Abstract
Background:
Interventional endoscopic ultrasound (EUS)-guided procedures such as EUS-guided celiac ganglia neurolysis (EUS-CGN) and EUS-guided broad plexus neurolysis (EUS-BPN) were developed to treat abdominal cancer-associated pain; however, these procedures are not always effective. The aim of this study was to explore predictors of pain response in EUS-guided neurolysis for pancreatic cancer-associated pain.
Methods:
This was a retrospective analysis of prospectively collected data of 112 consecutive patients who underwent EUS-BPN in our institution. EUS-CGN was added in cases of visible celiac ganglia. The neurolytic-spread area was divided into six sections and evaluated by post-procedural computed tomography scanning. Pain intensity was assessed using a visual analog scale (VAS), and a decrease in VAS scores by ⩾3 points after neurolysis was considered a good pain response. Univariable and multivariable logistic regression analyses were performed to explore predictors of pain response at 1 and 4 weeks, and complications.
Results:
A good pain response was obtained in 77.7% and 67.9% of patients at 1 and 4 weeks, respectively. In the multivariable analysis of these patients, the combination method (EUS-BPN plus CGN) was a significant positive predictive factor at 1 week (odds ratio = 3.69,
Conclusions:
EUS-BPN in combination with EUS-CGN was a predictor of a good pain response in EUS-guided neurolysis for pancreatic cancer-related pain. The larger number of neurolytic/contrast spread areas may lead to better outcomes in patients receiving combination treatment.
Keywords
Introduction
Visceral pain secondary to upper abdominal cancer is often difficult to control and poses a challenge to the physician. Celiac plexus neurolysis (CPN) consists of the chemical ablation of the celiac plexus (CP) and it can be used for the treatment of enduring pain caused by abdominal malignancies. Endoscopic ultrasound-guided CPN (EUS-CPN) was first described in 1996 [Wiersema and Wiersema, 1996] and is currently widely applied to treat upper abdominal cancer-associated pain [Gunaratnam et al. 2001; Collins et al. 2006; Michaels and Draganov, 2007; Puli et al. 2009; Penman and Rösch, 2009; Soweid and Azar, 2010; Kaufuman et al. 2010; LeBlanc et al. 2011; Sakamoto et al. 2011; Zou et al. 2012; Seicean, 2014; Seicean et al. 2013; Luz et al. 2014]. Current National Comprehensive Cancer Network guidelines (available at http://www.nccn.org) recommend EUS-CPN for the treatment of severe cancer-associated pain. Recently, different EUS approaches such as EUS-guided celiac ganglia neurolysis (EUS-CGN) [Levy et al. 2008] and EUS-guided broad plexus neurolysis (EUS-BPN) [Sakamoto et al. 2010] were developed to improve the efficacy of this technique. In a recent randomized, multicenter, controlled trial, EUS-CGN, a direct injection technique, was more effective than EUS-CPN in relieving pain [Doi et al. 2013]. Our group reported a single-center study comparing the pain-relieving efficacy of standard EUS-CPN with that of EUS-BPN that uses a 25-gauge needle to inject both sides of the superior mesenteric artery (SMA), and concluded that EUS-BPN provides better pain relief than EUS-CPN in patients with advanced pancreatic cancer [Sakamoto et al. 2010].
In several previous studies, EUS-CPN, -CGN, and -BPN showed satisfactory results and an excellent safety profile, indicating that they are promising methods; however, the efficacy of these techniques is not guaranteed. The aim of the current study was to explore predictors of pain response in patients undergoing EUS-guided neurolysis for abdominal pain caused by pancreatic cancer.
Patients and methods
Patients
This study was a retrospective analysis of prospectively collected data. Our database was reviewed to identify all patients who had undergone initial EUS-guided neurolysis for abdominal pain caused by pancreatic cancer between June 2008 and December 2014 in our institution. Patients who had been followed up at our institution for at least 4 weeks were eligible to enroll in the study. Relevant data were retrieved from the medical records of our institution. This study was approved by the Institutional Review Board of Kinki University Faculty of Medicine and written informed consent was obtained from all patients.
