Abstract
Background:
Hepatitis C virus (HCV) management is a challenge in patients with substance use disorder (SUD). This study aimed to describe an HCV screening and linkage to care program in SUD patients, and analyze the characteristics of this population in relation to HCV infection, particularly the impact of psychiatric comorbidities (dual diagnosis).
Methods:
This study was a prospective clinical cohort study using a collaborative, multidisciplinary model to offer HCV care (screening, diagnosis, and therapy) to individuals with SUD attending a dedicated hospital clinic. The characteristics of the participants, prevalence of HCV infection, percentage who started therapy, and adherence to treatment were compared according to the patients’ consumption characteristics and presence of dual diagnosis. HCV screening, diagnosis, treatment initiation, and sustained virologic response were analyzed.
Results:
528 individuals attended the center (November 2018–June 2019) and 401 (76%) accepted screening. In total, 112 (28%) were anti-HCV-positive and 42 (10%) had detectable HCV RNA, but only 20 of the latter started HCV therapy. Among the 253 (63%) patients with a dual diagnosis, there were no differences in HCV infection prevalence versus patients with SUD alone (p = 0.28). Dual diagnosis did not lead to a higher risk of HCV infection or interfere with linkage to care or treatment.
Conclusion:
This study found a high prevalence of dual diagnosis and HCV infection in SUD patients, but dual diagnosis was not associated with an increased risk of acquiring HCV or more complex access to care. Despite use of a multidisciplinary management approach, considerable barriers to HCV care remain in this population that would need more specific focus.
Keywords
Introduction
Hepatitis C virus (HCV) is the most prevalent viral hepatitis in Western countries and a major cause of chronic liver disease. 1 Recent epidemiological studies in the adult population of Spain have estimated that the prevalence of HCV infection with detectable HCV RNA is between 0.35% and 0.41%,2,3 and around 29% of these individuals are unaware of their infection. 4 Since the introduction of oral direct-acting antiviral agents (DAAs) the clinical care of patients with HCV-related liver disease has advanced considerably, enabling HCV cure in almost all cases regardless of viral genotype, degree of liver fibrosis, or comorbidity. 5 These huge improvements in HCV treatment have led the World Health Organization (WHO) to encourage elimination of HCV infection by 2030 by increasing the diagnosis, therapy, and prevention of this condition. 6
HCV infection is highly prevalent in people with substance use disorders (SUDs), especially those with a history of injecting drugs.5,7,8 An estimated 2 or 3 million individuals have a history of injecting drug use in Europe alone, and their anti-HCV antibody prevalence ranges from 15% to 84%. 9 A study in people who inject drugs in Catalonia (Spain) showed an overall HCV seroprevalence of 79.8%, and viremic infection in 58.5%. 10 However, there is little updated information for individuals using non-injected drugs. A study conducted in 2001 reported that the prevalence of anti-HCV antibodies in a sample of 529 non-injecting drug users (heroin, cocaine, or crack) ranged from 5% to 29%. 11
Approximately 50–75% of individuals with SUDs have comorbidities with other psychiatric disorders, a situation known as dual diagnosis. 12 These individuals report a greater social and clinical impact, including a poorer prognosis and higher healthcare costs.12,13 In a study performed in the United States in 2002, 62% of 33,824 patients with HCV infection had dual diagnosis, 14 but there is little recent information in this line. Dual diagnosis patients engage in more risky behaviors than the general population and have a higher probability of reinfection; hence, they are at considerable risk for HCV infection. Barriers to starting therapy in this population must be understood to improve HCV care and achieve HCV elimination.7,15
Considering the above and the limited available data on HCV in non-injecting drugs users and dual diagnosis patients, the objective of this study was to describe an HCV screening, diagnosis, and treatment program to facilitate linkage to care in all patients with SUDs attending a dedicated addiction and dual diagnosis center (ADDC). The characteristics of the participating individuals, the prevalence of HCV infection, the percentage of patients who started HCV therapy, and adherence to treatment were investigated in those with and without a dual diagnosis.
