Abstract
Inflammatory bowel diseases (IBDs), primarily encompassing ulcerative colitis and Crohn’s disease, represent a challenging spectrum of disorders with a multifaceted pathogenesis. Despite the array of available treatments, a demand for novel therapeutic options persists to achieve remission in a broader patient population. Research findings indicate that relying solely on a single biologic drug may limit future treatment choices, prompting consideration for a more suitable shift from step-up to top-down strategies in certain cases. In the backdrop of advancing drug development, reimagining the application of existing therapies presents a promising avenue. Among these innovative approaches is combination therapy. This review explores the outcomes of recent randomized clinical trials, systematic reviews, and case studies, focusing on dual biologic therapy. It underscores the effectiveness, safety, and tolerability of combining two biologic drugs in IBD, providing insights into a potentially impactful treatment strategy.
Introduction
Inflammatory bowel diseases (IBDs) are entities with a complex, multifactorial pathogenesis which comprise mainly ulcerative colitis (UC) and Crohn’s disease (CD). Besides genetic and environmental factors, immunological elements are known to be a significant hallmark in the development and maintenance of inflammation in IBD. Cytokines such as interleukin 10 (IL-10), IL-5, IL-9, IL-13, and IL-33 are more characteristic of UC. In CD, substantial levels of IL-10, IL-12, IL-27, and interferon (IFN)-γ are expressed. IL-6, IL-12, IL-17, IL-21, IL-23, and tumor necrosis factor α (TNF-α) are not specific and are found in both diseases.1,2 Among the cytokines, TNF-α is regarded as the most important player that drives inflammation. 3 Anti-TNF-α therapy was the first biologic drug to be introduced to treat IBD. 4 More than 20 years ago, the first chimeric antibodies against TNF-α were used to successfully treat CD and UC. However, there have been a lot of developments since then, such as the implementation of biosimilars, the widening of our understanding of IBD, and the establishment of new therapeutic goals. Thus, up to 40% of IBD patients fail to respond to the induction therapy with anti-TNF-α biologics assessed with mucosal healing, 5 and 23%–46% of the patients have a disease relapse in the maintenance phase after the initial response. 6 The pathogenetic factors such as diet, pollution, genetics, and microbiota disturbances contribute to the immunological changes inducing intestinal inflammation. The broad spectrum of immunological mechanisms such as immune cell trafficking encompasses a wide variety of integrins (i.e., α4β7, α4β1, β2), selectins (i.e., CD62L), chemokines (i.e., CCR9), and other mediators (sphingosine-1 phosphate, S1P) and their receptors. The complex immunology of IBD 7 may be partially the reason why single-target biologic therapy often fails. With the abundance of other biologic or small molecule drugs, such as vedolizumab (VED, anti-integrin α4β7 antibody), mirikizumab (anti-IL-23 antibody), risankizumab (anti-IL-23A antibody), ustekinumab (UST, anti-IL-12 and -IL-23 antibody), tofacitinib, filgotinib, upadacitinib (janus kinase inhibitors), ozanimod, etrasimod (sphingosine-1-phosphate receptor agonist), and more in development, it is easier to navigate and rotate the drugs in patients.
Despite the large number of available drugs, there is still a need for novel drugs to achieve remission in a bigger portion of patients. In addition, in some cases, a change to the second biologic may not be as effective as it would be when used as a first-line therapy. 8 Alongside the development of new drugs, the innovative utilization of existing therapies seems to be a promising idea. Some of the possible approaches are combination therapies with immunosuppressive agents and biologic drugs, two biologic drugs of different mechanisms, or biologic drugs with small molecule drugs.
The combination therapy already has a history in the treatment of IBD. In some studies, combining two or more drugs simultaneously was found to be more efficient than a single-drug treatment. The concept of combining two biological drugs emerged when Sands et al. 9 performed the study in which three infusions of natalizumab (antibody to α4 integrin) or placebo were added to the treatment with infliximab (IFX) in CD patients with persistent active disease. The combination therapy appeared safe and well tolerated in spite of the risk of progressive multifocal encephalopathy reported in patients treated with natalizumab. Despite the promising results of this randomized controlled trial (RCT), more recent reports come mainly from case studies describing the use of dual biologic therapy in hard-to-treat or refractory IBD as a rescue treatment. Nowadays, dual biologic therapy is gaining more and more interest as new RCTs of the combinations of different drugs emerge with encouraging results. The most common combination reported in case reports is the intention to link anti-TNF-α antibodies with IL inhibitors. Particular interest lies in VED with anti-TNFs and other biologic drugs due to its favorable safety profile and relatively slow onset of action. Thus, the addition of the compound characterized by a more rapid clinical response (particularly in the induction phase) may increase the treatment efficacy without an unnecessary drug rotation.
In this review, we summarize the existing data on the efficacy and safety profile of the combined biologic therapy in IBD.
Search strategy
PubMed, Google Scholar, Wiley, Springer, Scopus, Embase, and Web of Science databases were systematically and extensively searched for the bibliography. Clinical trials were also searched using the ClinicalTrials.gov database. The search included all the studies published up to January 2024, using the following keywords, alone or in combination: inflammatory bowel disease, Crohn’s disease, ulcerative colitis, biologic therapy, dual biologic therapy, combination, effectiveness, safety, adverse effects, vedolizumab, ustekinumab, anti-TNF, infliximab, natalizumab, guselkumab, and adalimumab. Articles concerning small molecules, including JAK inhibitors or S1PR modulators, were excluded. The searches were filtered to include only studies published in English. The titles and the abstracts were independently screened by the investigators, and the selected papers were subsequently discussed with all the authors.
