Abstract
Background:
Some chronic hepatitis C virus (HCV), genotype 1 infected patients treated with direct antiviral agents (DAAs) remain viremic at end of treatment (EOT+), yet go on to achieve sustained virological response 12 weeks after completion of therapy (SVR12). The incidence of EOT+/SVR in patients with genotype 1 and other genotypes, as well as whether such patients achieve SVR24 remain in question. The aims of this study were to evaluate the frequency and durability of EOT+/SVR12&24 and other response categories in HCV genotype 1, 2, or 3 infected patients treated with DAA in clinical practice.
Methods:
Data from patients treated with all oral sofosbuvir-based regimens at a university hepatology practice by 1 July 2015 were reviewed retrospectively. Responses were categorized based on virus levels during and post DAA treatment. HCV RNA levels were measured by Abbott RealTime HCV (ART) or by Roche CobasTaqMan v2.0 (RCTM) assays.
Results:
The study population included 89 patients. Participants were 62% genotype 1, 19% genotype 2 and 19% genotype 3, 54% cirrhotic and 46% treatment-experienced. A total of 45 received sofosbuvir–simeprevir, 38 sofosbuvir–ribavirin and 6 sofosbuvir–ledipasvir. The SVR12 rate was 82%. A total of 5 patients (6%), all with genotype 1, had EOT+ by ART assay and each achieved SVR12&24.
Conclusions:
A total of 9% of genotype 1 patients (6% overall) treated with DAAs were EOT+ by ART and all EOT+ cases achieved SVR24. EOT+/SVR was not observed with genotype 2 or 3 or by the RCTM assay. In patients treated with DAAs, EOT+ by the ART assay does not indicate treatment failure.
Introduction
For over a decade, the standard-of-care treatment for hepatitis C virus (HCV) infection was interferon (IFN)-α-based treatment combined with ribavirin. These therapies were associated with significant adverse effects while resulting in modest rates of sustained virologic response (SVR24, defined as undetectable HCV-RNA 24 weeks after completion of treatment) in patients with genotype 1 HCV infection [Fried et al. 2002; Manns et al. 2001]. With the introduction of direct acting antivirals (DAA) regimens, SVR24 has been superseded by SVR12 as the indicator of virologic cure. The DAAs have vastly improved SVR rates with shortened lengths of treatment and minimal adverse effects, even in populations that were historically considered difficult to treat [Terrault, 2015].
With pegylated (Peg)-IFN-based therapy, detection of HCV-RNA at the end of treatment (EOT) was an indicator of treatment failure [Ferenci et al. 2008; Jacobson et al. 2014; Poordad et al. 2011]. However, recent reports document that some patients (range 0.3–56%) treated with Peg-IFN-free DAAs regimens achieved SVR12 despite having detectable viremia at EOT (termed here EOT+/SVR12) [Harrington et al. 2015; Kohli et al. 2015; Sarrazin et al. 2015; Sidharthan et al. 2015a, 2015b]. Examination of the published data shows that high EOT+/SVR rates (56%) were documented only at the end of 6 weeks of treatment with sofosbuvir and ledipasvir in combination with the nonnucleoside NS5B inhibitor GS-9669 or the NS3 protease inhibitor GS-9451 [Kohli et al. 2015]. In addition, EOT+/SVR was detected more frequently when more sensitive HCV-RNA assays were used (e.g. 29% versus 3.1% of EOT+/SVR12 rates were found using ART versus RCTM) [Sidharthan et al. 2015a]. Earlier studies had relatively short-term follow up (SVR12), limited data from non-1 genotypes, and few patients with cirrhosis. Whether the reported EOT+/SVR12 subjects achieved SVR24 was not determined. Overall, the biological and clinical reasons for these surprising EOT+/SVR12 cases remain unknown.
Here we evaluated the frequency and the clinical characteristics of patients who had EOT+/SVR12 in our clinical practice, as well as which HCV measurement assays were used at EOT, and whether SVR12 cases achieved SVR24.
