Abstract
Background:
Peripheral blood lymphocytes play an important role in antitumour immunity. We examined the relationship between the minimum absolute lymphocyte counts (Min ALCs) during radiotherapy (RT) and clinical outcomes in patients with hepatocellular carcinoma (HCC).
Methods:
Data from a total of 69 HCC patients who had received RT were retrospectively analysed. Peripheral blood lymphocytes were measured before RT, weekly during RT and after RT. Regression and mixed-effect models were used to assess the relationships with and potential predictors of overall survival (OS). Receiver-operating characteristic (ROC) curve analysis was used to define optimal cut-off points of continuous variables for outcomes.
Results:
The median follow up was 30 months (range, 4–68 months). The median survival time (MST), 1-year OS rate and 2-year OS rate of the whole group were 25 months, 51% and 39%, respectively. The average circulating lymphocyte counts declined during RT (1493.19 versus 503.48 cells/µl, p < 0.001). A lower Min ALC was associated with worse OS (p = 0.001), with a cut-off value of 450 cells/µl (sensitivity and specificity, 50% and 70.6%, respectively). The MSTs, 1-year OS rates and 2-year OS rates were 15 months versus 47 months, 27% versus 78% and 4% versus 71% for patients with relatively lower (⩽450 cells/µl) and higher Min ALCs (>450 cells/µl), respectively (p < 0.001). After adjusting for potential confounders, multivariate Cox regression analysis demonstrated that Min ALC independently predicted patients’ OS (HR, 0.32; 95% CI, 0.15–0.69).
Conclusions:
Lower Min ALCs during RT may act as a worse prognostic factor for HCC after RT.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is one of the leading contributors to cancer incidence and mortality worldwide [Torre et al. 2015] and is particularly prevalent in China, given the recent rise in chronic hepatitis B virus (HBV) infection and cirrhosis [Cui and Jia, 2013]. Surgical resection is a major treatment for patients with early- and middle-stage HCC that produces good results. However, the majority of patients are medically inoperable at the time of diagnosis, and few meaningful therapeutic options are currently available. Traditionally, radiotherapy (RT) was infrequently used for treatment of HCC due to early reports that the radiation tolerance of the liver was far less than the therapeutic radiation dose [Koo et al. 2010]. Since three-dimensional conformal RT (3DCRT) was introduced for the treatment of HCC, it has achieved encouraging clinical outcomes [Sharma, 2014]. Due to its increased benefits, RT has been recommended for the treatment of all HCC tumours irrespective of their location (National Comprehensive Cancer Network Guidelines Version 1.2015).
Lymphocytes, which belong to a population of radiation sensitive cells, account for approximately 30% of the normal human white blood cell population and play an important role in antitumour immunity [Topalian et al. 2011]. In a study by Nagai and colleagues, peritransplant lymphopenia was found to predict HCC recurrence after liver transplantation [Nagai et al. 2014]. A low absolute lymphocyte count (ALC) was also used as an indicator of malnutrition and poor immune response in patients with chronic liver disease [Fernandez-Ruiz et al. 2009]. In addition, increasing evidence has shown that radiation-related lymphopenia is correlated with tumour progression and prognosis in several types of advanced cancers [Kitayama et al. 2011; Cho et al. 2016a, 2016b]. From these studies, we can hypothesize that peripheral ALCs will decline during RT, and the degree of decline may exhibit some clinical significance. However, the relationship between the minimum absolute lymphocyte count (Min ALC) during RT and clinical outcomes in patients with HCC has not been characterized. Therefore, the aim of this retrospective analysis was to validate our hypotheses and to investigate the clinical significance of Min ALCs during RT in HCC patients.
