Abstract
Pancreatic cancer serves as the third leading cause of cancer-associated morbidity and mortality in the United States, with a 5-year survival rate of only 12% with an expected increase in incidence and mortality in the coming years. Pancreatic ductal adenocarcinomas constitute most pancreatic malignancies. Certain genetic syndromes, including Lynch syndrome, hereditary breast and ovarian cancer syndrome, hereditary pancreatitis, familial adenomatous polyposis, Peutz–Jeghers syndrome, familial pancreatic cancer mutation, and ataxia telangiectasia, confer a significantly higher risk. Screening for pancreatic malignancies currently targets patients with germline mutations or those with significant family history. Screening the general population is not currently viable owing to overall low incidence and lack of specific tests. Endoscopic ultrasound (EUS) and its applied advances are increasingly being used for surveillance, diagnosis, and management of pancreatic malignancies and have now become an indispensable tool in their management. For patients with risk factors, EUS in combination with magnetic resonance imaging/magnetic resonance cholangiopancreatography is used for screening. The role of endoscopic modalities has been expanding with the increased utilization of endoscopic retrograde cholangiopancreatography, EUS-directed therapies include EUS-guided fine-needle aspiration and EUS-fine-needle biopsy (FNB). EUS combined with FNB has the highest specificity and sensitivity for detecting pancreatic cancer amongst available modalities. Studies also recognize that artificial intelligence assisted EUS in the early detection of pancreatic cancer. At the same time, surgical resection has been historically considered the only curative treatment for pancreatic cancer, over 80% of patients present with unresectable disease. We also discuss EUS-guided therapies of physicochemicals (radiofrequency ablation, brachytherapy, and intratumor chemotherapy), biological agents (gene therapies and oncolytic viruses), and immunotherapy. We aim to perform a detailed review of the current burden, risk factors, role of screening, diagnosis, and endoscopic advances in the treatment modalities available for pancreatic cancer.
Plain language summary
Pancreatic cancer serves as the third leading cause of cancer-associated morbidity and mortality with increasing incidence in the United States. At the time of presentation, these cancers are advanced and often unresectable with a dismal cumulative survival rate. Our review aims to shed light on endoscopic advances in diagnosing and managing pancreatic cancer. This review will help understand the role of screening in general and high-risk populations and review their associated risk factors. We also discuss the standing of established and emerging endoscopic modalities coupled with the era of existing chemotherapeutic regimens and surgical management and their performance metrics. We also explore the need for future studies in endoscopic ultrasound-guided therapeutic modalities that may pave as a treatment option for patients with pancreatic malignancies.
Keywords
Introduction
Pancreatic cancer is the 12th most common cancer worldwide but the 7th most common cause of cancer-related death, with incidence higher in men than women. 1 In the United States, it accounts for a third of cancer-associated morbidity and mortality. 2 At the time of presentation, these cancers are advanced and often unresectable with a dismal cumulative survival rate. In 2023, about 64,050 new cases of pancreatic cancer were detected and 50,550 attributed deaths in the United States with a dismal 5-year survival rate of 12%.3,4 This survival rate has increased from about 5% in 2000, which has been largely attributed to advances in chemotherapeutic modalities. 5 With its steadily increasing incidence, it is projected to be the second leading cause of cancer-related mortality in the United States by 2030. 6 Despite the combined increasing incidence and growing knowledge of the disease process, its pathophysiology, and treatment options, it remains cancer with an aggressive clinical course and poor outcomes. 7 The incidence of pancreatic ductal adenocarcinomas (PDAC) is five times higher in developed countries as compared to developing nations, with an average incidence of eight cases per 100,000 people in North America and an average age of 65 years at the time of diagnosis.8,9
PDAC constitutes over 90% of pancreatic malignancies, with other types being pancreatic acinar cell carcinoma, neuroendocrine tumors, and cystadenocarcinomas. Among PDACs, about two-thirds originate from the head and one-third arise from the body and tail of the pancreas.10,11
In this narrative review, the search strategy included studies published from inception to December 2023 in PubMed, EMBASE, and Cochrane library databases with keywords—“endoscopy,” “endoscopic ultrasound,” “endoscopic retrograde cholangiopancreatography,” and “pancreatic cancer,” “pancreatic ductal adenocarcinoma,” “pancreatic tumor,” “carcinoma of pancreas,” “endoscopic diagnosis/treatment/therapy/intervention/ablation/injection,” “celiac plexus neurolysis.” The search was restricted to articles in the English language and included case series, retrospective, prospective, randomized controlled trials, and animal and human studies. Abstracts and case reports were not included. Articles were screened by authors for appropriateness for inclusion in the review. References of included articles were also screened for thoroughness.
Our review aims to shed light on endoscopic advances, along with their safety and feasibility, in diagnosing and managing pancreatic cancer. This review will help understand the role of screening in general and high-risk populations and review their associated risk factors. We also discuss the standing of established and emerging endoscopic modalities coupled with the era of existing chemotherapeutic regimens and surgical management and their performance metrics.
Risk factors
A majority of diagnosed cases of pancreatic malignancy are sporadic due to its incidence and overall poor survival rates; however, there have been certain hereditary and non-hereditary risk factors that have been identified.