Materials and methods
The study protocol was approved by the Institutional Review Board at Loyola University Medical Center. Acquisition of informed consent was waived given the retrospective data from 106 consecutive treatment naïve or experienced adult patients who were initiated on interferon-free, DAA therapy for HCV in a university-based hepatology practice and had determination of SVR12 by 1 July 2015 were evaluated. One genotype 6 patient with persistent (quantifiable) HCV during and after treatment under sofosbuvir + ribavirin, 6 patients who did not complete treatment and 10 patients who did not have an EOT HCV RNA level, despite having week 12 post-treatment data were excluded, leaving 89 patients for analysis (Table 1). DAA agents included simeprevir (150 mg daily), sofosbuvir (400 mg daily) or combination ledipasvir–sofosbuvir (90–400 mg daily). A variety of treatment regimens were used (Table 2) reflecting the evolution of HCV therapy in clinical practice. Baseline characteristics were recorded including HCV genotype and pre-treatment viral load. Patients were categorized as having cirrhosis based on liver biopsy or serum fibrosis markers (FIBROSpect II®, Prometheus Laboratories, San Diego, CA) in conjunction with characteristic findings on cross-sectional imaging or clinical evidence of portal hypertension.
Demographic and pre-treatment patient characteristics (n = 89).

Eight patients previously received a protease inhibitor.
Treatment regimens, viral response and outcome.
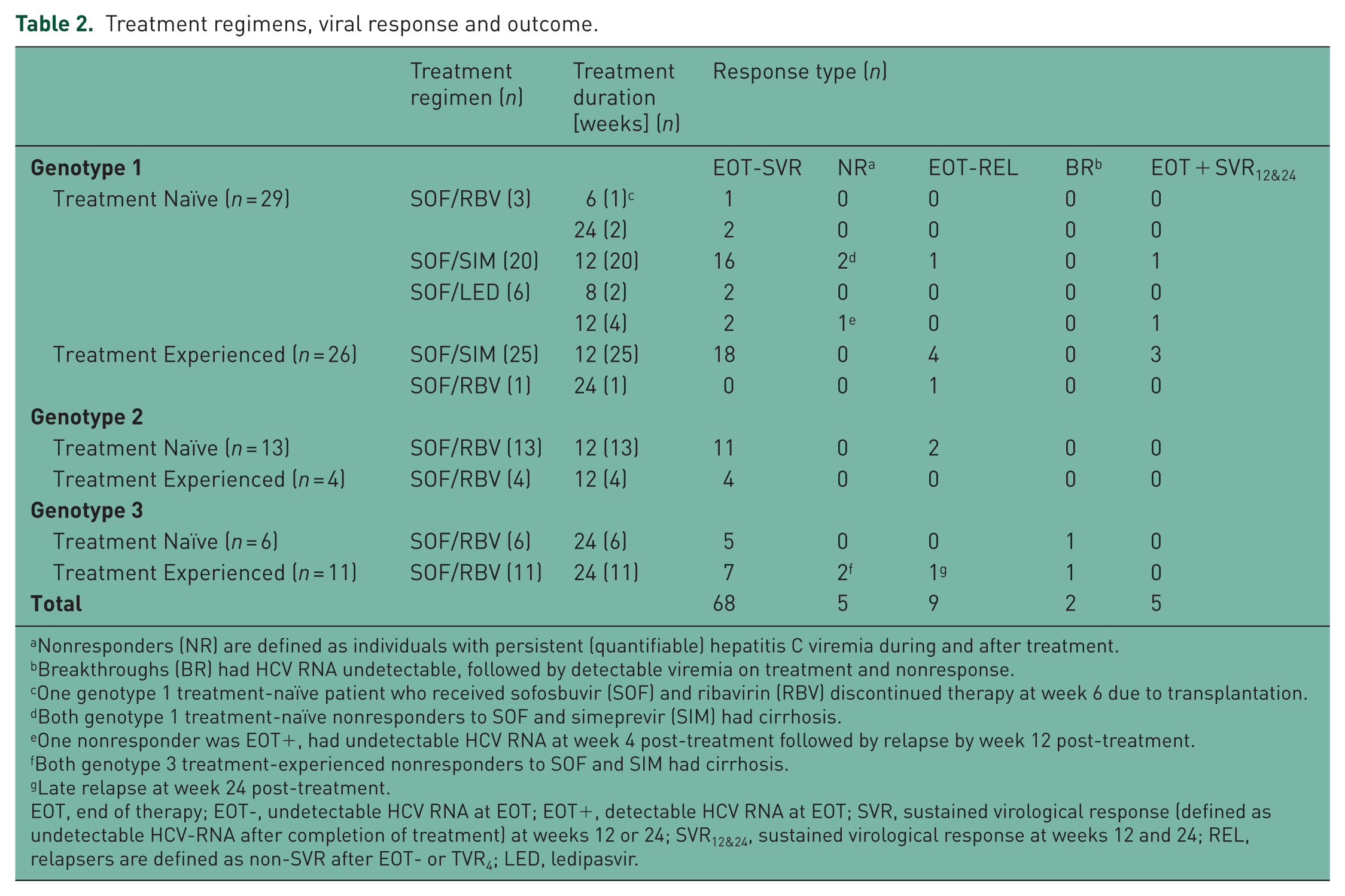
Nonresponders (NR) are defined as individuals with persistent (quantifiable) hepatitis C viremia during and after treatment.