Methods
Study populations
The study was conducted with the approval of the Shandong Tumour Prevention and Control Institutional Ethics Committee, Shandong Cancer Hospital affiliated to Shandong University. Written informed consent was obtained from the patients or their legal representatives. Data from patients who were diagnosed with HCC based on pathology results or who were diagnosed via the diagnostic criteria of the American Association of the Study of Liver Guideline [Fitzmorris et al. 2015] at Shandong Cancer Hospital affiliated to Shandong University between January 2009 and January 2015 were retrospectively analysed. The clinical data were obtained from medical records. All patients satisfied the following inclusion criteria: (i) ⩾18 years of age; (ii) Eastern Cooperative Oncology Group performance status of 0–1 [Gil-Alzugaray et al. 2013]; (iii) received RT with a conventional fraction at our institution; (iv) available baseline clinical, laboratory and follow-up data; (v) the peripheral white blood cell counts were all above 2000 cells/µl during RT treatment and didn’t receive any prophylactic or remedial treatment; and (vi) a survival time after RT of at least 3 months. Patients were excluded if they had a history of other malignancies, failed to complete RT, had evidence of distant metastasis, had received surgical treatment for HCC, had splenomegaly, a fever or an elevation of C-reactive protein during RT or had an HBV reactivation during RT that was attributed to HBV infection. A total of 69 patients with HCC were finally included and evaluated. All patients had undergone a standardized evaluation, including abdominal computed tomography (CT) scanning, chest X-ray or CT, brain magnetic resonance imaging (MRI) and bone radionuclide imaging, and the disease stage was identified according to the Barcelona Clinic Liver Cancer (BCLC) system. RT was delivered via sliding window 3DCRT or intensity-modulated radiation therapy. A total dose of 39.60–63.48 Gy (Gray) was administered at 1.8–2.0 Gy per fraction for 5 days a week. The gross tumour volume was delineated by CT according to the primary lesion or the lipiodol deposit from transarterial chemoembolization (TACE) and the positive enlargement of retroperitoneal lymph nodes. All plans were completed using the Varian Eclipse version 8.6.23 treatment planning system. Additionally, treatment plans including RT dose, number of fractions and the start and the end date of RT were recorded for each patient.
Assessment of absolute lymphocyte counts
Detailed clinical and laboratory data that were obtained prior to the initiation of RT were collected from enrolled patients and included demographic data, tumour status, haematologic parameters, biochemical parameters and serum alpha-fetoprotein (AFP) levels. Blood samples were obtained by venous puncture and were collected 0–3 days before the start of RT for the quantification of lymphocytes, as well as once a week during RT and after RT. Changes in lymphocyte counts during RT were evaluated. Additionally, the Min ALCs and the days when Min ALCs were measured were analysed.
Follow up and definition
Patients underwent a standardized follow up after RT until the end of December 2015, their last visit to our hospital or death; the follow-up evaluations included physical examinations, peripheral blood cell counts, liver function tests, serum AFP measurements, HBV-deoxyribonucleic acid test results (HBV-DNA test), X-rays every 3 months for the first year and every 6 months thereafter and abdominal and pelvic CT or MRI every 6 months. RT-related toxicity was documented during and after RT and was evaluated according to radiation therapy oncology group criteria. Radiation-induced liver disease (RILD) was defined as either an anicteric elevation of alkaline phosphatase levels of at least twofold and nonmalignant ascites (classic RILD) [Lawrence et al. 1995] or elevated transaminases of at least fivefold of the upper limit of normal or of pretreatment levels (grade 3 or 4 hepatic toxicity of the Common Toxicity Criteria Version 2.0 by the National Cancer Institute), in the absence of documented progressive disease (nonclassic RILD) [Trotti et al. 2000]. Patients who experienced disease progression or recurrence were treated appropriately with TACE, chemotherapy, or sorafenib. Overall survival (OS) was calculated from the time of the initiation date of RT to the date of death regardless of cause or the censored date of the last contact for surviving patients.