Hereditary risk factors
A number of inherited genetic alterations, including germline mutations, have been identified that increase the risk of pancreatic cancer when compared to the general population. Genetic conditions implicated in PDAC include hereditary breast and ovarian cancer syndrome (BRCA2 and BRCA1 mutation), Lynch syndrome (MMR genes mutation), hereditary pancreatitis (PRSS1 mutation), familial adenomatous polyposis (APC gene), Peutz–Jeghers syndrome (STK11 mutation), familial atypical multiple mole melanoma syndrome (CDKN2A mutation), familial pancreatic cancer mutation (FPC gene mutation), and ataxia telangiectasia (ATM) gene mutation. These genetic predispositions can have as high as a 132-fold increased risk of developing pancreatic malignancy when compared to the general population.12,13 Table 1 summarizes the most commonly implicated genetic conditions along with their involved genes (when applicable) and the relative risk associated with each of these syndromes linked to the development of pancreatic cancer during the patient’s lifetime.
Summary of genetic syndromes linked to increased risk of pancreatic cancer.

BRCA1/BRCA2, breast cancer type 1 and 2 proteins; CDKN2A/p16, cyclin-dependent kinase inhibitor 2A; CFTR, cystic fibrosis transmembrane conductance regulator; LKB1/STK1, liver kinase B1/serine-threonine kinase 11; MSH2, DNA mismatch repair protein Msh2; MLH1, MutL homolog 1, colon cancer, nonpolyposis type 2; MSH6, MutS homolog 6; PMS, PMS2, mismatch repair endonuclease; PRSS1, protease serine 1.
Non-hereditary risk factors
Non-hereditary risk factors encompass modifiable and non-modifiable risk factors that can play a role in cancer development. Non-modifiable risk factors include age with a peak incidence between the seventh and eighth decade of life, male sex, African-American ethnicity, and blood group A and B when compared to the O group.25,26
Over the recent decades, the scientific learning of PDAC has led to the well-established understanding of several non-hereditary modifiable risk factors that have the potential to be corrected with lifestyle modifications and, hence, prevent the development of pancreatic malignancies. These risk factors include smoking, obesity, alcohol abuse, type 1 or new-onset type 2 diabetes, chronic pancreatitis, nickel exposure, and previous history of peptic ulcer disease, specifically gastric ulcers, among others. 13
More recently, a better understanding of the association of both diabetes mellitus and chronic pancreatitis with increased risk of PDAC has been observed. In a meta-analysis conducted by Huxley et al., 27 36 studies showed a modest causal association between type 2 diabetes mellitus and PDAC. In this study, they searched from 1966 to 2005 to study 9220 patients with PDAC. Their analysis showed individuals with recently diagnosed (<4 years) diabetes had a 50% greater risk of the malignancy compared with individuals who had diabetes for more than 5 years (odds ratio 2.1 vs 1.5; p = 0.005), suggesting in some cases, diabetes may be an early manifestation of the tumor and not necessarily always linked to the duration of diabetes which has been suggested by an earlier understanding of the malignancy.
Type 3c diabetes is a form of pancreatic exocrine deficiency caused by numerous factors such as chronic pancreatitis, PDAC, hemochromatosis, cystic fibrosis, and previous PDAC surgery as defined by the American Diabetes Association.28,29 Patients with unclassified diabetes or type 3c diabetes mellitus (pancreatogenic) with or without chronic pancreatitis in several databases demonstrated an increased risk of developing PDAC (with a risk ratio of 4.7–12.1).28,30–32 Chronic pancreatitis remains the most common cause of type 3c diabetes forming about 80% of cases according to a large single-center study of 1868 patients. 33 It is now understood that both diabetes mellitus and chronic pancreatitis are independent risk factors for PDAC, but the combination might be associated with faster progression to malignancy. 32 While most studies point toward a modest increase in the risk of PDAC in patients with type 3c diabetes, in a population-based study from Taiwan, the risk of developing PDAC was significantly elevated with a hazard ratio of 33.5 in participants with both type 3c diabetes and chronic pancreatitis. This study also showed recently diagnosed diabetes (<2 years) to be associated with PDAC and again, an early manifestation of the malignancy. 34
Other modifiable risks with higher incidence include smoking and heavy alcohol which have been linked to a higher risk of acquiring PDAC as compared to the general population. Reinforcing lifestyle modifications of these modifiable risk factors has been a focus of many healthcare initiatives to curb the incidence of PDAC. 35
Clinical features
Pancreatic cancer, in its early stage, lacks clinically overt symptoms and signs and traditionally becomes clinically apparent as the disease progresses and begins to invade local tissues or metastasize to distant sites, making for its complicated clinical course. 36 Clinical features are frequently dictated by the tumor’s size, location, and stage at the time of presentation, which is often late in the illness’s natural course. Weight loss and anorexia are associated with up to 90% of the patients. About two-thirds of PDAC originate from the pancreatic head, leading to symptoms of painless jaundice, pruritis, and palpable non-tender distended gall bladder with the potential to present earlier in the course. PDAC arising from the body and tail of the pancreas can present later with a higher metastatic burden due to the inability to obstruct surrounding structures in earlier stages. As mentioned earlier, recent onset diabetes mellitus has also been implicated as a presenting symptom in PDAC.27,34 It is not uncommon for PDAC to cause complete pancreatic duct obstruction and present as recurrent acute pancreatitis. With metastatic disease, patients may develop symptoms of gastric outlet obstruction and thromboembolism from the direct extension of the tumor into surrounding structures.36,37
Role of screening and surveillance
Population-based consensus for screening of pancreatic cancer is lacking at this time due to the overall low risk of its development in the general population, which is estimated to be about 1.6% in their lifetime. 38 This, coupled with a lack of high-specificity tests, has made the direct screening of PDAC in the general population a challenge. While traditionally cross-sectional imaging such as computed tomography (CT) and magnetic resonance imaging (MRI) are employed for screening purposes, they may not be able to detect smaller pre-malignant lesions making early diagnosis a challenge and recommendation for endoscopic ultrasound (EUS) to be used in combination to these techniques. 39
However, in patients at higher risk of developing PDAC, consensus guidelines recommend surveillance to achieve early detection and potentially better outcomes. These include patients with germline mutations such as STK11 and CDKN2A or have germline mutations in at least one first-degree relative with BRCA1, BRCA2, PALB2, ATM, and MMR group of genes mutation. Family history of PDAC in two or more first-degree relatives or three or more relatives on the same side of the family also qualify for screening and surveillance of PDAC.40–42 Current recommendations for these select individuals recommend an EUS in combination with MRI/magnetic resonance cholangiopancreatography (MRCP) and a baseline HbA1c level, followed by a consideration to alternate EUS and MRI/MRCP at follow-up screening at a 12-month interval.40,41 Figure 1 demonstrates an EUS image of a pancreatic malignancy involving the head of the pancreas. Figure 2(a) and (b) demonstrate representative MRI images of pancreatic malignancy.

