therapies, dietary supplements, probiotics, and other physical or spiritual mind and body practices. 14 Although most people consider these therapies to be natural and safe, adverse effects can occur, and potential drug interactions with standard therapies should also be considered. 15 Most of these therapies and practices have a limited evidence base with regard to their efficacy and safety, and lack endorsement from regulatory bodies, in stark contrast to conventional medical therapies in the management of IBD, which undergo a stringent approval process.
The purpose of this narrative review is to provide an overview of the evidence, with randomized controlled trial (RCT) data, for the CAM, including curcumin, probiotics, wormwood, aloe vera and fish oil supplements, more commonly used by patients with IBD. We conducted a literature review of pre-clinical and clinical studies of these CAM using the electronic databases PubMed/MEDLINE and the Cochrane Library. Search terms used included “complementary medicine,” “complementary and alternative medicine,” and “inflammatory bowel disease.” A focused search was used for the five CAM reviewed in this article. The term “inflammatory bowel disease” was used together with “turmeric” and “curcumin,” “fish oil,” “wormwood,” “aloe vera,” and “probiotics”. Two authors independently reviewed all manuscripts published until 30 June 2019. All RCT trial data was included and other literature was included, based on a consensus decision of scientific merit by the reviewing authors (Figure 1 and Table 1). The cannabinoids are not discussed in this paper, as there is a separate publication by Picardo et al. dedicated to this topic. 16

Literature search results. CAM use in IBD.
Summary of the randomized placebo-controlled trials of complementary and alternative medications in IBD.
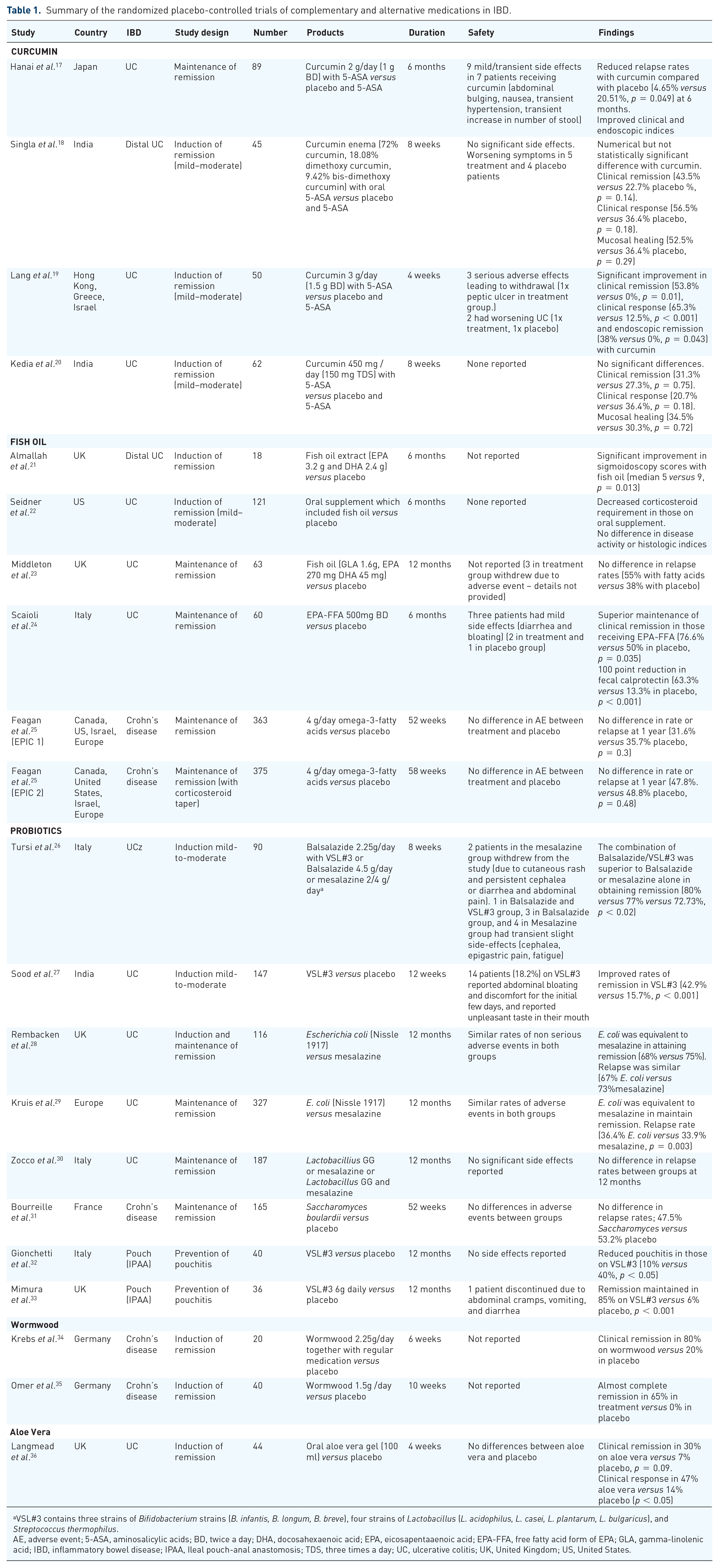
VSL#3 contains three strains of Bifidobacterium strains (B. infantis, B. longum, B. breve), four strains of Lactobacillus (L. acidophilus, L. casei, L. plantarum, L. bulgaricus), and Streptococcus thermophilus.
AE, adverse event; 5-ASA, aminosalicylic acids; BD, twice a day; DHA, docosahexaenoic acid; EPA, eicosapentaaenoic acid; EPA-FFA, free fatty acid form of EPA; GLA, gamma-linolenic acid; IBD, inflammatory bowel disease; IPAA, Ileal pouch-anal anastomosis; TDS, three times a day; UC, ulcerative colitis; UK, United Kingdom; US, United States.