Breakthroughs (BR) had HCV RNA undetectable, followed by detectable viremia on treatment and nonresponse.
One genotype 1 treatment-naïve patient who received sofosbuvir (SOF) and ribavirin (RBV) discontinued therapy at week 6 due to transplantation.
Both genotype 1 treatment-naïve nonresponders to SOF and simeprevir (SIM) had cirrhosis.
One nonresponder was EOT+, had undetectable HCV RNA at week 4 post-treatment followed by relapse by week 12 post-treatment.
Both genotype 3 treatment-experienced nonresponders to SOF and SIM had cirrhosis.
Late relapse at week 24 post-treatment.
EOT, end of therapy; EOT-, undetectable HCV RNA at EOT; EOT+, detectable HCV RNA at EOT; SVR, sustained virological response (defined as undetectable HCV-RNA after completion of treatment) at weeks 12 or 24; SVR12&24, sustained virological response at weeks 12 and 24; REL, relapsers are defined as non-SVR after EOT- or TVR4; LED, ledipasvir.
Patient demographics and pre-treatment variables are summarized in Table 1. Treatment regimens included sofosbuvir–simeprevir (50%), sofosbuvir–ribavirin (43%) and sofosbuvir–ledipasvir (7%). Treatment length varied from 8 to 24 weeks with the exception of one patient treated with sofosbuvir–ribavirin whose therapy was interrupted after 6 weeks when he underwent liver transplantation (Table 2).
HCV RNA measurement
HCV-RNA levels were measured by the Loyola University Clinical Laboratory using the Abbott RealTime PCR assay (ART) (Abbott Laboratories, Abbott Park, IL) with a lower limit of quantitation (LLOQ) of 12 IU/ml or when laboratories were drawn at other institutions by the COBAS TaqMan HCV Test v2.0 (RCTM) (Roche Molecular Diagnostics, Indianapolis, IN) with a LLOQ of 15 IU/ml. EOT+/SVR12 was defined as detectable HCV-RNA at completion of therapy with HCV-RNA undetectable at week 12 post-treatment.
Statistical analysis
All statistical analysis was performed using SPSS, version 19 (Chicago, IL). Descriptive statistics were used to assess for erroneous entries and to perform frequency counts. Student’s t tests were used to compare means. Categorical data were compared by chi-square tests or Fisher’s exact test where appropriate. Here p values < 0.05 were considered statistically significant.
