Abstract
All-oral, direct-acting antivirals (DAAs) have significantly improved the efficacy and safety of chronic hepatitis C (CHC) treatment but their effectiveness and safety among patients with chronic kidney disease (CKD) remains poorly understood. Our aim was to assess the efficacy and safety of DAAs for treatment of CKD patients. The National Library of Medicine through PubMed was searched for studies evaluating the efficacy of DAAs for the treatment of patients with CKD stages 4 or 5, as defined by the Kidney Disease Outcomes Quality Initiative guidelines [i.e. glomerular filtration rate (GFR) 15–29 ml/min per 1.73 m2 and GFR <15 ml/min per 1.73 m2, respectively, or hemodialysis or peritoneal dialysis]. Randomized clinical trials (RCTs) and relevant cohort studies were included if they were published in English and included sustained viral response after 12 weeks (SVR12) as a primary or secondary endpoint. After applying inclusion and exclusion criteria, eight studies (one RCT and seven cohort studies) following 350 patients were selected. For patients with CKD stage 4 or 5, ± hemodialysis, the overwhelming majority of DAA regimens were well-tolerated and resulted in SVR12 rates of 90–100%. Most studies were small, with the exception of one RCT evaluating elbasvir and grazoprevir. Overall, treatment of CHC in patients with CKD is highly effective with SVR12 rates similar to those seen in patients without CKD and with acceptable adverse event profiles. In patients with hepatitis C virus (HCV) genotype (GT) 1a, 1b or 4 and Stage 4 or 5 CKD, the best evidence available is for the use of elbasvir and grazoprevir. This combination as well as the combination of paritaprevir/ritonavir/ombitasvir/dasabuvir for HCV GT-1b are recommended. More studies are needed to assess efficacy and adverse effects of DAAs and their impact on CKD patients and to fully elucidate the effect of curing CHC on the natural history and sequelae of renal disease in CHC patients with CKD.
Keywords
Introduction
Globally, an estimated 170–180 million people are chronically infected with hepatitis C virus (HCV) leading to 500,000 deaths annually [Mohd Hanafiah et al. 2013]. In the United States (US), it is estimated that there are approximately 5 million people with chronic hepatitis C (CHC) [Edlin et al. 2015; Gish et al. 2015]. Untreated, CHC can progress to cause liver fibrosis, cirrhosis, hepatic decompensation, or hepatocellular carcinoma (HCC) [Hajarizadeh et al. 2013]. In addition, there are multiple extrahepatic manifestations of CHC, including kidney disease [Gill et al. 2016]. Although other renal diseases are sometimes seen with CHC, chronic kidney disease (CKD) in patients with CHC occurs most often in the presence of diabetes mellitus type 2. In addition, in patients with CHC, cryoglobulins found in 10–50% of patients [Lunel et al. 1996; Charles et al. 2009] may play a role in the development of CKD in select patients due to mixed cryoglobulinemia (MC). Cryoglobulins are immune complexes which are deposited in the mesangium of the kidneys, triggering glomerulonephritis [Azmi et al. 2015]. Direct deposition of HCV RNA and associated proteins may also contribute to the development of renal disease in CHC [Perico et al. 2009].
CKD is common in CHC patients. In one large population-based study, the prevalence of CKD was 16.5% in HCV-seropositive subjects, and there was a significantly higher prevalence of CKD stages ⩾3 (7.8%) in those who were HCV-seropositive compared with the population as a whole [Li et al. 2014]. Other large studies have confirmed that CHC is associated with a substantially increased risk for CKD and with more severe disease [Tsui et al. 2007; Lee et al. 2010; Chen et al. 2014]. CHC patients with other comorbidities, including diabetes and hyperlipidemia, are at even greater risk for CKD [Chen et al. 2014]. Conversely, the prevalence of CHC in patients with CKD, particularly those requiring hemodialysis (HD), is also high. In a survey of all US chronic HD centers anti-HCV was found in 7.8% of patients nationwide [Finelli et al. 2005], while in one urban outpatient HD center anti-HCV was found in 23.3% of patients, a finding strongly associated with a history of injection drug use [Sivapalasingam et al. 2002].