Statistical analysis
Statistical analysis was performed using SPSS statistical software (version 17, SPSS Inc., Chicago, IL, USA). Quantitative variables are presented as medians and ranges and compared using the Mann–Whitney U test (nonparametric). Qualitative variables are presented as frequencies and analysed using the Pearson chi-square test. Cut-off values for laboratory data demonstrated the maximum sensitivity and specificity for survival based on the receiver-operating characteristic (ROC) curve [Budczies et al. 2012]. The primary endpoint of this study was OS. Actuarial survival was obtained using the Kaplan–Meier method and was compared with the results of the log-rank test. We conducted univariate and multivariate analyses using the Kaplan–Meier method and the Cox proportional hazards model to assess factors related to patients’ survival. Potential risk factors with a p value of <0.1 in the univariate analysis were entered into a multivariate analysis to determine their independent effect. The sample size for the current study was not based on predetermined power calculations. The sample size from our analyses (n = 69) had 96% power to detect a hazard ratio (HR) for the effects of Min ALC on OS. A value of p < 0.05 in the two-tailed analysis was considered to indicate a statistically significant difference.
Results
Baseline characteristics of patients and clinical outcomes
Between January 2009 and December 2014, 154 patients were diagnosed with HCC and received RT at the Shandong Cancer Hospital affiliated to Shandong University. A total of 69 patients met the study criteria and were fully evaluated. The baseline characteristics of these patients are detailed in Table 1. The median follow-up period was 30 months (range, 4.0–68 months) for all patients and 37 months (range, 11–68 months) for living patients. After radiation, six patients developed RT-related complications and they all recovered after effective symptomatic treatment. As for postradiotherapy treatment, 65 patients received TACE (number of times were 2–5), two patients received chemotherapy and two patients received sorafenib as a result of distant metastasis.
Clinical characteristics of patients with hepatocellular carcinoma in the overall study cohort.

HCC, hepatocellular carcinoma; KPS, Karnofsky performance Status; HBV, hepatitis B virus; PVTT, portal vein tumor thrombosis; BCLC, Barcelona Clinic Liver Cancer; ALC, absolute lymphocyte count; PNI [albumin(g/l) + 0.005 × total lymphocyte (cells/ul)], prognostic nutritional index; GGT, γ-glutamyl transferase; AFP, alpha-fetoprotein; Min ALC, minimum absolute lymphocyte count; RILD, Radiation induced liver disease; TACE, transcatheter arterial chemoembolization; PEI, percutaneous ethanol injection.
ROC curve analysis was performed to determine the threshold Min ALC for the prediction of OS [Budczies et al. 2012]. The most predictive Min ALC was determined to be 450 cells/µl (sensitivity and specificity, 50% and 70.6%, respectively), and the area under the curve of the Min ALC was 0.56. To evaluate the prognostic role of Min ALCs, we divided the whole group into a lower Min ALC (⩽450 cells/µl) (n = 37) and a higher Min ALC (>450 cells/µl) group (n = 32). The characteristics of patients with respect to the cut-off Min ALC of 450 cells/µl are shown in Table 2. No significant differences were observed between the two groups for most baseline characteristics, except for the BCLC stage. The results indicated that the BCLC stage was positively associated with lower Min ALCs during RT in patients with advanced BCLC scores (p = 0.008).
Demographic and clinical characteristics of patients in the low minimum absolute lymphocyte count (Min ALC) and high Min ALC groups.

Min ALC, minimum absolute lymphocyte; KPS, Karnofsky performance status; BCLC, Barcelona clinic liver cancer; PNI [albumin(g/l) + 0.005 × total lymphocyte (cells/ul)], prognostic nutritional index; NLR (neutrophil count/lymphocyte count), neutrophil lymphocyte ratio; GGT, γ-glutamyl transferase; AFP, alpha-fetoprotein. *Statistically significant.