The inclusion criteria were as follows: (1) age older than 20 years; (2) enduring abdominal pain due to confirmed pancreatic cancer diagnosed by EUS-guided fine-needle aspiration (EUS-FNA), endoscopic biopsy or percutaneous biopsy; (3) presence of unresectable advanced pancreatic cancer. The contraindications included Eastern Cooperative Oncology Group performance status of 4, bleeding tendency (prothrombin time international normalized ratio > 1.5 and < 50,000 platelets) and presence of esophageal or gastric varices.
Pretreatment procedures
Patients were hydrated with an intravenous saline solution (500 ml) before the procedure to minimize the risk of hypotension. Patients were placed in the left lateral decubitus position with moderate sedation using intravenous midazolam with or without propofol. The level of sedation was titrated to optimize the tolerance to the procedure without compromising respiration, using a bispectral index-measuring monitor. Patients were continuously monitored during the procedure with an automated noninvasive blood pressure device, electrocardiogram tracing, and pulse oximetry.
Procedural techniques of EUS-BPN and EUS-CGN
In the present study, EUS-BPN was attempted in all patients and EUS-CGN was added in cases of visible celiac ganglia. EUS-BPN and CGN were performed using a linear array echoendoscope (GF-UCT 260; Olympus, Tokyo, Japan). US images were observed using an ALOKA ProSound SSD α-10 (ALOKA Co. Ltd., Tokyo, Japan). For EUS-BPN, at the level of the SMA, the probe was rotated clockwise toward the patient’s left until the SMA origin could no longer be visualized but the aorta could still be seen. A 25-gauge needle (Echo Tip Ultra, Cook Medical, Limerick, Ireland) filled with 0.9% saline solution was prepared and introduced through the biopsy channel and affixed to the hub. The 25-gauge needle was placed under direct EUS visualization adjacent and anterior to the lateral aspect of the aorta at a level above or next to the SMA. An aspiration test was then performed. A volume of 3 ml of 1% lidocaine was injected to prevent transient neurolytic agent-induced pain. Subsequently, a solution consisting of 9 ml of 99.5% absolute alcohol (Maruishi Pharmaceutical, Osaka, Japan) and 1 ml of contrast material (Iopamiron 300, Schering AG, Berlin, Germany; 300 mg d’Iode/ml) was injected up to 10 ml maximum. The needle was then withdrawn from the patient, flushed with 0.9% saline solution, and the same procedure was performed on the opposite side of the aorta (counter-clockwise rotation). Mainly, we chose to inject the neurolytic agents into both sides around the SMA. However, in some patients, the injection areas were decided depending on the locations of intervening vessels, the tumor, and the SMA. If there were intervening vessels with or without direct tumor invasion at the target area, we avoided injecting the neurolytic agents into the area and chose only the opposite side. If the injection target was below the SMA, we injected up to 10 ml of neurolytic agents into each side below the SMA and also around the celiac artery (CA). When we injected neurolytic agents in all four sites (two sides below the SMA and two sides around the CA), a maximum of 40 ml of neurolytic agent was used for EUS-BPN per patient.
One session consisted of EUS-BPN and a subsequent attempt to perform EUS-CGN. After visualization of the celiac trunk, the scope was rotated clockwise, enabling visualization and identification of the left adrenal gland. Most frequently, celiac ganglia could be visualized at the left of the CA, between the aorta and the left adrenal gland, at a level between the CA and the left renal artery. Ganglia were also visualized cephalad to the CA in some cases. Hypoechoic nodular structures linked by hypoechoic threads residing in the periphery of this region were defined as celiac ganglia. EUS-CGN was performed by direct ganglia injection. During EUS-CGN, direct ganglia injection was performed in as many visualized ganglia as possible. For each ganglion, 1–2 ml of the mixed solution described above containing pure alcohol and contrast medium was injected. All visualized celiac ganglia were subjected to the above procedure.
When only EUS-BPN was performed without EUS-CGN, the procedure was categorized as the single method. When EUS-BPN was performed in combination with EUS-CGN, the procedure was categorized as the combination method. Schematic images of EUS-BPN and -CGN are shown in Figures 1 and 2, respectively.
