Abstract
Endoscopic ultrasound-directed transgastric endoscopic retrograde cholangiopancreatography (EDGE) and endoscopic ultrasound-directed transenteric endoscopic retrograde cholangiopancreatography (EDEE) are innovative endoscopic techniques developed to overcome the challenges of biliary access in patients with surgically altered gastrointestinal anatomy. EDGE facilitates the creation of a gastro-gastric anastomosis, enabling endoscopic access to the excluded stomach and subsequent duodenum for endoscopic retrograde cholangiopancreatography (ERCP) procedures. Similarly, EDEE involves creating a gastro-jejunal anastomosis, allowing endoscopic access to the jejunum and hepaticojejunostomy for ERCP. These procedures are primarily indicated for patients with Roux-en-Y gastric bypass or other complex gastrointestinal surgeries that render traditional ERCP unfeasible. The major advantages of EDGE and EDEE include minimally invasive access to the biliary system, reduced procedural morbidity, and the ability to perform complex biliary interventions without additional surgeries. Using lumen-apposing metal stents in these procedures has further improved their safety and efficacy. This comprehensive review delves into EDGE and EDEE’s technical nuances, clinical outcomes, and safety profiles. Our extensive literature searches reveal high procedural success rates and low complication incidences, establishing these methods as viable alternatives to traditional surgical and percutaneous approaches. We also discuss recent technological advancements, including developing enhanced stents and endoscopic ultrasound-guided instruments, which have refined these techniques and expanded their applications. Moreover, the review examines the integration of EDGE and EDEE with other therapeutic modalities, such as cholangioscopy and intraductal lithotripsy, to optimize treatment outcomes. Future directions emphasize the need for larger, multicenter trials to validate these findings further and create standardized protocols to ensure consistent procedural efficacy and safety. This review highlights the transformative potential of EDGE and EDEE in therapeutic endoscopy, advocating for their broader adoption in clinical practice and ongoing innovation in this rapidly evolving field.
Plain language summary
Some patients who need treatment for bile duct or pancreatic problems have had surgeries that change the shape and connections in their digestive system. These changes make it difficult for doctors to reach the bile ducts using standard endoscopic procedures. Two new methods—called EDGE (Endoscopic Ultrasound Directed Transgastric ERCP) and EDEE (Endoscopic Ultrasound Directed Transenteric ERCP)—offer a solution. They allow doctors to create a new passage between parts of the digestive system using a special stent. This passage gives doctors access to the bile ducts with standard endoscopes, avoiding the need for open surgery. These techniques are especially useful for patients who have had a Roux-en-Y gastric bypass or other complex surgeries, where the standard route to the bile ducts is blocked. EDGE and EDEE are minimally invasive, meaning they involve less risk and shorter recovery times compared to surgery. This review article explains how these techniques work, their advantages, and how safe and effective they are. Studies show they have a high success rate and fewer complications than traditional surgical methods. They can also be used for repeated treatments if needed. The review also looks at new improvements in technology, such as better stents and instruments, and how these techniques can be combined with other treatments to improve results. Finally, the authors highlight the need for larger studies to confirm these promising results and support the use of EDGE and EDEE in routine medical practice.
Keywords
Introduction
The management of pancreaticobiliary diseases in patients with surgically altered anatomy (SAA) remains a challenging scenario for endoscopists. Traditional approaches, such as device-assisted enteroscopy (DAE)-guided endoscopic retrograde cholangiopancreatography (ERCP) and percutaneous transhepatic biliary drainage (PTBD), have been widely used but are often limited by suboptimal success rates, procedural complexity, and an increased risk of adverse events (AEs).1–3
Performing ERCP in patients with SAA is inherently difficult due to the loss of direct access to the ampulla of Vater or bilioenteric anastomosis. While conventional ERCP can be effectively performed in cases where the anatomical continuity between the stomach and duodenum is preserved, such as Billroth I (BI), sleeve gastrectomy (SG), or laparoscopic adjustable gastric band placement, other more complex surgical reconstructions pose significant technical hurdles. 4 Procedures like the Whipple procedure, Roux-en-Y gastric bypass (RYGB), Roux-en-Y hepaticojejunostomy (RYHJ), and Billroth II (BII) involve resection and/or reanastomosis, creating elongated afferent limbs, sharp angulations, and adhesions that complicate endoscopic navigation. 4 These anatomical modifications often make it extremely challenging, if not impossible, to reach the papilla or the bilioenteric anastomosis with a standard duodenoscope. Even when biliary access is achieved using DAE (single/double balloon), successful cannulation, stone extraction, or anastomotic evaluation can remain challenging due to limited maneuverability, the absence of an elevator, altered angulation, and restricted instrument stability.
DAE-guided ERCP, while minimally invasive, is limited by suboptimal diagnostic and therapeutic success rates of 69.4% and 61%, respectively, primarily due to the difficulty in accessing the papilla or bilioenteric anastomosis. 3 Additionally, AEs can occur in up to 6.5% of cases, with serious complications such as pancreatitis (2.2%), bleeding (0.4%), and perforation (0.8%). 3 PTBD, although more effective, carries a significantly higher AE rate of up to 60%, including catheter occlusion, dislodgement, and infection, often leading to frequent hospital readmissions.1,2
Surgical options, on the other hand, generally achieve high technical success rates but are associated with considerable morbidity, especially in patients with a history of multiple abdominal surgeries. Moreover, in cases of tumor recurrence, patients may not be suitable candidates for further surgical interventions due to poor overall prognosis and increased perioperative risks. 5
The advent of endoscopic ultrasound (EUS)-guided techniques has revolutionized the field by providing minimally invasive alternatives that overcome anatomical barriers.6,7
Among these innovations, EUS-directed transgastric ERCP (EDGE) and EUS-directed transenteric ERCP (EDEE) have emerged as promising techniques that facilitate endoscopic access to the biliary and pancreatic ducts in patients with complex postsurgical anatomy.8,9 These procedures employ lumen-apposing metal stents (LAMS) to create an anastomotic conduit, enabling standard duodenoscopes to access the papilla or bilioenteric anastomosis and reestablish a conventional retrograde approach for ERCP. While EDGE has been widely adopted for patients with RYGB, EDEE extends this concept to other types of SAA, including patients with hepaticojejunostomy (HJ) following Roux-en-Y reconstruction or those who have undergone pancreatic resection via the Whipple procedure, thereby expanding its clinical applicability. In this comprehensive review, we examined the evolution of EDGE and EDEE, with a focus on their technical approaches, clinical outcomes, and potential complications. As these techniques gain broader adoption, they are reshaping the management of pancreaticobiliary diseases in patients with SAA, offering effective and minimally invasive endoscopic alternatives in scenarios where traditional methods often fall short.
Materials and methods
In our review, we searched PubMed, PubMed Central, and Medline, including only English-language articles published up to the end of February 2025. We implemented a comprehensive search strategy using strings containing the following terms: “ERCP,” “surgically altered anatomy,” “EUS,” “endoscopic ultrasound,” “endoscopy,” “EUS-directed transgastric ERCP,” “EUS-directed transenteric ERCP,” “Roux-en-Y gastric bypass,” “Roux-en-Y hepaticojejunostomy,” “Billroth II,” and “Whipple procedure.” Additionally, we manually reviewed the references of the included studies and relevant reviews to identify any additional suitable publications.
