Abstract
Clinicians involved in the treatment of inflammatory bowel disease (IBD) increasingly come across patients with current or previous history of malignancies. With increasing and earlier use of immunosuppression and biologics in IBD patients, the question arises whether these treatments further increase the risk of new or recurrent cancers. A number of population-based observational studies have now reported the odds of development of new or recurrent cancers with thiopurines and antitumour necrosis factors (anti-TNFs). These data combined with data arising from treatment registries from other immune disorders such as rheumatoid arthritis are providing evidence of relative risks and safety profiles of these agents in the setting of active or prior cancer. Data from transplant literature give an indication for providing a drug-holiday period in patients with treated cancers. The risks of the treatment should be considered alongside the risk associated with withholding these effective treatments in patients with active IBD. In this review, we aim to summarize the current evidence in this area and provide a practical guidance.
Introduction
There is increased risk of intestinal and extra intestinal cancers in inflammatory bowel disease (IBD) patients.1,2 Likewise, as the population of patients with IBD ages, a proportion of these patients may develop de novo cancer.
In addition, there is increasing debate about the impact of IBD therapies in cancer development and progression.3,4 The evolution of newer treatment paradigms focusing on achievement of deep remission and even mucosal healing in IBD has resulted in increase in the use of immunosuppressive agents and biologics alone, or in combination, in IBD patients. While these treatments have significant efficacy data, the data on safety, particularly in relation to cancer risks, are limited.
With improvements in cancer survival rates for the majority of cancers, IBD clinicians are meeting increasing numbers of IBD patients with a prior history of cancer. Furthermore, patients and clinicians face challenges when faced with a situation of managing IBD in the presence of cancer. Unfortunately, patients with active cancers or those who had history of cancer in the preceding 5–10 years are excluded from randomized controlled trials or registration trials due to the unknown risk and concerns over provoking recurrence of cancer, or progression of active cancer, and patients who develop cancer while on newer therapies, such as biologics in IBD, are withdrawn from the studies. Hence, the data in relation to issues around cancer and IBD will only come from long-term observational studies and registries.
We aimed to review the relevant literature addressing these challenges and provide practical guidance based on the evidence.
Cancer risk with existing IBD therapies
Anti-TNFs
Concern about possible effects on malignancy has been present since their introduction to therapeutic armamentarium in IBD but the potential effects are difficult to predict accurately given the pleotropic effects of antitumour necrosis factors (anti-TNFs) and the complexity of the pathways involved in inflammation and tumour development. Most of current literature is about the risk of cancers in anti-TNF-treated patients. However, the effect of anti-TNF therapy on the progression of existing cancers, recurrence of cured cancers, the differential risks in different cancer types and the overall outcomes have not been well documented. The effects of anti-TNF therapy can be complex and often bidirectional on cancer incidence, outcome, recurrence and progression. TNFs have tumour-promoting activities, as a protumourigenic inflammatory cytokine such as nitric oxide induced deoxyribonucleic acid (DNA) mutations, promotion of autocrine growth signal and angiogenesis. 5 In addition, they can promote tumour invasion by inducing matrix metalloproteinases and also can induce resistance to cytotoxic drugs. On the other hand, as the name indicates, it also has tumour-inhibiting properties and has been used in the treatment of some cancers such as melanoma and sarcoma. 6
Accurate determination of the cancer risk of anti-TNFs is challenging, as a significant proportion of IBD patients have combined therapy, mostly with thiopurines. An initial meta-analysis on anti-TNF therapy in IBD reported a threefold increase in overall risk of lymphoma over general population but no increase in risk over therapy with immunomodulators. 7 However, this increased risk of lymphoma has not been replicated in the subsequent studies.8–11 In a Danish nationwide cohort study 8 with follow up, with patients on anti-TNF for 3.7 years, no increase in overall malignancy rates [relative risk 1.07; 95% confidence interval (CI) 0.85–1.36] or lymphoma rates (adjusted relative risk 0.90; 95% CI 0.42–1.91) was found. A retrospective study comparing patients on adalimumab monotherapy with those on combination therapy found no increase in lymphoma risk with monotherapy but there was an eightfold increase in lymphoma in combination therapy when compared with general population. 9 Similarly, data from the TREAT registry 10 indicate similar rates of malignancy in infliximab-treated patients compared with those who are infliximab naïve. Finally, a recent meta-analysis including 22 randomized controlled trials found no increase in lymphoma in IBD patients treated with anti-TNF therapy compared with the control population. 11 Furthermore, the previously feared complication of fatal hepatosplenic T-cell lymphoma has now been found to be distinctly associated with combination therapy with thiopurines in young patients with IBD rather than with monotherapy with anti-TNFs.