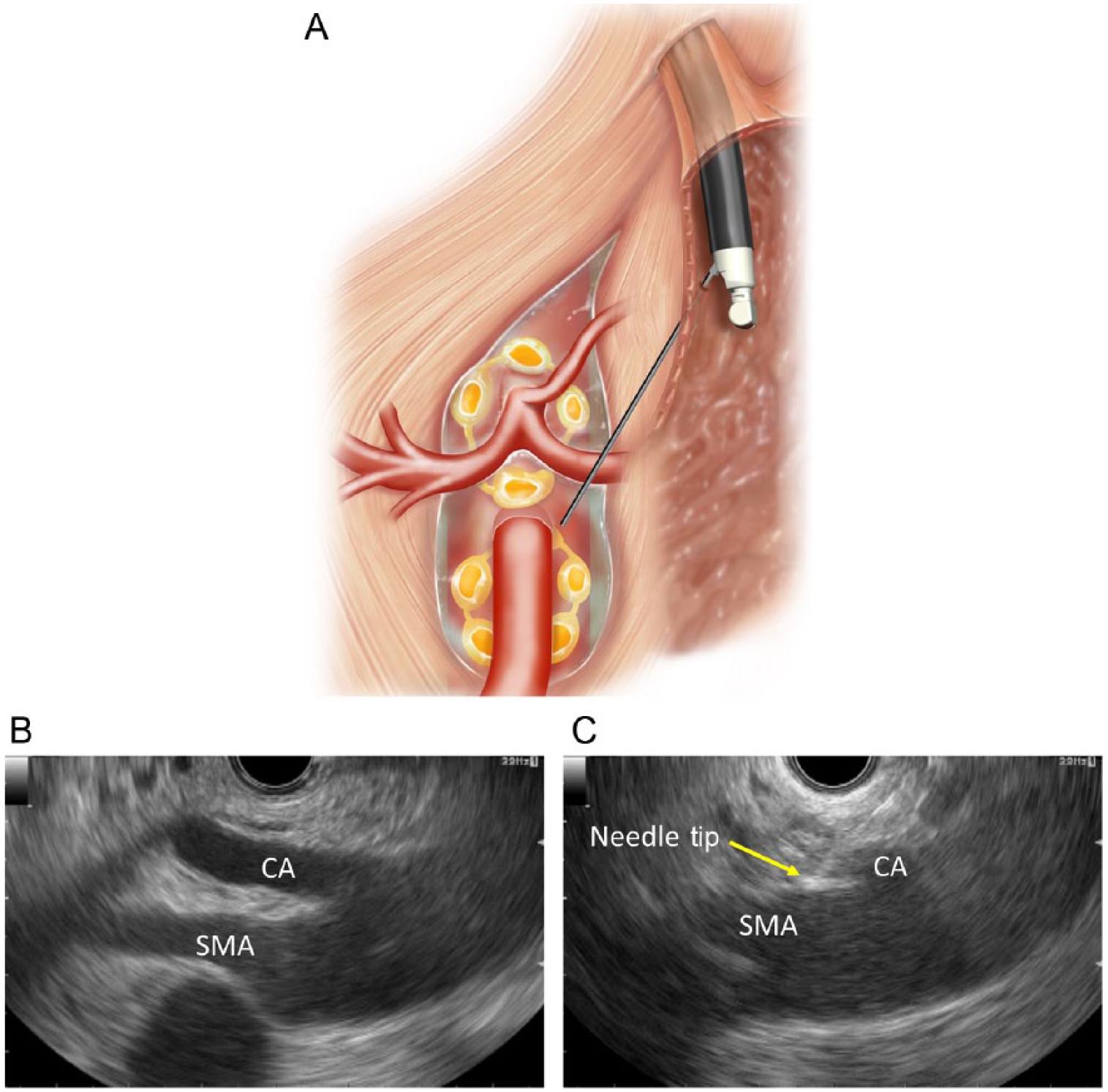
Endoscopic ultrasound-guided broad plexus neurolysis (EUS-BPN).

Endoscopic ultrasound-guided celiac ganglia neurolysis (EUS-CGN).
Pain scores
Pain scores were determined using a standardized 11-point continuous visual analog scale (VAS), with ‘0’ indicating no pain, ‘5’ indicating moderate pain and ‘10’ representing the worst pain ever. A good pain response was defined as a decrease in the VAS score by ⩾3 points without additional opioid medication at 1 or 4 weeks after neurolysis. To minimize subjective variations in the evaluation of VAS scores, the same physician (H. I.) explained the pain intensity scale to all patients. The physician was unaware of the detailed endoscopic procedures except that he was informed that the patients were suffering from abdominal pain caused by pancreatic cancer. At each follow-up visit, detailed instructions explaining how to assess the VAS were read aloud and the patients then informed the same physician of the VAS score that best reflected their pain status. The physician recorded the pain rating as well as patients’ responses in the analgesic questionnaire.