The CAM market is large and continues to grow, with the global market expected to reach US $115 billion by 2020. 37 The American National Centre for Complementary and Integrative Health and the National Center for Health Statistics reported that 38% of adults and 12% of children use some form of CAM. 38 The estimated prevalence of CAM is higher in patients with IBD, with most studies reporting rates of usage approximating 50%. An Israeli group surveyed 269 patients to determine the prevalence and reasons for CAM use in patients attending a gastrointestinal clinic. 9 The prevalence of CAM was 44%, with users more likely to be female (81% versus 56%, p < 0.01) and dissatisfied with conventional therapy (22% versus 8%, p < 0.01). In this latter study, 62% of patients reported improvement in their gastrointestinal symptoms with CAM use. A large multicenter controlled trial in Sweden reported usage of CAM in 48.3% of patients with IBD 39 ; 83.1% of users reported benefits with therapy, with only 14.4% reporting negative effects.
An Australian group looked at CAM use in 473 patients across three centers, utilizing a cross-sectional questionnaire 10 ; 45.4% of respondents reported regular CAM use. Significant predictors for use include reduction in conventional medication dose, adverse effects of conventional medication, seeking psychological treatment, CAM use by family or friends, dissatisfaction with patient–doctor communication, and lower levels of depression as measured by the Hospital Anxiety and Depression Scale.
Despite the significant use of CAM among IBD patients, most do not discuss this with their physicians.10,40 This is a concern, as CAM may have associated adverse effects, drug interactions, and organ specific toxicity. 15 CAM use is also associated with reduced compliance with conventional medical therapies. A study from Canada reported that IBD patients that used CAM were less adherent to medical therapy than non-users and general CAM users (70% versus 84% versus 81%, respectively, p < 0.05). 41 This raises concerns regarding disease control, disease progression, and the development of disease complications, which subsequently may be less responsive to medical therapies and may require more complicated medical options or surgical intervention.