EUS-directed transgastric ERCP
RYGB has become the predominant bariatric procedure, accounting for approximately 70%–80% of all bariatric surgeries. 10 RYGB, first described in 1994, involves the creation of a biliopancreatic limb measuring 75–150 cm and an alimentary (Roux) limb of 100–150 cm. The procedure involves creating a jejunojejunostomy and forming a 30-mL gastric pouch from the proximal stomach, followed by a gastrojejunal anastomosis that connects the Roux limb to the gastric pouch. 11 Approximately 29%–36% of post-bariatric patients develop gallstones, while 13% experience gallbladder sludge within 6–18 months after surgery.12,13 Due to the anatomical changes resulting from this surgery, approximately 150–300 cm of small intestine separates the gastric pouch from the ampulla, significantly hindering successful biliary cannulation using standard endoscopic techniques. 14 To overcome this challenge, EDGE was developed as a minimally invasive alternative for accessing the pancreaticobiliary region in patients with RYGB anatomy. 15 Kedia et al. 16 described the first version of the EDGE procedure in 2013 as a two-step approach. Initially, a 16-French gastrostomy tube was percutaneously inserted into the excluded stomach under EUS guidance to facilitate its identification and distension through the gastric pouch. In the second step, the gastrostomy tube was replaced with a fully covered self-expanding metallic stent (FC-SEMS), allowing antegrade ERCP using a side-viewing duodenoscope. 16 This method resembled the previously established EUS-guided percutaneous assisted transprosthetic endoscopic therapy (PATENT) technique. 17 With the introduction of the LAMS, Kedia et al. 16 in 2015 described a single-stage EDGE (ss-EDGE). This procedure establishes endoscopic access to the excluded stomach by creating a stable de novo gastro-gastric (GG) or jejuno-gastric (JG) anastomosis under EUS and fluoroscopic guidance using the LAMS 18 (Figure 1). The LAMS then serves as a conduit for the therapeutic endoscope, enabling access to the excluded stomach through the gastric pouch and facilitating ERCP or EUS when needed. The technique requires EUS visualization of the excluded stomach, followed by puncture with a 19-gauge fine-needle biopsy (FNB) needle to opacify and distend the target lumen. An LAMS is then deployed transmurally under EUS guidance to create the anastomosis. 19
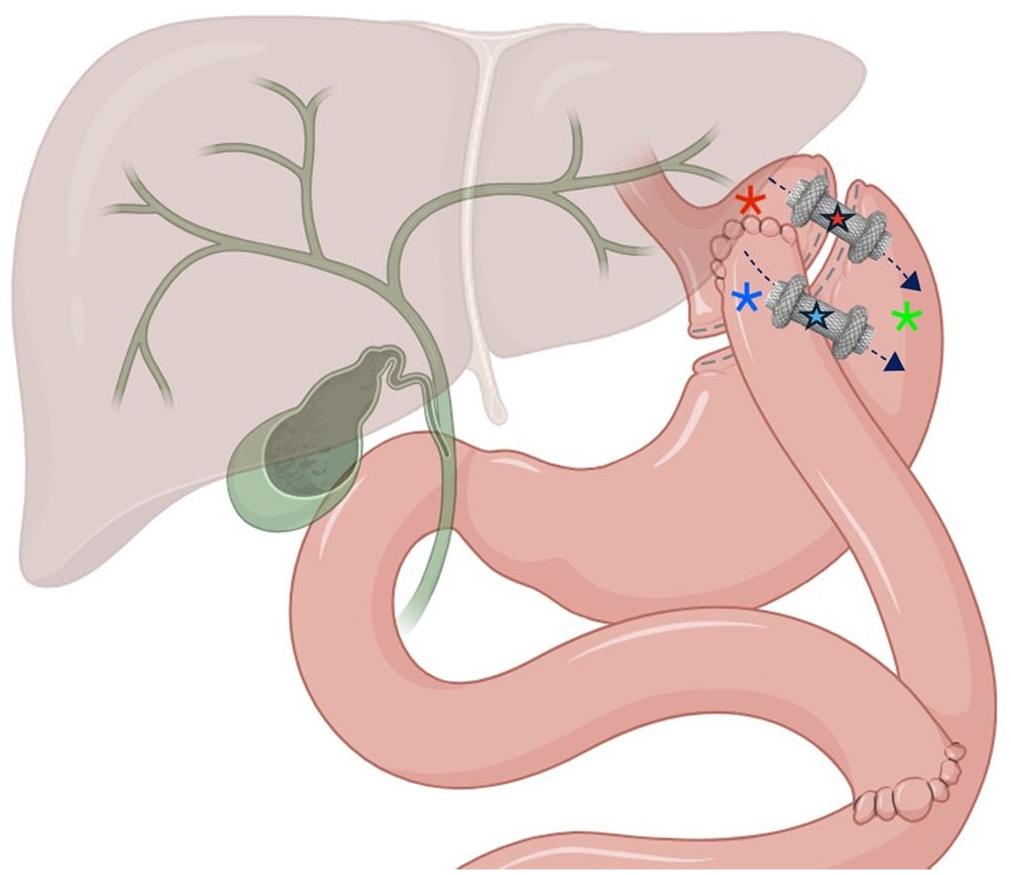
EDGE with gastro-gastric (red star) or jejuno-gastric (blue star) anastomosis. Gastric pouch (red asterisk); jejunal loop (blue asterisk); and gastric remnant (green asterisk).
The technique
For optimal planning of the EDGE procedure, preparatory steps are crucial to optimizing the procedural approach and minimizing the risk of complications. These steps involve obtaining detailed information about the patient’s prior surgery, which is essential, in addition to a thorough history, physical examination, and cross-sectional imaging to anticipate potential difficulties. 20 RYGB may have been performed via an open or laparoscopic approach, with possible revisions or complications, such as fistulas or perforations, that can impact the complexity of the endoscopic procedure.20,21 Additionally, a comprehensive anatomical evaluation with a frontal view endoscope should precede EUS to assess key anatomical factors, including the gastric pouch size, the location and patency of the gastrojejunal anastomosis, and the feasibility of safely deploying an LAMS within the pouch or, if necessary, from the jejunum. 20
EDGE procedure begins by advancing a linear echoendoscope into the gastric pouch. 22 The excluded stomach is identified under EUS guidance, either from the gastric pouch or the afferent jejunal limb, and subsequently accessed using a 19-gauge FNB needle (Figure 2).

Technical steps of EDGE. (a) Under EUS guidance, identification of the gastric remnant. (b) Puncture of the gastric remnant by a 19-gauge FNB needle for gastric lumen distension (red asterisk) with saline solution. (c–e) EUS, fluoroscopic, and endoscopic view of release of 20 × 10 mm electrocautery-enhanced LAMS. (f) Easy passage of the duodenoscope through the LAMS lumen (blue asterisk) to perform ERCP.
The choice of access point depends on several factors, including the presence of intervening vessels, the proximity of the access site to the excluded stomach (optimal distance less than 1–2 cm), the aim of positioning the distal flange of the LAMS within the gastric body or antrum, and the patient’s specific anatomical configuration. 14 However, a gastrostomy tract—connecting the gastric pouch to the remnant stomach body—is the preferred access point for EDGE, as it is associated with a lower risk of stent dislodgement and faster tract maturation compared to a jejuno-gastrostomy tract.14,23 When the FNB needle is secured inside the gastric lumen, usually a solution made with a contrast agent mixed with saline is injected to distend the stomach or afferent limb and visualize it under fluoroscopy. To facilitate this passage, the 19-gauge needle may be connected to a standard waterjet system to assist in distension. At this point, as with EUS-gastroenterostomy (EUS-GE), there are two technical options for LAMS deployment: the free-hand technique and the over-the-wire technique. 24 In the free-hand or Wireless Endoscopic Simplified Technique (WEST) approach, the electrocautery-enhanced LAMS (ec-LAMS; usually 20 or 15 mm in diameter) catheter is advanced directly into the distended gastric lumen without guidewire assistance.25,26 In the over-the-wire technique, a guidewire is first passed through the FNB needle into the gastric lumen, and the ec-LAMS catheter is then advanced over the guidewire for stent placement.24,27 The distal flange of the LAMS is deployed first into the excluded stomach under EUS and fluoroscopic guidance, followed by the deployment of the proximal flange into the gastric pouch or afferent jejunal loop under direct endoscopic and fluoroscopic control.