An image of EUS shows an irregular heterogeneous mass involving the head of the pancreas (marked yellow).
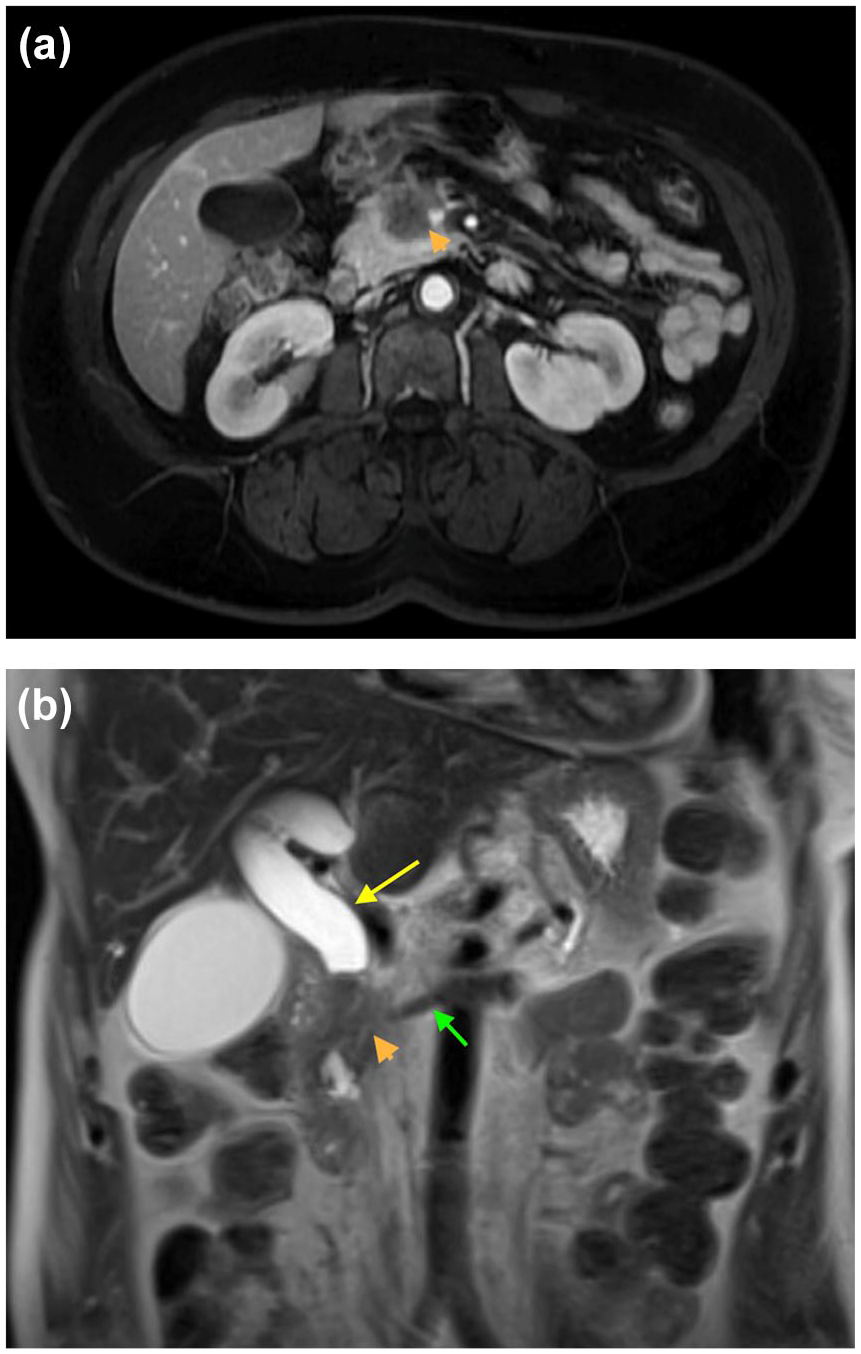
(a) An axial section of an MRI demonstrates a heterogeneous mass at the head of the pancreas (marked orange arrow). (b) Coronal section of an MRI demonstrates a heterogeneous mass at the head of the pancreas (marked orange arrow) with an upstream biliary (yellow arrow) and pancreatic duct (green arrow) dilation.
Performance metrics of endoscopic imaging compared to other listed modalities are discussed in later sections.