Turmeric (curcumin)
Curcumin is a naturally occurring substance, obtained from the spice turmeric, the root of Curcuma longa.42–44 It has been used in food preparation as well as in the field of medicine for centuries.42–44 Curcumin appears to possess a spectrum of pharmacological properties and has been used traditionally in a variety of inflammatory conditions. 43 In the last two decades, curcumin was found to be the active constituent of turmeric and has anti-inflammatory properties. This has driven research to examine its benefit in multiple fields of medicine. 45 Currently, curcumin is commercially available in supplements in a number of countries worldwide, for its presumed benefit. 43
Pathophysiology
Curcumin is one of the three components that form the curcuminoids complex found in turmeric. 46 It has a number of pharmacological properties, including anti-inflammatory effects that may be beneficial in the setting of IBD. It acts by suppressing multiple cytokines, namely interleukins (IL)-1, -2, and -12, tumor necrosis factor alpha (TNF-α), nuclear factor-kappa B (NFκB), as well as down-regulating the intracellular adhesion molecule-1 (ICAM-1) and Janus kinase.42,47 One of the main issues with its use is the limited systemic bioavailability of curcumin in humans, due to poor absorption, high metabolism, and rapid elimination. There have been a number of approaches to improve bioavailability, including the use of adjuvant agents such as piperine to reduce glucuronidation and combination with phospholipid complexes. 48 Several novel formulations, including packaging with nanoparticles, liposomes, and micelles, have demonstrated improved curcumin bioavailability both in vitro and in vivo. 49 Improving the bioavailability may further potentiate the anti-inflammatory effects of curcumin and enhance its efficacy in treating inflammatory conditions.
Clinical data in IBD
A pilot trial was conducted in 2005 to explore the therapeutic potential of curcumin in patients with IBD. 44 Five patients with Crohn’s disease and five patients with ulcerative proctosigmoiditis were administered an oral curcumin preparation (550 mg of curcumin twice daily for 1 month, then 550 mg three times daily for a further month). Patients with proctitis and Crohn’s disease demonstrated improvement in symptoms and disease activity scores. A Japanese group followed this up with a double-blind, placebo-controlled randomized trial in patients with UC. 17 They evaluated the potential beneficial effect of curcumin along with 5-ASA treatment for maintenance of disease remission over 6 months. A total of 89 patients were randomized to curcumin 1 g twice daily plus 5-ASA, or 5-ASA with a placebo. Clinical and endoscopic evaluations were undertaken every 2 months for a total of 6 months. There was a significant difference in relapse rates between groups (4.65% in the curcumin group versus 20.51% in the placebo group, p = 0.04). Curcumin was also associated with significant improvement in clinical and endoscopic indices. Some of the side effects reported with curcumin included nausea, abdominal distension, and transient stool frequency, but no patient needed to discontinue treatment because of adverse events.
Another study evaluated topical curcumin therapy in the form of an enema preparation in distal UC 18 ; 45 patients with mild-to-moderate distal UC were randomized to oral 5-ASA plus either curcumin enema or a placebo enema for 8 weeks. In the curcumin group, a larger proportion of subjects achieved response, remission, and improved endoscopic disease activity as compared with the placebo group; however, the differences in the aforementioned endpoints were not statistically significant in an intention-to-treat analysis. Per-protocol analysis revealed statistically significant superior results for the trial outcomes in those receiving curcumin compared with placebo with clinical response (92.9% versus 50%, p = 0.01), clinical remission (71.4% versus 31.3%, p = 0.03), and endoscopic improvement (85.7% versus 50%, p = 0.04).