Methods
Study design and patients
This was a prospective clinical cohort study designed to investigate HCV care in individuals with SUD or dual diagnosis, including HCV screening, diagnosis, and therapy with DAAs within the first 6 months after the diagnosis. The study included a prospective follow-up period of 6 months.
The study was conducted at the ADDC of Vall d’Hebron Hospital, Barcelona (Spain). All patients who attended the center from November 2018 to June 2019 were asked to participate. The inclusion criteria were age older than 18 years and a diagnosis of SUD with or without dual diagnosis. Patients with cognitive impairment (based on a Mini-Mental State Examination score <27) or low Spanish or English language proficiency that interfered with their ability to understand the study proposal were excluded. Having previously been diagnosed and cured of HCV was not an exclusion criterion as we were interested in studying the characteristics of those ever infected versus those never infected. The project (VHC-DAA-2018-01) was approved by the Ethics Committee of Vall d’Hebron Hospital. Patients did not receive any financial compensation. Written informed consent was obtained from all participants.
Procedure
After providing informed consent, all patients underwent a psychological evaluation to assess health-related quality of life (HRQoL) using the Short Form-36 Health Survey (SF-36), including the mental and physical dimensions, and depression status using the Beck Depression Inventory (BDI). A psychiatrist collected the sociodemographic and clinical features of each participant. A blood sample was obtained.
Patients who tested anti-HCV positive and had detectable HCV RNA underwent a clinical evaluation to assess suitability for treatment based on international guideline recommendations. Those eligible were offered treatment for 8 or 12 weeks according to the prescribed DAA scheme. To facilitate the start and follow-up of DAA treatment, a hepatologist went to the addiction center twice a week to visit the patients and coordinate their treatment with a multidisciplinary team that additionally included psychiatrists, psychologists, nurses, and assistants working together to facilitate linkage to care.
Patients accepting treatment were provided with the first 4 weeks of DAAs and were subsequently seen at 4-week intervals by the hepatologist to ensure adherence (determined by self-report), evaluate possible adverse events, and dispense the next 4 weeks of DAAs. Drug determination in urine was performed at the beginning of treatment and at each visit. At completion of treatment, and at week 12 after completion, patients attended a medical visit, a blood sample was obtained, and a new drug urinalysis was performed.
Instruments and variables
Sociodemographic and clinical features
Demographic data were collected at the time of enrollment (sex, age, nationality, civil status, housing, educational level, employment status, and criminal record). Information regarding clinical variables, such as self-reported medical conditions (e.g. cardiac, endocrine, metabolic, renal, and neurologic diseases, termed any medical history in the analysis), previous and current SUDs (such as alcohol, cannabis, benzodiazepines, cocaine and heroin; route of drug administration; age at start of SUD; polysubstance consumption defined as use of three or more substances), and previous SUD treatments, was also collected. Family history of substance use was excluded, as it was considered to have limited clinical relevance for the current study. Patients were asked of any known infectious disease such as HCV, HBV, or HIV. Psychiatric comorbidity with other mental disorders (dual diagnosis) was evaluated by a psychiatrist and established by clinical judgment, following the DSM-5 criteria. Mental disorders were grouped into psychotic, mood, anxiety, and personality disorders. All sociodemographic and clinical factors were categorical variables except for age.
Laboratory assessment
Blood tests included a complete blood count and standard biochemical panel. Non-invasive liver fibrosis assessment used the FIB-4 (based on age, platelet count, AST and ALT). FIB-4 results were interpreted according to two cut-off values: <1.45 indicated absence of cirrhosis, >3.25 indicated cirrhosis, 1.45–3.25 were considered inconclusive. HBsAg, anti-HIV, and anti-HCV antibodies were determined. In individuals testing positive for anti-HCV antibodies, HCV RNA and HCV genotype were determined. Drug urinalysis included benzodiazepines, methadone, cocaine, opioids, amphetamines, and cannabinoids.