Computed tomography assessment
Computed tomography (CT) scanning was performed immediately after the procedure to confirm the neurolytic/contrast spread area [Sakamoto et al. 2006]. Serial CT images were obtained between the upper and lower limits of the neurolytic/contrast spread. To evaluate the spread pattern, the region including the CA and SMA was divided on the frontal plane into six areas: upper right and left (above the CA), middle right and left (between the CA and SMA), and lower right and left (below the SMA) (Figure 3). The CP is located in the upper and middle areas, whereas the superior mesenteric plexus (SMP) and the inferior mesenteric plexus (IMP) are located in the middle and lower areas. The relationship between the number of areas of neurolytic/contrast spread and the subsequent level of pain response was then analyzed.

A. Division of the celiac, superior mesenteric, and inferior mesenteric regions into six areas: two upper areas (①, upper right; ②, upper left), two middle areas, (③, middle right; ④, middle left), and two lower areas (⑤, lower right; ⑥, lower left).
Statistical analysis
All statistical analyses were performed using SAS version 9.4 software (SAS Institute Inc., Cary, NC, USA). Univariable and subsequent multivariable analyses were performed using logistic regression to explore predictors of pain response at 1 and 4 weeks and to evaluate complications. The response variables for univariable and multivariable analysis were responders and nonresponders. The explanatory variables for univariable analysis were as follows: age, sex, performance status, initial VAS score, preintervention opioid dose (dose in morphine equivalents), session timing, presence of ascites, tumor size, tumor location, tumor staging, procedure method (combination or single), number of ganglia injected, and injected alcohol dose. The session timing means whether EUS-guided neurolysis was performed at initial cancer identification (early neurolysis) or during follow up (late neurolysis). For assessment of the tumor location, the cancers were categorized as pancreatic head