Such a selection process ensures that this review is based on relevant research. To date, only two randomized clinical trials have been published in this field. Most of the data come from case reports and observational studies, which offer a lower level of evidence, limiting the strength of conclusions that can be drawn. Nonetheless, we thoroughly examined each article, assessing the methodologies, results, and implications, which enabled us to summarize the current knowledge in the field, identify gaps, and propose directions for future research.
Potential beneficial mechanisms of the biologic combination therapy
The mechanisms of combination biologic therapy are based on drug interaction and patient-to-patient variability. 10 The beneficial impact of drug interaction includes additive and synergistic effects. The latter mechanism offers each patient a greater chance of clinical response to the assigned combination therapy by targeting different inflammatory pathways, which may be unobtainable in monotherapy.
The combination biologic therapy seems to be a favorable strategy in IBD patients with extraintestinal manifestations (EIMs), such as arthritis or psoriasis, to enhance the control of both luminal and extraintestinal disease activity. Indications for such a therapy can be directly borrowed from rheumatological and dermatological guidelines. According to the recommendations of the American College of Rheumatology, anti-TNF-α agents are the first-choice drugs in cases of spondyloarthritis, especially with the presence of axial involvement. 11 Combining the anti-TNF-α treatment with a gut-selective therapy like vedolizumab appears to be a reasonable strategy for IBD patients experiencing rheumatological EIMs. Anti-TNF-α, anti-IL-23, and anti-IL-17 therapies are all considered for the treatment of psoriasis and psoriatic arthritis. 12 Therefore, the combination of anti-TNF-α agents with the anti-IL-12/IL-23 agent UST may offer a promising treatment option for patients who develop paradoxical skin adverse events (AEs) from the anti-TNF-α therapy. Notably, this approach has been found effective in pediatric patients; however, in adult CD patients, it led to gastrointestinal remission but did not improve skin lesions.13,14
The main focus of interest in the combination biologic therapy includes blending the anti-TNF-α treatment with newer agents targeting interleukins (UST) or inhibiting integrin reactions (VED, natalizumab). The anti-TNF-α drugs (IFX, adalimumab, certolizumab pegol, golimumab (GOL)) are monoclonal antibodies that target TNF-α—a pro-inflammatory cytokine playing a central role in the IBD pathomechanism. Consequently, they induce the process of T-lymphocyte apoptosis in the gut mucosa or repress cytokine signaling.15,16 The anti-TNF-α agents are effective in remission induction and maintenance in both CD and UC. However, they are characterized by high rates of secondary response loss, mainly due to the development of anti-drug antibodies. 6 UST is a recombined human immunoglobulin that binds to the p40 subunit of IL-12 and IL-23. It modulates the function of Th1 and Th17 lymphocytes, influencing cytokine production. 17 Integrin antagonists are monoclonal antibodies designed to decrease the infiltration of lymphocytes into the gut endothelium by blocking the interaction between lymphocyte integrins and their endothelial ligands. VED selectively binds to the α4β7 integrin, preventing its interaction with mucosal addressin cell adhesion molecule 1 (MAdCAM-1). Natalizumab’s function is less specific, as it inhibits the binding of both α4β7 integrin to MAdCAM-1 and α4β1 integrin to vascular cell adhesion molecule 1.18,19 Figure 1 depicts the mechanisms of the chosen biologic drugs.
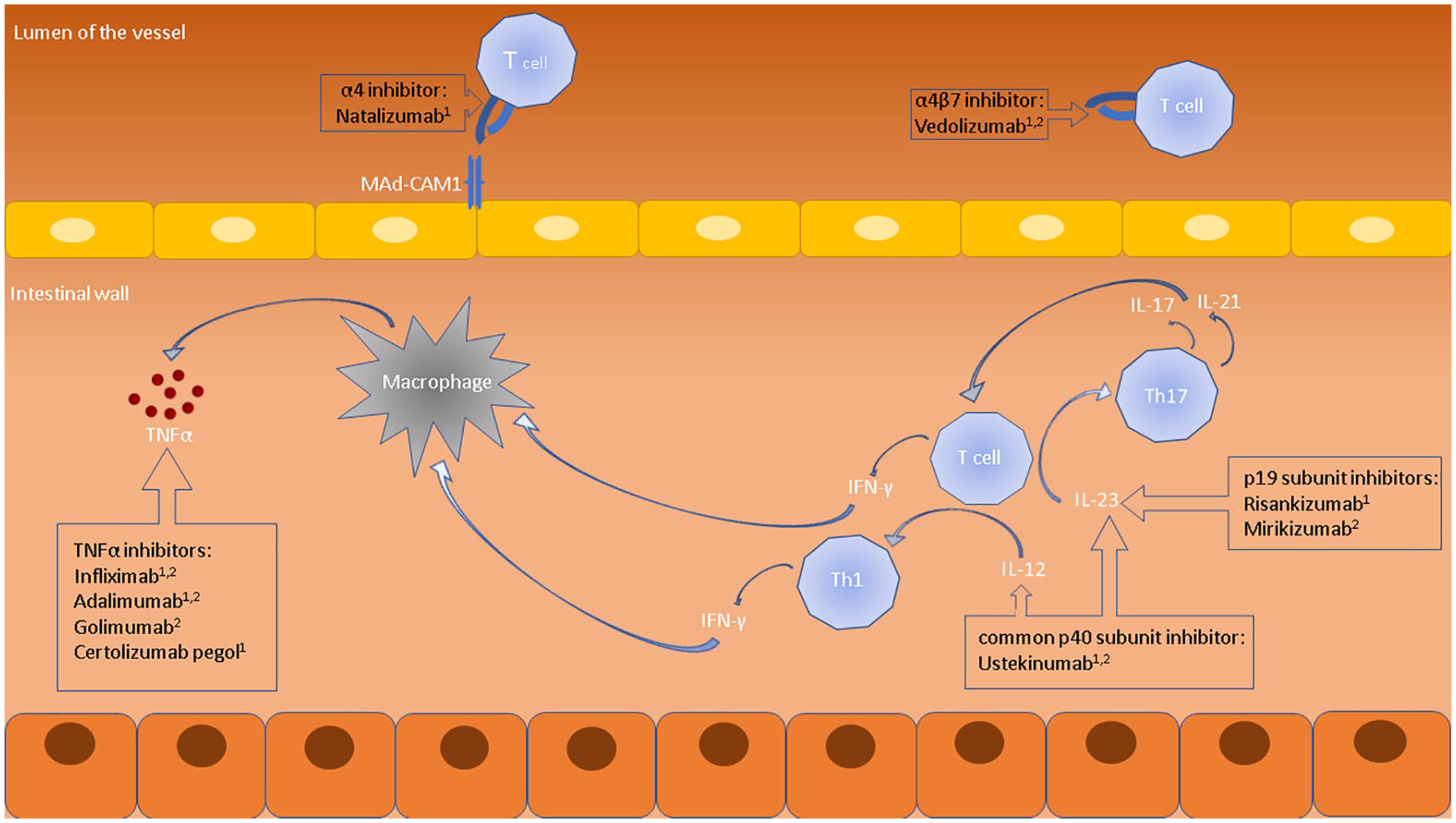
Available drugs and their mechanisms of action.