Questionnaires to assess depression and quality of life
The Spanish version of the Beck Depression Inventory (BDI-I) was used to assess the presence of depressive symptoms during the previous 2 weeks. The BDI-I is a 21-question multiple-choice self-report inventory. A value of 0–3 is assigned to each answer. The cut-off for depressive symptoms is 10, and higher total scores indicate more severe symptoms. 16 The SF-36 was used to measure functional health and well-being from the patient’s perspective during the last 4 weeks. Two summary measures of HRQoL were calculated: the physical component and mental component. Using algorithms, each scale is transformed into a 0–100 score, in which lower scores indicate greater disability.
Statistical analysis
Descriptive statistics (mean, standard deviation, frequency tables) of the main variables were calculated. The data were then analyzed at the bivariate level. The chi-square test was used to compare categorical variables and the Student t test for continuous variables between clinical groups. The chi-square test was not considered applicable when one or more of the cells had an expected count <5.
To reduce false-positive results, the Bonferroni correction for multiple tests was performed according to the number of tests in each bivariate analysis in order to avoid type 1 error. Variables that retained statistical significance and were considered clinically relevant were included in the logistic regression analysis. A conditional entrance method was used to select variables in the model. All statistical hypotheses were two-sided and p-values < 0.05 were considered statistically significant. SPSS version 20 (SPSS Inc., Armonk, NY, USA) for Windows was used for all analyses.
Results
Sample recruitment and sample features
During the recruitment period, 528 patients attended the ADDC and were asked to participate. Ultimately, 401 (75.9%) patients were included. Reasons for exclusion were refusal to take part in the study (n = 86), refusal to have blood drawn (n = 9), language barrier (n = 6), and cognitive impairment (n = 26).