The combination of curcumin and mesalamine in the induction of remission of mild-to-moderate UC, was also assessed in a RCT 19 ; 50 patients with active mild-to-moderate UC [defined by a Simple Clinical Colitis Activity Index (SCCAI) score of ⩾5 and <12] who did not respond to the maximized 5-ASA therapy, were randomized to receive curcumin capsules (3 g/day) or placebo in conjunction with their oral and topical 5-ASA treatment for 1 month. The primary end point of clinical remission (defined as SCCAI score ⩽2), was achieved in 53.8% (14 patients) receiving curcumin versus none (0%) in the placebo group. In the curcumin-treated group, 65.3% of patients achieved clinical response, and 38% achieved endoscopic remission (partial Mayo score ⩽1) at 4 weeks (p < 0.001 and p = 0.043, respectively, compared with placebo-treated patients). A further RCT conducted between 2003 and 2005, concluded a negative outcome for the induction of remission in patients with mild-to-moderate UC using combination of oral mesalamine and curcumin. 20 At baseline, 62 patients (curcumin: 29 and placebo: 33) were randomized. Out of these, only 41 patients (curcumin: 16 and placebo: 25) completed the trial and 21 patients did not complete. Over an 8-week period, 41 patients were randomized to oral mesalamine 2.4 g daily in addition to either curcumin (150 mg three times a day) or placebo. There was no significant difference between the two groups in terms of clinical remission, clinical response, or mucosal healing.
The current evidence demonstrates mixed results for the efficacy of curcumin in IBD. There may be a role for curcumin in the induction of active mild-to-moderate UC in those with an incomplete response to optimized 5-ASA therapy and as a complementary maintenance therapy. Further evaluation is required to establish its efficacy and determine an optimal dose. Strategies to improve systemic bioavailability of curcumin and the impact on its anti-inflammatory activity in IBD should also be evaluated. There is currently no evidence demonstrating the efficacy and safety of curcumin in Crohn’s disease.
Fish oil (omega-3 fatty acids)
Omega-3-fatty acids (n-3), which are part of a large family of polyunsaturated fatty acids (PUFA), are involved in the regulation of a number of body functions, including inflammation, blood pressure, coagulation, and neurological function. 50 They consist of lipids with two or more double bonds and are considered essential nutrients, as they are unable to be produced endogenously by the body. 51 They are found in a variety of food sources, including seeds, nuts, and fish, with the two most potent being eicosapentaaenoic acid (EPA) and docosahexaenoic acid (DHA). Omega-3-fatty acids are typically marketed and consumed as fish-oil supplements.
Pathophysiology
There has been increased interest in omega-3-fatty acids due to their anti-inflammatory and immunomodulatory effects. 52 Evidence from murine models have demonstrated a relationship with omega-3 levels and modulation of pro-inflammatory mediators.53,54 Several human studies have also shown that increased consumption of omega-3-fatty acids reduces the production of the eicosanoid inflammatory mediators (prostaglandins, thromboxanes, and leukotrienes), leading to shift to an anti-inflammatory profile.52,55 They also have been demonstrated to decrease the production of inflammatory cytokines including TNF, IL-1, and IL-6, and the expression of adhesion molecules on endothelial cells. Several clinical trials have reported beneficial effects of increased omega-3-fatty acids in coronary artery disease,56,57 obesity, 58 and rheumatic disorders.59–61
Clinical data in IBD
A large meta-analysis looked at the impact of dietary intake of fish and omega-3-fatty acids and risk of IBD. 62 This included 10 studies with a total sample size of 282,610, of which 2002 had IBD (1061 with Crohn’s disease and 937 with UC). Fish consumption was inversely associated with risk of Crohn’s disease, and there was a strong inverse association between dietary long chain omega-3-fatty acids and the risk of UC.
Supplementation of omega-3-fatty acids has demonstrated benefits in animal models with experimentally induced colitis.63,64 A pilot study in 18 patients with distal procto-colitis looked at the role of fish oil extract (EPA 3.2 g and DHA 2.4 g) or placebo (sunflower oil) over a period of 6 months. 21 They demonstrated a significant reduction in sigmoidoscopic scores in those that received the fish oil compared with placebo. Circulating levels of natural cytotoxic cells including natural killer (NK) and lymphokine-activated killer (LAK) cells were also measured using flow cytometric analysis, with a significant reduction in these cell types in those that received the fish oil extract.