A potential synergism of biologics would be particularly beneficial in anti-TNF-α refractory disease. The first widely applied combination therapy joining IFX with immunosuppressors, due to their improving effect on pharmacokinetics of anti-TNF-α agents. The addition of immunosuppressants such as thiopurines or methotrexate reduces the immunogenicity of anti-TNF-α antibodies, leading to higher serum drug concentrations.6,20 Evidence-based data were revealed in 2010 when the results of the SONIC trial were shown. 21 According to Colombel et al., 21 combining IFX with azathioprine was found to be superior to IFX alone in patients with CD, leading to a corticosteroid-free remission in a higher percentage of patients. This approach was implemented into practice and guidelines and is effectively used worldwide. IFX is characterized by higher immunogenicity rates compared to other anti-TNF-α agents; thus, the combination therapy with IFX seems to optimize the effectiveness of such a treatment. Schmitt et al. found that CD patients not responding to the anti-TNF-α therapy demonstrated a significant upregulation of the mucosal cytokines: IL-23p19, IL-23R, and IL-17A expression. High levels of IL-23 produced by intestinal CD14+ macrophages inhibit anti-TNF-α-induced apoptosis in mucosal T cells, preserving inflammation. Therefore, IL-23 may serve as a suitable molecular target in reducing resistance to anti-TNF-α therapy in CD patients. 22
The mechanisms of action of the anti-TNF-α agents and integrin antagonists provide a potential combination of these two therapies for sequential use. The anti-TNF-α therapy may be applied to induce inflammatory clearance through apoptosis, followed by the treatment with integrin antagonists. This precludes the inflammatory processes from perpetuating by blocking the infiltration of additional inflammatory cells in the gut mucosa. A common target of action for these two therapies is the MAdCAM-1 molecule. Clinical data have shown that the anti-TNF-α therapy leads to the downregulation of MAdCAM-1 in CD patients. 23 Furthermore, individuals who do not respond to IFX or adalimumab exhibit a high expression of MAdCAM-1, indicating a potential need for co-treatment with integrin antagonists. 24 Due to its gut selectivity and favorable safety profile, vedolizumab (VDZ) appears to be a preferable co-therapy agent, enhancing the efficacy of anti-inflammatory treatment without substantially increasing the infection risk. In addition, given VDZ’s slower onset of action compared to IFX, the initial induction treatment with systemic anti-TNF-α agents may serve as a bridge to remission, which can then be maintained with gut-specific VDZ monotherapy.
Data on the combination therapy of biologics and small molecules remain limited due to their relatively recent use in IBD treatment. However, a recent observational study by Miyatani et al. 25 retrospectively assessed the effectiveness of upadacitinib combined with UST in 10 patients with refractory CD. The indications for the combination therapy included active CD (n = 6), EIMs (joint pain; n = 2), or a combination of both (n = 2). The results showed that five out of six patients achieved clinical remission, with two arthritis patients experiencing the resolution of joint symptoms and one patient showing improvement in both conditions. Given that upadacitinib is effective for disorders such as rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, non-radiographic axial spondyloarthritis, and atopic dermatitis, adding it to the therapy may offer a promising option for IBD patients with EIMs, particularly joint-related symptoms. Notably, upadacitinib has the most rapid onset of action, reducing IBD symptoms as early as day 1 of the induction treatment. 26 Thus, utilizing upadacitinib alongside a slower-acting agent, for example, vedolizumab, during the induction period could enhance treatment outcomes, particularly in patients with contraindications to systemic steroids.