ERCP is typically performed in a subsequent session following EDGE to allow for tract maturation, as advancing the ERCP scope through the LAMS lumen too early may increase the risk of stent dislodgement and subsequent perforation. 28 However, a ss-EDGE is performed when ERCP is urgently needed, such as in the management of severe cholangitis. If the same session strategy is chosen, a pneumatic dilation of the LAMS lumen is performed to reduce friction or angulation during endoscope advancement and consequently the LAMS migration risk, resulting in a perforation of the GG or JG tract. To address the LAMS dislodgment risk, several endoscopic techniques have been developed to minimize the likelihood of stent displacement. 29 Through-the-scope clips (TTSCs) have previously been employed with moderate efficacy, demonstrating a reduction in stent migration rates from 34% to 13%. 30 The OverStitch endoscopic suturing system, introduced for full-thickness soft tissue approximation, has also been utilized to secure stents, resulting in improved migration outcomes.31,32 However, the widespread adoption of this technique has been hindered by limited data on optimal suturing methods, high procedural costs, and the requirement for specialized training. More recently, Ovesco® has introduced the Stentfix over-the-scope clip (OTSC) System, a dedicated OTSC specifically designed for stent fixation. The system features a smaller, more streamlined deployment mechanism that facilitates easier passage through the oropharynx and upper esophageal sphincter. Additionally, the redesigned cap is aligned parallel to the stent lumen, improving the clip’s ability to securely grasp both the stent mesh and adjacent tissue. The use of OTSCs has been associated with reduced procedure time and overall procedural costs, along with a trend toward fewer AEs. 33 After the completion of ERCP, the LAMS is removed based on the maturation of the tract, which typically occurs in 3–4 weeks. 34 After LAMS removal, usually with grasping forceps or snare, the fistulous tract may either be closed endoscopically or allowed to close spontaneously. Spontaneous closure can occur following simple stent removal or may be facilitated by the application of argon plasma coagulation (APC) to the fistula tract, promoting reepithelialization. Primary endoscopic closure, when performed, may involve the use of OTSC, TTSCs, or suturing systems. 35 In certain cases, the stents can be placed after the LAMS is removed to maintain the fistula tract for future use. 23
Indication
In patients with altered anatomy with Roux-en-Y reconstruction, traditional ERCP using a duodenoscope is difficult and often ineffective for accessing the ampullary region. Various techniques have been explored, including using a combination of pediatric colonoscopes and duodenoscopes, which have shown limited success rates of 33%–67%.36,37 The introduction of enteroscopy-assisted ERCP (EA-ERCP) improved biliary access rates to 60%–65%. 38 In 2002, the laparoscopic-assisted ERCP (LA-ERCP) technique was introduced, offering higher technical and clinical success rates compared to other approaches in patients with SAA.5,39 The procedure begins with laparoscopic identification and mobilization of the excluded stomach, followed by the creation of a gastrotomy on its anterior surface. 40 A 15–18-mm trocar is then introduced through the abdominal wall and positioned within the gastrotomy, which is secured using a purse-string suture. This maneuver facilitates anchoring of the excluded stomach to the anterior abdominal wall, providing a stable access point. A standard duodenoscope is subsequently advanced through the trocar into the gastric remnant, allowing retrograde endoscopic access to the biliary system. Fluoroscopic guidance is employed throughout to perform conventional ERCP maneuvers. Upon completion, the duodenoscope and trocar are withdrawn, and the gastrostomy is meticulously closed. 40 However, LA-ERCP remains resource-intensive, requiring coordination between endoscopy and surgical teams, and is associated with increased complication rates and costs. Although LA-ERCP presents several limitations, these include the requirement for advanced surgical and endoscopic expertise, increased resource utilization due to the need for an operating room rather than an endoscopy suite, the necessity for scope sterilization, and the logistical challenge of coordinating surgical and endoscopic teams.39,40 Surgical proficiency is particularly critical, especially in patients with high body mass index, extensive intra-abdominal adhesions, or prior abdominal surgeries, where accessing the excluded stomach can be technically demanding and time-consuming. Additionally, the endoscopist must be skilled in maneuvering the duodenoscope through the trocar and achieving appropriate positioning in the duodenum via the bypassed anatomy. 41 In the first decades of the 2000s, the widespread therapeutic EUS together with the introduction of LAMS fostered the development of the EDGE technique, which significantly revolutionized the management of pancreaticobiliary diseases in RYGB patients by eliminating the need for surgical or percutaneous access. It also facilitates repeat interventions for stent removal or exchange and eliminates the need to maintain a PEG tube for tract maturation, thereby contributing to lower complication rates. However, EDGE has not completely replaced previous traditional techniques. For example, the American Society for Gastrointestinal Endoscopy recommends EDGE as the preferred approach, citing its superiority over EA-ERCP across all outcome measures and its equivalence to LA-ERCP in terms of patient-centered outcomes and cost-effectiveness. 42 Also, the European Society of Gastrointestinal Endoscopy recommends that EDGE may be offered to patients with RYGB anatomy in specialized centers, following multidisciplinary evaluation, as a less invasive alternative to LA-ERCP. 43
Since its first description by Kedia et al. 16 in 2013, the technique has undergone substantial evolution, both in terms of clinical indications and technical approach, as documented by an increasing number of published studies. In the systematic review by Prakash et al., 44 the most frequent indication was choledocholithiasis (44%; 60/135), followed by pancreatitis (14%; 19/135), biliary obstruction (12%; 16/135), and cholangitis (10%; 14/135). Similarly, in the multicenter retrospective study by Runge et al., 45 choledocholithiasis remained the leading indication (97/178, 54%), followed by bile leak (18/178, 10%), benign biliary stricture (16/178, 9%), and recurrent acute pancreatitis (12/178, 7%). Ghandour et al. analyzed a cohort of RYGB patients undergoing EUS-guided transgastric interventions (EDGI) and found that pancreatic pathologies were the primary indication in 68.1% of patients, including pancreatic masses (16/47, 34%), pancreatic fluid collections (10/47, 21.3%), pancreatic cysts (4/47, 8.5%), and pancreatic duct dilation (2/47, 4.3%). In addition, 25.6% of patients underwent EDGI for evaluation or management of luminal gastroduodenal diseases. 46
Efficacy
In the systematic review by Prakash et al., which included 169 patients across 17 studies, GG fistula was performed in 61% and JG in 39% of cases. The 15-mm LAMS was more frequently used (132/169 cases, 78%) than the 20-mm version (37/169 cases, 22%), and cautery-assisted LAMS placement was preferred in 77% of cases. The TS rate for fistula creation was 99%, with an ERCP success rate of 98%. 44 Runge et al., in their multicenter study which included 78 patients, reported similar findings: 92% of cases involved ec-LAMS, with a preference for 15-mm stents (63%). Stent fixation was performed in 18% of cases, mostly using endoscopic suturing (91%). TS, defined as successful LAMS placement followed by ERCP completion, was achieved in 98% of cases. 45 Ghandour et al., in their multicenter study on 47 patients, confirmed the high TS of EDGI (96.3%). GG fistula was used in 78.7% of cases, with 20-mm LAMS used in 55.3% and 15-mm in 44.7%. Stent anchoring was performed in 38.3% of patients, mainly through endoscopic suturing in these cases as well. Reported technical failures were rare and related to stent misdeployment or migration during the procedure. 46
These data demonstrate that EDGE provides favorable therapeutic outcomes after ERCP procedures in patients with RYGB, as also shown by Su et al. in the only available meta-analysis published in 2023. This meta-analysis included 14 trials with a total of 574 patients who underwent 585 EDGE procedures, achieving cumulative technical success and clinical success rates of 98% and 94%, respectively, with no observed heterogeneity among the studies (p = 0.999, I2 = 0% for TS and p = 0.912, I2 = 0% for CS). 47
Furthermore, the effectiveness of EDGE has been evaluated in several case reports, particularly in complex anatomical settings, for example, in patients who underwent an SG that was later converted to an RYGB.48,49
Safety
Although EDGE is a promising procedure with a good safety profile, the most reported complications in the literature include perforation, stent migration, bleeding, and persistent fistula.