There is suggestion of association of anti-TNFs to increased risk of melanoma in one study. 12 This study found no association with nonmelanoma skin cancer (NMSC) and anti-TNF use. This apparent concern has not been confirmed in a national registry-based study 13 or in a metanalysis. 11
There are few studies in a palliative care setting where anti-TNF agents have been beneficial in improving cancer-related cachexia and chemotherapy tolerance.14–16 Furthermore, when cancers do happen in the setting of ongoing anti-TNF therapy, they do not appear as having a worse stage of disease and may even be protective in relation to metastasis.17,18
Thiopurines
Thiopurines are considered to be mutagenic due to their ability to induce somatic mutations and prevent DNA-repair mechanisms. 19 In addition, they interfere with the ability of the immune system to recognize the B cells infected by Epstein–Barr virus (EBV) leading to virus-related lymphoproliferative disorders. 20 Furthermore, thiopurines can induce oxidative stress and mutagenic oxidative DNA changes in conjunction with ultraviolet radiation, provoking skin malignancies. 21 Preliminary data about the cancer risk of thiopurines came from the renal transplant literature which suggested an association between thiopurine use and the risk of certain specific cancers, particularly NMSC and lymphoma. 22 The risk of lymphoma was confirmed in the prospective CESAME study which reported an adjusted hazard ratio for lymphoproliferative disorders of 5.28 (95% CI 2.01–13.90) in patients exposed to thiopurines compared with those patients who were thiopurine naïve. 23 A nested case control study from UK 24 also suggested a similar small risk of lymphoma in thiopurine-treated IBD patients with an odds ratio (OR) of 3.22 (95% CI 1.01–10.18). In a recent meta-analysis by Kotlyar and colleagues, 25 current use of thiopurines was associated with a significant risk of lymphoma with a pooled standardized incidence ratio in population-based studies of 5.71 (95% CI 3.72–10.1) with the risk becoming significant 1 year after initiation of treatment. The risk was higher in referral centre populations [standardized infection ratio (SIR) 9.25; 95% CI, 4.69–18.2] and in young patients under age 30 years (SIR 6.99, 95% CI, 2.99–16.4). In this study there was a reduction in risk with cessation of thiopurines (SIR 1.42; 95% CI 0.86–2.34).
Thiopurines increasing the DNA sensitivity to ultraviolet light and may predispose to skin cancers. 21 Thiopurines also have been associated with increased risk of skin cancers particularly NMSCs in a number of cohort studies in IBD patients receiving thiopurines.26,27 A subsequent meta-analysis 28 found a pooled hazard ratio of 2.28 (95% CI, 15–3.45), suggesting a significant yet modest increase in risk of NMSC in IBD patients treated with thiopurines. There is no obvious increase in risk of melanomas with thiopurines but there is additional increase in risk for other cancers such as urinary tract cancers, cervical cancer and colorectal cancer, although the data are somewhat limited. 28
Methotrexate
There are no convincing biological mechanisms that indicate that methotrexate may be carcinogenic.29,30
In a well-designed comparative effectiveness study 31 using propensity-score analysis for various disease-modifying drugs used for rheumatoid arthritis, methotrexate had a higher risk of cancer compared with TNF antagonists but lower risk compared with thiopurines. In addition, while there are reports of increased risk of a post-transplant lymphoproliferative-like condition in patients with rheumatoid arthritis and IBD treated with methotrexate, 30 the overall risk of lymphoma is the lowest with use of methotrexate, in a systematic review. 29 There is some evidence for increased risk of melanoma and of melanoma skin cancers in patients treated with methotrexate for psoriasis and rheumatoid arthritis. 32 However, this risk was not noted in IBD patients receiving methotrexate. 33
Newer biologics
There are limited, if any, data on malignancy risk of newer biologics such as vedolizumab. In the placebo-controlled studies and subsequent open-label studies, 19 cancers have been detected, accounting for 0.67% of participants in the trials. 34 There is theoretical concern regarding immunosurveillance in patients with gastrointestinal cancers when using vedolizumab, and although this has not been confirmed in pharmacovigilance data so far, vedolizumab is best avoided in patients with prior or current gastrointestinal cancer. Similarly, there are no data available so far on the cancer risk of ustekinumab-treated IBD patients. However, there was no additional risk for melanoma or NMSC in the Psoriasis Longitudinal Assessment and Register (PSOLAR) for treatment up to 12 months. 35
Mesalazine
There are no data to indicate increased risk of malignancies from mesalazine compounds. In fact meta-analysis of epidemiological studies have suggested that mesalazine has a chemopreventive role for colitis-associated colorectal cancer.36,37 The mechanisms behind the antineoplastic effect of mesalazine are incompletely understood, but it is likely they are mostly dependent on the ability of the drug to attenuate ongoing mucosal inflammation and its ability to inhibit the formation of reactive oxygen species from neutrophils.38,39
Steroids
Systemic glucocorticoids are potent immunosuppressants, potentially facilitating carcinogenesis. Sorensen and colleagues, from Denmark’s Aarhus University, identified almost 60,000 people enrolled in a prescription database who received long-term corticosteroids and reported an increased risk of lymphoma and NMSC. 40 Having 15 or more prescriptions for steroids filled over the 8-year period was associated with a 1.52-fold increase in basal cell carcinoma risk and a 2.45-fold increase in risk for squamous cell carcinoma. Non-Hodgkin’s lymphoma risk was found to be 2.68-fold higher for patients having 10–14 prescriptions filled over the study period. 40 A further more recent study also suggested the association with lymphoma and basal cell carcinoma. 41 However, studies in solid organ cancers have not confirmed this association; 42 the concept that the effect of steroids is primarily a reflection of the severity of the underlying inflammatory process is currently muted, as more recent studies did not show any association with increased risk of skin cancers or lymphoma. 43
A semiquantitative risk profile of the available drugs in relation to cancer recurrence is depicted in Figure 1.