As data on the combination biologic therapy are still scarce, it is not clear whether the potentially advantageous blend of two biologics refers to a specific synergistic action or cumulative effect of the intensified drug therapy.
Combination of TNF-α inhibitors with VED, UST, natalizumab, or guselkumab
Four systematic reviews assessing the combination biologic therapy in IBD have been published. In 2019, Ribaldone et al. 27 conducted a systematic review with a pooled analysis comprising 7 studies, including 18 patients (44% with active UC). Fifteen of them were treated with a combination of a TNF-α inhibitor and VED, with combination therapy duration ranging from 6 to 21 months. Clinical improvement was observed in all the patients, and 93% showed endoscopic improvement suggesting the utility of combination biologic treatment in IBD. 27 A systematic review with a meta-analysis conducted by Ahmed et al. 28 in 2022 confirmed the usefulness of combination biologic treatment as a highly selected therapeutic option for refractory IBD patients. The study, with a 24-week median treatment duration, included various combination therapies. The pooled rates of clinical and endoscopic remission were 59% (95% CI, 42%–74%) and 34% (95% CI, 23%–46%), respectively, during a median follow-up of 32 weeks. Remarkably, better responses were observed when the combination therapy was used for concomitant EIMs rather than refractory intestinal disease. 28 Alayo et al. 29 performed another systematic review with a meta-analysis to evaluate the safety and effectiveness of combining biologics in IBD. The most common combination biologic therapy was a TNF-α inhibitor combined with VED. The pooled rates of clinical response and remission among subjects treated with VED/TNF-α inhibitor combined treatment were 77.9% (95% CI, 51.3–97.2; 8 studies; 53 therapeutic trials (TTs); I2 66%) and 55.1% (95% CI, 19.6–88.5; 8 studies; 53 TTs; I2 81%), respectively. By contrast, the pooled rates of endoscopic/radiologic response and remission were 38.2% (95% CI, 19.5–58.4; 5 studies; 35 TTs; I2 0%) and 18.0% (95% CI, 1.6–41.8; 5 studies; 35 TTs; I2 32%), respectively. However, the authors emphasized the low certainty of results due to the observational nature of the studies and significant imprecision regarding the GRADE framework. Berinstein et al. 30 concluded, based on their systematic review, that combining biologic therapies may be a promising option for partially or non-responsive patients with IBD. However, the study’s results were primarily based on low-quality case reports and case series, posing a risk of article bias favoring extreme evidence. The first multicenter RCT was published in 2007 by Sands et al. 9 In this RCT, the researchers determined the safety, tolerability, and efficacy of IFX combined with natalizumab in subjects with active CD despite the ongoing therapy with IFX solely. Seventy-nine patients were randomly assigned to obtain three intravenous infusions of natalizumab 300 mg (52 patients) or placebo (27 patients) every 4 weeks. Moreover, the IFX was administered at the dose of 5 mg/kg intravenously every 8 weeks for at least 10 weeks before randomization and throughout the trial. Although patients receiving the combination biologic therapy showed a trend toward clinical improvement, the differences were not statistically significant. It was demonstrated by a mean decrease in Crohn’s disease activity index (CDAI), while the IFX monotherapy did not reduce CDAI (−37.7 vs +3.5; p = 0.084). In addition, the patients who were administered the combination biologic therapy achieved remission more frequently compared to the placebo group at any time (46% vs 41%). In week 6, the mean rise in Inflammatory Bowel Disease Questionnaire (IBDQ) score was moderately higher in the group with natalizumab and IFX compared to the group receiving only IFX (12.3 vs 9.1; p < 0.605), and in week 10 the results were similar in both groups (18.7 vs 17.3; p = 0.811). Interestingly, the mean platelet count, a marker of the inflammatory response, decreased in the group receiving IFX and natalizumab, while it remained relatively unchanged in the patients treated with IFX alone. 9 In the largest randomized, double-blind, controlled, phase II trial (NCT03662542), Feagan et al. 31 assessed the combination therapy with golimumab and guselkumab (GUS) in moderately to severely active UC. The trial participants were not previously treated with TNF, IL-12/23, or IL-23p19 antagonists, and were characterized by an inadequate response or intolerance to the oral or intravenous corticosteroids or immunosuppressants. An earlier therapy with VED was permitted if it was discontinued for ⩾18 weeks prior to week 0. The scheme of combination therapy comprised subcutaneous golimumab 200 mg at week 0 followed by subcutaneous golimumab 100 mg at weeks 2, 6, and 10, and intravenous guselkumab 200 mg at weeks 0, 4, and 8, followed by subcutaneous guselkumab monotherapy 100 mg every 8 weeks for 32 weeks. The monotherapy with golimumab was subcutaneously administered at the dose of 200 mg at week 0 followed by a dose of 100 mg at week 2 and every 4 weeks thereafter for 34 weeks. In turn, the monotherapy with guselkumab was intravenously applied at the dose of 200 mg at week 0/4/8, followed by subcutaneous administration of 100 mg every 8 weeks thereafter for 32 weeks. The combination biologic therapy with golimumab and guselkumab demonstrated higher efficacy compared to the treatment with either antibody alone, with 83% of subjects achieving a ⩾30% decrease from baseline in the full Mayo score at week 12 (adjusted treatment difference to golimumab monotherapy 22.1% (80% CI 12.9–31.3); nominal p = 0.0032 and 8.5% (−0.2 to 17.1); nominal p = 0.2155 to guselkumab). 31