Absolute lymphocyte counts during radiation
We examined the peripheral lymphocyte counts during RT. This is a retrospective study, and the timing and frequency of blood tests were therefore different in each patient. We analysed the medical data of all patients according to the number of days from the initiation of RT. As expected, the average circulating lymphocyte counts declined after RT (1493.19 versus 503.48 cells/µl, p < 0.001) (Figure 1a). The Min ALCs during RT and on the days when Min ALCs were measured are plotted in Figure 1(b). According to this graph, it is clear that most of the Min ALCs appeared between 20 and 50 days after the initiation of RT.

(a) Alteration of serum absolute lymphocyte counts (ALCs) during radiotherapy (RT) for all cases. Serum ALCs were significantly declined in all cases after RT (b) The minimum (Min) ALCs and the days when Min ALCs were measured during RT.
Associations between patient characteristics and survival
At the end of the follow-up period, 22 (31.9%) patients were still alive. The median survival time (MST), 1-year OS rate and 2-year OS rate in the whole group were 25 months, 51%, and 39%, respectively. The MSTs of different subgroups and the results of univariate and multivariate analyses are detailed in Table 3. Survival was worse in patients with relatively lower Min ALCs (⩽450 cells/µl) than in those with higher Min ALCs (>450 cells/µl); the MSTs, 1-year OS rates and 2-year OS rates were 15 months versus 47 months, 27% versus 78% and 4% versus 71%, respectively, (p < 0.001) in the lower and higher Min ALC groups. No correlation of survival with age, gender, Karnofsky performance status (KPS), prognostic nutritional index (PNI), γ-glutamyl transferase (GGT), radiation dose or postradiotherapy treatment was found. The OS of patients with BCLC stage C, lower serum ALCs, higher serum neutrophil–lymphocyte ratio (NLR) and AFP levels before RT or lower serum Min ALCs during RT was significantly worse than that of patients with a BCLC stage of A/B, higher serum ALCs, lower serum NLR and AFP levels or higher serum Min ALCs (p values were <0.001, 0.018, 0.001, <0.001 and <0.001, respectively; see Figure 2).