The age at first screening in high-risk populations has been debated and differs between societies. The International Cancer of the Pancreas Screening Consortium recommends that surveillance should begin at age 50 or later, except for 10 years earlier than the youngest relative with PDAC 43 while some European societies recommend first screening take place between 40 and 50 years of age, or 10–15 years earlier than the youngest relative with PDAC in the case of familial disease. 44
The role of serological tests as an adjunct diagnostic modality has been well studied. The carbohydrate antigen 19-9 (CA19-9) is currently the only U.S. Food and Drug Administration-approved serum biomarker. Its use has been only suggested as an adjunct to clinical context, imaging, and histopathological diagnosis due to its low sensitivity (80%, 95% confidence interval (CI) = 72%–86%) and low specificity (75%, 95% CI = 68%–80%). 45 Many other biomarkers are described in the literature. However, none of them have the diagnostic accuracy to be utilized for screening and diagnosis of PDAC individually, and further validation studies are needed.
Current guidelines are geared to screen patients with germline mutations and familial PDAC; however, none exist targeting populations with sporadic pancreatic malignancies. Recent studies have highlighted a fewfold increase in acquiring PDAC in populations with diabetes, obesity, smoking, and chronic pancreatitis in an era of increasing prevalence of these risk factors as well as of pancreatic malignancies.46–48 The relatively low incidence in the general population and lack of cost-effectiveness seem to conflict with the severity of pancreatic malignancies at presentation and the impact on survival of emerging early intervention.
Endoscopic diagnostic modalities
While the most widely used modality includes dynamic contrast-enhanced CT scan due to its availability, EUS fine-needle aspiration (FNA) biopsy has been deemed a more sensitive and specific test for the diagnosis PDAC as shown by a study by Eloubeidi et al. 49 where an average of 4 needle passes showed a sensitivity of 95%, specificity 95%, positive predictive value of 100%, and negative predictive value of 85.2% in 101 patients.
EUS helps to elucidate the extent of tumor involvement by direct visualization of the pancreatic malignancy and the neighboring vascular structures, assess and biopsy surrounding lymphadenopathy, and reliably obtain tissue for histopathological confirmation.
One of the challenges with EUS FNA remains, at times, the inability to obtain sufficient tissue to differentiate PDAC from other conditions such as chronic pancreatitis, autoimmune pancreatitis, lymphoma, and tuberculosis. EUS fine-needle biopsy (FNB) has outperformed EUS FNA in this regard by providing core tissue with preserved architecture of glandular and stromal tissue due to the advent of newer biopsy needles allowing for additional immunohistochemistry, DNA sequencing, or RNA-based marker studies. 50 Recent multiple multicentered trials highlighted EUS FNB’s superiority in obtaining core samples and the need for fewer needle passes due to its wide bore design, shorter procedure time, and excellent histological yield compared to EUS FNA.51–53 Figure 3(a) and (b) shows stepwise access to a pancreatic mass with an EUS FNB needle.
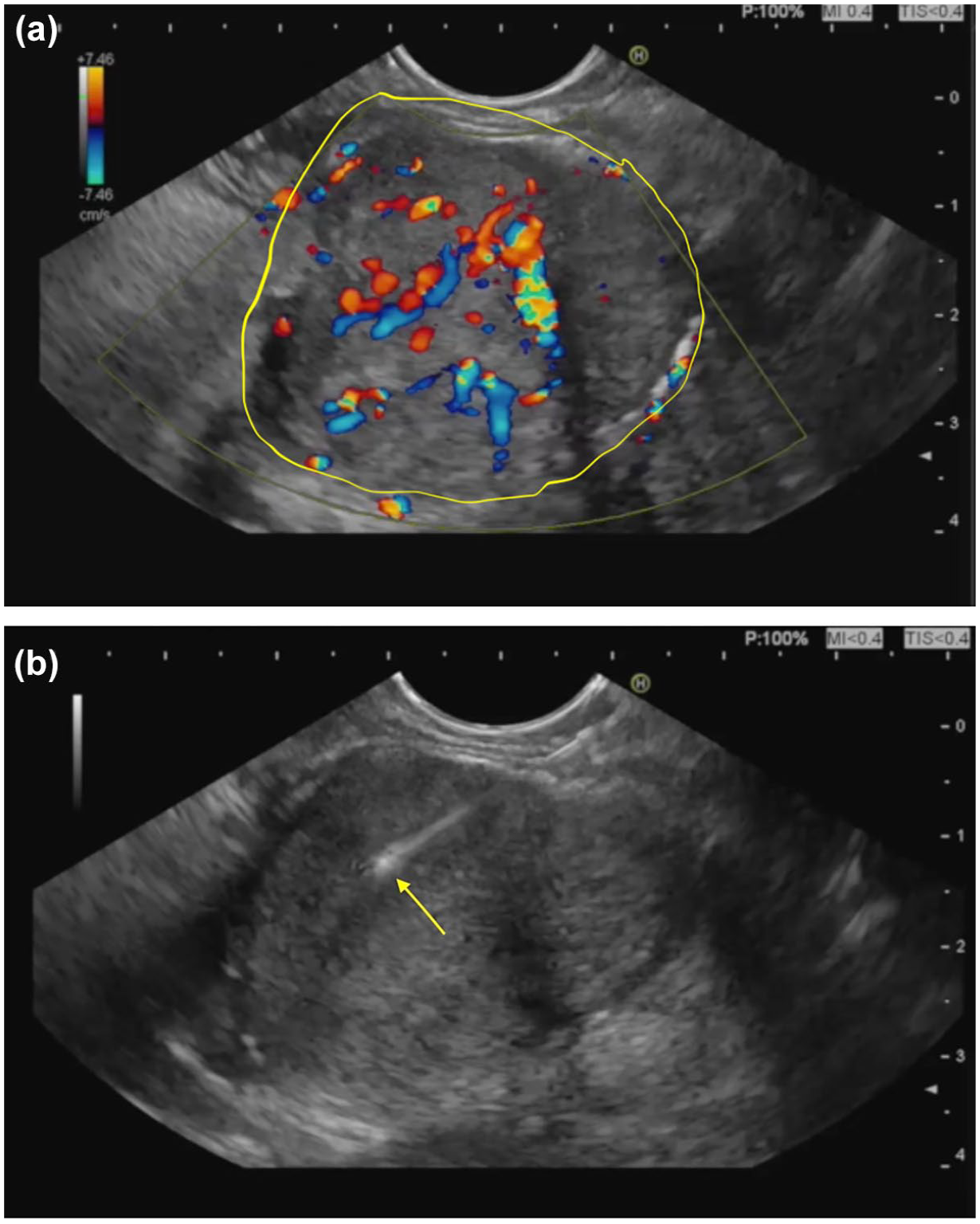
(a) EUS image shows a pancreatic mass under Doppler examination with aberrant vasculature. A standard practice is looking for an avascular plane before introducing an EUS FNB needle. (b) EUS FNB needle (yellow arrow) being introduced into the same pancreatic mass (from (a)).