Retrospective studies on combination biologic therapy in IBD are limited but provide valuable insights. Yang et al. 32 studied patients with refractory CD who received combination biologic therapy with VED, UST, or IFX. The combination of anti-TNF-α with VED or UST showed endoscopic improvement (defined as > 50% reduction in Simplified Endoscopic Score-Crohn’s disease (SES-CD)) in 33% of trials and endoscopic remission (defined as SES-CD < 3) in 27% of trials. Clinical response was observed in 40% and clinical remission, defined by CD-PRO-2 < 8, was seen in 33% of trials. 30 Moreover, the improvement in aforementioned outcomes was accompanied by a decrease in CRP level (pre-treatment 17.0 (11.0–24.0) to post-treatment 9.0 (4.0–14.0), p = 0.02), indicating that combination biologic therapy may be the promising option for selected patients with refractory CD. A retrospective study by Privitera et al. 33 assessed the efficiency of the combination targeted biologic therapy in Italian patients with refractory IBD. Nine patients received TNF-α inhibitor combined with VED or UST, showing clinical response in all patients by the end of the induction. 33 Nine patients, including seven with CD, were administered combination targeted therapy with TNF-α inhibitor (certolizumab at the dose of 200 mg every 4 weeks, adalimumab at the dose of 40 mg every week, or IFX in the dose of 5 mg/kg every 8 weeks) combined with either VED (300 mg every 4 or 8 weeks) or UST (90 mg every 8 or 12 weeks). The duration of the therapy differed between the cases and ranged from 4 to 28 months. A similar retrospective multicenter study in Finland by Eronen et al. 34 focused on patients with CD who failed three to five different monotherapies with biologics. The combination of adalimumab with UST was the most commonly used therapeutic option and the majority of patients manifesting a response to combination biologic therapy were treated with these agents (56%). Altogether four patients among the subjects (4/11; 36%) were in clinical and endoscopic remission at the end of the follow-up and in one case response to the combination biologic therapy was partial with the relief of clinical symptoms. These changes were accompanied by a statistically significant reduction in fecal calprotectin concentration. 34 Kwapisz et al. 35 conducted a medical record-based retrospective study at Mayo Clinic, where the established CD or UC patients were treated with IFX, adalimumab, certolizumab pegol, golimumab, VED, or UST in combination. The disease worsening was the indication for combination therapy, and all the subjects were naïve to the changed, second biologic medication. Ten of 15 patients received the TNF-α antibody with VED or UST, resulting in symptomatic improvement, clinical response, and dose reduction of corticosteroids in the majority of patients. Among the TNF-α antibodies, golimumab appeared to be the most effective (100%). Unfortunately, the authors did not reveal detailed data about the doses and frequency of the administered drugs. 35 A European retrospective observational study by Goessens et al. 36 assessed the utility of combination biologic therapy in three groups of patients: those with active IBD, those with active IBD concomitant with active immune-mediated inflammatory diseases/EIM, or those with active immune-mediated inflammatory diseases/EIM irrespective of IBD activity. The combination of the anti-TNF-α with the anti-integrin biologics was the most effective therapeutic option, inducing complete or partial endoscopic (reduction in SES-CD/Mayo score) improvement in both CD (64%) and UC (73%). 36
RCTs and observational studies on combination biologic therapy in IBD are still limited, and most of the available results come from case series and case reports. Notable case reports include VED with golimumab in ileocolonic and perianal CD, VED with golimumab in severe UC associated with spondyloarthropathy, and VED with IFX or adalimumab in extensive colitis in UC after the failure of the TNF-α antibody.37–39 However, more recent and interesting studies have been detailed in the current paper. In 2020, Fumery et al. 40 presented their experience in combination biologic therapy in seven patients with IBD, including five with a combination of the TNF-α antibody (golimumab, adalimumab, or IFX) and UST. The luminal and perianal CD, the coexisting of CD, and ankylosing spondylitis, as well as the presence of paradoxical AEs, were the indications to use the combination therapy. Four of five patients receiving the combination biologic therapy achieved steroid-free, clinical remission, with the duration of the biologic combination ranging from 3 to 30 months. 40 In 2022, Abdullah et al. 41 published a case series assessing the efficacy of combination biologic or small molecule therapy for achieving endoscopic remission in seven patients with refractory IBD. TNF-α antibody with VED was administered in three subjects with CD. The therapies resulted in steroid-free clinical remission (2, 2, and 3 points in Harvey–Bradshaw Index for the first, second, and third patients, respectively) and endoscopic remission (2, 1, and 1 point in SES-CD for the first, second, and third subjects, respectively), indicating that combining therapies may be a valuable option in critical conditions and complications of CD. 41 Le et al. 42 presented the case of a 38-year-old man with refractory CD who experienced complications, including right hemicolectomy or surgical interventions due to colonic obstruction, enterocutaneous fistulas, and perforations. In addition, the patient was a primary non-responder to the treatment with VED and adalimumab. Switching to the combination biologic therapy with IFX and UST resulted in steroid-free clinical remission, suggesting that combining therapies may be a valuable option in critical conditions and complications of CD. 42
At least two prospective trials (Clinical Trials.gov Identifier: NCT02764762 43 and NCT00055536) were conducted to assess the effectiveness and safety of combination biologic therapy with TNF-α antibody. In the NCT00055536 phase II trial, the combination of IFX and natalizumab was tested in the individuals diagnosed with active CD currently taking IFX. Unfortunately, detailed results were not posted. In the NCT02764762 open-label phase IV study, the efficacy and safety of triple combination therapy with intravenous VED, subcutaneous adalmumab, and oral methotrexate in the early treatment of patients with CD were assessed. The results demonstrated that 54.5% of the participants achieved clinical remission and 34.5% achieved endoscopic response (defined as CDAI score < 150 and SES-CD ⩽ 2, respectively) at week 26. 43
Combination of UST and VED