Univariate and multivariate analysis of prognostic factors of overall survival.

MST, median survival time; HR, hazard ratio; CI, confidence interval; KPS, Karnofsky performance status; BCLC, Barcelona clinic liver cancer; ALCs, absolute lymphocyte counts; PNI [albumin (g/l) + 0.005 × total lymphocyte (cells/ul)], prognostic nutritional index; NLR (neutrophil count/lymphocyte count), neutrophil-to-lymphocyte ratio; GGT, γ-glutamyl transferase; AFP, alpha-fetoprotein; Min ALCs, minimum absolute lymphocyte counts during radiotherapy; TACE, transcatheter arterial chemoembolization. *There are no results; $statistically significant.
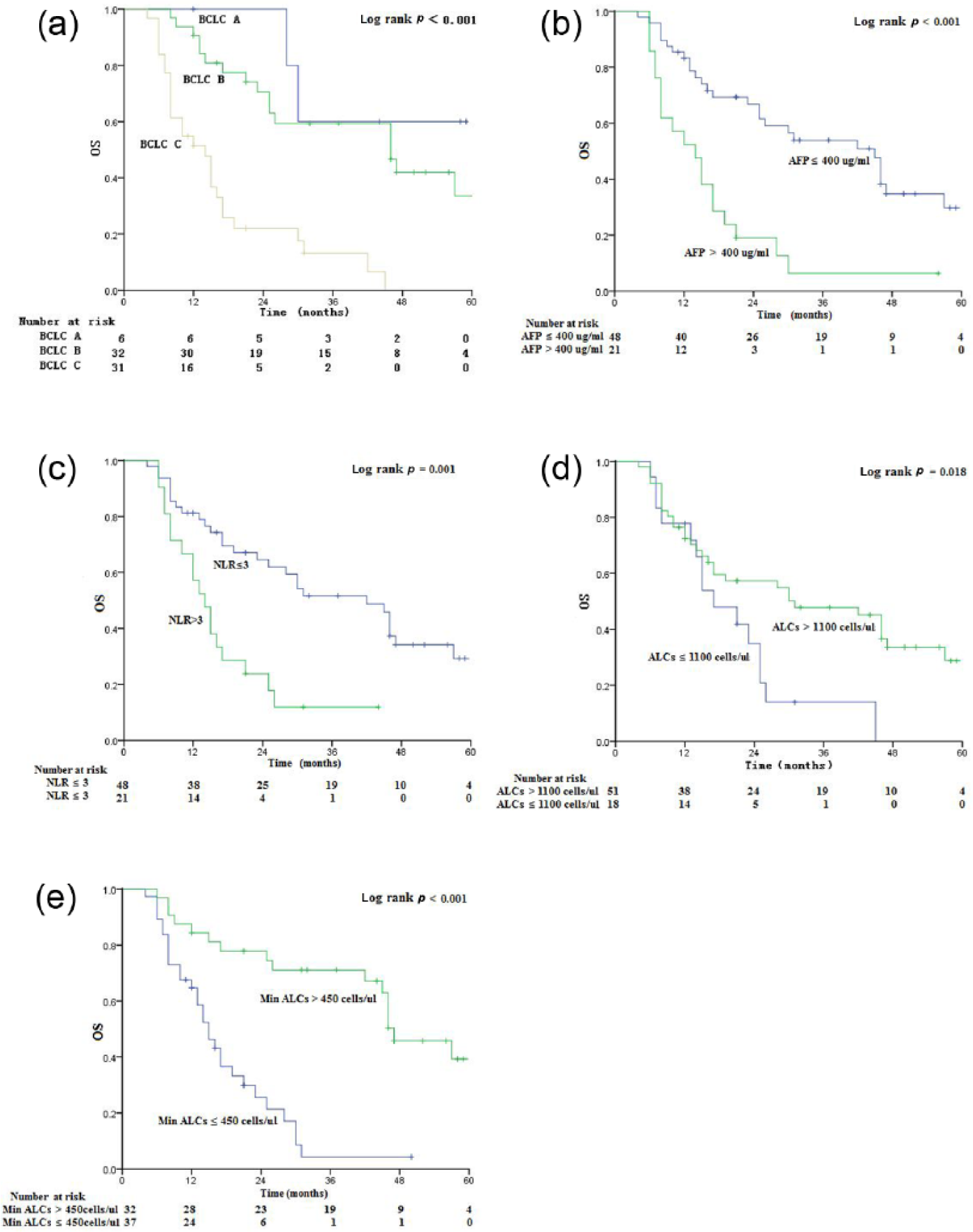
Survival curves according to the Kaplan–Meier method in the patients who underwent radiotherapy. (a) Overall survival (OS) curve with a comparison of the survival rates in different Barcelona Clinic Liver Cancer (BCLC) score; (b) OS curve with a comparison of the survival rates in the low- and high-alpha-fetoprotein (AFP) groups; (c) OS curve with a comparison of the survival rates in the low- and high neutrophil–lymphocyte ratio (NLR) groups; (d) OS curve with a comparison of the survival rates in the low- and high-absolute lymphocyte count (ALC) groups; (e) OS curve with a comparison of the survival rates in the low- and high-minimum (Min) ALCs groups.
A Cox regression model was used to further assess the independent prognostic ability of the above risk factors. After adjusting for potential confounders, multivariate Cox regression analysis demonstrated that Min ALC independently predicted patients’ OS [hazard ratio (HR), 0.32; 95% confidence interval (CI), 0.15–0.69] (see Table 3). However, the prognostic significance of the ALC and NLR before RT disappeared in the multivariate regression (p = 0.173 and p = 0.064, respectively).