Artificial intelligence (AI) has been increasingly studied in the field of gastroenterology in varied fields, such as survival models and prognostication models for inflammatory bowel disease, colon cancer, and pancreatic malignancies.54,55 Recently, there have been studies acknowledging the recognition of AI-assisted EUS in the early detection of PDAC. In addition to achieving high accuracy, sensitivity, and specificity levels, AI-assisted EUS also helps better differentiate radiological mimickers, such as chronic and autoimmune pancreatitis from pancreatic malignancy.55,56
Endoscopy-guided therapeutic interventions
The role of endoscopic interventions transcends its diagnostic capabilities but extends well into therapeutic realms owing to its minimally invasive nature and ability to provide a comprehensive and personalized approach to PDAC care.
Role of endoscopy in relief of obstruction
Surgical resection is the only curative treatment for PDAC; however, endoscopic advances have been instrumental in relieving acute and sub-acute biliary obstruction, downstaging tumors, and providing palliative therapy for obstruction and pain relief.
Endoscopic retrograde cholangiopancreatography (ERCP) and EUS-guided ERCP are frequently employed when patients present with an obstruction of the biliary drainage due to the pancreatic malignancy to alleviate symptoms and is the preferred method of decompression when compared to operative bypass. 57 Plastic stents across the biliary system are preferred when the patient has obstruction with symptoms such as pruritis or cholangitis, the diagnosis is uncertain, or when an anticipated delay in surgical intervention is expected. On the contrary, metallic stents are preferred in patients with established diagnoses who are not obvious surgical candidates. However, the placement of a metallic stent does not preclude surgical resection.
ERCP with the placement of a self-expandable metal stent is the recommended method for palliation in patients with malignant extrahepatic biliary obstruction. 58 When the pancreatic malignancy does not allow access to native papilla in the ampullary region, EUS-guided ERCP can be employed with great technical success ranging between 75% and 100%. Different routes such as (transgastric or transduodenal) for EUS-guided biliary drainage can be utilized to achieve internal biliary drainage with different techniques such as rendezvous, hepaticogastrostomy, choledochoduodenostomy.59–61 Introduction of lumen-apposing metal stent (LAMS) has helped reduce the complexity and adverse events associated with these procedures. 62 A multicenter randomized control trial by Teoh et al. showed that among 155 patients randomized to receive either EUS-guided choledochoduodenostomy or ERCP drainage for unresectable malignant biliary obstruction, the clinical success and 1-year stent patency rates were similar in both groups. However, EUS-guided choledochoduodenostomy was associated with greater technical success and reduced procedure time. 63 Figure 4(a)–(d) depicts a stepwise approach to EUS-guided ERCP highlighting a choledochoduodenostomy for relief of obstruction from PDAC.

(a) EUS image of a dilated CBD (yellow arrow) as viewed from the duodenal bulb in a patient with an inaccessible native ampulla (not visualized here). (b) EUS image of an access needle (yellow arrow) passed through the duodenal bulb into the CBD. (c) EUS image of a deployed proximal phalange of a lumen apposing metallic stent (AXIOS) (yellow arrow) inside the CBD. (d) EUS-directed choledochoduodenostomy. A fluoroscopy image shows a lumen-apposing metallic AXIOS stent (yellow arrow) connecting the duodenal bulb to the CBD (orange arrow).
LAMS can also be used to relieve gastric outlet obstruction associated with pancreatic malignancies. When used for gastroenterostomy, LAMS has been shown to have a technical success rate of 87%–100% and a clinical success rate of 84%–100%.64,65
EUS-directed therapy
The need for newer neoadjuvant therapeutic options has become dire as over 80% of patients with PDAC at presentation are unresectable, that is, either metastatic or locally advanced pancreatic cancer (LAPC). 66 LAPC is defined as >180 degrees of involvement of the superior mesenteric artery or common hepatic artery, involvement of the aorta, un-reconstructable portal, and mesenteric vein due to tumor infiltration as defined by the National Comprehensive Cancer Network. 67 The 15%–20% of patients that have resectable cancer status at the time of diagnosis are treated with neoadjuvant chemotherapy and surgery. In a study by Hammel et al., the overall survival rate of LAPC has been reported between 11 and 13 months after diagnosis with chemoradiotherapy or chemotherapy alone. 68 Unfortunately, a sizable remainder of the patients are not surgical candidates.