Perforation is a recognized AE, with incidence rates varying across studies 50 . It can occur both during and after the procedure, often in conjunction with LAMS migration. Perforation may occur during initial LAMS release or following dislodgement during duodenoscope passage through the LAMS lumen, and it happens more frequently after JG LAMS placement than after GG placement. 45
LAMS migration is a frequent complication of the EDGE procedure, with reported rates ranging from 15% to 33%, and it can occur during or after subsequent ERCP.9,18,51 Krafft et al. in their retrospective study compared the risk of LAMS dislodgement between SS-EDGE and dual-session EDGE (DS-EDGE) with a shortened interval of 2–4 days between anastomosis creation and ERCP. During the study period, 11 patients underwent SS-EDGE, with 64% (7/11) having a GG-fistula and 36% (4/11) a JG-fistula. Meanwhile, 10 patients underwent shortened-interval DS-EDGE, with 60% (6/10) having a GG-fistula and 40% (4/10) a JG-fistula. 52 The results showed that SS-EDGE carried a significantly higher risk of LAMS dislodgement (5/11, 45%) compared to the dual-session approach (1/10, 10%), with a relative risk 8.17 times higher (p = 0.05). Moreover, while minor AEs occurred in 2/11 (18%) of patients in the SS-group, no AEs were reported in the DS-group. These findings suggest that a 2–4-day interval between the two steps of the procedure allows for partial fistula maturation, reducing the risk of complications without significantly delaying ERCP. 52
Techniques such as LAMS fixation, balloon dilation, and the use of larger-diameter LAMS (20 mm) can help reduce the risk of stent migration. However, limited data are available comparing these strategies in terms of clinical outcomes and associated AEs. Shinn et al. 53 conducted a retrospective cohort study to identify predictive factors for stent migration during the EDGE procedure. Among the 129 procedures reviewed, 11 cases of intraprocedural LAMS migration were observed, resulting in a migration rate of 8.6%. Univariate analysis revealed that the use of smaller (15 mm diameter) LAMS was a significant predictor of migration. Fewer intraprocedural migrations occurred with the 20-mm LAMS (3.5%) compared to the 15-mm LAMS (18.6%), with a risk ratio of 5.271 (95% confidence interval (CI): 1.47–18.87; p = 0.007). 53 Factors that did not significantly influence stent migration included LAMS dilation (p = 0.084), access route (transgastric vs transjejunal, p = 0.502), and use of electrocautery-enhanced stents (p = 0.141). Notably, none of the LAMSs that were secured with endoscopic suturing experienced migration, whereas 3 out of 11 non-sutured LAMSs did. This finding suggests that endoscopic suturing is a statistically significant predictor of reduced intraprocedural migration (p = 0.033). In contrast, double-pigtail stents were used to anchor LAMS in 11 cases, but this method did not significantly reduce the risk of migration (p = 0.504). 53
A pilot study by Khan et al. 54 evaluated the use of a TTS-endoscopic suturing system (TTS-ESS) to prevent stent migration in six patients who had undergone RYGB, all of whom underwent an SS-EDGE procedure. CS and TS were achieved in all patients without any procedure-related AEs. There were no instances of stent migration, pneumoperitoneum, or GI perforation. At the 4-week follow-up, the stents remained anchored, with no signs of migration. 54
One of the main factors limiting the widespread adoption of the EDGE procedure is the risk of developing a persistent fistula after LAMS removal. This complication can potentially lead to weight regain by reversing the gastric bypass and may also contribute to marginal ulcer formation.44,55 Reported rates of persistent fistula development following LAMS removal in EDGE procedures vary widely, ranging from 0% to 41%, with most studies reporting an average incidence of around 10%.18,56,57 This variability may be attributed to differences in technique, including LAMS diameter, the use or absence of primary closure, and inconsistencies across centers in the methods used to objectively assess fistula persistence after stent removal. In a multicenter-matched case-control study, Ghandour et al. sought to identify factors associated with the development of persistent fistulas. The study included 25 patients who were found to have a persistent fistula during follow-up surveillance and compared them with 50 patients who showed no signs of a fistula. 58 Objective testing to assess fistula presence was conducted, on average, 15.6 weeks after LAMS removal, though with a wide range of variability (±14.1 weeks). 58 Regarding the type of stents used, a 20-mm LAMS was placed in 60% of patients (45 cases), while a 15-mm LAMS was used in the remaining 40% (30 cases). The stents facilitated the creation of a GG-fistula in 77.3% of patients (58 cases) and a JG-fistula in 22.7% (17 cases). The mean dwell time of the LAMS before removal was 74.7 days, with substantial variability (±106.2 days). Following LAMS removal, 46 out of 75 patients (61.3%) underwent APC for fistula de-epithelialization to promote healing. In addition, primary closure of the fistula was performed in 20 patients (26.7%) using various endoscopic techniques: TTS-ESS in 17 patients (22.7%), endoscopic tacking in 2 patients (2.7%), and a combination of OTSCs and TTS-ESS in 1 patient (1.3%). A comparison between patients who developed a persistent fistula and those who did not revealed no significant differences in key factors such as fistula creation site (GG 84% vs JG 74%, p = 0.3), LAMS size (20 mm: 56% vs 62%, p = 0.6), deployment technique (free-hand: 68% vs 78%, p = 0.3), or procedural approach (SS-EDGE: 40% vs 58%, p = 0.1). Additionally, no significant differences were observed in the use of primary endoscopic closure (36% vs 22%, p = 0.2) or APC treatment (76% vs 54%, p = 0.07) following LAMS removal. However, LAMS dwell time was significantly longer in patients who developed a persistent fistula compared to those who did not (127.3 ± 163.6 vs 48.4 ± 42.7 days, p = 0.02). The risk of developing a persistent fistula increased by 9.5% for every additional 7 days the LAMS remained in place. Notably, a dwell time exceeding 40 days was identified as a strong predictor of persistent fistula formation (79.2% vs 45.8%; odds ratio (OR): 4.5; 95% CI: 1.5–14.0, p = 0.01). 58
In a recent retrospective bicenter study, Cui et al. 59 explored the effectiveness of closing the GG or GJ-fistula with the X-tack System (Boston Scientific, Marlborough, MA, USA) at the time of stent removal in 19 RYGB patients who had undergone SS-EDGE with a 20-mm LAMS. 59 The primary outcome was to assess whether fistulas persisted at least a month after the stent was removed. On average, patients had their LAMS in place for approximately 32 days (median = 19). At the time of stent removal, all patients underwent closure of the anastomosis. In 10 cases, the procedure included APC applied to the rim of the fistula before performing TTS tack-based suturing, while the remaining nine patients underwent TTS-ESS alone. Importantly, follow-up studies confirmed that every patient had achieved full fistula closure, highlighting the potential effectiveness of this approach. 59 Finally, Aghaie Meybodi et al. in their case report described a novel approach for closing a persistent 25-mm GG fistula. In a patient who had failed multiple prior endoscopic interventions, closure was successfully achieved using a postinfarct muscular ventricular septal defect occluder, demonstrating an innovative off-label application of cardiac device technology in complex gastrointestinal fistula management. 60