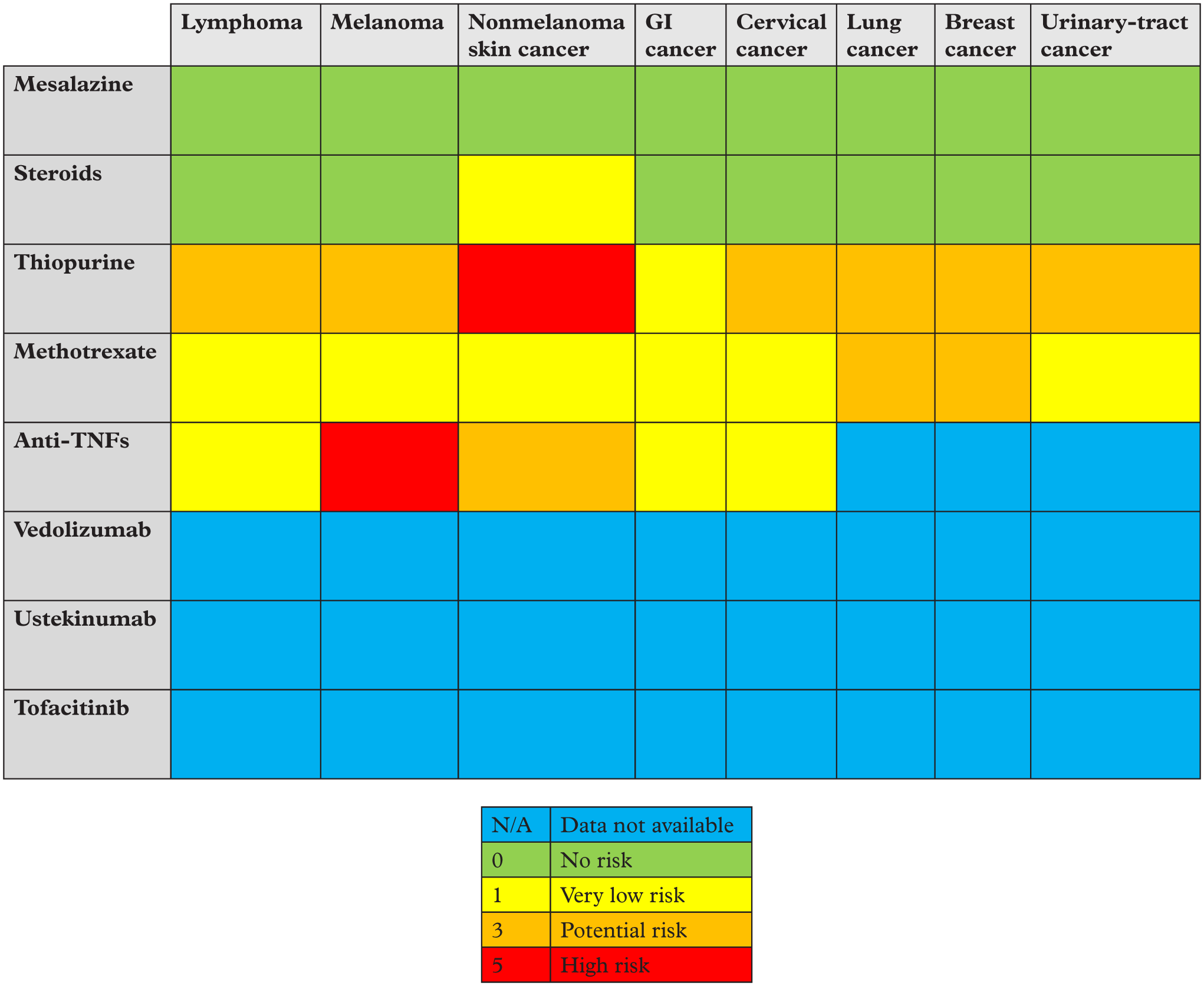
Semi quantitation malignancy risk of drugs used for IBD.