EUS-guided fine-needle injection (FNI) is an emerging modality that enables endoscopists to directly administer therapies to locally advanced tumors and to reduce systemic side effects. This can be further divided into physicochemical, molecular biological, and immunological therapies based on therapeutic mechanisms. 69 Figure 5 shows a flowchart classification of available EUS FNI therapies, and Table 2 summarizes their mechanism of action.

A flowchart depicts various available modalities delivered via EUS-guided FNI for treating pancreatic cancer.
Summary of EUS-guided injection therapies.

EUS, endoscopic ultrasound.
Physicochemical therapies
Physicochemical therapies include ablation, brachytherapy, and intratumor chemotherapy.
Radiofrequency ablation
EUS radiofrequency ablation (RFA) is an emerging modality that has shown promise in the therapy and palliation of PDAC. This technique involves the delivery of thermal energy using probes at the tip of the endoscope, typically between 60°C and 100°C, to achieve localized coagulation necrosis. This, in turn, allows the tumor to become more porous and allows systemic therapy to penetrate the tissue. This technique causes local disruption of the tumor milieu using heat, and, in turn, the coagulative necrosis releases a varied spectrum of tumor antigens, potentially triggering a systemic antitumor response at local (RFA-applied sites) and distant tumor sites, referred to as abscopal effect.70,71 Figure 6 shows the EUS-directed RFA of an LAPC.

EUS image shows EUS-directed RFA delivery into a locally advanced pancreatic cancer.
Commercially available EUS-RFA probes come in two different types, monopolar and bipolar probes. For the purpose of EUS-directed therapy, monopolar probes are utilized. Their mechanism involves a generator that releases high-intensity current through a delivery electrode and a dispersive electrode that acts as an earthing pad.72,73
The efficacy and safety of EUS RFA have been reported to be as high as 100% with minimal procedure-related adverse events in multiple prospective and retrospective studies.74,75 EUS RFA offers the ability to often precisely visualize and localize target lesions to deliver precise treatment and serves as a minimally invasive technique with minimal procedure-related adverse events which is increasingly being adopted as an active adjunct for the treatment of locally advanced pancreatic malignancy.76,77
Microwave ablation
Microwave ablation (MWA) technology represents an alternative modality to EUS RFA, which utilizes electromagnetic microwaves to deliver thermal energy (900–2500 MHz) by agitating the water molecules in the target tissue, producing friction and heat, causing cellular death and coagulative necrosis.78,79 This technology has certain theoretical benefits over other thermoablative technologies including higher intratumor temperatures, larger ablation volumes, and faster ablation times. 79 MWA has shown promise in delivering therapy in patients with PDAC using open surgical, laparoscopic, or percutaneous approach in two studies, however with a small sample size.80,81 While still in its infancy, this approach needs validation through larger studies.
Cryotherm ablation
The role of this technology has been established in the local treatment of numerous malignancies involving the breast, skin, prostate, and kidneys. 73 As the name suggests, it combines the freezing properties of cryogenic gas along with the thermal radiofrequency effect to achieve increased devitalization of targeted tissue. When compared to bipolar RFA, it offers higher efficacy owing to the combined thermal effect. 82 Like EUS RFA, EUS-CTA (cryo-therm ablation) also demonstrates an abscopal effect, as explained in the RFA section, to achieve antitumoral immunity at local and distant tumor sites.83,84 Despite its promising mechanism, it has not been demonstrated in limited trials. In a study by Arcidiancono et al. where they treated 16 (of the 22 enrolled) patients using EUS-CTA and reported minor adverse events in 43.7% of patients, including amylase elevations (3), abdominal discomfort (3), and duodenal bleeding (1) managed endoscopically. Unfortunately, they reported complications related to tumor progression in four patients (25%) with a median post-treatment survival of 6 months. 85 Similarly, a study by Petrone et al. treated 26 (74.3%) of 35 enrolled patients with EUS-CTA. They reported the technique to ablate 34.9% of neoplastic tissue to find a favorable correlation between ablation duration and necrosis volume (R = 0.66, p = 0.013) with the median post-ablation survival time of about 5–9 months when treated with more than one ablation cycle. 86 Based on current data, EUS-CTA has the potential to be a feasible and safe alternative therapy; however, it needs technical improvement in the delivery mechanism. Robust randomized, controlled trials demonstrating an overall survival benefit in patients with LAPC would also be crucial in solidifying its role.