Several larger human trials in patients with UC yielded mixed results. One RCT, which included 121 patients with mild-to-moderate UC, administered an oral supplement enriched with fish oil or placebo over period of 6 months. 22 This supplement also included several other constituents including fructo-oligosaccharides, gum arabic, vitamin C, vitamin E, and selenium. Although the supplement was associated with decreased corticosteroid requirements at 6 months, there were no significant differences in disease activity and histologic indices between groups.
A further RCT was performed in 63 patients with quiescent UC. 23 Patients were randomized to receive a trial medication, (1.6 g gamma-linolenic acid, 270 mg eicosanpentaenoic acid, and 45 mg of docosahexaenoic acid) per day or placebo (sunflower oil 500 mg/day.) Patients were followed up for 12 months with similar rates of relapse in disease activity between groups. A third RCT in patients with UC in clinical remission (partial Mayo score < 2), but with an elevated fecal calprotectin ⩾150 μg/g, were randomized to receive a free fatty acid form of EPA (EPA-FFA), 500 mg twice daily or placebo 24 ; 60 patients were followed up for 6 months. Patients randomized to the EPA-FFA group had higher rates of 100 point reduction in calprotectin levels (63.3% versus 13.3%, p < 0.001) as well as maintenance of clinical remission (76.6% versus 50%, p = 0.035).
There have been two large multinational placebo-controlled trials, the Epanova Program in Crohn’s Study 1 and 2 (EPIC 1 and EPIC 2) looking at the oral administration of n-3 PUFA in prevention of relapse of Crohn’s disease. 25 In these trials, 363 and 375 patients with quiescent Crohn’s, CDAI score of less than 150, were randomized to receive either 4 g of omega-3-fatty acids a day or placebo for up to 58 weeks. Patients were not on any other treatment modality for their Crohn’s disease. Their rates of relapse were similar between groups, EPIC 1 (31.6% omega-3-fatty acids versus 35.7% placebo, p = 0.3) and EPIC 2 (47.8% omega-3-fatty acids versus 48.8% placebo p = 0.48). There were also no differences in adverse events between groups. These suggested that omega-3-fatty acids are not effective in the prevention of relapse in Crohn’s disease. A Cochrane review from 2014 also concluded that omega-3-fatty acids are ineffective for Crohn’s disease. 65
Although safe and well tolerated, omega-3-fatty acids lack efficacy in maintenance of remission in Crohn’s disease. There are mixed results in patients with UC, with further larger studies required to demonstrate a benefit.