Treatment of IBD in those with prior history of cancer
With reduction in cancer mortality rates for the majority of cancers globally and increased incidence of IBD, it is inevitable that IBD clinicians will be seeing progressively increasing numbers of patients who had a cancer diagnosis in the past. There is emerging evidence that a diagnosis of cancer may significantly influence clinicians and patients in their decision of the future therapeutic strategy for IBD. Clinical guidelines based on expert consensus often recommend significant restrictions in medications such as immunosuppression and biologics and as a result, clinicians have historical reluctance to commence immunosuppressive and biological therapy in patients with prior history of cancer, hence potentially increased risk of surgery as a treatment in those who have significant active disease. If continued treatment or initiation of treatment with anti-TNFs or immunosuppressants or both have no or limited impact or indeed, a beneficial effect on cancer outcomes, it is difficult to justify depriving the severe IBD patients the effect they have on their disease.
The concerns about the risk of using immunosuppressants originate from the large experience in the post-transplant setting. 44 In a much-quoted study of 1137 renal transplant recipients with prior malignancy, 45 21% of patients developed recurrent cancer, the most common types being myeloma, NMSC, bladder cancer, breast cancer and renal cancers. The majority of these recurrences occurred within the 2 years postmalignancy with only 13% relapsing after 5 years. A further study confirmed these findings in 939 cancer patients who received transplantation, with 22% recurrence rates, with half of them occurring in patients whose cancer was treated within 2 years of transplantation. 46 While transplant patients often receive multiple immunosuppressants and hence the risk may be heightened, based on these figures, one can divide cancers into risk categories for recurrence following immunosuppression (Table 1).
Recurrence risk of common cancers.

The risk of a new incident or a recurrent previous cancer in IBD patients and receiving or continuing thiopurines has been studied in a number of trials. The seminal study of these come from a subgroup analysis of the CESAME study which is a prospective observational cohort study of over 17,000 IBD patients in France. 47 This analysis included 405 IBD patients with history of previous cancer, of which 93 patients were exposed to thiopurines and 312 were not exposed and found that there was no additional risk of incident cancers in those exposed to immunosuppressants (27/1000 patient years of exposure) compared with those not exposed (19.2/1000 patient years of exposure). Importantly, this study mainly focused on thiopurines and had 50% of their patients with prior cancer within the preceding 5 years, and all the patients with prior cancer and who developed recurrent cancers were exposed to thiopurines and not to biologics. Most of the patients with previous cancer who had a new cancer development, had it affecting a different or the same organ but a different histological type. Furthermore, the most common type of recurrent cancer in patients with a history of cancer was NMSC. Increased risk of recurrent NMSC has also been reported in studies on patients with all autoimmune conditions, including IBD. 48 Expert opinions suggest that thiopurines can be continued in the setting of NMCSs as long as they are fully excised. 49 The scenario is different when there is an EBV-related lymphoproliferative disease, where thiopurines should not be used and patients offered treatment with anti-TNFs or methotrexate. 50
Most randomized controlled trials using anti-TNFs in IBD excluded patients with prior history of cancer and hence the data of recurrent cancer in anti-TNF-treated patients with prior history of cancer come from observational cohort studies and disease registries. A study from New York reported 30% of their cohort of IBD patients with prior cancer developing new or recurrent cancer. 51 In this study, exposure to anti-TNF agents alone or in combination with thiopurines was not associated with risk of new or recurrent cancer in the 5-year follow-up period following diagnosis of cancer, even after adjusting for the baseline risk of recurrence of the index cancer. 51 These data are corroborated in studies using large national registries in rheumatoid arthritis patients exposed to anti-TNF agents.52–54 In the study using the British Rheumatology biologic register by Dixon and colleagues, 52 there was no difference in the risk of development of new or recurrent cancers in anti-TNF-exposed patients who had a prior history of cancer. Furthermore, the rate of malignancy was lower in the anti-TNF-exposed group compared with those who used other disease-modifying drugs (25.3 versus 38.3 per 1000 patient-years). However, the risk of recurrent melanoma was higher in the anti-TNF group, thereby suggesting that it is better to avoid anti-TNFs in the setting of metastatic or recurrent melanoma. Similar data are reported from the prospective cohort study of rheumatology patients in Germany, 53 with no statistical difference in crude cancer incidence rates between those exposed to anti-TNFs versus other immunosuppressants. More recently, a Swedish case control study reported similar breast cancer recurrence regardless of anti-TNF exposure among patients with rheumatoid arthritis and a previous history of cancer. 54
A further French retrospective study evaluated the risk of incident cancers in 79 IBD patients receiving anti-TNF therapy having had a cancer history within the preceding 5 years. 55 In this study, 15 (19%) patients developed new or recurrent cancers with a crude incidence rate of 84.5% (95% CI 83.1–85.8) per 1000 patient-years. In the whole cohort, the survival without cancer at 1, 2, and 5 years was 96%, 86% and 66% respectively. This study also analysed the crude incidence rate in the interval from cancer diagnosis, and anti-TNF treatment was more than and equal to 2 years and found a numerical difference in crude cancer rates (97.7 versus 64.4 per 1000 patient-years) suggesting that it is best to avoid anti-TNF therapy for the first 2 years after cancer. The crude incidence rate of new or recurrent cancer is higher in this study compared with what is recorded in rheumatology literature.52,53 The German biologic register 53 and the British rheumatology biologics registry 52 reported crude incidence rates of 45.5 per 1000 patient-years and 25.3 per 1000 patient-years, respectively, in those with prior cancer and receiving anti-TNFs for rheumatoid arthritis. The difference may be due to the fact that in these studies, the median interval between cancer and anti-TNF exposure was longer at 10 years. In addition, these studies used methotrexate as a concomitant immunomodulatory in the majority of patients, as opposed to the abovementioned French study in which 72% of patients were on thiopurines.