Prakash et al. 44 in their systematic review reported an AE’s rate of 27.8% (47/169) of EDGE procedures. Of these, 41/169 (24.2%) were directly related to the EDGE procedure, while 6/169 (3.5%) were typical complications of ERCP. Minor AEs occurred in 18% (31/169) of cases, including intraprocedural stent migration (n = 19/169, 11.2%), stent misdeployment (n = 8/169, 4.7%), and post-procedure abdominal pain (n = 4/169, 2.3%). Stent migration often occurred during ERCP and was managed endoscopically using LAMS replacement or by stent-in-stent technique using LAMS or esophageal FC-SEMS placement. Moderate AEs were observed in 5.3% (9/169) of patients, comprising bleeding (n = 5/169, 2.9%), persistent fistula (n = 2/169, 1.1%), and perforation (n = 2/169, 1.1%). Bleeding was attributed to fistula creation (n = 1), LAMS exchange (n = 1), or unknown causes (n = 3). Persistent fistulas, one GG, and one JG, were detected at 6 weeks and 7 months post-LAMS removal. One severe AE was reported, involving a perforation caused by a malfunction of an LAMS delivery system, requiring emergent surgery. 44
In the study by Runge et al., early AEs occurred in 15.7% of patients (28/178). Perforation was reported in 6/178 cases (3%), mostly during EUS-guided transjejunal (n = 4/178, 2.2%) stent placements, with one perforation occurring during ERCP following sphincterotomy. 45 5/6 (83.3%) of these perforations were successfully managed with endoscopic closure (n = 4) or conservative treatment, while one patient (16.7%) required surgery. Intraprocedural LAMS migration occurred in 2/178 patients (1.1%), leading to the abortion of the EDGE procedure without reattempting it. Additionally, LAMS misdeployment was reported in 9/178 cases (5%), but all were resolved using the stent-in-stent technique, allowing the procedures to be completed. Among the 90 patients (59% of those who underwent LAMS removal) who were evaluated for persistent fistula, 10% (9/90) were diagnosed with a persistent fistula at least 8 weeks after stent removal, with no significant difference in persistent fistula rates between GG (4/45, 9%) and JG (5/45, 11%) LAMS placements (p = 0.73). Notably, diabetes mellitus was significantly more prevalent among patients who developed persistent fistulas (56% vs 21%, p = 0.02). Although no preventive closure was performed at the time of LAMS removal, five out of nine patients with persistent fistulas subsequently underwent successful endoscopic treatment. 45
In the study by Ghandour et al., 46 AEs were reported in 10.6% of patients (5/47). The most frequent AE was intraprocedural LAMS migration, occurring in 6.4% of cases (3/47). Additionally, two patients (4.3%) experienced delayed spontaneous LAMS migration, both without significant clinical consequences. Most LAMS migration cases (4/5) were managed endoscopically or conservatively, while one patient required surgery due to JG tract dehiscence and perforation. Notably, all cases of LAMS migration occurred in patients without stent anchoring, with a significantly higher migration rate observed in this group compared to those with anchoring (27.8% vs 0%; p = 0.001). Although smaller LAMS diameters were associated with a higher migration risk than larger stents, the difference was not statistically significant (19% vs 3.8%; p = 0.15). Migration was also more frequent in DS-EDGE compared to SS procedures (13.3% vs 5.9%; p = 0.6), though this was not statistically significant. Regarding follow-up, LAMS removal was performed in 28 patients (59.6%) with a mean dwell time of 84.3 ± 87.6 days. Among these, primary fistula closure was attempted in 64.3% of cases using endoscopic suturing (n = 12), TTSCs (n = 3), OTSCs (n = 2), or endoscopic tacking (n = 1). The remaining 35.7% of patients experienced spontaneous fistula closure. Objective assessments to evaluate persistent fistulas were conducted in 17/47 patients (36.1%) at a median of 16 weeks post-LAMS removal. Persistent fistulas were detected in two patients (11.7%). One case was managed with successful endoscopic closure using APC and suturing, while the other fistula was intentionally left open to aid in weight gain. 46
In a multicenter study conducted by Kedia et al., 61 the long-term outcomes of EDGE were assessed, with a particular focus on the rate of fistula persistence rate. A total of 172 patients were included in the study, with the majority (82%) having biliary stricture as the primary indication for the procedure. Among these patients, 125 (73%) underwent GG fistula creation. A 20-mm LAMS was used in 81% of cases, with a 15-mm LAMS in the remaining 19%, and LAMS fixation system was used in 19% of cases. The TS rate was 99.4%, with only one case of failure. The most common AE was stent migration or dislodgement, occurring in 28 patients (16.3%). Of these, 19 cases (68%) were peri-procedural and were managed with the stent-in-stent technique, although two required surgical intervention. In the remaining 9 cases (32%), complications occurred in the post-procedural period; 11 were managed through stent repositioning, or replacement, while one patient required surgical removal due to small bowel obstruction caused by stent migration. Bleeding was reported in 11 patients (6%), all of whom were successfully managed endoscopically. LAMS removal was performed in 142/172 patients (83%), with a mean indwell time of 69 days. At the time of LAMS removal, the fistula had closed in 49% of cases, with various closure techniques used, including suturing (52 cases), APC (39 cases), and OTSC (17 cases). Among the 163 patients with follow-up data, 61% had follow-up longer than 3 months, and 34% had follow-up beyond 6 months. Fistula closure was reassessed in 62 patients (43.6%) at an average of 256 days after LAMS removal. Of these, 31% (19 patients) were found to have persistent fistulas. The only factor significantly associated with fistula persistence was the total LAMS dwell time, with the persistent group having a mean indwell time of 86 days compared to 50 days in those without fistula persistence (p < 0.004). Other factors, such as LAMS deployment location, diameter, and methods used for fistula closure, did not show a statistically significant difference. Of the 19 patients with persistent fistulas, 11 underwent repeat endoscopic procedures, achieving 100% TS using a combination of suturing, APC, and OTSCs. 61
Comparison with other techniques
In patients with RYGB anatomy, conventional ERCP is technically unfeasible due to the inability to reach the second portion of the duodenum through standard endoscopic routes. Historically, LA-ERCP has been the preferred approach in this population, particularly advantageous in patients who require a simultaneous cholecystectomy, as both procedures can be completed in a single surgical session under general anesthesia. However, LA-ERCP is associated with a relatively high rate of AEs, reaching up to 36% in some series, and may not be optimal for patients expected to need multiple ERCP sessions.5,62
EA-ERCP offers a nonsurgical alternative by using a balloon-assisted or spiral enteroscope to access the biliary tree through the altered anatomy. While it addresses the challenge of extended limb length, its TS remains inferior to LA-ERCP. This is largely due to the limitations of using forward-viewing enteroscopes without the elevator and the lack of dedicated ERCP accessories compatible with long working lengths. 63
More recently, EDGE has emerged as a highly promising technique. By creating a GG or JG fistula using an LAMS, EDGE enables endoscopists to gain direct access to the excluded stomach and perform ERCP using standard duodenoscopes. This technique bypasses the anatomical challenges posed by the Roux limb and eliminates the need for coordination between surgical and endoscopic teams. One of EDGE’s key advantages is its minimally invasive nature, reducing the reliance on general anesthesia and avoiding surgical intervention. Additionally, the transgastric access created can be reused for multiple procedures, making EDGE particularly advantageous for patients with suspected ampullary lesions, biliary malignancies, or those requiring serial interventions for staging, palliation, or surveillance. Other benefits include shorter hospital stays, lower overall healthcare costs, and reduced morbidity compared to LA-ERCP. Nevertheless, LA-ERCP remains a valuable alternative in cases where surgical access is already planned or when a safe and appropriate window for EDGE creation cannot be identified. 42 Figure 3 presents the main advantages and disadvantages of the three techniques (Figure 3).

Pros and cons of the three techniques.