Patients with CKD, whether requiring or not requiring HD, have been less responsive to traditional, interferon (IFN)-based regimens, with sustained viral response after 12 weeks (SVR12) rates of approximately 30–50% [Sporea et al. 2006; Liu et al. 2008]. In addition, toxicities due to IFN or ribavirin (RBV) are often magnified in patients with CKD, too commonly resulting in premature discontinuation of therapy, higher dropout rates and treatment-associated mortality [Azmi et al. 2015]. The advent of all-oral, direct-acting antivirals (DAAs) has improved the efficacy, safety, and ease of administration of HCV treatment for most patients, with rates of sustained virologic response, i.e. an undetectable HCV RNA level using a sensitive assay (typically with a lower limit of 25 IU/ml) 12 weeks after completion of therapy (SVR12; i.e. ‘cure’) in patients with HCV genotypes 1–4 of over 90–95% [Afdhal et al. 2014; Ferenci et al. 2014; Feld et al. 2015; Zeuzem et al. 2015]. Treatment of subpopulations of CHC patients, however, including those with CKD and other comorbid diseases and conditions such as human immunodeficiency virus (HIV) and liver transplantation, requires special consideration, given that treatment of these subgroups has historically been more difficult, with increased adverse effects (AEs) on therapy, potential for drug interactions, dosing concerns and decreased efficacy of treatment [Rodriguez-Luna et al. 2004; Torriani et al. 2004; Neumann et al. 2006; Azmi et al. 2015].
Understanding the efficacy, safety, and tolerability of novel DAAs in patients with CKD is important for clinicians treating CHC patients with this common comorbidity. The aim of this review was to evaluate and assess the efficacy and safety of DAA therapies in the treatment of CHC for patients with concomitant CKD.
Methods
Using PubMed, a comprehensive literature search was performed for studies published from 2014 to 2016 that evaluated the efficacy of DAAs for the treatment of CHC and included patients with CKD. References of identified articles were searched for additional relevant articles. Randomized clinical trials and relevant cohort studies were included if they were published in English and included SVR12 as a primary or secondary endpoint. Studies presenting information exclusively about patients undergoing liver transplant, IFN-containing regimens, the retreatment of patients who failed previous DAA-only therapy, retrospective analyses, patients with decompensated cirrhosis, or in which SVR was not a primary outcome, or which did not include SVR12 data, were excluded. Data including study design, participant demographics, stage of liver disease, genotype information, previous treatment history, treatment regimens, treatment durations, and SVR were extracted by coauthors and recorded on a standardized electronic data collection sheet.