A comprehensive systematic review and meta-analysis of immune-suppressive therapies after a prior diagnosis of cancer in a variety of immune-mediated diseases including IBD has been recently conducted by Shelton et al. 56 In the meta-analysis, they included 11,702 patients accounting for over 31,000 person-years of follow up after a prior diagnosis of cancer. In this study, the rates of recurrent cancers were similar in those receiving anti-TNF therapy (33.8/1000 person-years), those receiving immunosuppressants (36.2/1000 person-years) or no immunomodulation at all (37.5/1000 person-years). Although there was a numerically higher recurrent cancer risk found in those receiving combined immunosuppression (54.5/1000 person-years), this was not statistically significant, and the analysis was limited by including only three studies with combined immunosuppression. When analysed separately, including 3706 IBD patients contributing 10,332 person-years of follow up, there were 539 cases of incident or recurrent cancers. The pooled incidence rates among included subjects with a new or recurrent cancer on immunosuppressants or biologics was not significantly different to those receiving no immunosuppression (37.9, 48.5 and 35.7, respectively, p > 0.3 for all). The authors also used the random-effects model to evaluate the rate of recurrent or new cancer between anti-TNF therapy compared with immunosuppressive therapy and found numerical difference in favour of anti-TNFs, although not statistically significant. When the analysis was limited only to those with index skin cancers, those taking immunomodulators but not biologics were significantly higher than those taking no immunosuppressants (71.6/1000 person-years versus 50.8/1000 person-years). While the authors conducted sensitivity analysis to overcome some of the heterogeneity issues in their metanalysis, none of the included studies included propensity-score matching, thereby indicating that there will be selection bias among practitioners treating the included patients in these studies, favouring avoidance of immunosuppression in those with high risk of cancer recurrence. 57
All of these studies and the abovementioned meta-analysis are limited by the small numbers of incident or recurrent cancers, making firm conclusions and clinically meaningful recommendations difficult. In particular, all these studies have pooled together cancer of various sites and varying severity and hence, may not be an accurate reflection of the natural biologic evolution of carcinogenesis. Identification of site-specific risk could lead to improved surveillance and pharmacovigilance. Hidden bidirectional patterns of risk should also be considered. For example, it may be that anti-TNFs may reduce the risk of lymphoma by reducing inflammation not only in patients who respond but in those who don’t respond; it may increase the risk by its direct tumour-promoting effect. This can only be teased out by studying changing patterns in subtypes of lymphoma; in particular, those closely associated with inflammation or those associated with oncogenic viruses.
There are no studies looking at the safety of immunomodulators or biologics in the setting of carcinoma in situ or premalignant conditions such as Barrett’s oesophagus. However, there are accumulating data for increased risk of abnormal pap smears and cervical intraepithelial neoplasia in IBD patients,58,59 and the possible increased risk of introduction or continuation of immunosuppressants, particularly thiopurines, needs to be evaluated in future studies.
In summary, in contrast to the data from the post-transplant population, observational studies in IBD patients with prior malignancy do not appear to be at significantly increased risk of new or recurrent cancer. IBD treatment decisions following diagnosis of cancer are complex and must take into account the type of cancer, its natural history, the interval from the cancer diagnosis and its treatment completion, and the need for the IBD treatment decision and importantly the severity and predicted course of IBD and the alternatives to immunosuppressants and biologics. In practice, in most cases with a prior history of cancer, immunosuppressants and anti-TNF initiation should be delayed for at least 2 years, with the delay extended to 5 years for a cancer with high risk of recurrence, such as cancers of renal tract, endometrial cancer melanoma and lung cancer.Thiopurines should be avoided in cases of prior EBV-related lymphoma, human-papillomavirus-related carcinoma and cancer of the urinary tract.