In a meta-analysis published in 2024 by Gangwani et al., 64 EDGE was compared with LA-ERCP and EA-ERCP. The overall TS rate was 94.8% for EDGE, defined as the successful creation of a transmural fistula using an LAMS, followed by completion of the ERCP procedure. For LA-ERCP, the TS rate was 95.0%, achieved through coordinated surgical access and successful ERCP completion. In contrast, EA-ERCP demonstrated a lower TS rate of 66.3%, with success defined as completion of the ERCP using either single- or double-balloon enteroscopy, depending on device availability. There was no statistically significant difference in TS between the EDGE and LA-ERCP groups (OR = 0.768, 95% CI: 0.196–3.006, p = 0.704, I2 = 14.13%). However, EDGE demonstrated a significantly higher success rate than EA-ERCP (OR = 4.507, 95% CI: 2.201–9.266, p < 0.001, I2 = 0.08%). Similarly, LA-ERCP showed a statistically significant advantage over EA-ERCP (OR = 0.159, 95% CI: 0.081–0.313, p < 0.001, I2 = 15.38%). The AE rates were 14.9% for EDGE, 20.3% for LA-ERCP, and 14.7% for E-ERCP. When comparing these techniques, no statistically significant differences emerged. For instance, the comparison between EDGE and LA-ERCP yielded an OR of 0.720 (95% CI: 0.305–1.696, p = 0.452, I2 = 21.18%), indicating comparable safety profiles. Similarly, the comparison between EDGE and EA-ERCP resulted in an OR of 0.902 (95% CI: 0.364–2.234, p = 0.824, I2 = 42.67%), further supporting the absence of a significant difference in AEs rates. Moreover, when evaluating EA-ERCP versus LA-ERCP, the data also showed no significant difference in complication rates. Overall, these findings suggest that EDGE, LA-ERCP, and E-ERCP offer similar safety profiles. 64
In a previous meta-analysis (76 studies), Gkolfakis et al. 65 compared the three techniques (EDGE, EA-ERCP, LA-ERCP) and provided an in-depth analysis of the various approaches used during EA-ERCP. Most patients (n = 4934, 72.9%) underwent Roux-en-Y anastomosis, with 38.9% of these cases (n = 1919) involving bariatric procedures using RYGB. They reported a pooled TS rate, defined here as reaching the area of interest (ampulla or anastomosis), of 87.3% (95% CI: 85.3–89.4) for EA-ERCP. However, when breaking down the results by modality, first-generation manual spiral enteroscopy-assisted (SEA)-ERCP showed a lower success rate of 70.3% (95% CI: 55.1–85.6). In contrast, single-balloon enteroscopy-assisted (SBEA)-ERCP allowed for optimal positioning in front of the ampulla or anastomosis in 88.1% of cases (95% CI: 85.5–90.6), and double-balloon enteroscopy-assisted (DBEA)-ERCP achieved a similar rate of 89.8% (95% CI: 87.1–92.4). EDGE, on the other hand, demonstrated a superior pooled TS rate of 97.9% (95% CI: 96.4–99.4), while LA-ERCP enabled sufficient positioning in front of the ampulla in 99.1% of patients (95% CI: 98.6–99.7). The analysis clearly showed that both EDGE and LA-ERCP were superior to EA-ERCP in terms of TS (p < 0.001), yet there was no significant difference between EDGE and LA-ERCP regarding the ability to achieve adequate positioning (p = 0.43). Additionally, among the various EA-ERCP modalities, the TS of first-generation manual SEA-ERCP was significantly lower compared to DBEA or SBEA (p < 0.001). In contrast, the success rate of balloon enteroscopy-assisted ERCP (BEA-ERCP) did not significantly differ between DBEA and SBEA (p = 0.65). 65
Connell et al. 66 evaluated the use of EDGE specifically for the management of choledocholithiasis in patients post-RYGB, comparing its outcomes to those of LA-ERCP, BEA-ERCP, laparoscopic common bile duct exploration (LCBDE), EUS-guided hepatic access with antegrade clearance (EGHAC), percutaneous transhepatic biliary drainage (PTBD), and rendezvous guidewire-assisted (RGA) ERCP. The study found exceptionally high stone clearance rates across several techniques. LA-ERCP achieved a perfect clearance rate (1.00; 95% CI: 0.99–1.00, p = 0.47), and EDGE demonstrated similarly high efficacy (0.97; 95% CI: 0.90–1.00, p = 0.54). Other modalities, including IGS ERCP, PTHBD, and LCBDE, also performed remarkably well, with clearance rates of 1.00 (95% CI: 0.87–1.00), 1.00 (95% CI: 0.96–1.00), and 0.99 (95% CI: 0.93–1.00, p < 0.001), respectively. In contrast, BEA and EGHAC showed significantly lower stone clearance rates of 61.5% (95% CI: 44.3–76.3, p = 0.188) and 74.0% (95% CI: 42.9–91.5, p = 0.124), suggesting that these techniques may require further refinement or more selective patient application to achieve comparable results. EDGE was associated with the highest complication rate among the modalities studied, at 24.3%, with stent dislodgement or malpositioning being the most reported issue. BEA had the lowest complication rate (5.9%), while LA-ERCP and IGS ERCP had similar rates of AEs, at 12.8% and 14.3%, respectively. 66
Another crucial aspect to consider is the economic aspect. Wang et al. 51 compared EA-ERCP, LA-ERCP, and EDGE not only in terms of clinical efficacy but also regarding safety, including parameters such as hospitalization duration and overall costs. The analysis revealed that patients undergoing EDGE had the lowest post-procedure hospitalization rate, at 44%, compared to 77% for DAE and 100% for LA-ERCP (p < 0.01). Additionally, GATE was associated with the shortest hospital stay, with a median duration of 0 days, compared to 2 days for DAE and 3 days for LA-ERCP (p < 0.0001). From a cost-effectiveness perspective, LA-ERCP was the most expensive approach, with the highest total procedural and hospitalization costs based on Medicare/Medicaid reimbursement data. In contrast, EDGE and EA-ERCP showed comparable procedural and hospitalization costs (p = 0.76). These findings highlight the potential economic and clinical advantages of EDGE, particularly in minimizing hospital stays and associated costs. 51
EDGE has increasingly emerged as the preferred approach over LA-ERCP in select patient populations, particularly those with suspected ampullary lesions, biliary malignancies, or a high likelihood of requiring repeated biliary interventions. One of EDGE’s primary advantages is its minimally invasive nature, which often eliminates the need for surgical access and general anesthesia. The technique enables the creation of a durable transgastric fistula allowing for repeated ERCP sessions without the need for new access each time. This is especially valuable in patients undergoing oncologic staging, palliative treatment, or long-term biliary surveillance. Furthermore, EDGE is generally associated with shorter hospital stays, lower procedural costs, and reduced postoperative morbidity compared to LA-ERCP. In contrast, LA-ERCP may still be preferred in specific clinical scenarios, such as in patients requiring a concomitant cholecystectomy. In such cases, LA-ERCP allows both ERCP and cholecystectomy to be performed under a single sedation, streamlining patient care and minimizing cumulative procedural risk. Additionally, when a safe anatomical window for EDGE cannot be identified—due to unfavorable gastric pouch orientation, interposed vasculature, or distorted anatomical landmarks—LA-ERCP, as well as EA-ERCP, remain important and viable alternatives. Ultimately, the choice between EDGE and LA-ERCP should be individualized, considering procedural feasibility, patient comorbidities, anatomical considerations, and the anticipated need for repeat interventions. 42 Table 1 summarizes the key data on technical outcomes and AEs associated with EDGE, EA-ERCP, and LA-ERCP, as reported in major comparative retrospective studies (Table 1).
Summary of technical outcomes and adverse events of EDGE, EA-ERCP, and LA-ERCP in major comparative retrospective studies.

Indicates a statistically significant difference.
AE, adverse events; EA-ERCP, enteroscopy-assisted endoscopic retrograde cholangiopancreatography; EDGE, endoscopic ultrasound-directed transgastric ERCP; LA-ERCP, laparoscopic-assisted ERCP.
EUS-directed transenteric ERCP
EDEE is an advanced endoscopic technique that builds upon the success of EUS-guided biliary drainage (EUS-BD). This procedure involves the creation of a transgastric or transenteric anastomosis between the proximal gastrointestinal tract and a small bowel loop containing the biliary orifice. 73 By utilizing an LAMS, this anastomotic connection enables the endoscope to bypass the altered anatomy and access the papilla or the biliary anastomosis in a standard retrograde manner, allowing the use of conventional ERCP accessories 74 (Figure 4).

Endoscopic ultrasound transenteric endoscopic retrograde cholangiopancreatography.
EDEE was first described in 2019 by Bukhari et al., 73 who performed the procedure on a 41-year-old woman with a history of orthotopic liver transplantation (OLT) and Roux-en-Y reconstruction for primary sclerosing cholangitis. The patient presented with recurrent cholangitis due to biliary cast syndrome, with previous attempts at EA-ERCP failing due to extensive biliary casts. 73 EDEE successfully created a direct route to the biliary tree, enabling effective treatment.
The technique
EDEE requires meticulous planning to determine the shortest and most favorable route to the papilla or HJ, enhancing the success of future cannulation attempts and biliary interventions. 74 Preprocedural imaging and surgical history should be carefully reviewed to evaluate the anatomical layout and identify the optimal site for transenteric anastomosis. In cases of HJ, determining whether a single or dual anastomosis is present is crucial for procedural planning. 74
As with EUS-GE, which has three main techniques, EDEE also has different technical variations, primarily influenced by patient anatomy, clinical setting, and device availability.24,25 Concerning patient anatomy, EDEE is most commonly performed in patients who underwent pancreaticoduodenectomy according to the Whipple Procedure, also in pylorus-preserving variant, total or partial gastrectomy, biliary resection, or OLT with HJ, all with Roux-en-Y reconstruction, and duodenal switch.
In the first EDEE technical variant, similar to the WEST of EUS-GE, the procedure begins with the identification and distension of the biliopancreatic limb. 73 An upper endoscopy is typically performed by therapeutic endoscope with a large operative channel to evaluate the postsurgical anatomy and, if feasible, access the biliopancreatic limb directly. 74 If successful, jejunal loop adequate distension can be achieved by placing an orojejunal tube (usually 8,5-10Fr in diameter) or by direct instillation by the working channel of the frontal viewing endoscope. 74
Once the target limb is adequately distended with a contrast medium-saline-dye (indigo carmine/methylene blue) solution, a linear echoendoscope is introduced 74 (Figure 5).