The final sample of 401 patients had a mean age of 45.4 ± 11.5 years and 301 (75.1%) were men. In total, 253 patients (63.1%) had a dual diagnosis and 148 (36.9%) had only SUD without psychiatric comorbidities. The most frequent SUDs involved alcohol (73.6%), cocaine (58.4%), cannabis (47.1%), heroin (38.2%), and benzodiazepines (30.9%). The sociodemographic, clinical, psychometric, and therapeutic features of patients with and without dual diagnosis are summarized in Table 1. The dual diagnosis group had a higher percentage of women (32%), Spanish natives (87.4%), and unemployed individuals (80.2%). In addition, these patients had a family history of psychiatric disorders more often (47%), were more prone to sedative use disorder (37.5%), had required more medical treatments for SUD control (88.8%), and had been previously hospitalized more often for detoxification (56.2%). Overall, dual diagnosis patients showed more depressive symptoms (according to the BDI-I) and had a poorer HRQoL.
Baseline characteristics of patients with and without a dual diagnosis and follow-up of those with hepatitis C.
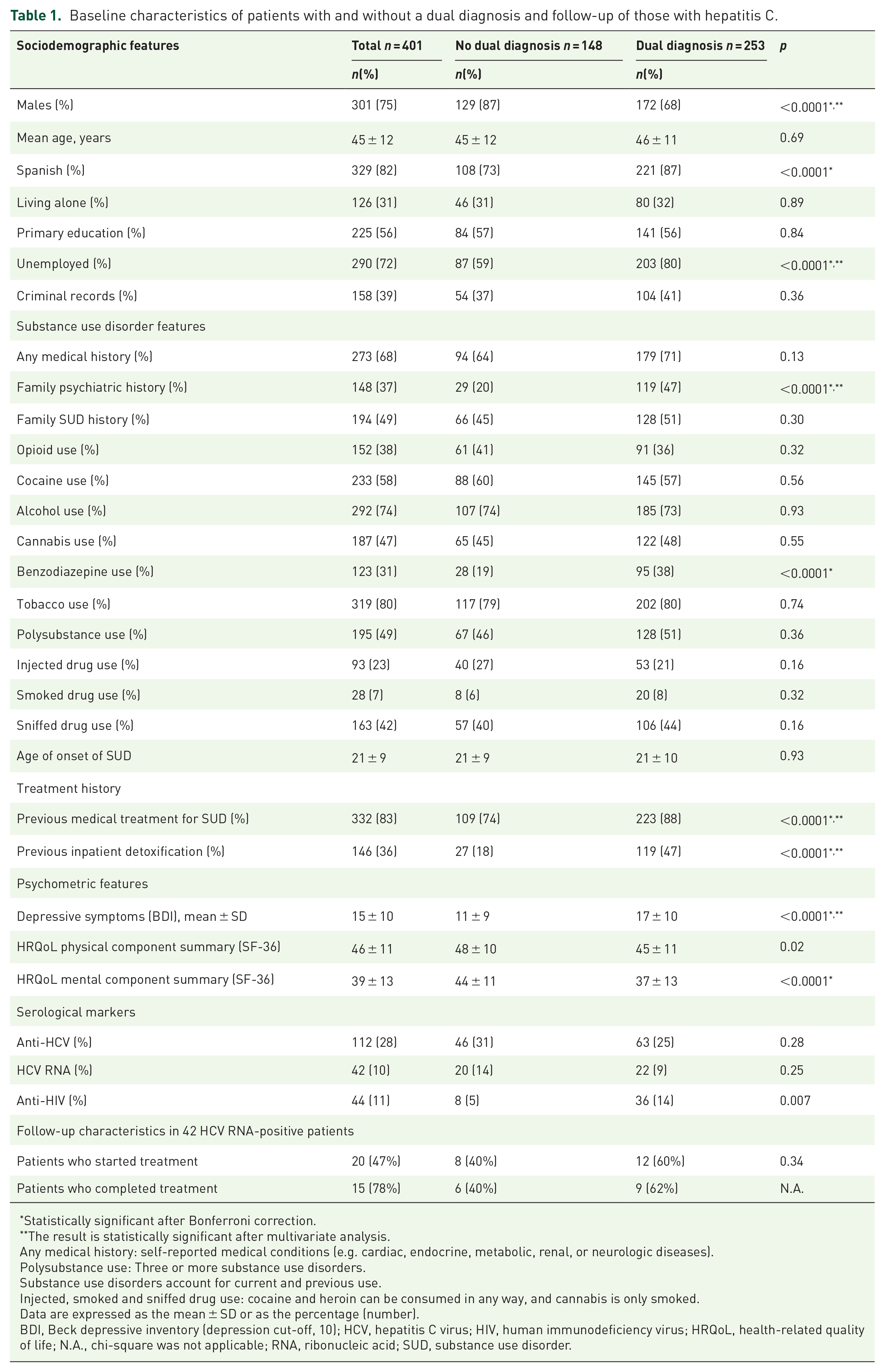
Statistically significant after Bonferroni correction.
The result is statistically significant after multivariate analysis.
Any medical history: self-reported medical conditions (e.g. cardiac, endocrine, metabolic, renal, or neurologic diseases).
Polysubstance use: Three or more substance use disorders.
Substance use disorders account for current and previous use.
Injected, smoked and sniffed drug use: cocaine and heroin can be consumed in any way, and cannabis is only smoked.
Data are expressed as the mean ± SD or as the percentage (number).
BDI, Beck depressive inventory (depression cut-off, 10); HCV, hepatitis C virus; HIV, human immunodeficiency virus; HRQoL, health-related quality of life; N.A., chi-square was not applicable; RNA, ribonucleic acid; SUD, substance use disorder.
The following variables were independently associated with dual diagnosis status: female sex [Odds ratio (OR):1.50], benzodiazepine use disorder (OR 2.48), previous medical treatment for SUD (OR 5.85), unemployment (OR 0.40), and depressive symptoms (OR 1.10) (Nagelkerke R2 = 0.32; chi-square = 64.27; p < 0.0001) (Table 2).
Multivariable analysis identifying factors independently associated with (a) dual diagnosis (b) anti-HCV antibody positivity.

Any medical history: self-reported medical conditions (e.g. cardiac, endocrine, metabolic, renal and neurologic diseases).
Substance use disorders account for current and previous use.
BDI, Beck Depression Inventory; CI, confidence interval; OR, odds ratio; SUD, substance use disorders.