One of the key questions concerning therapy options in IBD patients with prior history of cancer is the length of drug-holiday period required for immunosuppressants and biologics after the treatment of the index cancer. The ECCO consensus guidance 60 in this topic recommends an interval of 2 years before starting immunosuppression and perhaps waiting up to 5 years for malignancies with intermediate or high risk of recurrence, such as of the urinary tract, gastrointestinal cancer, leukaemia and multiple myeloma. A similar interval is suggested in many expert opinion papers published.61,62
Therapies for IBD and for active cancer: effect on cancer and IBD
Most clinicians will alter management strategy of IBD when diagnosed with a new cancer, particularly with cessation of immunosuppressants and with lesser use of immunosuppressants and anti-TNFs. 63 However, increasing data indicate that patients with active IBD at cancer diagnosis may actually benefit from the cancer treatment with cytotoxic therapy and achieve remission.64–66 If the IBD is not well controlled despite the chemotherapy for the active cancer, aminosalicylates (5ASAs) and steroids are considered as the first option, and anti-TNFs considered as second-line therapy in nonresponders, based on the expert opinion statements and treatment guidelines reporting the theoretical concern about worsening of cancer outcomes.60,62
Therapy of IBD may impact the course of cancers. In colorectal cancer, immunosuppressants (azathioprine, methotrexate) and anti-TNFs have been found to have a negative impact on the disease-free survival and overall survival. 67 However, the cancers and lymphomas that occur following anti-TNF therapy are not associated with any worse stage at diagnosis or worse prognosis.18,68,69 In some studies of patients with active or recent myeloma, with anti-TNFs there was risk of progression to invasive melanomas,70,71 and expert guidelines recommend withholding anti-TNFs in this setting. 60 It is not clear whether methotrexate has similar issues in relation to tumour progression or prognosis. Lymphoma-specific survival was found not to differ based on methotrexate usage, but prognosis was worse than for lymphoma in the general population. 72 However, methotrexate is currently used in the treatment of certain cancers at high doses as in the case of breast cancer and urinary tract cancers.73,74 Given the theoretical risks related to DNA changes and also myelosuppression, it is entirely reasonable to withhold thiopurines during the treatment of active cancer. 60
When patients with IBD are diagnosed with cancer, the focus of attention of the clinician and patient undoubtedly turns towards treatment of cancer. The impact of cancer therapies on the course of IBD is important, as an active IBD may complicate the choices of therapies and potential outcomes. It is reported that associated IBD may have a negative impact on the survival of patients with extraintestinal cancer, independent of the cancer staging.70,75,76
In a study of 84 patients with extraintestinal solid organ malignancy from Massachusetts General Hospital, 51 17% of patients who had inactive IBD at the time of cancer diagnosis flared during a follow up of 6 months, with the risk of flare higher in young patients with Crohn’s disease with prior history of anti-TNF therapy before diagnosis of cancer, and in those receiving an extended period of hormonal therapies. On the contrary, the authors found that of the 15 patients who had active IBD at the time of cancer diagnosis, 66.7% achieved IBD remission with cytotoxic cancer chemotherapy, while the reminder who did not achieve remission had hormonal therapy alone or in combination with cytotoxic chemotherapy. This study gives early indicators to anticipate disease flare and develop treatment plans in patients undergoing cancer treatments in the presence of active or inactive IBD. In this study 63.1% of the patients maintained their IBD medication through their cancer treatment and there was no significant difference in the proportion of patients who were maintained on immunosuppressants and biologics (22% and 14%) after diagnosis of cancer. However, in another study from France, 63 the diagnosis of extraintestinal cancer prompted a change in management of IBD with lesser use of thiopurines (19% versus 25%, p = 0.001) and increased use of surgery (4% versus 2.5%, p = 0.05), yet there was no change in activity of IBD. In this study, thiopurines were often stopped or switched to methotrexate, while anti-TNFs were avoided in cases of severe malignancies; in a further small study exploring the clinical course of Crohn’s disease following chemotherapy, IBD flare was noted in 40% of patients. 66 The impact of radiotherapy on cancer patients with IBD is not well known and many oncologists prefer to avoid pelvic radiotherapy on in the presence of IBD. However, in a retrospective study from Mount Sinai Hospital, New York, 5-year survival of rectal cancers in patients with IBD treated with pelvic radiation was similar to those with no prior IBD and there was no increase in gastrointestinal toxicity. 77
In cases where there is breakthrough of IBD symptoms, it is important to ensure that the clinician confirms IBD flare as the reason for symptoms, as many chemotherapeutic agents may have diarrhoea as a side effect. In addition, superadded infections should also be considered. 78 Hence a flare of IBD should be confirmed by endoscopy and once the diagnosis of IBD flare is confirmed, oncologists often prefer to use corticosteroids as a first option because of their effect on the tumour and tumour-related symptoms but there are some data supporting the postulation that steroids may enhance tumour-cell resistance to apoptosis and reduce immunosurveillance. In addition, some population-based studies suggest increased risk of nonmelanoma lymphomas in those on prolonged corticosteroids,41,42 but whether this risk is related to the severity of underlying IBD is uncertain. Despite this, the overall general consensus and expert opinion is that corticosteroids are a safer option than immunosuppressants. While there are no comparative studies of steroids versus anti-TNFs in the management of uncontrolled IBD in cancer patients on treatment, anti-TNFs may well be a backup plan, currently. In milder disease flares following cancer treatment, 5ASAs and enteral nutrition may be options.