Patients were divided into five subgroups according to the number of neurolytic/contrast spread areas (six, five, four, three, and two or less areas). In the subgroup analysis, response rates were calculated and compared between the subgroups using logistic regression analysis. The numbers of neurolytic/contrast areas were also measured for the two procedure methods (single or combination) using logistic regression analysis. Statistical significance was set at a
Results
Between June 2008 and December 2014, 112 consecutive patients underwent initial EUS-guided neurolysis and all of them were followed up for at least 4 weeks after the procedure at our institution. Patient demographics, disease, and treatment characteristics are shown in Table 1 for the 112 patients with available data for analysis. All patients had malignant tumors histologically confirmed by EUS-FNA (
Demographics and clinical characteristics of patients who underwent endoscopic ultrasound-guided neurolysis (
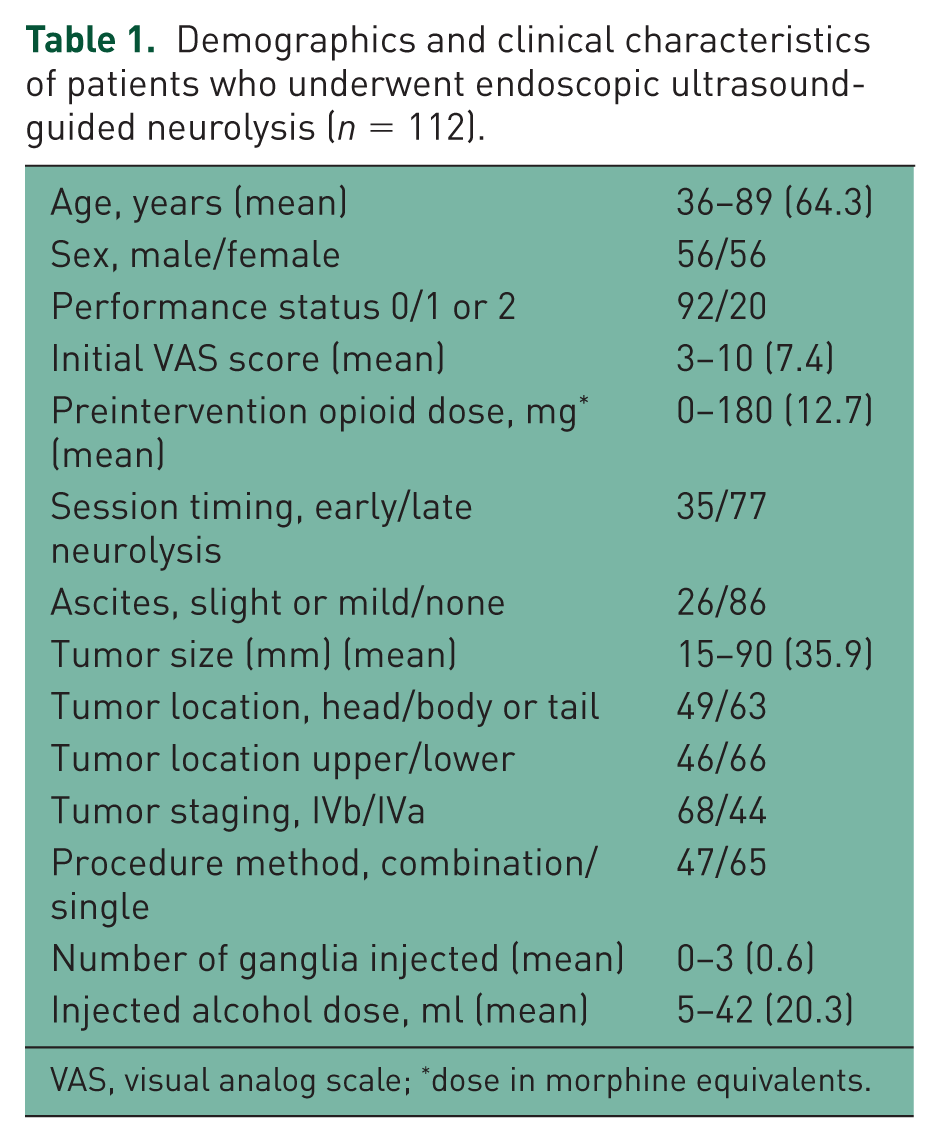
VAS, visual analog scale; *dose in morphine equivalents.
A. Univariable analysis of factors associated with pain response after 1 week in the enrolled cohort of 112 patients.
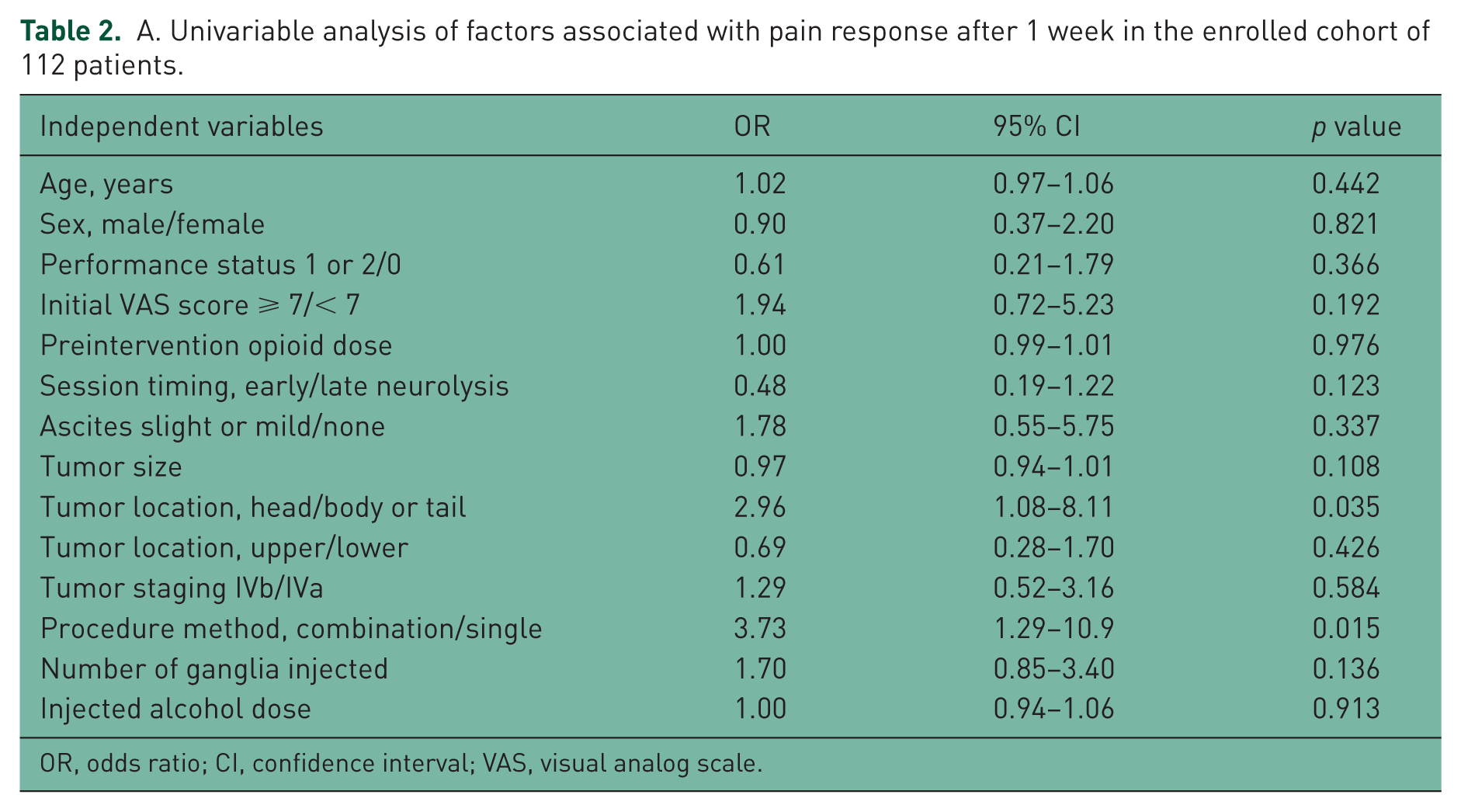
OR, odds ratio; CI, confidence interval; VAS, visual analog scale.
B. Multivariable analysis of factors affecting pain response after 1 week.

OR, odds ratio; CI, confidence interval; EUS, endoscopic ultrasound; BPN, broad plexus neurolysis; CGN, celiac ganglia neurolysis.
A. Univariable analysis of factors associated with pain response after 4 weeks in the enrolled cohort of 112 patients.

OR, odds ratio; CI, confidence interval; VAS, visual analog scale.
B. Multivariable analysis of factors affecting pain response after 4 weeks.

OR, odds ratio; CI, confidence interval; EUS, endoscopic ultrasound; BPN, broad plexus neurolysis; CGN, celiac ganglia neurolysis.