Results
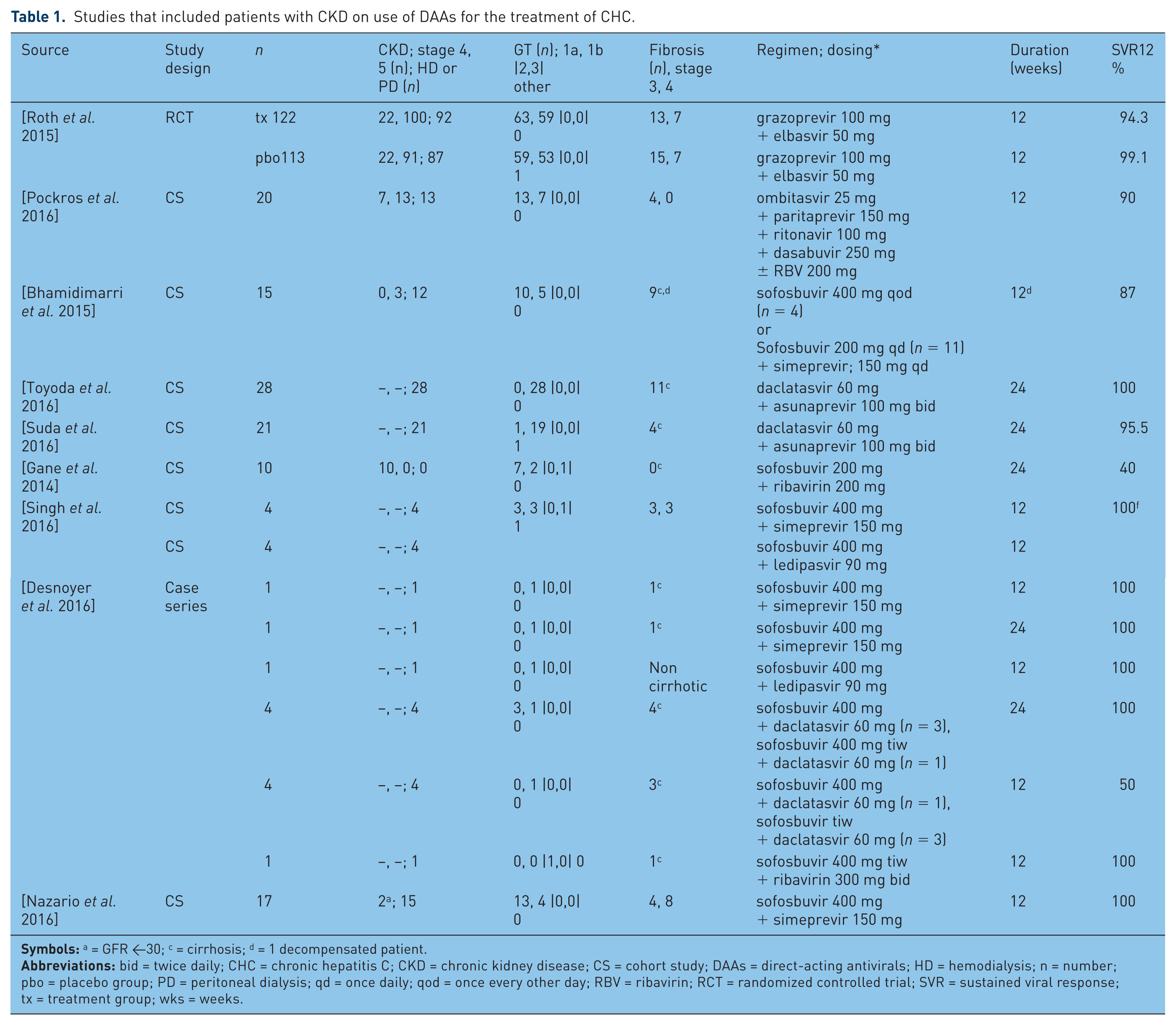
After applying inclusion and exclusion criteria, nine studies, including one randomized controlled trial (RCT) and seven cohort studies, were identified that evaluated the use of DAAs for the treatment of CHC and included patients with CKD. These nine studies enrolled only patients with CKD stage 4 or 5 [National Kidney Foundation, 2002] or on HD or peritoneal dialysis (PD). Results of each of these studies are summarized in Table 1.
Studies that included patients with CKD on use of DAAs for the treatment of CHC.
Evidence for the use of elbasvir-grazoprevir in chronic kidney disease
Elbasvir-grazoprevir (Zepatier, Merck & Co. Inc., Whitehouse Station, NJ, USA) is an oral, fixed-dose combination of a novel NS5A replication complex inhibitor (elbasvir) and a later generation novel HCV NS3/4A protease inhibitor (grazoprevir), both of which have antiviral activity in HCV GTs 1a and 1b. One RCT evaluated the use of elbasvir-grazoprevir for 12 weeks in patients with CKD [Roth et al. 2015]. In this study, 235 GT1 CHC patients with CKD stage 4 or 5 were enrolled. A total of 76% of patients were on HD; 18% of patients included had advanced stage 3 or 4 liver disease. Overall, 96.6% achieved SVR12. Of the seven patients who did not achieve SVR12, six discontinued treatment and one patient with HCV GT1b and CKD stage 5 relapsed. The most common AEs reported were headache (17%), nausea (15%), and fatigue (12%).
Evidence for the use of a combination of ombitasvir, paritaprevir, ritonavir and dasabuvir in chronic kidney disease
Ombitasvir is an NS5A inhibitor, paritaprevir an acylsulfonamide inhibitor of the NS3/4A serine protease and dasabuvir a nonnucleoside NS5B polymerase inhibitor, all of which have antiviral activity in HCV GTs 1a and 1b. Because ritonavir is a potent inhibitor of CYP3A4 enzymes it is used as a pharmacologic booster for paritaprevir. The regimen can be used with or without RBV. The four-drug combination is available in the Viekira Pak (AbbVie, Lake Bluff, IL, USA) in which ombitasvir, paritaprevir, and ritonavir are combined as a fixed-dose tablet, with dasabuvir taken as a separate tablet.