Photodynamic therapy
Photodynamic therapy (PDT) converts a non-cytotoxic agent to a cytotoxic substrate using an appropriate light wavelength for excitation and inducing selective cellular apoptosis and necrosis in the targeted tissue. This mechanism involves the use of a tumor-localizing photosensitizing agent, followed by activation of the agent by a preset wavelength of light and sequential photochemical and photobiological steps that cause irreversible photodamage to the tumor cells. 87 In the treatment of PDAC, after activation with the photosensitizer is achieved, EUS-guided illumination using a small diameter optical fiber is performed to deliver a 630 nm laser light dose to achieve localized tissue necrosis without causing significant surrounding inflammation due to lack of thermal ablation. 73 Choi et al. conducted the first EUS-PDT for patients with LAPC in 2015 in a single-center prospective trial. They enrolled four patients in the study; one had LAPC and the rest were other solid organ tumors. They reported a technical success rate of 100% with no intraprocedural complications and a median follow-up of 5 months. 88 In 2018, DeWitt et al. 89 performed another single-center prospective trial with 12 LAPC patients treated with EUS-PDT to find an increase in tumor necrosis in 6 (50%) patients compared to baseline. In a recent study by Hanada et al. 90 in 2021, they enrolled eight patients with LAPC who were treated with EUS-PDT using verteporfin (a newer PDT agent) to report a zone of necrosis in the malignancy in five (62.5%) of the patients. All the reported studies had a technical success rate of 100% with no intraprocedural complications suggesting the safety profile of this modality.88–90 It has appeared to be a viable option for patients with advanced pancreatic malignancies who are poor surgical candidates. However, more robust studies need to back this claim.
Brachytherapy
Brachytherapy includes the placement of radioactive seeds in the tumor using CT or ultrasound guidance to deliver radiation to the tumor for local control of the tumor. EUS FNI is increasingly being utilized to place the seeds accurately. Radioactive seeds commonly used during brachytherapy include iodine-125, iridium-192, and palladium-103. 91 Sun et al. utilized EUS-guided brachytherapy in 15 patients and placed 22 radioactive seeds per patient. 27% of patients had a partial response, and 20% had a minimal response, with stable disease in 33% of patients. 92 An open-label EUS-guided brachytherapy using phosphorus-32 microparticles with chemotherapy (gemcitabine ± nab-paclitaxel) in patients with locally advanced PDAC is currently underway. 93
Intratumor chemotherapy
Intratumor chemotherapy has shown some promise in animal models. Intralesional injection of OncoGel (a formulation of paclitaxel) in eight Yorkshire pigs showed sustained concentration of the drug in the pancreas on day 14. 94 In canine models, pancreatic injection of 5-fluorouracil in the pancreas showed necrosis of the pancreatic tissue. 95 These studies have formed the basis for trialing intratumor chemotherapy in patients with advanced unresectable malignancies. Levy et al., in a single-center prospective study with 36 patients with stage II (3), stage III (20), and stage IV (13) disease, utilized gemcitabine (38 mh/mL) EUS-FNI without any reported adverse events. They reported an overall survival of 78% and 44% at 6- and 12-month follow-ups, respectively, with a mean overall survival of 10.4 months. Four patients with stage 3 disease (20%) got downstaged and underwent surgical resection. 96 Most recently, Yang et al. showed the utility of a novel drug delivery system that consisted of gemcitabine in combination with a thermos-sensitive hydrogel (GEM/PPP) in mice, and effects were compared with gemcitabine, ethanol, and polidocanol. GEM/PPP gel was injected using EUS-FNI, and tumor weight decreased by 75.96%, increasing survival by 14.4 days. No significant adverse effects or systemic toxicity were noted. 97 Further research is needed to replicate the feasibility of this therapy in the clinical setting.
Molecular biological therapies
Various molecular biological therapies have been studied for pancreatic malignancies. Matsuda et al. 98 showed that patients with increased expression of carbohydrate sulfonyltransferase 15 (CHST15), responsible for matrix remodeling in PDAC, had worse overall survival than patients with a lower expression. STNM01 is an RNA oligonucleotide that inhibits CHST15 gene expression. In phase I/IIa trial of patients with unresectable PDAC refractory to gemcitabine plus nab-paclitaxel therapy, EUS FNI using STNM01 was studied in 22 patients in 5 centers in Japan, published in January 2023. The overall survival was 7.8 months with 8 grade 3 adverse events reported. 99
The KRAS mutation pathway is another established method for tumor genesis. An open-label phase I/IIa study targeted inoperable LAPC using EUS-guided injection of si-G12D-LODER, a miniature biodegradable implant, releasing siRNA against KRAS. 100 The performance of this molecule was studied in combination with chemotherapy, which showed a decrease in tumor marker CA19-9 in 70% (7/10) of patients. However, 80% (10/12) had either stable or partial response with a median survival of 15 months. In addition to this, one-third of the patients experienced severe adverse events (5/15). While the response was modest, further studies looking at safer delivery with increased penetration might be warranted.
Oncolytic viruses, including adenovirus, reovirus, parvovirus, measles virus, and herpes virus, have been studied in clinical and preclinical settings for their ability to lyse tumor cells. 101
Adenoviruses including TNFerade (GenVec Inc., Gaithersburg, Maryland, USA), which is an adenovirus vector carrying the human tumor necrosis factor-alpha gene, and ONYX-015 (Onyx Pharmaceuticals, San Francisco, USA), which is an oncolytic adenovirus that is tumor-selective and induces cell death, can be delivered using EUS-FNI. 102 In the Stage III clinical trial of TNFerade, which compared the standard of care (including fluorouracil and radiotherapy) with the standard of care and TNFerade therapy, there was no difference in median survival in the two groups. 103 In the phase I/II trial of intratumor injection of ONYX-015 along with IV Gemcitabine in patients with unresectable PDAC, 11 out of 21 patients had to stop the study because of disease progression or toxicity, limiting its utility. 104 Other oncolytic viruses that have been studied in clinical settings include HF10 which is derived from herpes simplex virus-1 and adenovirus-mediated double-suicide gene therapy with modest benefits.105,106 However, more studies are warranted to establish the safety and efficacy of this modality.