The literature review indicates that the majority of adult patients with dual biologic therapy implemented for IBD received anti-TNF-α with either VED or UST. The combination of UST with VED is a less common therapeutic option, likely due to the broader licensed therapeutic indications of TNF-α antibodies compared to VED and UST. However, the ongoing studies suggest that combining VED and UST may be a valuable option due to their greater safety profile and lesser concern about loss of response.44,45 A systematic review with a meta-analysis from 2022 demonstrated that the combination of UST and VED may be an effective form of treatment for patients with refractory IBD. The pooled clinical response and remission rates among the subjects treated with VED and UST were 83.9% (95% CI, 66.4–96.8; 7 studies; 38 TTs; I2 0%) and 47.0% (95% CI, 14.5–80.7; 7 studies; 38 TTs; I2 64%), respectively. 29 A retrospective cohort study on patients with refractory CD revealed that the combination of UST and VED after inefficient therapy with VED, UST, or IFX contributed to the improvement of the disease’s clinical course. Endoscopic improvement was achieved in 63% of the combination biologic trials, endoscopic remission occurred in 25% of the trials, and clinical response was observed in 71% of the combination therapy trials. 32 Another retrospective cohort study assessing the use of combination biologic or small molecule therapy in IBD demonstrated that this therapeutic option may be effective for subjects with refractory disease or concomitant autoimmune diseases inadequately controlled by a biologic monotherapy. UST and VED were the most commonly used combination, with 47.2% of subjects receiving this combination biologic therapy. The median duration of combination therapies was 8 months, and all the subjects’ cumulative outcomes showed more frequent clinical and endoscopic remission at follow-up compared to baseline. 46
Biscaglia et al. 47 presented two case reports demonstrating the utility of VED in combination with UST in patients with IBD and dermatological comorbidities. The 24-month combination therapy with VED and UST contributed to the remission of disease activity and psoriasis in adult male patients with CD without causing adverse effects. In a 57-year-old man with UC and psoriasis, the combination of VED and UST resulted in remission of diseases after a 21-month treatment with no observed side effects. 47 Several case reports also present the efficacy of VED with UST in mitigating the disease course, inducing clinical and endoscopic remission, and causing colonic mucosa healing. These cases include refractory, aggressive penetrating CD with enterocutaneous perianal fistulas, and refractory ileocolonic CD after the failure of various biologics.41,48,49
Selected studies investigating the effectiveness of combination biologic therapy in IBD are summarized in Table 1.
Studies investigating the effectiveness and safety of dual biologic therapy in IBD.

ADA, adalimumab; APR, apremilast; CD, Crohn’s disease; CERT, certolizumab; DBT, double biological therapy; ETA, etanercept; FC, fecal calprotectin; GOL, golimumab; GUS, guselkumab; IBD, inflammatory bowel disease; IFX, infliximab; IQR, interquartile range; MD, median; MTX, methotrexate; NAT, natalizumab; OCRE, ocrelizumab; SEC, secukinumab; TNF-α, tumor necrosis factor α; UC, ulcerative colitis; UST, ustekinumab; VED, vedolizumab.
Safety profile of combination biologic therapy
Despite the fact that safety analyses were performed in all the aforementioned studies, they are incomparable. The occurrence rate differs from 0 (when including case studies) to 92% (in RCT performed by Sands et al. 9 ). Only two RCTs have been performed until now9,31 while others were observational or case studies. Strict AE reporting in RCTs is not relatable to reporting in the latter studies. However, in RCT by Sands et al., 9 the safety of combination therapy is apparent as 92% (48/52) of the patients receiving natalizumab and IFX experienced AEs compared to 100% (27/27) of the patients receiving placebo and IFX. The most common AE reported in both groups was a headache. Also, the proportion of infection-associated AEs in both groups was similar: 27% (14/52) in the experimental arm versus 30% (8/27) in the placebo arm. Similar results were reported in the most recent RCT performed by Feagan et al. 31 where the patients were treated with golimumab and guselkumab solely or in combination. AE analyses showed that 76% of the patients (55/72) in the GOL group and 65% of the patients (46/71) in the GUS group reported at least one AE compared to 63% of the patients (45/71) in the combination therapy group during the 50-week therapy. The most common AEs among any group were infections. Notably, infections are one of the most common events that the patients report during biologic therapy outside research, in clinical practice. In the meta-analysis by Bonovas et al., 56 the use of biologic agents of any kind was associated with a significant risk of developing the infection among the patients with IBD (OR, 1.19; 95% CI 1.10–1.29) and opportunistic infection (OR, 1.90; 95% CI, 1.21–3.01). Contrarily, the risk of developing malignancy was not profound in the assessed studies (OR, 0.90; 95% CI, 0.54–1.50). Whether it is relatable to combination biologic therapy is questionable; however, in the RCT by Sands et al. 9 neither opportunistic infections nor lymphomas were noted either in the interventional or placebo group. It seems that a greater risk of developing an AE (an infection in particular) may be induced by the combination of a biological drug with another immunosuppressive agent rather than by the addition of another biological. In a retrospective cohort study by Glassner et al. 46 among the 23 AEs, 9 AEs (39%) occurred in the patients who were administered immunomodulator in addition to the combination biological/biological + small molecule therapy and 18 AEs (78%) in those who were also taking steroids. Hence, the earlier withdrawal of immunomodulators or quicker tapering of steroids should be taken into account when considering combination biologic therapy. The specific AE distribution in the studies reported in the literature is shown in Table 2.