Probiotics
Probiotics are defined by the World Health Organization as, “live microorganisms which when administered in adequate amounts confer a health benefit on the host”. 66 Probiotics have been used for a number of medical conditions, with growing evidence about their potential benefits in gastrointestinal diseases, including IBD.67–72 Probiotics were found to be the most common supplements used (64%) among CAM users that attended a gastroenterological clinic. 9
Pathophysiology
Multiple probiotic bacterial strains have been studied in IBD; these include, but are not limited to, Bifidobacterium strains (B. infantis, B. longum, B. breve), Lactobacillus (L. acidophilus, L. casei, L. plantarum, L. bulgaricus), Escherichia coli Nissle 1917, and Streptococcus thermophilus.28,68,73 These non-pathogenic live bacterial strains are hypothesized to overpopulate the gastrointestinal tract with potentially beneficial microbes that confer anti-inflammatory and immunomodulatory effects.67–72,74 Studies have demonstrated that they can ameliorate intestinal epithelial barrier protective function,75,76 down-regulate inflammatory cytokines and Toll-like receptor expression,68,76 suppress TNF-α, 77 and induce protective cytokines such as IL-10. 68
Clinical data in IBD
In UC, probiotic use was examined for the treatment of mild-to-moderate disease activity in combination with 5-ASA therapy. 26 The combination of low-dose balsalazide (2.25 g/day) and VSL#3 [contains three strains of Bifidobacterium strains (B. infantis, B. longum, B. breve), four strains of Lactobacillus (L. acidophilus, L. casei, L. plantarum, L. bulgaricus), and Streptococcus thermophilus] was more effective than either balsalazide alone (4.5 g/day) or mesalazine alone (2.4 g/day) in the treatment of mild-to-moderate active UC. A significant difference in remission at 8 weeks in the intention-to-treat analysis was observed (80% versus 77% versus 72.73%, p < 0.02) in the combination of low-dose balsalazide (2.25 g/day) plus VSL#3, balsalazide (4.5 g/day) or mesalazine (2.4 g/day), respectively. An open-label trial of 32 patients with mild-to-moderate UC, VSL#3 use for 6 weeks resulted in induction of remission (53%) measured by Ulcerative Colitis Disease Activity Index (UCDAI). 78 In this study, subjects were allowed to continue treatment with corticosteroids or mesalamine in oral or rectal forms. Similarly, a double-blinded, placebo-controlled trial conducted in India showed significant induction of remission in 147 patients with mild-to-moderate UC treated with VSL#3 for 12 weeks, compared with placebo (42.9% versus 15.7%, p = <0.001). 27 Concomitant medications were not allowed, except for stable doses of azathioprine for at least 3 months prior.
Two trials compared the probiotic preparation of E. coli Nissle 1917 with mesalazine for maintenance of remission in UC over 1 year, with both demonstrating these were equivalent in maintaining remission disease activity.28,29 Lactobacillus GG was also examined for the maintenance of remission of UC in a RCT. 30 Although Lactobacillus GG was better than standard therapy in providing longer relapse-free time, no significant difference was observed between Lactobacillus GG alone or in combination with mesalazine versus mesalazine, at 6 and 12 months. Systematic reviews concluded inconsistent results about their benefit in UC.74,79,80
There is no clear evidence to support the use of probiotics for the induction or maintenance of remission in Crohn’s disease. A recent meta-analysis concluded that they were ineffective in the induction of remission of active disease or preventing relapse in both quiescent disease or in the postoperative setting. 80 An RCT evaluated Saccharomyces boulardii use versus placebo for the maintenance of Crohn’s disease following treatment with steroids and salicylates in 165 patients. It found no benefit in maintaining remission with high rates of relapse among both groups. 31 Similarly, VSL#3 was not effective in maintaining remission or improving quality of life in Crohn’s disease in another RCT, published in abstract form. 81 In line with these trials, probiotics were also not effective in preventing postoperative recurrence after surgery for Crohn’s disease in several trials.82,83
Ileal pouch-anal anastomosis (IPAA) is the preferred procedure after proctocolectomy for medical refractory UC; 80% of patients will have at least one episode of pouchitis at 30 years after IPAA. 84 The strongest evidence for the use of probiotics in IBD, is the use of VSL#3 for preventing recurrent pouchitis. For preventing the first episode of pouchitis during the first year after IPAA, one RCT of 40 patients were given VSL#3 versus placebo daily for 1 year immediately after ileostomy closure. 32 Only two patients (10%) in the VSL#3 group developed pouchitis compared with 40% in the placebo group (p < 0.05). VSL#3 was also found to induce remission for mildly active pouchitis; 23 consecutive patients with mild active pouchitis were treated with two sachets of VSL#3 (3,600 billion bacteria/day) over a 4-week period. 85 In this study, 69% of patients achieved remission as defined by Pouchitis Disease Activity Index clinical (PDAI) score of ⩽2, endoscopic PDAI of ⩽1, and total PDAI score of ⩽4.