Viral hepatitis and HIV infection
Among the 401 participants, 112 (27.9%) had anti-HCV antibodies and, of these, 42 (10.5% of the sample) had detectable HCV RNA. Of the 70 anti-HCV-positive patients with undetectable HCV RNA, 34 (48.6%) had been previously treated for HCV infection. Eight of the 42 patients (19%) testing HCV RNA positive were unaware of the infection. The most prevalent HCV genotypes (G) in patients with detectable HCV RNA were G1a (38.1%), G3 (31%), G1b (16.7%), G4 (7.1%), and G2 (2.4%). HIV prevalence was higher in the dual diagnosis group (14.3% versus 5.4% p = 0.007).
Results in relation to the presence of anti-HCV antibodies
Sociodemographic, clinical, psychometric, and therapeutic features in patients with and without anti-HCV antibodies are presented in Table 3. There were no significant differences in the prevalence of HCV infection between patients with and without a dual diagnosis. Patients testing anti-HCV-positive had a lower educational level and a higher percentage of criminal records. HIV infection was more frequent in anti-HCV positive patients (35.1% versus 1.7%, p < 0.0001). Use of any substance including non-injecting drug use (alcohol, cannabis, tobacco, and benzodiazepine), polysubstance use, injecting drug use, and early onset of SUD were related to a higher risk of anti-HCV-positive status. In addition, anti-HCV antibodies were more common in SUD patients who had anxiety or personality disorders, previous medical treatment for SUD, and previous detoxification hospitalizations. Regarding liver injury, no significant differences were seen in FIB-4 levels between anti-HCV-positive than anti-HCV-negative patients.
Baseline characteristics of patients according to the presence of anti-HCV antibody or HCV RNA.

Statistically significant after Bonferroni correction.
Any medical history: self-reported medical conditions (e.g. cardiac, endocrine, metabolic, renal or neurologic diseases).
Polysubstance use: three or more substance use disorders.
Substance use disorders account for current and previous use.
FIB-4 >3.25 indicated cirrhosis.
Data are expressed as the mean ± SD or as the percentage.
ALT, alanine transaminase; AST, aspartate transaminase; BDI, Beck depressive inventory (depression cut-off, 10); HCV, hepatitis C virus; HRQoL, health-related quality of life (lower scores indicate more disability); RNA, ribonucleic acid; SUD, substance use disorder.
On multivariable analysis, the following variables were independently associated with anti-HCV antibody positive status: opioid use disorder (OR: 27.8), cocaine use disorder (OR: 6.3), any medical history (OR: 21.75), and injecting drug use history (OR: 15) (Nagelkerke R2 = 0.62; chi-square = 303.69; p < 0.0001) (Table 2). The current results should be cautiously interpreted because there may be collinearity between opioid use disorder and injecting drug use. However, both variables are clinically relevant, and therefore they were maintained in the model.
Active hepatitis C infection and linkage to care
Among the 112 individuals testing positive for anti-HCV antibodies, HCV RNA was detected in 42 (37.5%) cases. No significant differences were found regarding dual diagnosis between HCV RNA-positive and HCV RNA-negative individuals (52% versus 63% p = 0.25). HCV RNA-positive individuals were younger (41.1 ± 7.3 versus 46.1 ± 8.3 p = 0.001), had received fewer previous SUD medical treatments (98.6% versus 85.7% p = 0.007), and had more pronounced liver injury [higher AST (72 IU/L versus 25 IU/L, p < 0.0001) and ALT levels (75 IU/L versus 21 IU/L, p < 0.0001)] than HCV RNA-negative patients (Table 3). There were no significant differences between these groups on multivariable analysis. The FIB-4 score was determined in all participants, with no significant differences between the clinical groups. FIB-4 was >3.25 in 3 of the 20 patients who started antiviral therapy. None of them had a history of liver decompensation.
Despite the centralized management, only 20 of the 42 treatment-eligible participants attended the first visit with the hepatologist. Patients did not attend for the following reasons: 11 were lost to follow-up, six returned to their country of origin, two did not want to start treatment, two were followed by hepatologists in other centers, and one was imprisoned.
In the comparison of HCV RNA-positive patients who did not start DAA treatment and those who did, patients who did not start reported use of a larger amount of cocaine during the last month (2.64 ± 3.74 versus 0.55 ± 1.8 g/week; p = 0.014), had a lower academic level (52.6% versus 22.7%; p = 0.047), and were more often unemployed (26.3% versus 4.5% p = 0.049). No differences were found for any of the other sociodemographic or clinical variables, or for dual diagnosis status.