In a cohort study of 20 GT1 CHC patients with advanced stage 4 or 5 CKD, with or without a need for HD, patients were treated with ombitasvir, paritaprevir, ritonavir, and dasabuvir for 12 weeks [Pockros et al. 2016]. For patients with GT1a, RBV was added to the regimen. A total of 35% of patients had stage 4 CKD; 65% had stage 5 CKD; and 65% were on HD. Overall, two-thirds of the patients were GT1a; 35% were GT1b; and 20% had advanced stage 3 liver fibrosis. Overall, 18 of the 20 patients (90%) achieved SVR12. One patient died before reaching SVR12 due to left ventricular systolic dysfunction which was unrelated to either HCV or treatment with the DAAs. There was one patient with GT1a who had suboptimal compliance with the treatment regimen, with less than 92% adherence based on pill counts, and stopped taking RBV on day 58. Ultimately, this patient relapsed at post-treatment week 4. Most of the AEs reported were mild or moderate including fatigue, diarrhea, nausea, headache and dizziness; overall, 62% of HCV GT1a patients who took the regimen requiring RBV developed anemia as compared with no GT1b patients (regimen not requiring RBV). A total of four of the patients who developed anemia required erythropoietin and one developed a hemoglobin <8 g/dl.
Evidence for the use of daclatasvir and asunaprevir in chronic kidney disease
Daclatasvir (Daklinza, Bristol-Myers Squibb, New York, NY, USA) is an inhibitor of the HCV nonstructural protein NS5A. Asunaprevir (Sunvepra, Bristol-Myers Squibb, New York, NY, USA) is an inhibitor of the hepatitis C serine protease NS3/4A. Daclatasvir and asunaprevir have in vitro activity against HCV GTs 1–6 and 1, 4, 5 and 6, respectively [Manns et al. 2014]. The combination of asunaprevir and daclatasvir in patients with CKD was evaluated in two cohort studies [Suda et al. 2016; Toyoda et al. 2016]. One case-controlled study of Japanese patients evaluated the efficacy of asunaprevir and daclatasvir for 24 weeks in patients with HCV GT1b [Toyoda et al. 2016]. A total of 28 CHC patients on HD and 193 control CHC patients without renal dysfunction treated simultaneously with DAAs were enrolled. Overall, 56 of these controls were matched based on a propensity score to patients with CKD. All patients were infected with HCV GT1b and 39% of patients with CKD had compensated cirrhosis at baseline. All patients with CKD achieved SVR12. AEs were similar in patients with and without CKD; in particular, drug-induced liver injury was common and similar (21.4% versus 26.8% respectively) with a rapid normalization of alanine transaminase (ALT) after drug discontinuation.
In a second cohort study of asunaprevir and daclatasvir for 24 weeks, 21 Japanese patients on HD were included [Suda et al. 2016]. Overall, 90% of patients were infected with HCV GT1b, and four patients (19%) had cirrhosis. A total of 20 (95.5%) patients achieved SVR12 and 1 noncirrhotic patient (4.5%) relapsed at post-treatment week 4. Overall, one patient discontinued therapy at week 12 due to elevated ALT > 10 times the upper limit of normal, but nonetheless achieved SVR12. The most common AEs were nasopharyngitis, anemia and increased ALT in 28.6%, 28.6%, and 14.3% of patients, respectively. Of note, anemia was thought to be related to the underlying CKD. There is also a recent report of improvement of renal function in a CHC patient with liver cirrhosis and severe leg edema due to chronic renal dysfunction who was treated with daclatasvir and asunaprevir combination therapy [Tsuge et al. 2016]. During the course of therapy, the serum creatinine level nearly normalized and the leg edema gradually improved. The patient achieved SVR12, after which these improvements continued.
Evidence for the use of sofosbuvir and ribavirin in chronic kidney disease
Sofosbuvir (Sovaldi, Gilead Sciences, Inc., Foster City, CA, USA) is a potent inhibitor of the HCV NS5B region, encoding the HCV viral RNA polymerase. In one cohort study [Gane et al. 2014] sofosbuvir and RBV were used together for 24 weeks in 10 patients with stage 4 CKD. A total of seven patients were infected with HCV GT1a, two with GT1b, and one patient with GT3. Despite rapid on-treatment viral suppression, only 40% of patients achieved SVR12. Among the six patients who failed therapy, two patients discontinued therapy, including one patient who withdrew consent and one who was not compliant. The reported AEs were anemia (50%), headache (40%), pruritus (30%), and rash (30%).