Taking into account the effects of the tumour staging on OS, staging was stratified to further clarify the prognostic significance of Min ALCs (Figure 3). We did not perform a comparison for the BCLC A group because only six patients were included in that group. For patients with BCLC B staging, subjects with higher Min ALCs exhibited longer OS than subjects in the lower Min ALC group (p = 0.003; Figure 3a) and patients with BCLC B staging (p = 0.047; Figure 3b).

Survival curves according to the Kaplan–Meier method in the patients who underwent radiotherapy. The graphs represent the assessment of the prognostic role of minimum absolute lymphocytes counts (Min ALC) during RT according to Barcelona Clinic Liver Cancer (BCLC) score B (a) and BCLC score C (b).
Discussion
A decline in the ALC after treatment has been identified as a prognostic biomarker for several malignancies. Saroha and colleagues suggested that a low ALC is associated with aggressive features and inferior survival in patients with clear cell renal cell carcinoma [Saroha et al. 2013]. Balmanoukian and colleagues reported that lymphopenia was an independent predictor for survival in patients with resected pancreatic adenocarcinoma [Balmanoukian et al. 2012]. A decline in peripheral lymphocyte counts after RT was first described in the 1970s [Stratton et al. 1975], but the clinical significance has not been well studied. This retrospective analysis of HCC patients who received RT demonstrated that lymphocyte counts declined after RT, and this observation has also been found in other cancer types [Santin et al. 2000; Pinkawa et al. 2014]. In our study, a multivariate analysis revealed that Min ALCs were a predictor of OS that was independent of the BCLC score and the serum AFP level. Patients who developed lower lymphopenia (Min ALCs ⩽ 450 cells/µl) after RT had worse OS than those who exhibited relatively higher Min ALCs (>450 cells/µl) (p = 0.002; HR = 0.31; 95% CI, 0.15–0.64). This result suggests that a decrease in peripheral lymphocyte counts would result in a poor prognosis for HCC patients. The mechanisms behind this finding are multifactorial and are not yet completely understood. Some theories may explain this association. First, some studies have suggested that lymphopenia during RT might be caused by the apoptosis of lymphocytes that are caught in the radiation field [Yovino and Grossman, 2012]. Second, the correlation between the increasing infiltration of CD4+ T lymphocytes at tumour margins and a better prognosis of HCC patients has been demonstrated [Ma et al. 2016]. Third, in vivo studies have shown that tumour cells that are dead or dying due to RT with or without chemotherapy can present as tumour-associated antigens to host immune cells and thereby activate antitumour immune responses [Apetoh et al. 2007]. Some clinical data have revealed the presence of radiation-induced antitumour immunity in humans [Schaue et al. 2008]. Lymphocytes exert an antitumour effect in the tumour microenvironment and affect cancer development and progression [Fu et al. 2013]. HCC patients with severe lymphopenia have worse immune system function and antitumour immunity. This association has also been verified in other types of cancer [Cho et al. 2016a, 2016b]. Consequently, a lower serum Min ALC during RT, as a reflection of the host immunity network, may be used to predict the result of cancer treatment. However, we cannot ignore the effect of various factors such as host nutrition and chronic infection on the peripheral lymphocyte counts. HBV activation and other infections were not detected in any patients in our study during the course of RT; therefore, we could state that this value reflects the overall condition of the host to fight the tumour. The Min ALC may be a good marker of the host immune reaction to the tumour.