Relationship between neurolytic/contrast spread areas and pain response rate after 1 week (A) and 4 weeks (B). The upper two-bar graph shows a comparison of response rates between patients with at least four and those with less than four neurolytic/contrast spread areas. The lower five-bar graph shows the comparison of response rates between patients with six, five, four, three, and two or less neurolytic/contrast spread areas.
Complications occurred in 22.3% of 112 patients. Most of the complications were minor and self-limited, and included transient inebriation (8.0%), transient hypotension (4.5%), transient increase of pain (3.6%), and transient diarrhea (3.6%). Major complications occurred in one patient (0.9%), who developed acute paraplegia after the single method (EUS-BPN alone). In this patient, a total volume of 20 ml was injected into both sides. The number of neurolytic/contrast spread areas was six. MRI performed the next day demonstrated diffuse intramedullary T2 hyperintensity below the T-11 level to the conus medullaris, although the CT scan performed immediately after endoscopic treatment revealed no spread of the neurolytic/contrast agent into the spine. In the univariable analysis, there were no significant predictors of complications.
Discussion
Two meta-analyses of the utility of EUS-guided neurolysis for unresectable abdominal cancer-related pain showed an alleviation rate of approximately 80%, with a treatment duration of approximately 4–5 weeks [Puli et al. 2009; Kaufuman et al. 2010]. In the present study, cancer-associated pain was relieved by EUS-guided neurolysis in approximately 78% and 68% of patients at 1 and 4 weeks, respectively. The remaining patients did not respond to EUS-guided neurolysis. Analysis of the predictors of pain response in patients undergoing EUS-guided neurolysis showed that the combination method was the best predictor of pain response at both 1 and 4 weeks after EUS-guided neurolysis.