Checkpoint-inhibitor colitis
Checkpoint inhibitors are immune-stimulatory antibodies that have transformed the management and prognosis of individuals with metastatic melanoma, metastatic lung cancer and renal cell cancers. Checkpoint-inhibitor-induced colitis is an increasingly recognized immune-related adverse event that shares many of the same phenotypical and histological characteristics of IBD. 79 Detailed evaluation and management of checkpoint-inhibitor colitis are outside the scope of this paper and have been subject to excellent reviews elsewhere.80,81 Diarrhoea and colitis may present approximately 6 weeks into immune-checkpoint-inhibitor therapy and appear to be dose dependent with ipilimumab. For severe grade toxicity, with stool frequency more than seven per day or with systemic-features admission, rehydration and endoscopic confirmation of colitis are recommended. Treatment with high-dose steroids improves the colitis in the majority of ptients. 82 Those who do not respond to steroids within a reasonable time frame should be offered anti-TNF therapy using infliximab. 83 There are emerging data on use of vedolizumab in this setting. 84
Practical guidance
Our current markers predicting outcomes of IBD patients at diagnosis and also risk of IBD relapse for patients in remission are evolving, and this will also help in identifying those who are needing early initiation or reintroduction of immunosuppressants and biologics after an index cancer diagnosis.
Data from ongoing, adequately powered studies which are prospectively collecting data on disease phenotypes, treatments and past or incident cancers such as the European ICARE study
[ClinicalTrials.gov identifier: NCT02377258] will inform more clearly of safety in a patient with prior cancer, but in the interim, exercising caution in outpatients with a very recent history of cancer, particularly those with high-risk index cancers, is advised.
The overall purpose of this review was to summarize the evidence to help and guide in the problematic scenario of needing to make treatment decisions in patients with IBD in the setting of current or previous cancer. As detailed here, it is unlikely we will have direct evidence from randomized controlled trials and hence currently, we must rely on evidence from observational studies, with the caveat that observational studies are only ever a reflection of the clinical practice that led to that data. On the basis of the available data, a suggested stepwise approach to managing patients with prior history of cancer (Figure 2) and active cancer (Figure 3) is presented.
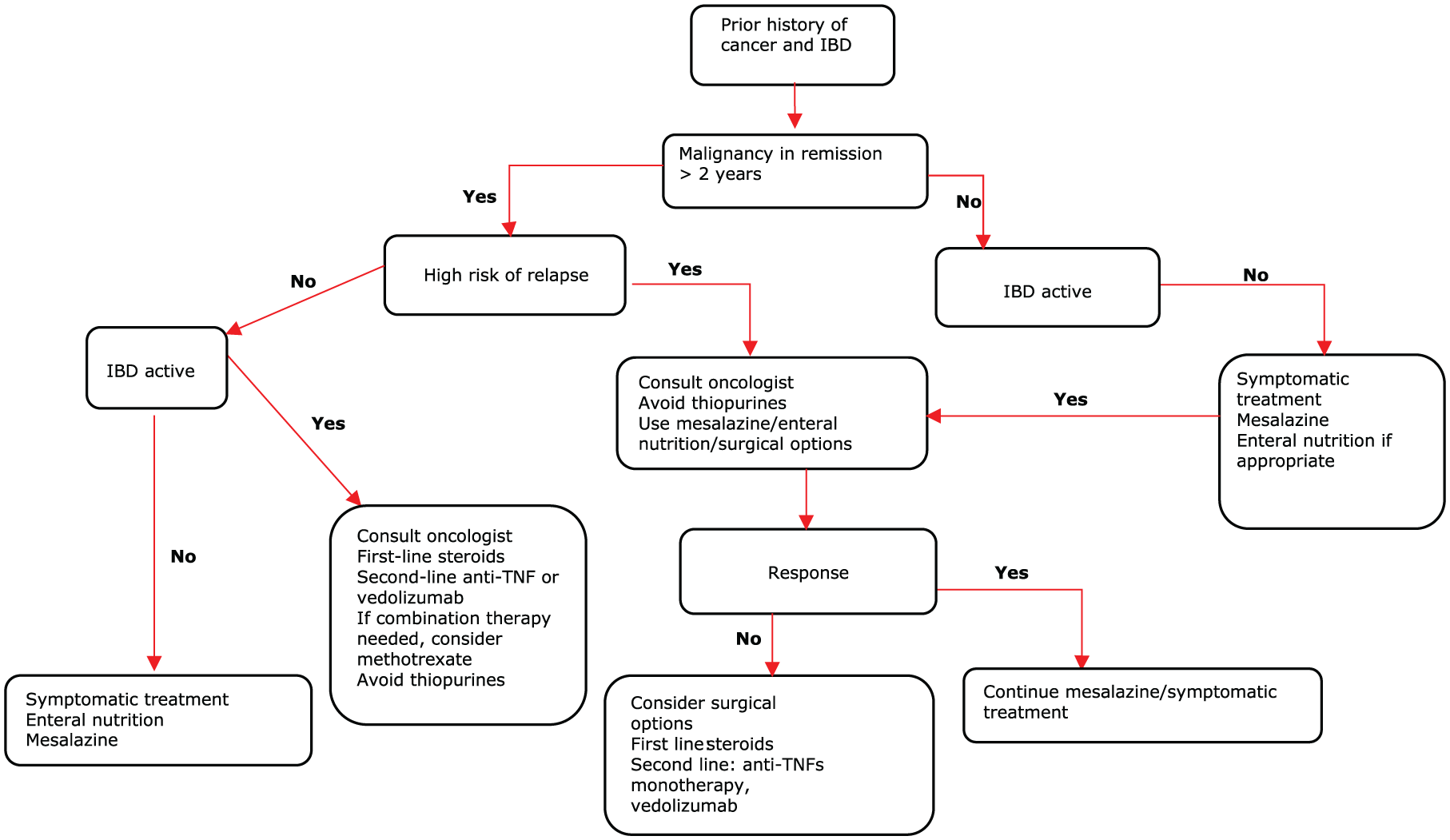
Suggested algorithm in IBD and previous cancer.