Results
End of treatment positive/sustained virologic response
Response patterns are detailed in Table 2, which provides treatment regimens and duration of therapy by genotype and previous treatment status. A total of 5 patients had EOT+/SVR12, all of whom were HCV-RNA undetectable at week 4 post-treatment and achieved SVR12 and SVR24. The EOT+/SVR12&24 patients had HCV genotype 1 (three genotype 1a and two genotype 1a or 1b). In total, 5/55 (9%) patients with genotype 1 had EOT+/SVR12&24. Four had evidence of cirrhosis. Two were treatment naïve and three were previously treated. All received 12 weeks of therapy, including four (of 45) who were treated with sofosbuvir–simeprevir and one (of six) who had sofosbuvir–ledipasvir. None received sofosbuvir–ribavirin, although only four genotype 1 patients were treated with sofosbuvir–ribavirin. All five patients had quantifiable HCV-RNA at week 4 of treatment and detectable, but not quantifiable HCV-RNA at EOT. In total 49/89 (55%) EOT HCV measurements were performed by ART, including 32/55 (58%) genotype 1 cases and all five EOT+/SVR cases. While power was limited due to the small number of EOT+/SVR12&24 cases, there were no significant differences in demographic or clinical features between the EOT+/SVR12&24 patients and either the 16 treatment failures or the 68 EOT-/SVR12 cases. One additional genotype 1 patient who had detectable but not quantifiable HCV-RNA at EOT by the ART assay had an undetectable HCV-RNA level at week 4 post-treatment. However, the patient relapsed with quantifiable viremia at week 12 post-treatment.
Comparison of patients with sustained virologic response to treatment failures
To identify potential determinants of treatment failure, features of all 73 patients who achieved a SVR12 were compared with the 16 treatment failures. Patients with a pre-treatment HCV-RNA level <800,000 IU/ml were more likely to have a SVR12 (25/26) than patients with a pre-treatment level >800,000 IU/ml (47/63) [p = 0.019]. Patients with cirrhosis trended toward being less likely to achieve SVR12 (36/48) than those without cirrhosis (37/41) [p = 0.062]. There was a trend toward a higher SVR12 rate in women (36/40) than men (37/49) [p = 0.077]. There were no significant differences between the groups with regard to age, race, BMI, HCV genotype distribution, history of prior treatment, or whether HCV RNA was detectable at week 4 of therapy. A total of 85 patients had a week 4 HCV-RNA measurement, at which time 51% had target not detected, 20% detected, but not quantifiable, and 29% had quantifiable HCV-RNA.
Discussion
The current study evaluated patients treated in clinical practice with approved DAA regimens for a standard duration of time and HCV-RNA measurements were made by sensitive assays (ART and RCTM with LLOQ of 12 and 15 IU/ml, respectively). The findings add to the evolving understanding of response patterns to DAA regimens. The 24-week post-treatment HCV-RNA measurements showed that EOT+/SVR12 was durable in all cases. Interestingly, EOT+/SVR12&24 was only observed in patients with HCV genotype 1 and only by ART. The prevalence of EOT+/SVR12&24 was 9% in patients with genotype 1. EOT+/SVR12&24 patients had advanced fibrosis; four out of five had cirrhosis and one had evidence of F2–F4 fibrosis by FIBROSpect II (Prometheus Laboratories, San Diego, CA). All EOT+/SVR12&24 cases had quantifiable HCV-RNA at week 4 of therapy and had detectable HCV RNA at EOT by the ART assay, which previously was found under Peg-IFN/ribavirin/protease-inhibitor to be more sensitive at the detection limit than the RCTM assay [Fevery et al. 2014; Maasoumy et al. 2014; Vermehren et al. 2014]. Interestingly, a recent study by Maasoumy and colleagues [Maasoumy et al. 2016] compared the performance of RCTM and ART in patients treated with sofosbuvir-based regimens. They found that 20% of genotype 1 patients and 18% of genotype 3 patients treated with sofosbuvir–simeprevir ± ribavirin and sofosbuvir–daclatasvir ± ribavirin were EOT+ by ART with 92% and 100% SVR rates, respectively.
All EOT+/SVR cases in the current study were detected by ART in agreement with Maasoumy and colleagues [Maasoumy et al. 2016]. It is not known whether the greater sensitivity, the amplification method, or other characteristics of the ART assay make it more likely to detect HCV RNA at EOT than with the RCTM assay. In the present series, the ART assay was used in 32 genotype 1 cases and RCTM was used in the remaining 23 at EOT. Thus, 16% (5/32) of genotype 1 patients evaluated by ART were EOT+/SVR, which is close to the 20% rate reported by Maasoumy and colleagues [Maasoumy et al. 2016] but somewhat lower than the 29% indicated by Sidharthan and colleagues [Sidharthan et al. 2015a] by ART. Similar to previous studies, EOT+/SVR was not identified in genotype 2 patients [Maasoumy et al. 2016; Zeuzem et al. 2014]. The ART assay was found to have a slightly lower limit of detection for HCV genotype 2 compared with genotype 1 (see http://www.accessdata.fda.gov/cdrh_docs/pdf10/P100017B.pdf). However, a difference in sensitivity for the ART assay by genotype does not explain the absence of EOT+/SVR cases in patients with HCV genotype 2, as a lesser limit of detection for genotype 2 would be expected to make identification of EOT+/SVR cases more likely.