Twenty patients started HCV treatment, but one patient was lost to follow-up at week 8 of therapy. Treatment adherence was excellent in 17 patients, whereas one patient stopped treatment 28 days before completion, and one other missed doses for a total of 6 days. In all 19 treated patients, HCV RNA was undetectable at treatment end. Fifteen patients attended the week 12 post-treatment visit and all had achieved sustained virological response (SVR), but the remaining four were lost to follow-up (Figure 1). In total, 90 visits had been scheduled up to week 12 post-treatment. Patients did not show up or showed up some days later in 27 (30%) of these visits.

Flow chart study enrollment.
The only adverse effects detected during HCV treatment were a mild increase in anxiety and asthenia, but none of the patients required discontinuation or modification of their concomitant psychiatric medication
Fifteen patients were tested for active drug use during antiviral therapy. Eleven of them tested positive for methadone or benzodiazepines, six for methadone or benzodiazepines plus certain illegal drugs (cocaine, opioids, or amphetamines), and three for illegal drugs alone.
Discussion
In accordance with the WHO effort to eliminate HCV infection worldwide, our hospital launched a multidisciplinary program to facilitate HCV screening and linkage to care in SUD patients, a population at high risk for acquiring this infection. This study describes the characteristics of these patients and the initial experience in screening, diagnosing, and treating them. We particularly focused on determining whether there would be differences as related to HCV between patients with SUD alone and those with dual diagnoses. The results show that dual diagnosis status did not lead to a higher risk of HCV infection or interfere with linkage to care or treatment compared with SUD alone. The similar rates of HCV infection between both groups of patients could be explained if the main factor for acquisition of hepatitis C is risk behavior related to substance abuse and not own psychiatric disease.14,17 Patients with dual diagnoses accounted for 63% of the cohort, a value consistent with findings from previous studies performed in similar settings.18–21
On screening, anti-HCV antibody tested positive in 27.9% of patients, and 10.5% of them had detectable HCV RNA. The HCV RNA prevalence found here in SUD patients is 20 times higher than the values reported in the general Spanish population,2,3 but it is lower than values reported in other SUD studies,6,11,22 mainly because of the contribution of injecting drug users in these previous reports. We found no differences in the prevalence of HCV viremia between individuals with a dual diagnosis and those with SUD alone.
In our study, HCV RNA-positive patients were younger and had received fewer previous medical treatments for SUD than those testing negative. These findings are reasonable, as older patients are likely to have had more frequent contact with medical care, and therefore more opportunities to be diagnosed and treated for all their diseases. 23 Hospital admission offers an opportunity to link these individuals to HCV care, as has been shown in a previous study. 24
Now that DAAs have provided effective antiviral treatment, the main issue in the HCV-infected population is improving access to care, a challenge well illustrated by the data from this study. We found that HCV diagnosis and management remains difficult in the SUD population despite the use of a centralized, multidisciplinary approach for care. All patients in the cohort were offered HCV testing based on reflex HCV RNA testing by venipuncture which, in some cases, discouraged study participation (7% of those who refused screening). Although the percentage is not extremely high, this is a modifiable factor that can be improved. Point-of-care HCV RNA testing by non-invasive dried blood spot analysis has shown high sensitivity and specificity10,25 and could be a feasible way to increase screening rates in the SUD population.
Lack of screening is the first barrier against eradication of HCV infection and may be one of the most important obstacles to achieving the WHO objective. 26 In Spain, the percentage of HCV RNA-positive individuals in the general population unaware of their infection has been estimated at 29.4%. 27 Studies conducted in SUD patients have reported values of 35% to 43%.10,28 In our sample, only 19% of HCV RNA-positive patients with SUD were unaware of the infection. This may be because of the current heightened awareness and management of this infection in our hospital outpatient clinics. Although this is an improvement, there remains a considerable reservoir of potentially transmissible infection that should be brought to light. A universal screening approach incorporating anti-HCV or HCV RNA reflex testing in emergency rooms and outpatient clinics when blood tests are performed for other purposes could be of value in this regard.