Technical steps of EDEE. (a) Biliary loop distension by a solution of saline mixed with contrast medium using the previously placed PTBD. (b) EUS view of the dilated biliary loop. (c, d) EUS and endoscopic view of the 15 × 10 mm electrocautery-enhanced LAMS release. (e) Contrast medium flows through the LAMS lumen from the stomach to the biliary loop lumen. (f) Final fluoroscopic view after ERCP with placement of a fully covered self-expanding metal stent for treating stenosis of the hepatico-jejunostomy.
Color Doppler ultrasound is utilized to rule out intervening vessels, and the distance between the two luminal walls is measured to ensure feasibility for anastomosis creation (usually, it should be <10–15 mm). In select cases, the target loop can be accessed by a 19-gauge FNB needle for contrast medium injection or aspiration of the blue solution to confirm appropriate positioning. 75 Another crucial aspect before proceeding with LAMS deployment is accurately identifying the biliary loop under EUS guidance. This technical step begins at the hepatic hilum, with EUS guidance used to follow the common hepatic duct down to the biliodigestive anastomosis or the papilla. 7 When using an orojejunal catheter for biliary loop distension, the loop with the catheter inside should be identified under EUS guidance to confirm the correct visualization before proceeding. Following confirmation, an ec-LAMS is deployed under EUS and fluoroscopic control, creating a gastro-jejunal (GJ) or jejunum-jejunal anastomosis. 8 The diameter of the LAMS should allow the passage of the duodenoscope/therapeutic endoscope; for this reason, preferably a 15- or 20-mm LAMS is chosen. The correct placement is confirmed by the rapid flow of the solution through the LAMS lumen, together with the direct visualization of the jejunal mucosa through the LAMS lumen. Depending on the clinical setting (urgent vs elective procedure), either an SS or a DS approach is selected. In urgent cases (e.g., severe cholangitis/cholecystitis), the LAMS lumen can be dilated using a pneumatic balloon catheter, allowing the endoscope to be advanced for immediate ERCP. 8 Although strong evidence on this aspect is lacking, LAMS fixation with TTSCs or a dedicated OTSC may also be considered to enhance stability. 29
In contrast, if a DS approach is planned, ERCP is performed after a waiting period of 3–4 weeks to allow for anastomotic maturation. 74 In cases where an internal PTBD has already been placed, the biliary-jejunal loop distension could be obtained by the instillation of contrast medium solution directly by the existing drainage (Figure 5(a)). In afferent limb syndrome cases with significant jejunal loop dilation caused by downstream stenosis, the target loop is often easily identified through the stomach by EUS guidance and then accessed by ec-LAMS.76,77
Once the stent is in place, a therapeutic frontal viewing endoscope or a duodenoscope is advanced through the LAMS lumen to access the biliary loop. Fluoroscopic guidance is essential to maintain the correct direction, as the upstream and downstream limbs may appear similar. Once the biliary orifice is accessed, standard ERCP techniques can be applied for diagnostic and therapeutic interventions.
Indication
Indications for EDEE include cases where standard ERCP, EA-ERCP, and/or EUS-BD have failed or are not feasible. 78
This approach is beneficial in patients with SAA, such as RYHJ, Whipple procedure, BII, or duodenal switch anatomy.8,75
EDEE is especially valuable when postsurgical small bowel loops are long and prohibit endoscopic reach to the biliary-enteric anastomosis using DAE. It is also advantageous in scenarios requiring advanced therapeutic interventions—such as electrohydraulic lithotripsy, cholangioscopy, or stent revisions—which may not be feasible through conventional EUS-HGS, which could be limited by the absence of adequate bile duct dilation or the need for larger working channels. Additionally, EDEE provides an important alternative in patients for whom PTBD is contraindicated or undesirable, or in whom EUS-HGS is technically challenging, such as those with a history of total gastrectomy, hepatic resection, or OLT. 74
In the most extensive report available, Mutignani et al. evaluated EDEE in 80 patients with SAA. More specifically, HJ with a Roux-en-Y loop was the most common type of reconstruction (91%), followed by hepatectomy with biliary resection and HJ on a Roux-en-Y loop, OLT with HJ on a Roux-en-Y loop, and Whipple resection. 79 The primary indications included biliary single-duct anastomotic stricture in 74% of patients, choledocholithiasis in 7.2%, biliary multiple-duct anastomotic stricture in 5.7%, uncovered self-expanding metal stent entrapment in 5.7%, recurrent cholangitis in 2.9%, dehiscence of the biliary anastomosis in one case (1.4%), biliary leak following left hepatectomy in a prior OLT in 1.4%, and biliary fistula after pancreaticoduodenectomy in 1.4%. 79
Similarly, a retrospective multicenter study by Ichkhanian et al. 8 (31917168) analyzed data from 7 hospitals between 2014 and 2018, involving 18 patients. The leading indications were malignant biliary strictures (44.4%), biliary stones (27.8%), benign strictures (22.2%), and a retained pancreatic stent causing recurrent pancreatitis (5.6%). The most frequent surgical reconstructions encountered were Whipple procedures (55.5%) and RYHJ (33.3%), followed by BII and duodenal switch (both 5.6%). 8
Furthermore, EDEE may be integrated into broader endoscopic strategies. For instance, when an EUS-GE is performed for malignant gastric outlet obstruction (GOO), the created anastomosis can provide future access to the papilla, further expanding endoscopic treatment options. 74 A recent retrospective analysis by Kesar et al. included nine patients with concurrent GOO and biliary obstruction. All patients underwent gastroduodenostomy using a 20 × 10 mm LAMS, followed by ERCP through the gastroduodenal stent, resulting in complete resolution of jaundice in 100% of cases. 80
Efficacy
Limited research has been conducted on the outcomes of EDEE, but existing studies suggest encouraging results (Table 2).
This table summarizes the outcomes of the largest series (⩾5 patients) on EDEE published so far.

EDEE, endoscopic ultrasound-directed transenteric ERCP; ERCP, endoscopic retrograde cholangiopancreatography; FC-SEMS, fully covered self-expandable metal stent; LAMS, lumen-apposing metal stent.