Two explanations have been offered for the phenomenon of EOT+/SVR [Kohli et al. 2015; Sidharthan et al. 2015b]. A virologic hypothesis, supported by in vitro data, is that some DAAs promote the production of noninfectious viral particles, leading to the transient presence of RNA-detectable noninfectious virus at EOT with a smaller residual of (or theoretically in some cases complete absence of) infectious virus at EOT [Sansone et al. 2014]. An immunologic hypothesis is that immune-mediated clearance of residual infectious virus results in SVR after completion of DAA therapy [Meissner et al. 2014]. The feasibility of immune clearance is consistent with an elegant longitudinal study showing that HCV-specific T-cell responses can successfully clear small amounts of circulating HCV-RNA identified years after interferon-based therapy [Veerapu et al. 2011]. The virologic and immunologic hypotheses are not mutually exclusive and both mechanisms could contribute to a varying extent in any given EOT+/SVR case. That is, noninfectious particles and a small number of infectious virions could coexist at EOT, with subsequent immune-mediated clearance (or degradation) of residual infectious virus. It is not possible to discern whether either or both mechanisms played a role in the five EOT+/SVR12&24 cases in the current retrospective series. However, the EOT+ case, which was HCV RNA undetectable at week 4 post-treatment and then relapsed between weeks 4 and 12 of follow up is consistent with incomplete immune suppression of residual infectious virus. A third explanation for the EOT+/SVR cases is that the EOT+ measurements represent false positives. Given the sizeable and growing amount of evidence that some patients treated with DAAs have residual viremia at EOT [Maasoumy et al. 2016], we feel that the EOT+ measurements are unlikely to be false positives.
An analysis of 779 patients treated with DAAs in registry trials found that 99.7% of patients who achieved SVR12 had SVR24 [Yoshida et al. 2015]. In the current study, 1/74 (1.4%) who had an SVR12 relapsed by week 24, which provides a conservative measure of late relapse as 29 patients did not have week 24 post-treatment HCV-RNA measurements. Identification of relapse between 12 and 24 weeks post-treatment, although infrequent, provides a rationale to continue to assess for SVR24. The observed nonresponder and breakthrough cases reflect outcomes in clinical practice and differ from the experience in optimized clinical trial populations where end treatment response was uniform [Harrington et al. 2015]. All four of the nonresponder patients and one of the breakthroughs had cirrhosis.
In summary, the clinical implication of this study is that EOT+ by ART does not equal treatment failure in the era of DAAs. Moreover, EOT+/SVR12 was durable to SVR24. The profile of EOT+/SVR cases in the current study included use of the ART assay, genotype 1, advanced fibrosis and slow treatment response (quantifiable HCV-RNA at treatment week 4 as in Maasoumy et al. [2016]), with a prevalence of 9% in patients with genotype 1. It is notable that EOT+/SVR has not been reported in genotype 2 patients. While this case series cannot distinguish between the virologic and immune mechanisms hypothesized to explain EOT+/SVR, the EOT+ case, which was found to have undetectable HCV RNA at week 4 post-treatment and then relapsed between week 4 and 12 of follow up is consistent with an incomplete immune response against residual infectious HCV RNA particles in one particular patient.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIH grants R01-AI078881, R01-OD011095 and P20-GM103452 and the US Department of Energy contract DE-AC52-06NA25396.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Miguel Malespin has served as a consultant for Gilead Pharmaceuticals; Alan Perelson has served as a consultant for Gilead, Bristol-Myers Squibb, Merck and Achillion Pharmaceuticals.