Immunological therapies
Immune cells play a key role in cancer control. Hence, immunotherapy has been studied for PDAC. Dendritic cells (DCs) are antigen-presenting cells that induce a T-cell response. They can recognize and process tumor antigens and hence elicit an immune response to these antigens. 106 Irisawa et al. studied the response to intratumoral injection of immature DCs using EUS FNI in patients with unresectable stage IV PDAC who failed to respond to gemcitabine. The median survival was 9.9 months and the procedure was well tolerated. 107 In the phase I/II trial, Hirooka et al. demonstrated that comprehensive immunotherapy, which included zoledronate-pulsed DCs, gemcitabine (GEM), and αβT cells in patients with locally advanced pancreatic carcinoma, 15 patients showed an overall survival of 12 months. 108 Cytoimplant (allogeneic mixed lymphocyte culture) delivered into the tumor using EUS FNI was studied by Chang et al. 109 with two patients showing partial response and one patient showing minor response with a median survival of 13.2 months. DNA plasmids have also been studied in unresectable locally advanced PDAC. Hanna et al. used BC-819, a DNA plasmid that was injected in the tumor in nine patients, using CT guidance in three and EUS in six patients. The tumor was downstaged in two patients after receiving chemotherapy or chemoradiation, and partial response was noted in three patients. 110 Further studies are needed to assess the combination of immunotherapy with chemotherapy and chemoradiation in advanced PDACs.
Pain management using EUS-guided neurolysis
Pancreatic malignancies can be a source of intractable pain, specifically after the invasion of surrounding neurovascular structures. EUS-guided celiac plexus neurolysis (CPN) is a useful adjunct to opiates for the management of intractable pain. A meta-analysis of 980 patients demonstrated pain reduction in 70% of the patients with PDAC undergoing celiac plexus ganglion neurolysis under EUS guidance with good tolerability showing its promise as a dependable pain alleviation strategy for this patient population. 111
Typically, pain control is achieved by injection of 98% absolute alcohol for neurolysis and 0.25% bupivacaine for analgesia into the celiac plexus under direct visualization through EUS. Injection techniques for CPN include direct injection into the ganglia or CPN, bilateral injections at the root of the celiac artery, and central injection anterior to the root of the celiac artery.112,113 CPN can further be combined with an injection in the free retroperitoneal space (combined CPN). A systematic review showed no difference in response rates between the three techniques. 113
Though usually well tolerated with <10% of patients experiencing side effects such as diarrhea, hypotension, and temporary pain exacerbation, serious complications such as GI bleed, spinal stroke, perforation of the stomach, and aorta have been reported.113,114 Figure 7(a) and (b) shows the EUS-guided neurolysis of a celiac ganglion.

(a) EUS image shows the celiac ganglion (yellow arrow). (b) EUS-directed celiac ganglion neurolysis (yellow arrow).
Future directions
Due to its delayed presentation, aggressive nature, poor mortality, and rising incidence of pancreatic malignancies, future research should prioritize the development of effective screening strategies, especially targeting high-risk patients. Investigations into underlying genetic and acquitted risk factors in patients with “sporadic” forms of PDACs, which constitute the majority of cases, require further exploration.
Studies exploring the role of novel serum biomarkers with high sensitivity and specificity for PDAC are needed. In addition, screening programs’ clinical utility, feasibility, cost-effectiveness, and survival benefits should be thoroughly assessed to aid in the early recognition of pancreatic malignancies. The role of risk factors, such as diabetes mellitus, alcohol use, and smoking in PDAC, as well as their pathophysiology, needs to be elucidated in more detail with a focus on whether controlling these factors impacts long-term incidence.
The role of EUS FNB has become pivotal in obtaining a tissue diagnosis for PDAC. Exploring the integration of AI with EUS to achieve timely and accurate diagnosis is an avenue for future studies. Since most pancreatic tumors are unresectable at the time of diagnosis, studies are needed to develop effective neoadjuvant chemotherapy and immunotherapy regimens which can be used along with endoscopic-guided therapeutic interventions to help increase survival rates. Studies looking at the combined role of neo-adjuvant chemotherapy and EUS-directed therapies would be clinically useful. EUS RFA is showing promise in locally advanced PDAC with promising results and minimal side effects. Large-scale, multi-center clinical trials are needed to study to establish the role of this and other EUS-guided therapies at a population level.
Concerted efforts to educate patients, primary care physicians, and gastroenterologists would be pivotal to identify and screen patients at high risk of developing PDAC. Ultimately, as with other kinds of malignancies, the goal should be to provide individualized and directed therapy to patients by integrating information about their genetics, epigenetics, and tumor signature, which would require a multidisciplinary approach to help patients with pancreatic cancer.
Conclusion
Pancreatic malignancies are often unresectable and advanced at the time of presentation, with a very poor 5-year survival rate. Screening the general population remains unviable. However, directed screening is offered to those with known risk factors. EUS and related modalities are increasingly used for screening, surveillance, diagnosis, and managing pancreatic malignancies. EUS combined with FNB is a newer emerging tool with higher accuracy and a safer profile for the diagnosis of PDAC. EUS-guided ERCP is used to relieve obstruction of biliary drainage, playing an important role in symptom management and palliation in advanced cancers. EUS RFA is an emerging modality used for therapy and palliation of PDAC with desirable outcomes and safe tolerability. It could serve as an area of promising future direction in the management of pancreatic malignancy.