Evidence for the use of sofosbuvir and simeprevir in chronic kidney disease
Simeprevir (Olysio, Janssen Therapeutics, Titusville, NJ, USA) is an NS3/4A protease inhibitor with efficacy against HCV GT1 and GT4. A total of three cohort studies evaluated the use of sofosbuvir and simeprevir in patients with CKD [Bhamidimarri et al. 2015; Nazario et al. 2016; Singh et al. 2016]. In one cohort study, sofosbuvir and simeprevir were used to treat 15 patients (11 receiving HD, one receiving PD, and three patients with CKD stage 5 and no dialysis) [Bhamidimarri et al. 2015]. A total of 60% (n = 9) of patients had cirrhosis, of whom one was decompensated. Overall, 93% (n = 14) of patients were treated for 12 weeks with sofosbuvir and simeprevir. The dosage of sofosbuvir varied; 73% (n = 11) of patients received 200 mg daily while the rest, 26% (n = 4), received 400 mg daily. Among patients treated for 12 weeks, 93% achieved SVR12. Overall, one patient with cirrhosis was treated for 24 weeks but relapsed. Overall, SVR12 was 87%. In a small subgroup analysis, SVR12 was higher in patients without cirrhosis (100%) as compared with patients with cirrhosis (78%).
In a second cohort study, 17 patients were treated with 12 weeks of sofosbuvir and simeprevir [Nazario et al. 2016]. A total of 76% of patients were infected with HCV GT1a, and 24% with GT1b. Overall, 88% of patients were on HD; 12% were not on HD but had a GFR < 30 ml/min. A total of 47% of patients were cirrhotic and 24% had stage 3 liver fibrosis. All patients achieved SVR12. Nearly 25% of the cohort (n = 4) reported mild AEs, with insomnia being the most common (12%). There were no discontinuations.
In a third cohort study, four patients on HD were treated with sofosbuvir and simeprevir for 12 weeks [Singh et al. 2016]. In a combined analysis of these patients and four others treated with the fixed-dose combination ledipasvir-sofosbuvir (Harvoni, Gilead Sciences, Inc., Foster City, CA, USA), all patients who completed therapy achieved SVR12. Half of the cohort (n = 4) experienced AEs while undergoing treatment, including nausea/vomiting, headache and insomnia. Overall, one patient experienced a decrease in hemoglobin >2 g/dl not requiring discontinuation.
Evidence for the use of sofosbuvir and ledipasvir in chronic kidney disease
In one cohort study, four patients on HD were treated with sofosbuvir and ledipasvir for 12 weeks [Singh et al. 2016]. As described above, in a combined analysis of these patients with four other patients treated with sofosbuvir and ledipasvir, all patients who completed therapy achieved SVR12.
Evidence for the use of sofosbuvir and daclatasvir in chronic kidney disease
In one cohort study [Desnoyer et al. 2016], eight patients on HD were treated with sofosbuvir and daclatasvir. A total of four patients were treated for 4 weeks, all of whom achieved SVR12. However, only two of the four patients treated for 12 weeks achieved SVR12 (50%). Both patients who relapsed were cirrhotic.
Discussion
Overall, treatment of CHC in patients with CKD is highly effective, with SVR12 rates similar to those seen in patients without CKD (Figure 1). It is important to note that to date most studies of DAAs in this population have been small, with the exception of elbasvir-grazoprevir for which the most rigorous evidence exists. Overall, AEs in CKD patients receiving DAAs is similar to those seen in patients without CKD, with the exception of anemia, which is increased in CKD patients requiring RBV-containing regimens. Additional studies are needed to assess the efficacy and full AE profile of DAAs and the impact of CHC cure on CKD for CHC patients with CKD.

Rates of SVR12 by regimen: data combined from all included trials.
Historically, HCV therapy in the setting of renal dysfunction has been challenging. With IFN-containing regimens, dosing adjustments, increased prevalence of moderate to severe AEs, and lower efficacy rates were barriers to treatment of patients with CKD [Deltenre et al. 2011; Azmi et al. 2015; Carvalho-Filho et al. 2015]. However, when possible, ‘cure’ of CHC has been associated with improvements in CKD [Ble et al. 2014; Tsuge et al. 2016]. Although DAAs have very substantially improved CHC ‘cure’ rates for patients without CKD, RCTs are needed to evaluate most of the available DAA regimens in CKD patients since these have not yet been carried out.