Whether bilateral injection of neurolytic agents is superior to central injection during EUS-CPN is controversial. LeBlanc and colleagues randomized 50 patients with pancreatic cancer to central or bilateral EUS-CPN groups, and found no difference in efficacy between the two groups [LeBlanc et al. 2013]. Sahai and colleagues evaluated the efficacy of bilateral injection compared with central injection in 160 patients, and found that bilateral injection was the only predictor of >50% of pain reduction by day 7 [Sahai et al. 2009]. However, these two reports did not compare the neurolytic spread between the bilateral and central injection methods.
Immediate assessment of the neurolytic spread using CT scanning is useful for the prediction of pain relief, as a wide neurolytic/contrast spread area is associated with a high response rate [Sakamoto et al. 2006, 2010]. In another study by Iwata and colleagues, limited distribution of alcohol to the left side of the CA was a significant factor associated with a negative response to EUS-CPN [Iwata et al. 2011]. In the present study, the response rates at 1 and 4 weeks correlated with the number of neurolytic/contrast spread areas.
In EUS-CGN, celiac ganglia are visualized by EUS in 62.5–89.4% of patients [Gerke et al. 2006; Gleeson et al. 2007; Ha et al. 2008; Kaufuman et al. 2010; Ascunce et al. 2011; Wang et al. 2012]. A retrospective study by Ascunce and colleagues showed that visualization of the celiac ganglia with direct injection into the ganglia is the best predictor of pain improvement after EUS-CPN [Ascunce et al. 2011]. In a recent randomized multicenter trial by Doi and colleagues, EUS-CGN was more effective than EUS-CPN in providing pain relief [Doi et al. 2013]. In the present study, additional EUS-CGN (the combination method in this study) led to a better pain response than that achieved with the single method. The number of neurolytic/contrast spread areas was higher in patients receiving combination treatment (EUS-BPN plus CGN) than in those treated with the single method (EUS-BPN alone). The larger number of neurolytic/contrast spread areas may lead to better outcomes in patients receiving combination treatment.
Three articles on the correlation between tumor location and the response to CPN have been published, although the tumor location associated with better pain relief remains controversial [Rykowski and Hilgier, 2000; Ascunce et al. 2011; Iwata et al. 2011]. Ascunce and colleagues reported that tumors located outside the head of the pancreas are weakly associated with a good response to EUS-CPN and EUS-CGN [Ascunce et al. 2011]. On the other hand, Rykowski and Hilgier reported that the posterior transcutaneous CPN technique is more effective in tumors involving the head of the pancreas than in those affecting the body and tail of the pancreas [Rykowski and Hilgier, 2000]. In the present study, multivariable analysis revealed that patients with tumors located at the pancreatic head tended to respond better than those with tumors at the pancreatic body or tail after 1 week, which is consistent with the results described by Rykowski and Hilgier. Iwata and colleagues reported that EUS-CPN is less effective in patients with direct invasion of the CP [Iwata et al. 2011]. Pain caused by direct invasion of the CP is less likely to be induced by tumors of the head of the pancreas than by those of the body or tail [Iwata et al. 2011]. Direct cancer invasion from the pancreatic body to the CP may restrict the spread of neurolytic solution and limit the subsequent pain relief.
The CP extends down from the origin of the CA to the origin of the SMA. The SMP and IMP are situated on the lateral and anterior aspects of the aorta between the origin of the SMA and the inferior mesenteric artery. The CP, SMP and IMP are composed of a network of nerve fibers that originate from both sympathetic and parasympathetic nervous systems. Therefore, we hypothesized that pain relief might be achieved in patients with unresectable pancreatic cancer by using EUS-guided broad plexus neurolysis, because this could interrupt extensive nociceptive impulses from the abdomen. In a previous study from our group, EUS-BPN provided patients with advanced abdominal cancer with better pain relief than standard EUS-CPN without incurring serious complications, especially in cases in which the cancer expanded extensively within the abdominal cavity beyond the distribution of the CP [Sakamoto et al. 2010]. Only 19% of the EUS-CPN patients achieved satisfactory, long-lasting pain relief, whereas the EUS-BPN procedure was significantly more effective in lower cancer patients, for whom 79% achieved pain relief [Sakamoto et al. 2010]. In another study using CT guidance, broader plexus neurolysis, including that of the celiac, inferior mesenteric, and superior hypogastric plexuses produced effective immediate pain relief in all patients without serious adverse events, although 48.6% experienced transient diarrhea [Kitoh et al. 2005]. Therefore, in the present study, we performed EUS-BPN as the first line procedure and used tumor location (upper
In the randomized pilot study by LeBlanc and colleagues comparing the effects of 10 and 20 ml of alcohol injected during neurolysis, there was no difference in complete pain response between the two groups [LeBlanc et al. 2011]. Similarly, in the present study, the dose of injected alcohol did not affect the pain response.