AEs reported in the studies on dual biologic therapy in IBD.
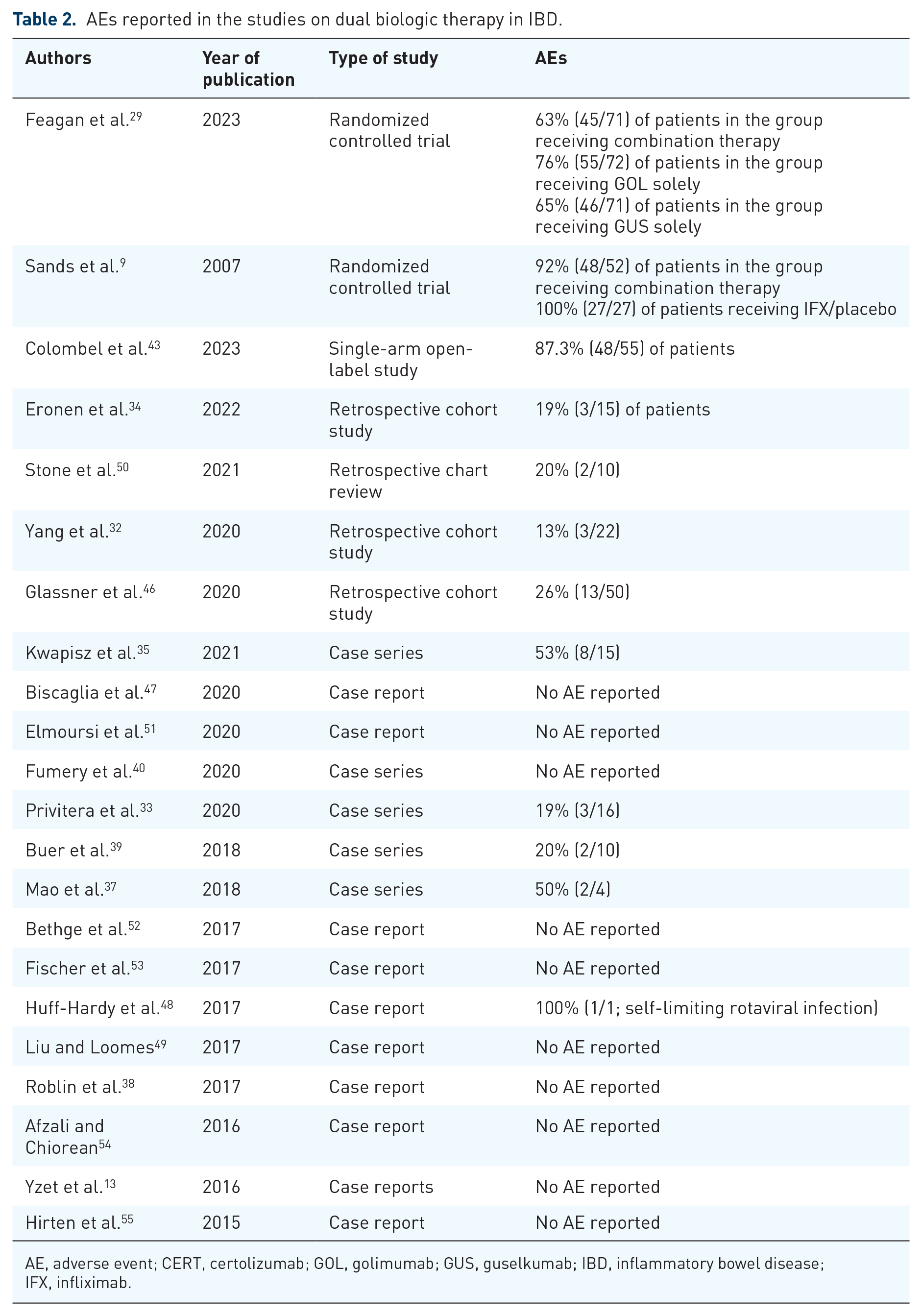
AE, adverse event; CERT, certolizumab; GOL, golimumab; GUS, guselkumab; IBD, inflammatory bowel disease; IFX, infliximab.
The safety of combination biologic therapy is further underscored by the attempts to implement it in pediatric patients with IBD. In 2022, Wlazlo et al. 57 reported the results of a study in which a combination therapy of anti-TNF-α with either VED or UST was utilized in 14 children following the failure of monotherapy with a single biologic agent. Over the course of 4 months, three patients experienced AEs (21.4%), including anal abscess, cardiac complications following COVID-19 infection, and colectomy. The age range of the patients in the aforementioned study was 3–17, indicating that it is relatively safe to initiate combination biologic therapy as early as possible to mitigate complications typical of pediatric patients with IBD. It is worth noting that the observation period in the study was short (4 months); thus, further RCTs are necessary to evaluate the long-term efficacy and safety.