The single RCT done so far with CKD patients is the C-SURFER study which evaluated elbasvir-grazoprevir in patients with Stage 4 or 5 kidney disease, the majority of whom (76%) were on HD, and showed SVR12 rates similar to those seen in patients without CKD (99%) and without additional AEs [Roth et al. 2015]. It should be noted that in this study all patients were treated with 12 weeks of elbasvir and grazoprevir. Patients with HCV GT1a did not have baseline testing performed for the presence of NS5A resistance-associated variants (RAVs) (at the M28, Q30, L31 and Y93 positions) which have subsequently been shown to lead to reduced rates of SVR12 (70% versus 98% in patients with and without RAVs, respectively) [Merck & Co., 2016]. The impact of baseline RAVs in patients with CKD on SVR12 is not known. The American Association for the Study of Liver Diseases (AASLD) and the Infectious Diseases Society of America (IDSA) recommend that patients with CKD and HCV GT 1a, 1b or 4 be treated with 12 weeks of elbasvir-grazoprevir without RBV [American Association for the Study of Liver Diseases and Infectious Diseases Society of America, 2016] based on data from the C-SURFER study [Roth et al. 2015]. This is in contrast to their recommendations for patients with HCV GT1a without CKD, for whom the recommendation to extend treatment duration to 16 weeks with the addition of RBV has been made based on small studies showing increased SVR12 rates (100% with 16 weeks elbasvir + grazoprevir + RBV versus 70% with 12 weeks elbasvir + grazoprevir) [American Association for the Study of Liver Diseases and Infectious Diseases Society of America, 2016]. Further studies and postmarketing surveillance are required to understand if a prolonged duration or RBV should also be used for GT1a patients with CKD as well. Certainly, if RBV is deemed to be beneficial in the GT1a CKD patients with RAVs (known to occur in approximately 10% of patients with HCV GT1a), an increased incidence of anemia can certainly be expected [Saxena et al. 2016]. The impact of dose-reduction of RBV, as may be clinically indicated in CKD patients, on SVR12 rates for this patient population is not known.
Small numbers of patients with CKD were included in the trials evaluating the DAA combinations of ombitasvir, paritaprevir, ritonavir and dasabuvir with or without RBV, sofosbuvir and simeprevir, and the combination of asunaprevir and daclatasvir. However, it is encouraging that SVR12 rates were similar to those seen in clinical trials in patients without CKD [Ferenci et al. 2014; Kwo et al. 2016; Ogawa et al. 2016; Wei et al. 2016]. Although anemia occurred in over half of the patients who required the addition of RBV to this four-drug regimen, there were no discontinuations. It should be noted that no patients with cirrhosis were included in this study.
Asunaprevir and daclatasvir were evaluated only in patients with HCV GT1b and in a Japanese patient population who have historically good predictors of response to HCV treatment (GT1b, low body mass index, favorable IL28B CC GT), limiting the broad interpretation of results to other patient populations [Suda et al. 2016; Toyoda et al. 2016]. Although now approved in Japan, Bristol-Myers Squibb has decided that it will not pursue US Food and Drug Administration (FDA) approval of the daclatasvir-asunaprevir regimen for the treatment of HCV GT1b patients in the US. This regimen was complicated by high rates of ALT elevations, attributed to asunaprevir, in both patients with and without CKD.
Sofosbuvir and RBV in patients with HCV GT1 and CKD stage 4 also showed similar SVR12 rates to those seen in clinical trials with non-CKD patients [Osinusi et al. 2013; Gane et al. 2014]. While this regimen is not commonly used in patients with HCV GT1 given the availability of more effective DAA regimens, it is still commonly used for HCV GTs 2 and 3 [American Association for the Study of Liver Diseases and Infectious Diseases Society of America, 2016]. A higher incidence of anemia in CKD patients (~50%) compared with patients without CKD (~30%) can be expected given the requirement for RBV [Osinusi et al. 2013; Gane et al. 2014]. Only one study of sofosbuvir and ledipasvir in patients with CKD has been published and included only four patients, all of whom achieved SVR12 [Singh et al. 2016].