VSL#3 was also studied for the maintenance of remission in refractory or recurrent pouchitis. After 4 weeks of antibiotic induced remission, 36 patients were randomized to VSL#3 6 g/day or placebo. At 1 year, remission [defined as a clinical (PDAI) score of ⩽2], endoscopic PDAI of ⩽1 was significantly higher in VSL#3 group compared with placebo (85% versus 6%; p < 0.0001). 33 A trial in the United States (US) looking at maintenance of remission was, however, negative. 86
The probiotic VSL3# has demonstrated efficacy, alone and in combination with 5-ASA therapy, in inducing remission in patients with active UC. Several probiotic strains including VSL3# may be effective in preventing a relapse in UC activity. The current limited evidence suggests probiotics are not effective in Crohns disease; however, further larger RCTs are required to establish if they play a role. Current clinical data, however, does support the use of VSL3# as an effective therapy for both induction and maintenance of pouchitis in patients with an IPAA.
Wormwood (Artemesia absinthium)
The genus Artemesia comprises over 400 species of herbaceous plants that have traditionally been used to treat a variety of digestive ailments.87,88 Artemesia absinthium, the most well known, commonly referred to as wormwood, is usually prepared with a distillation of neutral alcohol and other herbs. It has a broad spectrum of bioactivity and has been demonstrated to exhibit anti-inflammatory, anti-tumor, anti-microbial, and anti-oxidant activity.
Pathophysiology
The phytoconstituents of the Artemesia spp., including flavonoids and sesquiterpene molecules, demonstrate anti-inflammatory activity through the synthesis and release of inflammatory mediators including histamine, serotonin, bradykinins, and prostaglandins.89–91 The flavonoid 5,6,3′,5′-tetramethoxy 7,4′-hydroxyflavone (p7F) extracted from wormwood has been found to suppress the serum level of TNF-α in mice. 92 It also inhibits the expression and production of other pro-inflammatory mediators including COX-2/PGE2 and iNOS/NO. A further constituent of wormwood, artemisin, and its metabolite artesunate may also play a role in its anti-inflammatory effect. Artesunate was found to decrease the secretion of the cytokines IL-1b, IL-6 and IL-8 in TNF-α stimulated human rheumatoid arthritis fibroblast-like synoviocytes. 93
Clinical data in IBD
There have been two small placebo-controlled trials evaluating the therapeutic effects of wormwood in patients with Crohn’s disease. One study enrolled 20 patients with active Crohn’s disease (CDAI greater than 200) to receive dried powdered wormwood (3 × 750 mg) or placebo for 6 weeks. 34 Patients on anti-TNFα agents were excluded in order to measure effect of wormwood on serum TNFα levels. Clinical remission (CDAI score below 170 or reduction by 70 points) was seen in 8 of 10 (80%) patients in the wormwood group, compared with only 2 (20%) in the placebo group. Average serum TNF-α levels fell from 24.5+/–3.5 pg/ml at baseline to 8.0+/–2.5 pg/ml at week 6 in the wormwood group.
A second multicenter trial from five sites in Germany recruited 40 patients with active Crohn’s disease who had been receiving a daily dose of prednisone for at least 3 weeks, to receive a herbal blend containing wormwood (3 × 500 mg each day) or placebo for 10 weeks. 35 Patients were evaluated at 2-weekly intervals during the 10 weeks, and then followed up for a further 10 weeks after the end of the intervention. The initial dose of steroids was maintained for 2 weeks, followed by a defined wean whereby all patients were free of steroids by week 10. At week 8, 65% of patient’s receiving wormwood had almost complete remission of symptoms (based on CDAI score) as compared with none in the placebo group, with remission persisting until the end of observation. Wormwood also demonstrated a steroid-sparing effect and improved quality-of-life scores. These two small trials suggest efficacy for wormwood in Crohn’s disease; however, one major limitation of both was the non-reporting of adverse events. Wormwood contains a component, Thujone, that has been associated with dose-dependent neurotoxicity in humans and can induce convulsions and psychosis. 94 Further large RCTs evaluating the safety and efficacy of wormwood are required before any recommendations can be made about its utility in the management of Crohn’s disease. Wormwood has not been evaluated in UC to date.