With respect to the timing of EUS-guided neurolysis sessions, Wyse and colleagues reported that early EUS-CPN performed at the time of diagnostic- and staging-EUS, provides better pain relief than conventional management, in addition to preventing progressive increases in morphine consumption [Wyse et al. 2011]. In the present study, the timing of EUS-guided neurolysis did not affect its efficacy. These results suggest that EUS-guided neurolysis may be effective not only at the time of initial cancer detection (early neurolysis), but also during follow up (late neurolysis), although our study included a smaller number of patients who underwent early neurolysis.
A recent review that included 15 studies of EUS-guided neurolysis found that complications occurred in 21% of 661 patients [Alvarez-Sánchez et al. 2014]. Most of the reported complications were minor and self-limited, usually lasting less than 48 hours, and were attributed to disruption of sympathetic activity. In the present study, minor complications occurred in 22.3% of 112 patients and all minor complications were transient and self-limited. There was only one major complication consisting of acute spinal cord infarction with paraplegia in a patient treated by the single method (EUS-BPN alone). Serious complications after EUS-guided neurolysis are uncommon [O’Toole and Schmulewitz, 2009; Alvarez-Sánchez et al. 2014]. A recent overview of the safety and complications of EUS-guided neurolysis reported serious complications in 0.2% of EUS-guided neurolysis cases [Luz et al. 2014; Alvarez-Sánchez et al. 2014], and two cases of acute paraplegia after EUS-guided neurolysis have been reported [Fujii et al. 2012; Mittal et al. 2012]. In the present study, the complication rate was not related to the predictor of good pain response (procedure methods), suggesting that the combination method provides better pain relief without serious complications.
The present study had several inherent limitations. One potential limitation was that this study was retrospectively performed in a single institution. The second potential limitation of the present study was the lack of double blinding for the selection of patients for the two procedures. Additional EUS-CGN was performed only when celiac ganglia were detected on EUS, and visible celiac ganglia (42.0%) were detected in a lower proportion of patients than that reported previously (62.5–89.4%). The visibility of some ganglia can be impaired by the hyperechoic appearance of the alcohol injected during EUS-BPN. We feared that the hyperechoic appearance of the ganglia located on the puncture line for EUS-BPN might impair the visibility of the SMA if the ganglia had been treated before EUS-BPN. However, an alternative method using EUS-CGN before EUS-BPN might facilitate visualization of the ganglia. The third limitation was the subjective evaluation of pain. Because pain is difficult to measure objectively, a quantitative analysis of the efficacy of the prospective method is required. The fourth limitation was the short duration of follow up. It was difficult for us to follow up patients for longer than 4 weeks, because some patients were transferred to a hospice and had a short survival time.
Conclusion
The combination method (EUS-BPN plus CGN) was a predictor of better pain relief, suggesting that the combination method might improve the efficacy of EUS-guided neurolysis. The larger number of neurolytic/contrast spread areas might contribute to the better efficacy of the combination method. Further prospective, randomized control studies to compare EUS-BPN with and without EUS-CGN are needed to evaluate the efficacy of the different approaches to EUS-guided neurolysis.
Footnotes
Author contributions
Masayuki Kitano and Hiroki Sakamoto: manuscript writing, drafting conception and design, performing the endoscopic procedure. Kosuke Minaga and Takeshi Miyata: manuscript writing, analyzing the data and providing clinical advice. Hajime Imai, Kentaro Yamao, Ken Kamata, Shunsuke Omoto, Kumpei Kadosaka, Toshiharu Sakurai and Naoshi Nishida: contribution to writing the manuscript and providing clinical advice. Yasutaka Chiba: statistical analysis of data. Masatoshi Kudo: contribution to writing the manuscript, drafting conception and design.
Funding
The present study was supported by grants from the Japan Society for the Promotion of Science (grant numbers 22590764 and 25461035).
Conflict of interest statement
The authors declare that there is no conflict of interest.