Mutignani et al. 81 evaluated EDEE in 32 patients with either RYHJ or Whipple anatomy. The technical and clinical success rate was 96.9%, with one failure due to challenges in optimal echoendoscope positioning. In another case, the same problem occurred but was solved by using a 15 × 10-mm LAMS. 81
Ichkhanian et al. 8 analyzed EDEE in 18 patients with non-RYGB anatomy. Technical success rate was defined as successful completion of all steps of EDEE, while clinical success was defined as successful completion of EDEE and the intended interventions without the need for subsequent percutaneous drainage or surgery. Enteroenteric anastomosis creation was successful in all 18 cases (100%). ERCP was successfully performed in 17 patients after creation of the enteroenteric anastomosis for an overall technical success rate of 94.4%. Clinical success was achieved in 17 of 18 patients (94.4%). The one case in which both clinical and technical failures occurred after EUS-guided GJ creation was due to a stricture 10 cm distal to the GJ, and thus ERCP could not be completed. Fourteen patients underwent EDEE in separate sessions, with a median interval of 21 days between procedures. Four patients (22.2%) underwent same session enteroenteric anastomosis creation and ERCP. 8
Donatelli et al. 82 performed a retrospective study aimed at evaluating the efficacy, and short- and long-term outcomes of EUS-guided duodeno or jejuno-jejunal anastomosis with permanent placement of LAMS for the management of hepaticobiliary disorders following pancreaticoduodenectomy, total gastrectomy, or HJ with Roux-en-Y-reconstruction. Technical success for LAMS deployment was defined as successful creation of an EUS-guided anastomosis, and technical success for ERC was defined as successful completion of biliary treatment. Clinical success was defined as stricture resolution at radiological examination and improvement of liver chemistry tests. The technical success of LAMS placement was 91% (10/11 patients), with one failure related to the excessive distance of the target jejunal loop. Technical success for the ERCP treatment was 100%. Clinical success was achieved in all patients. 82
In the recently published large retrospective series by Mutignani et al., 79 the authors defined TS as the successful creation of the anastomosis and the ability to perform biliary interventions. CS was defined as the resolution of the biliary condition and improvement in the patient’s clinical status. In this series, jejunal loop distension was achieved using a previously placed PTBD in 50% of patients, and a 15 × 10 mm ec-LAMS was used in most cases. TS was achieved in 79 out of 80 patients (98.7%), with the only technical failure resulting from an inability to maintain a stable position with the EUS scope. Single-session ERCP (SS-ERCP) was performed in 57 patients (71.25%), and the overall CS rate was also 98.7%, with one clinical failure due to the inability to perform ERCP caused by the long distance between the HJ and the created anastomosis. 79
Additionally, a recent international, multicenter study by Mangiavillano et al. 83 included 270 patients with SAA undergoing LAMS placement across 25 tertiary care centers (through November 2023). While the overall technical and clinical success rates were high (98% and 97%, respectively), only three patients in this cohort underwent EDEE. Despite the limited sample size, EDEE achieved a 100% technical and clinical success rate, with only one reported AE, a stent misdeployment, which was successfully managed. These findings reinforce the potential efficacy of EDEE while also highlighting the need for further dedicated studies evaluating its outcomes on a larger scale. 83
Safety
EDEE is generally considered safe when performed by experienced therapeutic endoscopists at high-volume centers. 24 Periprocedural AEs’ rates range from 3% to 6%, while overall complications occur in approximately 18%–28% of cases.8,81 A retrospective study of 18 patients undergoing EDEE reported one case of post-procedural abdominal pain, which resolved with conservative management, and four cases requiring endoscopic reintervention due to stent occlusion. 8 Another systematic review of 32 patients identified one instance of moderate, self-limiting bleeding, along with 5 late AEs, including 4 cases of stent migration and 1 persistent biliocutaneous fistula in a patient with a percutaneous transhepatic drain. 81
In the large series by Mutignani et al., 79 14 AEs were reported among 80 patients, corresponding to an overall AE’s rate of 17.5%. These included seven early and seven late AEs. Among the early AEs, the authors documented three cases of moderate self-limited bleeding, two cases of partial intraprocedural stent misdeployment, one case of acute respiratory failure, and one case of asymptomatic intraperitoneal free air detected under the diaphragm during ERCP, attributed to partial detachment of the jejunal loop. The most common late AEs were spontaneous LAMS displacements, occurring in six patients (four complete and two partial), all of which were successfully managed using the LAMS-in-LAMS technique.79,84,85
Among the most concerning AEs is stent misdeployment, occurring in around 8% of cases. 50 Recently, a classification system for stent misdeployment in EUS-GE has been introduced, categorizing misplacement into four types based on location. 86 Since EDEE involves similar technical principles to EUS-GE, comparable risks of stent-related complications may be anticipated. To mitigate migration risk, a staged approach with ERCP performed 3–4 weeks after LAMS placement is preferred to ensure anastomotic maturation. 74 If a single-session EDEE is pursued, using a large-caliber (e.g., 20 mm) LAMS with additional stent fixation techniques, such as suturing, is strongly advised. 74 Employing a small-caliber diagnostic duodenoscope when available may further reduce the risk of displacement. When withdrawing the endoscope post-ERCP, maintaining a trailing guidewire can facilitate luminal access for salvage stent placement if displacement occurs. 74
Early identification of stent dislodgement is crucial, as it is functionally equivalent to perforation. If detected promptly, rescue strategies include re-stenting over a retained guidewire or utilizing a stent-in-stent technique with a FC-SEMS placed coaxially within the LAMS to maintain anastomotic patency.84,85,87 Late-stage migration after tract maturation can generally be managed with simple stent replacement.
Future perspectives and conclusion
The introduction of LAMS has significantly expanded the therapeutic potential of EUS, enabling the creation of novel gastrointestinal anastomoses and revolutionizing the management of patients with SAA. Both EDEE and EDGE have emerged as highly promising techniques, offering minimally invasive alternatives to traditional ERCP, EA-ERCP, or PTBD. By establishing transenteric or transgastric access to the biliary system, these approaches provide repeatable and durable access routes, improving the feasibility of complex biliary interventions and reducing the need for surgery or multiple access procedures.
Current evidence—primarily derived from retrospective series and expert-driven case reports—suggests that EDEE and EDGE are relatively safe and effective when performed by experienced therapeutic endoscopists in high-volume referral centers. However, their widespread adoption is still limited by a lack of high-quality prospective data and standardized procedural frameworks. To address these gaps, future research should focus on the design and execution of multicenter, randomized controlled trials with well-defined inclusion criteria and standardized outcome measures. These studies should evaluate technical and clinical success, AE rates, fistula persistence after LAMS removal, need for reintervention, and long-term cost-effectiveness. Predefined endpoints and stratification by type of reconstruction (e.g., RYGB, RYHJ, Whipple), procedural setting (single-session vs two-step), and clinical indication (benign vs malignant disease) will be crucial in determining which patient subgroups benefit most from each approach.
Several key unmet needs remain unaddressed. One major area is the absence of standardization in critical technical aspects, including the optimal LAMS caliber. While 15-mm LAMS are frequently used, the best diameter may vary depending on anatomical factors, procedural goals, and the need for repeated access or passage of large accessories. Another unresolved issue is the optimal timing for stent removal and the management of the resulting fistulous tract. There is currently no consensus on whether spontaneous closure is sufficient or whether endoscopic closure—using OTSCs, suturing systems, or novel sealants—should be systematically pursued. Given that persistent fistulas may lead to delayed AEs or require additional procedures, the development of evidence-based algorithms for fistula closure is a pressing need.
Equally important is the lack of dedicated devices tailored specifically for EDGE and EDEE. Current tools, including LAMS and delivery systems, were designed for other indications such as pseudocyst drainage and may not fully meet the demands of transgastric or transenteric ERCP access. Future innovations should focus on stents with improved anchoring mechanisms, adjustable calibers, and anti-migration features, as well as delivery platforms optimized for difficult anatomical orientations and long endoscope working distances.
Finally, robust cost-effectiveness analyses are urgently needed to evaluate EDGE and EDEE against alternative strategies such as EUS-HGS, surgical revision (e.g., Roux-en-Y), or percutaneous interventions. These analyses should consider not only procedural costs but also long-term outcomes such as hospital readmissions, quality of life, reintervention burden, and survival—especially in oncologic patients or those with complex benign disease.
In conclusion, the adoption of EDEE and EDGE represents a paradigm shift in the management of pancreaticobiliary diseases in patients with altered anatomy. As the prevalence of bariatric surgery, liver transplantation, and complex GI resections continues to grow, so too will the demand for advanced endoscopic solutions. To fully realize the potential of these innovative techniques, a concerted effort is needed to standardize procedural protocols, reach consensus on key technical aspects such as fistula closure and stent selection, and drive the development of dedicated devices and training programs. Only through such coordinated advancements can EDEE and EDGE move from specialized innovations to standardized, widely accessible tools in therapeutic endoscopy.88,89
Finally, robust cost-effectiveness analyses are urgently needed to evaluate EDGE and EDEE against alternative strategies such as EUS-HGS, surgical revision (e.g., Roux-en-Y), or percutaneous interventions. These analyses should consider not only procedural costs but also long-term outcomes such as hospital readmissions, quality of life, reintervention burden, and survival—especially in oncologic patients or those with complex benign disease.
In conclusion, the adoption of EDEE and EDGE represents a paradigm shift in the management of pancreaticobiliary diseases in patients with altered anatomy. As the prevalence of bariatric surgery, liver transplantation, and complex GI resections continues to grow, so too will the demand for advanced endoscopic solutions. To fully realize the potential of these innovative techniques, a concerted effort is needed to standardize procedural protocols, reach consensus on key technical aspects such as fistula closure and stent selection, and drive the development of dedicated devices and training programs. Only through such coordinated advancements can EDEE and EDGE move from specialized innovations to standardized, widely accessible tools in therapeutic endoscopy.
Footnotes
Acknowledgements
All Anesthesiologists, nursing, technical, administrative, social and health care staff of the Gastroenterology and Digestive Endoscopy Units of IRCCS San Raffaele Hospital (Milan) and IRCCS Policlinico San Donato (Milan) and of the Unité d’Endoscopie Interventionnelle of the Hôpital Privé des Peupliers (Paris) for their indispensable contribution in the realization of these complex operative endoscopy procedures.