Suggested algorithm IBD and current cancer.
The first step in relation to this in a patient presenting with IBD is to ascertain current or previous cancer status. If there is current cancer, the clear priority will be to determine the treatment of cancer under the multidisciplinary team of oncology and surgery when appropriate. In addition to the effectiveness and safety of cancer therapy, due consideration should be given in relation to IBD about the risk of worsening the IBD with therapy and chemotherapy side effects which may mimic an IBD flare. Furthermore, the potential for cytotoxic therapy to maintain IBD remission should also be considered. With regards to existing IBD treatments in the setting of active cancer, it is advised to withhold immunosuppression, particularly thiopurines. For patients already on anti-TNFs, they can be continued except in the setting of melanoma if the IBD status risk assessment indicates high risk of IBD flare. With patients flaring during active cancer on treatment, first-line treatment should be conventional, using mesalazine (if appropriate), enteral nutrition (if appropriate), and steroids with immunosuppressants and biologics avoided. However, if there is nonresponse to steroids then biologics can be considered as second line in discussion with the oncologist. Given the gut selective mechanism, vedolizumab may be considered above anti TNFs, although the latter may be required for severe and refractory flares. Surgery should also be considered as an option.
In the setting of IBD diagnosis with a prior history of cancer, the three important considerations are the interval from the cancer treatment, the risk stratification of the cancer itself and the severity of IBD. It is essential to determine the duration of complete recovery from cancer following its treatment. Based on the available data from the observational studies, the meta-analysis and prior evidence from transplant literature, a minimum interval of 2 years is required before considering immunosuppressive and biologic therapy in cancers with low to intermediate risk of recurrence. The one possible exception will be NMSC which has been completely removed surgically, where methotrexate or anti-TNF monotherapy may be use as required without the need for a 2-year interval. For high-risk cancers with late chances of metastasis (breast, melanoma and renal cell carcinoma), a minimum interval of 5 years from recovery of cancer is required and an approach similar to that instituted for those with current or active cancer may be required in cancers with delayed risk of recurrence such as breast or renal cancers. After 2 years, the risk of malignancy recurrence or new malignancy appears favourable for methotrexate when compared with thiopurines. Thiopurines should only be considered if no other options are available and following opinion from an oncologist and also after the minimum interval following cancer treatment. Anti-TNFs, when used as monotherapy, appear to have a very favourable risk profile in relation to cancer development based on the current data from registries and observational studies in IBD and rheumatological disorders. In general, combination therapy with thiopurines in the setting of prior cancer should be avoided, and if combination therapy is essential, methotrexate, in combination with anti-TNFs, would be preferable to using thiopurines as combination therapy. While newer gut specific biologics appear an attractive option in this setting, firm conclusions on the role of specific biologics in relation to each other cannot be made, based on current data.
Summary and conclusion
Clinical guidelines based on expert consensus often recommend significant restrictions in medications, such as immunosuppression and biologics, and as a result, clinicians are historically reluctant to commence on immunosuppressive and biological therapy in patients with current cancer or prior history of cancer. Current evidence will favour a rationale for a 2-year drug holiday, with particular reference to thiopurines and cautious use of biologics in situations when IBD activity is significant. It is essential that a case-by-case decision in consultation with the oncologist, taking into account the risks of cancer progression and site-specific risk of recurrence, is warranted. An important aspect not to overlook is the potential, on treatment of IBD, for consequences, including future cancer risk. If continued treatment or initiation of treatment with anti-TNFs or immunosuppressants or both have no or limited impact or indeed a beneficial effect on cancer outcomes, it is difficult to justify depriving the severe IBD patients the effect they have on their disease.
Footnotes
Author Note
The author, Shaji Sebastian is also affiliated to IBD Unit, Department of Gastroenterology Hull York Medical School,Faculty of health Science, University of Hull and York, Hull, UK.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