It remains questionable whether the combined therapy induced any changes in fertility and pregnancy. As more and more is known about the safety of biologic monotherapy in men and women in the reproductive age, this issue will eventually arise. To date, no data have been known on the possible disturbances in the reproductive system in patients with IBD treated with a combination therapy. The analyses of 1850 cases of maternally IFX-exposed pregnancies showed that the prevalence of complicated pregnancy or detrimental infant outcomes (up to the age of 2) did not differ from the general population. 58 A similar safety profile is expected from VDZ and UST, conversely to JAK inhibitors which have shown embryotoxicity and teratogenicity in preclinical studies. 59 Although combining two drugs that already have a limited but known safety profile in pregnant women would be predicted not to induce any harm, mechanisms including the cross-talk on a molecular level, idiosyncrasy, and others have to be taken into consideration. Thus, further studies should address this important issue as most IBD patients are of reproductive age.
Financial aspects and insurance issues
Quoting the analysis by Wang et al., in 2019 there were 4.9 million cases of IBDs worldwide and this number is constantly increasing. 60 Therefore, the cost of the maintenance of these diseases and their complications significantly burdens global healthcare systems. According to the population-based study among 1289 European patients with IBDs published by Burisch et al. 61 in 2020, the mean cost of the treatment per patient-year was €2609. Inceptively, hospitalizations and diagnostic procedures accounted for more than 50% of healthcare costs. However, in subsequent years after the diagnosis, increasing expenditure on biologics was observed, reaching 73% of spending in CD and 48% in UC in the fifth year of the follow-up. The dominance of anti-TNF-α therapy in healthcare costs was already noticed a few years earlier by van der Vack et al. 62 According to their study, the incremental use of biological therapy in IBDs led to the shift in medical spending, resulting in the reduction of the participation of hospitalizations and surgery in the healthcare costs. Interestingly, a systemic review by van Linschoten et al. 63 proved that the expected decrease in inpatient costs was not achieved despite the higher clinical effectiveness of biologics over non-biologic therapy. Moreover, Feng et al. 64 upon the end of their cohort study noted that the introduction of the biosimilars was not clearly associated with the lowering of out-of-pocket costs for commercially insured patients.
The financial aspect might be the factor limiting the availability of combination biologic therapy, especially in countries in which the healthcare system is largely dependent on private insurance facilities. In 2017, an analysis was carried out by Yadav et al. 65 on the topic of American insurance policies for patients with IBD. According to the report, most of the policies did not comply with the current American Gastrointestinal Association guidelines for treating CD and UC. Ninety-eight percent of the policies required the preceding failure of conventional drugs before the approval of an anti-TNF therapy.
To date, no data in the context of the funding of implementation of the combination biologic therapy have been available. However, judging by the foregoing approach of the insurance companies, access to this modern therapy might be restricted due to financial reasons.
Future directions
One of the challenges that every clinician faces in case of a refractory disease is the selection of a balanced therapy that takes both the established effectiveness and acceptable safety profile into consideration. For numerous patients, biologic therapy remains the last resort treatment before surgery. Unfortunately, there is still a vast cohort of patients who are primarily not responsive to the monotherapy with currently available drugs or who lost the response to them after the initial improvement. A potential solution to this issue is the development of new biologic drugs but until then, new applications of already existing agents are required.
The first remarks regarding the combination biologic therapy in IBD emerged over a dozen years ago, yet the combination of two biologics has not been commonly applied so far. Hopefully, in the near future, thanks to the expanding data, the qualification of eligible subjects for combination biologic therapy will be more ubiquitous. Interestingly, the clinical trial NCT06453317 aiming at the assessment of the effectiveness and safety of the combination therapy with IFX and UST began in July 2024 at the Medical University of Lodz, Poland.
Another factor that might influence the availability of combination biologic therapy in the forthcoming years is the introduction of biosimilars after the expiration of patent protection of original drugs. According to the analysis published recently by Chen et al., 66 the advent of biosimilars has led to a substantial decrease in the cost of biologic medications worldwide, including the United States.
Conclusion
As the number of IBD patients with primary or secondary refractory disease is constantly increasing, new approaches in the therapy are required. According to the most up-to-date studies, combination biologic therapy might be one of the most promising directions in modern treatment, as it proved to be both effective and safe in comparison to biologic monotherapy.
The findings suggest that combining biologics with different mechanisms of action, for example, anti-TNF-α antibodies with anti-integrin or anti-interleukin agents, may provide more patient-tailored care aimed at a broad spectrum of symptoms, both luminal and extraintestinal. Fortunately, in the last decade, the number of available biologics has increased; therefore, numerous combinations are feasible. Most certainly, in the following years, the possible conjoinings will be even more multitudinous thanks to the continual development of new agents.
However, the implementation of the combination biologic treatment might be limited due to financial issues, as the cost of production of biologics is one of the highest among drugs available for the therapy of IBDs. The impact of this factor might be particularly significant in countries where the healthcare system is based on a mix of private facilities, public programs, and out-of-pocket payments.
Due to the fact that the majority of data so far have come from retrospective studies or case reports, further research in the area of combination biologic therapy is needed. In particular, randomized clinical trials are desirable to fully establish the optimal combinations of biologic agents regarding their safety, efficacy, and availability among different subgroups of IBD patients.