Aloe vera
Aloe vera (Xanthorrhoeaceae) is a tropical plant that has been used for thousands of years with a number of purported medical benefits. It is one of the most common natural remedies used by patients with IBD. 95 It has been reported to have anti-ulcer as well as immunomodulatory effects.96,97
Pathophysiology
Aloe vera has been studied in an animal model of colitis. An aloe vera gel preparation was demonstrated to ameliorate colonic tissue injury in dextran-sulfate-sodium-induced colitis in rats, by down-regulating inflammatory mediators and attenuating immune cell recruitment. 98 Both TNFα and plasma leukotriene B4 were significantly decreased in the rats supplemented with aloe components compared with a colitis control group.
Clinical data in IBD
A trial in the United Kingdom (UK) randomized 44 patients with UC to receive an oral aloe vera gel (10ml twice daily for 4 weeks) or placebo in a 2:1 ratio. 36 The primary endpoint of clinical remission (Simple Clinical Colitis Activity Index ⩽ 2) was seen in 30% of those receiving aloe vera, compared with 7% in those on placebo (p = 0.09). A clinical response (Simple Clinical Colitis Activity Index ⩽ 2 or decrease in score of ⩾3) occurred in 47% in those receiving aloe vera compared with 14% in the placebo group (p < 0.05). There were, however, no differences in endoscopic scores. The aloe vera gel was well tolerated, with no significant differences in adverse events between treatment and placebo groups. Further studies are required to assess the efficacy and safety of aloe vera in the management of UC.
Clinical guidelines
The European Crohn’s and Colitis Organization (ECCO) recently published a Topical Review on Complementary Medicine and Psychotherapy in Inflammatory Bowel Disease and provided clinical guidance based on the current available evidence. 99 They outlined that curcumin, in combination with 5-ASA, may be effective in inducing remission in mild-to-moderate UC as well as potentially being effective as a complementary maintenance therapy in UC. They concluded that omega-3 fatty acids may be beneficial in maintaining remission in Crohn’s disease; however, study quality and heterogeneity of trials limit these findings. They found no evidence for the use of probiotics in the induction or maintenance of remission in Crohn’s disease. The probiotic E. coli Nissle 1917, however, may be effective in inducing remission and is effective in maintaining remission in patients with UC.
Conclusion
CAM are commonly used among patients with IBD. Although the use of CAM is generally considered safe, evidence for their efficacy and safety in the management of IBD is lacking. Curcumin may be beneficial in the induction and maintenance of UC. n-3 PUFA may be also be beneficial in UC; however, n-3 PUFA lacks efficacy in Crohn’s disease. Probiotics are not effective for Crohn’s disease, but certain strains have demonstrated efficacy in UC and pouchitis. There is limited data that suggests wormwood may be beneficial in Crohn’s disease, and that aloe vera may improve clinical outcomes in UC. Further large randomized controlled studies, however, are required to validate the efficacy of all these complimentary therapies in IBD. Physicians should be aware of the current evidence behind the various CAM in order to better counsel their patients in the management of IBD.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
Cynthia H. Seow has served as a consultant, speaker and has served on advisory boards for Janssen Pharmaceuticals, Abbvie, Takeda, Ferring, Shire, and Pfizer.
Shane M. Devlin has served as a consultant, speaker and has served on advisory boards for Janssen Pharmaceuticals, Abbvie, Takeda, Ferring, Shire, and Pfizer.
Sherman Picardo has served as a speaker for Janssen Pharmaceuticals, Abbie, Pfizer and Takeda.