Overall, SVR rates using DAA regimens in patients with severe CKD stage 4 or 5, or on HD, have been high, mimicking results in patients without CKD. In these studies, no dose adjustments were made to the included DAAs with the exception of sofosbuvir. Sofosbuvir is unique in that it undergoes intracellular activation to an active triphosphate form that cannot be detected in the plasma, and then ultimately to an inactive, renally-eliminated metabolite [Kirby et al. 2015]. Given the renal elimination of this drug, the dosing of sofosbuvir in patients with CKD is unclear. In three of the five small studies included here, a full dose (400 mg daily) was used in patients, including those on HD, without any apparent AEs. Conversely, dose reduction of sofosbuvir (200 mg daily or 400 mg every other day) was used in two other small studies, without apparent reductions in SVR12. Further data are required to understand the optimal dosing and potential side effects of sofosbuvir in patients with CKD [AbbVie, 2016; Bristol-Myers Squibb, 2016; Merck & Co., 2016]. To date, anemia has been the only AE that is noticeably increased in patients with CKD who required RBV-containing regimens; however, we are limited by small sample sizes in all studies except for the elbasvir-grazoprevir trial. In addition, few patients with cirrhosis have been included in these studies.
The efficacy and tolerability of these DAAs, in particular elbasvir-grazoprevir, which has the most robust data on safety and efficacy, opens up for the first time the possibility of treatment of CHC patients requiring HD or who are on the renal transplant waiting list. This was previously not possible or poorly tolerated given the toxicities of IFN-containing regimens. While transplantation of CHC patients with kidneys from HCV-positive donors has been common in the past, the increased treatment and cure of CHC in potential donors may decrease the pool of these positive donors, thereby increasing the importance of HCV treatment pretransplant. In addition, the early treatment of CHC patients with CKD may prevent progression of renal disease in patients whose renal disease is related to HCV alone or in combination with other comorbidities.
Clinicians initiating treatment with DAA regimens in patients with severe renal impairment should monitor patients closely for AEs, as the complete profile is unclear at this time. In patients with HCV GT1a, 1b or 4 and Stage 4 or 5 CKD, the best evidence available is for the use of elbasvir and grazoprevir. This combination as well as the combination of paritaprevir/ritonavir/ombitasvir/dasabuvir for HCV GT1b is recommended by the AASLD/IDSA guidelines for HCV treatment [American Association for the Study of Liver Diseases and Infectious Diseases Society of America, 2016]. Additional studies of these regimens in patients with CKD, including in those with and without cirrhosis, are required to understand the complete safety and efficacy profiles of these regimens, and to fully elucidate the impact of CHC treatment and cure on halting the progression of renal disease in CHC patients, and perhaps even initiating CKD regression.
Footnotes
Acknowledgements
The authors wish to express their gratitude to Kartik Joshi for his editorial assistance and to Lark Lands, PhD for her invaluable assistance in preparing the manuscript for publication.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Dr Anita Kohli, MD has current activity with the scientific or clinical advisory boards of Gilead Sciences and Alexion Pharmaceuticals. Dr Kohli is a member of the Speakers Bureau and received honoraria from Merck & Co.
Dr Ali Alshati, MD and Dr Fawaz Georgie, MD have no conflict of interest.
Dr Richard Manch, MD has performed as a consultant or advisor to Gilead Sciences, AbbVie, Bristol-Myers Squibb, and Merck & Co. Dr Manch is a member of the Speakers Bureau for Gilead Sciences, AbbVie, Bristol-Myers Squibb, and Merck & Co. Dr Manch has had grants/research support from Gilead Sciences and AbbVie.
Dr Robert G. Gish, MD has had grants/research support from AbbVie, Benitec Biopharma, Gilead Sciences, and Merck & Co. Dr Gish has performed as a consultant or advisor to AbbVie, Akshaya Pharmaceuticals, AstraZeneca, Bristol-Myers Squibb, Genentech, Gilead Sciences, Hoffman-LaRoche, Ltd., Ionis Pharmaceuticals, Janssen, Merck & Co., Nanogen Biopharmaceutical, and Presidio Pharmaceuticals. Dr Gish has current activity with the scientific or clinical advisory boards of AbbVie, AstraZeneca, Genentech, Gilead Sciences, Janssen, Merck & Co., and Nanogen Biopharmaceutical. Dr Gish is a member of the Speakers Bureau for AbbVie, Bristol-Myers Squibb, Gilead Sciences, and Merck. Dr Gish is a minor stock shareholder of Cocrystal Pharma.