The difficulties to linkage to care and treatment seen here have been reported in other studies, where unemployment and, particularly, low educational level have been associated with low healthcare retention rates.29–31 In our cohort, these factors associated with failure to treat, as well as the use of large amounts of cocaine, were more common in patients refusing than accepting DAA therapy. These findings are not surprising. Cocaine users are more impulsive and often have a severe addiction, which leads to poorer clinical outcomes. 32 A low educational level interferes with the use of resources and decreases awareness of any risk. 33
Spain has a publically funded universal healthcare system in which everyone is entitled to treatment. With this in mind, the low percentage of SUD patients starting HCV therapy is worrisome. Among the 42 patients eligible for DAA therapy, 20 (47.6%) were treated: 15 (36%) achieved SVR, and five were lost to follow-up. These findings are in line with a study conducted in Spain assessing HCV therapy in an opioid treatment program, where 38% of 249 patients eligible for DAAs achieved SVR. 34 These results indicate that treatment uptake needs to be enhanced in these patients by close follow-up and motivational programs, but it should also be accompanied by preventive efforts such as educational interventions, counseling, and needle exchange programs. 35
Our study has several limitations. One concern is the small percentage of participants with detectable HCV RNA in our sample, which limited the power of some analyses. As the study was performed in daily clinical practice, the sample contained both injecting and non-injecting drug users, and previous treatment for HCV infection was not an exclusion criterion, factors that could explain this low percentage. A further limitation was that treatment could not be started in half the HCV RNA-positive individuals, mainly because they did not return to the ADDC and were lost to follow-up. Finally, self-reported instruments were used for certain factors studied; hence, there could be some recall bias.
The strengths of our study include its prospective design, real-world setting of daily practice, and enrollment of a large sample including all patients seen in a dedicated unit; that is, SUD patients using non-injecting as well as injecting drugs and dual diagnosis patients, a population with scarce data in the literature. In addition, centralized management with a multidisciplinary team provided more complete access and specialized care on several levels for these patients, although it did not achieve a high rate of treatment. The extensive evaluations included data on the psychiatric characteristics assessed by trained staff using validated instruments. Finally, HCV-related variables were well documented by reliable laboratory analyses.
In summary, the results of this study highlight a high prevalence of dual diagnosis and HCV in SUD patients. However, in general, dual diagnosis was not associated with a higher risk of HCV infection or more complex linkage to care for this condition. We found that despite centralized management by a multidisciplinary team, there are still considerable barriers to HCV care in this population, particularly regarding early screening and treatment. It is essential to address these barriers to achieve the WHO objective of HCV elimination.
Footnotes
Acknowledgements
This study was an investigator-initiated study funded by AbbVie. The funder had no role in the study design, or collection, analysis and interpretation of the data.
Author contributions
Conflict of interest statement
Lara Grau-López: No personal or financial conflicts of interest.
Cristina Marcos-Fosch: No personal or financial conflicts of interest.
Constanza Daigre: No personal or financial conflicts of interest.
Raúl Felipe Palma-Alvarez: No personal or financial conflicts of interest.
Ariadna Rando-Segura: No personal or financial conflicts of interest.
Jordi Llaneras: No personal or financial conflicts of interest.
Marta Perea-Ortueta: No personal or financial conflicts of interest.
Francisco Rodriguez-Frias: No personal or financial conflicts of interest.
Nieves Martínez-Luna: No personal or financial conflicts of interest.
Mar Riveiro-Barciela: Has received research grants from Gilead, and served as speaker for Gilead and Grifols. No personal conflicts of interest.
Josep Antoni Ramos-Quiroga: No personal or financial conflicts of interest.
Joan Colom: No personal or financial conflicts of interest.
Rafael Esteban: Has received research grants from Gilead and has served as advisor for Gilead, Bristol-Myers Squibb, and Novartis. No personal conflicts of interest.
María Buti: Has received research grants from Gilead and has served as advisor for Gilead, Bristol-Myers Squibb, and Novartis. No personal conflicts of interest.