In the present study, we found that the ALC and NLR before RT were correlated with OS according to the univariate analysis, similar to the results of previous studies [Templeton et al. 2014]. In fact, accumulated evidence has suggested that a higher NLR is an independent prognostic factor in various solid tumours, including HCC [Kishi et al. 2009; Ubukata et al. 2010; Huang et al. 2011]. In addition, some studies have shown that a high NLR may suggest an impaired host immune response of HCC patients to the tumour [Zhong et al. 2006]. Taken together, these results indicated that an elevated NLR can indirectly reflect OS. Our results showed that patients with an NLR ⩽ 3 exhibited significantly better OS than those with an NLR > 3 (p = 0.001). However, the association between a high NLR and poor survival is likely complex and largely unclear. The increased number of neutrophils and the decreased number of lymphocytes could result in a high NLR. Patients with lymphocytopaenia have a weaker lymphocyte-mediated immune response to the tumour as noted above. As for patients with neutrophilia, Grivennikov and colleagues suggested that systemic and local inflammatory responses to the tumour may play a role in tumour invasion [Grivennikov et al. 2010]. In addition, neutrophils have been shown to be involved in angiogenesis, which affects tumour development and is associated with a poor prognosis in HCC patients [Zhong et al. 2006]. Taken together, these findings indicate that an elevated NLR can indirectly reflect the tumour burden, invasion and OS. Therefore, in-depth research into the mechanism of such an effect is warranted. A lower ALC and an elevated NLR before RT were correlated with a poor prognosis, but were not found to be independent predictors of OS according to the multivariate analysis in our study, while a lower serum Min ALC (⩽450 cells/µl) during RT was an independent predictor of OS. Therefore, a lower serum Min ALC during RT may be a more suitable and reliable long-term prognostic predictor for HCC patients.
The effect of the RT dose on the probability of tumour control and survival is well established [Ben-Josef et al. 2005]. Dawson and colleagues found that patients who were treated with >70 Gy had an improved MST compared with those who received <70 Gy (16.4 months versus 11.6 months, p = 0.0003) [Dawson et al. 2000]. In our study, no correlation between RT dose and OS was found. The radiation doses were all 39.60–63.48 Gy, and no patient received a radiation dose that was >70 Gy. Therefore, the RT dose that patients received in our study was far less than the lethal dose that obtained the maximum antitumour effect in HCC patients. The main barrier to dose escalation is the presence of adjacent critical structures, especially a normal liver parenchyma. The issue of how to maximize therapeutic effects by escalating the radiation dose while simultaneously sparing the adjacent normal liver is a major subject of ongoing investigations.
Considering that RT-related lymphopenia appears to be common, severe and closely associated with a poor prognosis in HCC patients, prospective studies are needed to further discover and test treatment approaches that preserve or restore immunologic function. With regard to checkpoint inhibitors, such as antiprogrammed-death 1, programmed-death ligand 1 and cytotoxic T-lymphocyte antigen 4 therapies, clinical trials have suggested that treatment with these agents could restore immunologic function [Hiniker, 2012] and that the levels of peripheral lymphocytes are predictive of efficacy [Ménard et al. 2008]. The results could steer future treatment approaches towards a reduction in the toxicities of RT in lymphocytes and produce a definition of the proper timing and use of immunologic interventions.
Some major limitations of this study should be addressed. First, the survival analysis was limited by the retrospective analysis and small number of patients. The data were collected over a long period, from a single centre, and most patients had HBV-related diseases. Second, no data showed that lymphocyte subsets (i.e. B, helper T and cytotoxic T cells) or the alterations of different subsets during or after RT could predict survival. Third, some patients received additional treatments such as TACE or sorafenib after the completion of RT. We believe that these treatments may also effect the outcomes. Clearly, these results warrant a largescale meta-analysis or prospective studies to validate our findings.
The results of this retrospective analysis demonstrated that in addition to BCLC stage and serum AFP levels before RT, Min ALCs during RT have an independent predictive value for OS. The Min ALC during RT, which is an immunity-based prognostic marker, may act as a prognostic factor for HCC patients who are undergoing RT. In addition, peripheral lymphocyte counts are simple to measure in clinical practice and are routinely assessed in patients during treatment. Therefore, examination of the dynamic changes in lymphocyte counts may be helpful in the design of individualized treatments for HCC patients.
Footnotes
Acknowledgements
Qianqian Zhao and Xiaoqing Xu contributed equally to this work.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
